Pollen Molecular Identification from a Long-Distance Migratory Insect, Spodoptera exigua, as Evidenced for Its Regional Pollination in Eastern Asia
Abstract
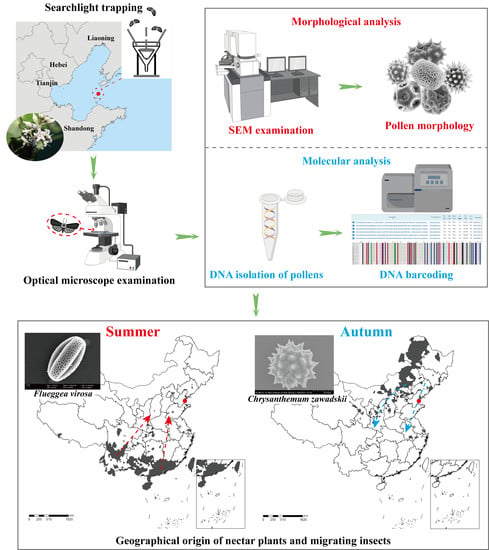
:1. Introduction
2. Results
2.1. Morphology of the Proboscis and Pollen Detection
2.2. Plant Hosts Inferred from Pollen
2.3. Sex, Annual, and Seasonal Differences in Pollen Adherence Ratio
2.4. Annual and Seasonal Shifts in Pollen Taxa
2.5. Characteristics of Pollen-Bearing Plants
2.6. Diversity of Attached Pollen across Different Noctuid Moths
3. Discussion
4. Materials and Methods
4.1. Moth Collection
4.2. Microscopic Examination of Pollen
4.3. Molecular Analyses
4.4. Pollen and Plant Host Identification
4.5. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Magallón, S.; Gómez-Acevedo, S.; Sánchez-Reyes, L.L.; Hernández-Hernández, T. A metacalibrated time-tree documents the early rise of flowering plant phylogenetic diversity. New Phytol. 2015, 207, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Labandeira, C.C.; Dilcher, D.; Ren, D. Florivory of early cretaceous flowers by functionally diverse insects: Implications for early angiosperm pollination. Proc. R. Soc. B 2021, 288, 20210320. [Google Scholar] [CrossRef] [PubMed]
- Sauquet, H.; von Balthazar, M.; Magallón, S.; Doyle, J.A.; Endress, P.K.; Bailes, E.J.; Schönenberger, J. The ancestral flower of angiosperms and its early diversification. Nat. Commun. 2017, 8, 16047. [Google Scholar] [CrossRef]
- Bernays, E.A.; Bright, K.L.; Gonzalez, N.; Angel, J. Dietary mixing in a generalist herbivore: Tests of two hypotheses. Ecology 1994, 75, 1997–2006. [Google Scholar] [CrossRef]
- Mitter, C.; Farrell, B.; Futuyma, D.J. Phylogenetic studies of insect-plant interactions: Insights into the genesis of diversity. Trends Ecol. Evol. 1991, 6, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Rader, R.; Cunningham, S.A.; Howlett, B.G.; Inouye, D.W. Non-bee insects as visitors and pollinators of crops: Biology, ecology, and management. Annu. Rev. Entomol. 2020, 65, 391–407. [Google Scholar] [CrossRef]
- Requier, F.; Pérez-Méndez, N.; Andersson, G.K.; Blareau, E.; Merle, I.; Garibaldi, L.A. Bee and non-bee pollinator importance for local food security. Trends Ecol. Evol. 2022, 38, 196–205. [Google Scholar] [CrossRef]
- Wagner, D.L. Moths. In Encyclopedia of Biodiversity; Levin, S.A., Ed.; Academic Press: San Diego, CA, USA, 2001; Volume 4, pp. 249–270. [Google Scholar]
- Kawahara, A.Y.; Plotkin, D.; Espeland, M.; Meusemann, K.; Toussaint, E.F.; Donath, A.; Breinholt, J.W. Phylogenomics reveals the evolutionary timing and pattern of butterflies and moths. Proc. Natl. Acad. Sci. USA 2019, 116, 22657–22663. [Google Scholar] [CrossRef]
- Devoto, M.; Bailey, S.; Memmott, J. The ‘night shift’: Nocturnal pollen-transport networks in a boreal pine forest. Ecol. Entomol. 2011, 36, 25–35. [Google Scholar] [CrossRef]
- LeCroy, K.A.; Shew, H.W.; van Zandt, P.A. Pollen presence on nocturnal moths in the Ketona Dolomite glades of Bibb County, Alabama. S. Lepid. News 2013, 35, 136–142. [Google Scholar]
- Macgregor, C.J.; Pocock, M.J.; Fox, R.; Evans, D.M. Pollination by nocturnal Lepidoptera, and the effects of light pollution: A review. Ecol. Entomol. 2015, 40, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Fox, R. The decline of moths in Great Britain: A review of possible causes. Insect Conserv. Divers. 2013, 6, 5–19. [Google Scholar] [CrossRef]
- Bryant, V.M.; Pendleton, M.; Murry, R.E.; Lingren, P.D.; Raulston, J.R. Techniques for studying pollen adhering to nectar-feeding corn earworm (Lepidoptera: Noctuidae) moths using scanning electron microscopy. J. Econ. Entomol. 1991, 84, 237–240. [Google Scholar] [CrossRef]
- Lingren, P.D.; Bryant, V.M.; Raulston, J.R.; Pendleton, M.; Westbrook, J.; Jones, G.D. Adult feeding host range and migratory activities of com earworm, cabbage looper, and celery looper (Lepidoptera: Noctuidae) moths as evