Assessment of the Antibiofilm Performance of Chitosan-Based Surfaces in Marine Environments
Abstract
1. Introduction
2. Results
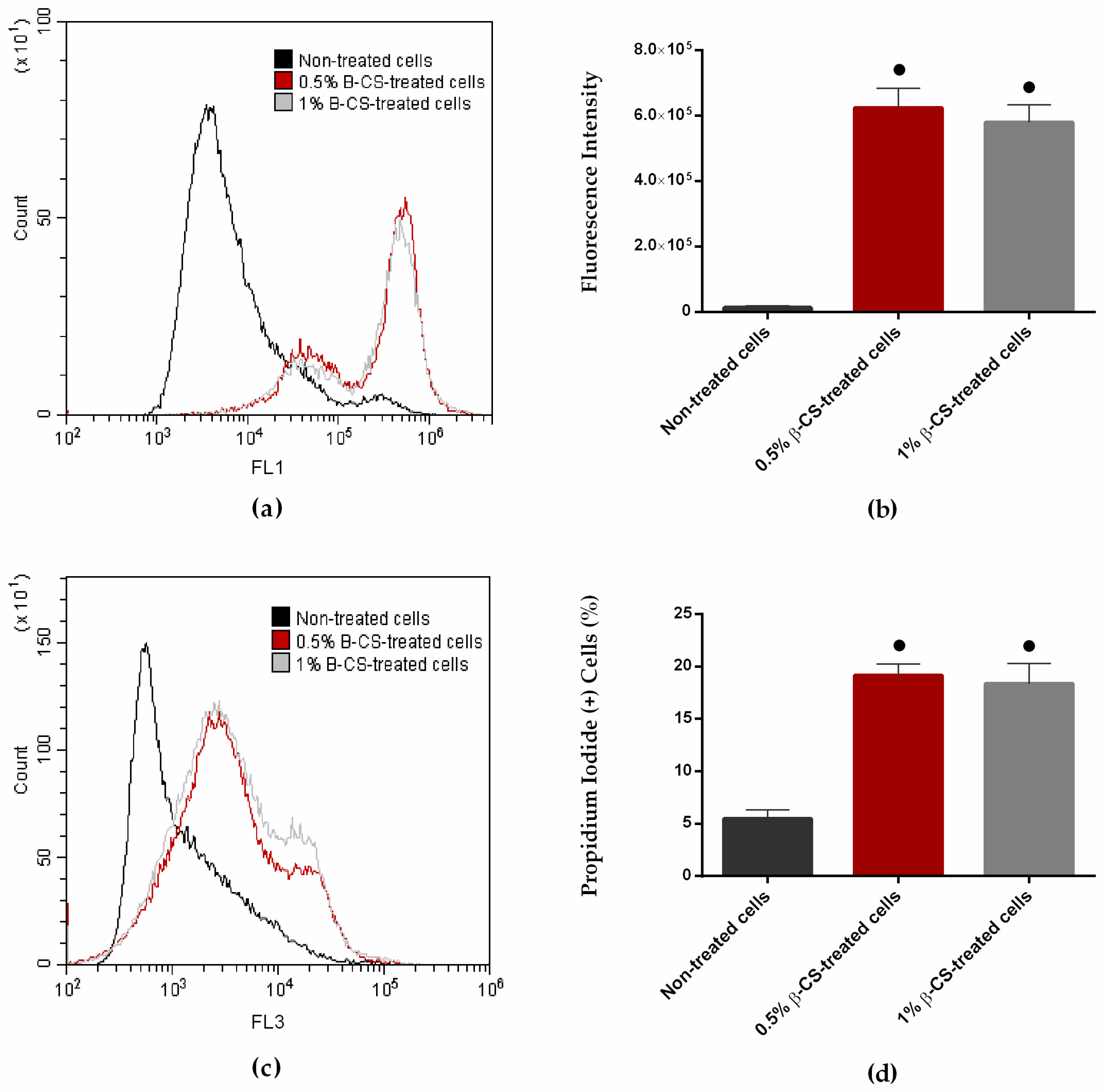
2.1. Chitosan and Its Mechanism of Action
2.2. Synthesis and Characterization of Poly (Lactic Acid)-Chitosan Surfaces
2.3. Antifouling Activity of CS-Based Surfaces
3. Discussion
4. Materials and Methods
4.1. Chitosan and Its Derivates
4.2. Bacteria Strains and Culture Conditions
4.3. Characterization of CS Mechanism of Action
4.4. Functionalization of Poly (Lactic Acid)-Chitosan Surfaces
4.5. Surface Characterization
4.5.1. Water Contact Angle Measurements
4.5.2. White Light Profilometry
4.6. Antifouling Activity of Chitosan-Based Surfaces
4.6.1. Biofilm Formation
4.6.2. Biofilm Quantification
Colony-Forming Unit Counts
Biofilm Thickness and Structure
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Faria, S.I.; Teixeira-Santos, R.; Gomes, L.C.; Silva, E.R.; Morais, J.; Vasconcelos, V.; Mergulhão, F.J.M. Experimental Assessment of the Performance of Two Marine Coatings to Curb Biofilm Formation of Microfoulers. Coatings 2020, 10, 893. [Google Scholar] [CrossRef]
- Amara, I.; Miled, W.; Slama, R.B.; Ladhari, N. Antifouling processes and toxicity effects of antifouling paints on marine environment. A review. Environ. Toxicol. Pharmacol. 2018, 57, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Mathew, N.T.; Kronholm, J.; Bertilsson, K.; Despeisse, M.; Johansson, B. Environmental and Economic Impacts of Biofouling on Marine and Coastal Heat Exchangers. In EcoDesign and Sustainability II: Social Perspectives and Sustainability Assessment; Kishita, Y., Matsumoto, M., Inoue, M., Fukushige, S., Eds.; Springer: Singapore, 2021; pp. 385–398. [Google Scholar]
- Kuliasha, C.A.; Fedderwitz, R.L.; Finlay, J.A.; Franco, S.C.; Clare, A.S.; Brennan, A.B. Engineered Chemical Nanotopographies: Reversible Addition–Fragmentation Chain-Transfer Mediated Grafting of Anisotropic Poly(acrylamide) Patterns on Poly(dimethylsiloxane) To Modulate Marine Biofouling. Langmuir 2020, 36, 379–387. [Google Scholar] [CrossRef]
- Lacoursière-roussel, A.; Bock, D.G.; Cristescu, M.E.; Guichard, F.; McKindsey, C.W. Effect of shipping traffic on biofouling invasion success at population and community levels. Biol. Invasions 2016, 18, 3681–3695. [Google Scholar] [CrossRef]
- Delauney, L.; Compère, C.; Lehaitre, M. Biofouling protection for marine environmental sensors. Ocean Sci. 2010, 6, 503–511. [Google Scholar] [CrossRef]
- Caruso, G. Microbial Colonization in Marine Environments: Overview of Current Knowledge and Emerging Research Topics. J. Mar. Sci. Eng. 2020, 8, 78. [Google Scholar] [CrossRef]
- Vinagre, P.A.; Simas, T.; Cruz, E.; Pinori, E.; Svenson, J. Marine Biofouling: A European Database for the Marine Renewable Energy Sector. J. Mar. Sci. Eng. 2020, 8, 495. [Google Scholar] [CrossRef]
- Yebra, D.M.; Kiil, S.; Dam-Johansen, K. Antifouling technology—past, present and future steps towards efficient and environmentally friendly antifouling coatings. Prog. Org. Coat. 2004, 50, 75–104. [Google Scholar] [CrossRef]
- Turner, A. Marine pollution from antifouling paint particles. Mar. Pollut. Bull. 2010, 60, 159–171. [Google Scholar] [CrossRef]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef]
- Oyatogun, G.M.; Esan, T.A.; Akpan, E.I.; Adeosun, S.O.; Popoola, A.P.I.; Imasogie, B.I.; Soboyejo, W.O.; Afonja, A.A.; Ibitoye, S.A.; Abere, V.D.; et al. Chapter 11—Chitin, chitosan, marine to market. In Handbook of Chitin and Chitosan; Gopi, S., Thomas, S., Pius, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 341–381. [Google Scholar]
- Zeidberg, L. Doryteuthis opalescens, Opalescent Inshore Squid. In Advances in Squid Biology, Ecology and Fisheries. Part I–Myopsid Squids; Rosa, R., O’Dor, R., Pierce, G., Eds.; Nova Publishers: New York, NY, USA, 2013; pp. 159–204. [Google Scholar]
- Yan, N.; Chen, X. Sustainability: Don’t waste seafood waste. Nature 2015, 524, 155–157. [Google Scholar] [CrossRef]
- Hülsey, M.J. Shell biorefinery: A comprehensive introduction. Green Energy Environ. 2018, 3, 318–327. [Google Scholar] [CrossRef]
- Kumar, S.; Ye, F.; Dobretsov, S.; Dutta, J. Chitosan Nanocomposite Coatings for Food, Paints, and Water Treatment Applications. Appl. Sci. 2019, 9, 2409. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Kim, K.D.; Chun, S.C. Antibacterial Activity of Chitosan Nanoparticles: A Review. Processes 2020, 8, 1173. [Google Scholar] [CrossRef]
- Li, Z.; Yang, F.; Yang, R. Synthesis and characterization of chitosan derivatives with dual-antibacterial functional groups. Int. J. Biol. Macromol. 2015, 75, 378–387. [Google Scholar] [CrossRef]
- Hosseinnejad, M.; Jafari, S.M. Evaluation of different factors affecting antimicrobial properties of chitosan. Int. J. Biol. Macromol. 2016, 85, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.C.; Chen, C.Y. Antibacterial characteristics and activity of acid-soluble chitosan. Bioresour. Technol. 2008, 99, 2806–2814. [Google Scholar] [CrossRef]
- Teixeira-Santos, R.; Lima, M.; Gomes, L.C.; Mergulhão, F.J. Antimicrobial coatings based on chitosan to prevent implant-associated infections: A systematic review. iScience 2021, 24, 103480. [Google Scholar] [CrossRef]
- Gomes, L.C.; Faria, S.I.; Valcarcel, J.; Vázquez, J.A.; Cerqueira, M.A.; Pastrana, L.; Bourbon, A.I.; Mergulhão, F.J. The Effect of Molecular Weight on the Antimicrobial Activity of Chitosan from Loligo opalescens for Food Packaging Applications. Mar. Drugs 2021, 19, 384. [Google Scholar] [CrossRef]
- Lima, M.; Teixeira-Santos, R.; Gomes, L.C.; Faria, S.I.; Valcarcel, J.; Vázquez, J.A.; Cerqueira, M.A.; Pastrana, L.; Bourbon, A.I.; Mergulhão, F.J. Development of Chitosan-Based Surfaces to Prevent Single- and Dual-Species Biofilms of Staphylococcus aureus and Pseudomonas aeruginosa. Molecules 2021, 26, 4378. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, E.; Bonnet, C.; Lemarchand, K. Biofouling growth in cold estuarine waters and evaluation of some chitosan and copper anti-fouling paints. Int. J. Mol. Sci. 2009, 10, 3209–3223. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Lelchat, F.; Cérantola, S.; Brandily, C.; Colliec-Jouault, S.; Baudoux, A.C.; Ojima, T.; Boisset, C. The marine bacteria Cobetia marina DSMZ 4741 synthesizes an unexpected K-antigen-like exopolysaccharide. Carbohydr. Polym. 2015, 124, 347–356. [Google Scholar] [CrossRef]
- Tang, X.; Xu, K.; Han, X.; Mo, Z.; Mao, Y. Complete genome of Cobetia marina JCM 21022T and phylogenomic analysis of the family Halomonadaceae. J. Oceanol. Limnol. 2018, 36, 528–536. [Google Scholar] [CrossRef]
- Azemar, F.; Faÿ, F.; Réhel, K.; Linossier, I. Ecofriendly silicon-poly(lactic acid) hybrid antifouling coatings. Prog. Org. Coat. 2020, 148, 105841. [Google Scholar] [CrossRef]
- Chiang, H.Y.; Pan, J.; Ma, C.; Qian, P.-Y. Combining a bio-based polymer and a natural antifoulant into an eco-friendly antifouling coating. Biofouling 2020, 36, 200–209. [Google Scholar] [CrossRef]
- Darya, M.; Abdolrasouli, M.H.; Yousefzadi, M.; Sajjadi, M.M.; Sourinejad, I.; Zarei, M. Antifouling coating based on biopolymers (PCL/PLA) and bioactive extract from the sea cucumber Stichopus herrmanni. AMB Express 2022, 12, 24. [Google Scholar] [CrossRef]
- Montalvão, G.; Moshrefi-Torbati, M.; Hamilton, A.; Machado, R.; João, A. Behaviour of 3D printed PLA and PLA-PHA in marine environments. Earth Environ. Sci. 2020, 424, 012013. [Google Scholar] [CrossRef]
- Wang, J.; Wu, Y.; Cao, Y.; Li, G.; Liao, Y. Influence of surface roughness on contact angle hysteresis and spreading work. Colloid Polym. Sci. 2020, 298, 1107–1112. [Google Scholar] [CrossRef]
- Aufrecht, J.A.; Fowlkes, J.D.; Bible, A.N.; Morrell-Falvey, J.; Doktycz, M.J.; Retterer, S.T. Pore-scale hydrodynamics influence the spatial evolution of bacterial biofilms in a microfluidic porous network. PLoS ONE 2019, 14, e0218316. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.; Adeleye, A.S.; Page, H.M.; Kui, L.; Lenihan, H.S.; Keller, A.A. Nano and traditional copper and zinc antifouling coatings: Metal release and impact on marine sessile invertebrate communities. J. Nanopart. Res. 2020, 22, 1–15. [Google Scholar] [CrossRef]
- Silva, E.R.; Ferreira, O.; Ramalho, P.A.; Azevedo, N.F.; Bayón, R.; Igartua, A.; Bordado, J.C.; Calhorda, M.J. Eco-friendly non-biocide-release coatings for marine biofouling prevention. Sci. Total Environ. 2019, 650, 2499–2511. [Google Scholar] [CrossRef] [PubMed]
- Blain, S.; Guillou, J.; Tréguer, P.; Woerther, P.; Delauney, L.; Follenfant, E.; Gontier, O.; Hamon, M.; Leilde, B.; Masson, A.; et al. High frequency monitoring of the coastal marine environment using the MAREL buoy. J. Environ. Monit. 2004, 6, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Bakker, D.; Plaats, A.; Verkerke, G.; Busscher, H.; van der Mei, H. Comparison of Velocity Profiles for Different Flow Chamber Designs Used in Studies of Microbial Adhesion to Surfaces. Appl. Environ. Microbiol. 2003, 69, 6280–6287. [Google Scholar] [CrossRef]
- Silva, E.R.; Tulcidas, A.V.; Ferreira, O.; Bayón, R.; Igartua, A.; Mendoza, G.; Mergulhão, F.J.M.; Faria, S.I.; Gomes, L.C.; Carvalho, S.; et al. Assessment of the environmental compatibility and antifouling performance of an innovative biocidal and foul-release multifunctional marine coating. Environ. Res. 2021, 198, 111219. [Google Scholar] [CrossRef]
- Vázquez, J.A.; Noriega, D.; Ramos, P.; Valcarcel, J.; Novoa-Carballal, R.; Pastrana, L.; Reis, R.L.; Pérez-Martín, R.I. Optimization of high purity chitin and chitosan production from Illex argentinus pens by a combination of enzymatic and chemical processes. Carbohydr. Polym. 2017, 174, 262–272. [Google Scholar] [CrossRef]
- Vázquez, J.A.; Ramos, P.; Valcarcel, J.; Antelo, L.T.; Novoa-Carballal, R.; Reis, R.L.; Pérez-Martín, R.I. An integral and sustainable valorisation strategy of squid pen by-products. J. Clean. Prod. 2018, 201, 207–218. [Google Scholar] [CrossRef]
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial Actions and Applications of Chitosan. Polymers 2021, 13, 904. [Google Scholar] [CrossRef]
- Spengler, C.; Nolle, F.; Mischo, J.; Faidt, T.; Grandthyll, S.; Thewes, N.; Koch, M.; Müller, F.; Bischoff, M.; Klatt, M.A.; et al. Strength of bacterial adhesion on nanostructured surfaces quantified by substrate morphometry. Nanoscale 2019, 11, 19713–19722. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Q.; Yan, T.; Jiang, Z.; Zhang, X.; Zuo, Y.Y. Quantitatively predicting bacterial adhesion using surface free energy determined with a spectrophotometric method. Environ. Sci. Technol. 2015, 49, 6164–6171. [Google Scholar] [CrossRef] [PubMed]
- Abiraman, T.; Ramanathan, E.; Kavitha, G.; Rengasamy, R.; Balasubramanian, S. Synthesis of chitosan capped copper oxide nanoleaves using high intensity (30kHz) ultrasound sonication and their application in antifouling coatings. Ultrason. Sonochem. 2017, 34, 781–791. [Google Scholar] [CrossRef]
- Mahto, L.; Pal, D. Preparation and characterization of amphiphilic polymer coating for marine biofouling control. Mater. Sci. Eng. 2020, 798, 012005. [Google Scholar] [CrossRef]
- Bulwan, M.; Wójcik, K.; Zapotoczny, S.; Nowakowska, M. Chitosan-based ultrathin films as antifouling, anticoagulant and antibacterial protective coatings. J. Biomater. Sci. Polym. Ed. 2012, 23, 1963–1980. [Google Scholar] [CrossRef]
- Kara, F.; Aksoy, E.A.; Yuksekdag, Z.; Hasirci, N.; Aksoy, S. Synthesis and surface modification of polyurethanes with chitosan for antibacterial properties. Carbohydr. Polym. 2014, 112, 39–47. [Google Scholar] [CrossRef] [PubMed]
- De-la-Pinta, I.; Cobos, M.; Ibarretxe, J.; Montoya, E.; Eraso, E.; Guraya, T.; Quindós, G. Effect of biomaterials hydrophobicity and roughness on biofilm development. J. Mater. Sci. Mater. Med. 2019, 30, 77. [Google Scholar] [CrossRef] [PubMed]
- Ururahy, M.S.; Curylofo-Zotti, F.A.; Galo, R.; Nogueira, L.F.; Ramos, A.P.; Corona, S.A. Wettability and surface morphology of eroded dentin treated with chitosan. Arch. Oral Biol. 2017, 75, 68–73. [Google Scholar] [CrossRef]
- Stoleru, E.; Dumitriu, R.P.; Munteanu, B.S.; Zaharescu, T.; Tănase, E.E.; Mitelut, A.; Ailiesei, G.-L.; Vasile, C. Novel procedure to enhance PLA surface properties by chitosan irreversible immobilization. Appl. Surf. Sci. 2016, 367, 407–417. [Google Scholar] [CrossRef]
- Thomas, M.S.; Pillai, P.K.S.; Faria, M.; Cordeiro, N.; Kailas, L.; Kalarikkal, N.; Thomas, S.; Pothen, L.A. Polylactic acid/nano chitosan composite fibers and their morphological, physical characterization for the removal of cadmium(II) from water. J. Appl. Polym. Sci. 2020, 137, 48993. [Google Scholar] [CrossRef]
- Bonilla, J.; Fortunati, E.; Vargas, M.; Chiralt, A.; Kenny, J. Effects of chitosan on the physicochemical and antimicrobial properties of PLA films. J. Food Eng. 2013, 119, 236–243. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J.; Burgess, J.G. Chitosan-zinc oxide nanocomposite coatings for the prevention of marine biofouling. Chemosphere 2017, 168, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Sanpo, N.; Tharajak, J. Antimicrobial Properties of Transition Metals Substituted Chitosan against Cobetia marina. Appl. Mech. Mater. 2016, 848, 123–126. [Google Scholar] [CrossRef]
- Romeu, M.J.; Alves, P.; Morais, J.; Miranda, J.M.; de Jong, E.D.; Sjollema, J.; Ramos, V.; Vasconcelos, V.; Mergulhão, F.J.M. Biofilm formation behaviour of marine filamentous cyanobacterial strains in controlled hydrodynamic conditions. Environ. Microbiol. 2019, 21, 4411–4424. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, S.; Miura, Y.; Kaneko, H.; Nishimura, S.I.; Nishi, N.; Iwase, M.; Tokura, S. Molecular Weight Dependency of Antimicrobial Activity by Chitosan Oligomers. In Food Hydrocolloids: Structures, Properties, and Functions; Nishinari, K., Doi, E., Eds.; Springer: Boston, MA, USA, 1993; pp. 71–76. [Google Scholar]
- Jumaa, M.; Furkert, F.H.; Müller, B.W. A new lipid emulsion formulation with high antimicrobial efficacy using chitosan. Eur. J. Pharm. Biopharm. 2002, 53, 115–123. [Google Scholar] [CrossRef] [PubMed]
- No, H.K.; Young Park, N.; Ho Lee, S.; Meyers, S.P. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int. J. Food Microbiol. 2002, 74, 65–72. [Google Scholar] [CrossRef]
- Jena, G.; Anandkumar, B.; Vanithakumari, S.C.; George, R.P.; Philip, J.; Amarendra, G. Graphene oxide-chitosan-silver composite coating on Cu-Ni alloy with enhanced anticorrosive and antibacterial properties suitable for marine applications. Prog. Org. Coat. 2020, 139, 105444. [Google Scholar] [CrossRef]
- Elshaarawy, R.; Mustafa, F.H.A.; Herbst, A.; Farag, A. Surface functionalization of chitosan isolated from shrimp shells, using salicylaldehyde ionic liquids in exploration for novel economic and ecofriendly antibiofoulants. RSC Adv. 2016, 6, 20901–20915. [Google Scholar] [CrossRef]
- Al-Naamani, L.S. Antifouling properties of chitosan coatings on plastic substrates. J. Agric. Mar. Sci. 2019, 23, 92–98. [Google Scholar] [CrossRef]
- Dobretsov, S.; Abed, R.M.M.; Muthukrishnan, T.; Sathe, P.; Al-Naamani, L.; Queste, B.Y.; Piontkovski, S. Living on the edge: Biofilms developing in oscillating environmental conditions. Biofouling 2018, 34, 1064–1077. [Google Scholar] [CrossRef]
- Al-Belushi, M.A.; Myint, M.T.Z.; Kyaw, H.H.; Al-Naamani, L.; Al-Mamari, R.; Al-Abri, M.; Dobretsov, S. ZnO nanorod-chitosan composite coatings with enhanced antifouling properties. Int. J. Biol. Macromol. 2020, 162, 1743–1751. [Google Scholar] [CrossRef]
- Simůnek, J.; Brandysová, V.; Koppová, I.; Simůnek, J., Jr. The antimicrobial action of chitosan, low molar mass chitosan, and chitooligosaccharides on human colonic bacteria. Folia Microbiol. (Praha) 2012, 57, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Tayel, A.A.; Moussa, S.; Opwis, K.; Knittel, D.; Schollmeyer, E.; Nickisch-Hartfiel, A. Inhibition of microbial pathogens by fungal chitosan. Int. J. Biol. Macromol. 2010, 47, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Benhabiles, M.S.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.F.A.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 2012, 29, 48–56. [Google Scholar] [CrossRef]
- Zheng, L.-Y.; Zhu, J.-F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr. Polym. 2003, 54, 527–530. [Google Scholar] [CrossRef]
- Hou, J.; Wang, C.; Rozenbaum, R.T.; Gusnaniar, N.; de Jong, E.D.; Woudstra, W.; Geertsema-Doornbusch, G.I.; Atema-Smit, J.; Sjollema, J.; Ren, Y.; et al. Bacterial Density and Biofilm Structure Determined by Optical Coherence Tomography. Sci. Rep. 2019, 9, 9794. [Google Scholar] [CrossRef]
- Drescher, K.; Shen, Y.; Bassler, B.L.; Stone, H.A. Biofilm streamers cause catastrophic disruption of flow with consequences for environmental and medical systems. Proc. Natl. Acad. Sci. USA 2013, 110, 4345–4350. [Google Scholar] [CrossRef] [PubMed]
- El-saied, H.A.-a.; Ibrahim, A.M. Effective Fabrication and Characterization of Eco-friendly Nano Chitosan Capped Zinc Oxide Nanoparticles for Effective Marine Fouling Inhibition. J. Environ. Chem. Eng. 2020, 8, 103949. [Google Scholar] [CrossRef]
- Elshaarawy, R.F.M.; Mustafa, F.H.A.; van Geelen, L.; Abou-Taleb, A.E.A.; Tadros, H.R.Z.; Kalscheuer, R.; Janiak, C. Mining marine shell wastes for polyelectrolyte chitosan anti-biofoulants: Fabrication of high-performance economic and ecofriendly anti-biofouling coatings. Carbohydr. Polym. 2017, 172, 352–364. [Google Scholar] [CrossRef]
- Aybar, M.; Perez-Calleja, P.; Li, M.; Pavissich, J.P.; Nerenberg, R. Predation creates unique void layer in membrane-aerated biofilms. Water Res. 2019, 149, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.E.; Caiazza, N.C.; O’Toole, G.A. Rhamnolipid surfactant production affects biofilm architecture in Pseudomonas aeruginosa PAO1. J. Bacteriol. 2003, 185, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Chang, I.; Gilbert, E.S.; Eliashberg, N.; Keasling, J.D. A three-dimensional, stochastic simulation of biofilm growth and transport-related factors that affect structure. Microbiology 2003, 149, 2859–2871. [Google Scholar] [CrossRef] [PubMed]
- Allan, G.G.; Peyron, M. Molecular weight manipulation of chitosan II: Prediction and control of extent of depolymerization by nitrous acid. Carbohydr. Res. 1995, 277, 273–282. [Google Scholar] [CrossRef]
- Maréchal, J.-P.; Culioli, G.; Hellio, C.; Thomas-Guyon, H.; Callow, M.E.; Clare, A.S.; Ortalo-Magné, A. Seasonal variation in antifouling activity of crude extracts of the brown alga Bifurcaria bifurcata (Cystoseiraceae) against cyprids of Balanus amphitrite and the marine bacteria Cobetia marina and Pseudoalteromonas haloplanktis. J. Exp. Mar. Biol. Ecol. 2004, 313, 47–62. [Google Scholar] [CrossRef]
- Holmström, C.; James, S.; Neilan, B.A.; White, D.C.; Kjelleberg, S. Pseudoalteromonas tunicata sp. nov., a bacterium that produces antifouling agents. Int. J. Syst. Bacteriol. 1998, 48 Pt 4, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Combarros-Fuertes, P.; Estevinho, L.M.; Teixeira-Santos, R.; Rodrigues, A.G.; Pina-Vaz, C.; Fresno, J.M.; Tornadijo, M.E. Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli. Microorganisms 2019, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Kramer, B.; Muranyi, P. Effect of pulsed light on structural and physiological properties of Listeria innocua and Escherichia coli. J. Appl. Microbiol. 2014, 116, 596–611. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, A.; Bourbon, A.; Quintas, M.; Coimbra, M.; Vicente, A. Κ-carrageenan/chitosan nanolayered coating for controlled release of a model bioactive compound. Innov. Food Sci. Emerg. Technol. 2012, 16, 227–232. [Google Scholar] [CrossRef]
- Bateni, A.; Susnar, S.S.; Amirfazli, A.; Neumann, A.W. A high-accuracy polynomial fitting approach to determine contact angles. Colloids Surf. A: Physicochem. Eng. Asp. 2003, 219, 215–231. [Google Scholar] [CrossRef]
- Whitehead, K.; Kelly, P.; Li, H.; Verran, J. Surface topography and physicochemistry of silver containing titanium nitride nanocomposite coatings. J. Vac. Sci. Technol. B 2010, 28, 180–187. [Google Scholar] [CrossRef]
- Skovager, A.; Whitehead, K.; Siegumfeldt, H.; Ingmer, H.; Verran, J.; Arneborg, N. Influence of flow direction and flow rate on the initial adhesion of seven Listeria monocytogenes strains to fine polished stainless steel. Int. J. Food Microbiol. 2012, 157, 174–181. [Google Scholar] [CrossRef]
- Telegdi, J.; Trif, L.; Románszki, L. Smart Anti-Biofouling Composite Coatings for Naval Applications; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, M.; Gomes, L.C.; Teixeira-Santos, R.; Romeu, M.J.; Valcarcel, J.; Vázquez, J.A.; Cerqueira, M.A.; Pastrana, L.; Bourbon, A.I.; de Jong, E.D.; et al. Assessment of the Antibiofilm Performance of Chitosan-Based Surfaces in Marine Environments. Int. J. Mol. Sci. 2022, 23, 14647. https://doi.org/10.3390/ijms232314647
Lima M, Gomes LC, Teixeira-Santos R, Romeu MJ, Valcarcel J, Vázquez JA, Cerqueira MA, Pastrana L, Bourbon AI, de Jong ED, et al. Assessment of the Antibiofilm Performance of Chitosan-Based Surfaces in Marine Environments. International Journal of Molecular Sciences. 2022; 23(23):14647. https://doi.org/10.3390/ijms232314647
Chicago/Turabian StyleLima, Marta, Luciana C. Gomes, Rita Teixeira-Santos, Maria J. Romeu, Jesus Valcarcel, José Antonio Vázquez, Miguel A. Cerqueira, Lorenzo Pastrana, Ana I. Bourbon, Ed D. de Jong, and et al. 2022. "Assessment of the Antibiofilm Performance of Chitosan-Based Surfaces in Marine Environments" International Journal of Molecular Sciences 23, no. 23: 14647. https://doi.org/10.3390/ijms232314647
APA StyleLima, M., Gomes, L. C., Teixeira-Santos, R., Romeu, M. J., Valcarcel, J., Vázquez, J. A., Cerqueira, M. A., Pastrana, L., Bourbon, A. I., de Jong, E. D., Sjollema, J., & Mergulhão, F. J. (2022). Assessment of the Antibiofilm Performance of Chitosan-Based Surfaces in Marine Environments. International Journal of Molecular Sciences, 23(23), 14647. https://doi.org/10.3390/ijms232314647











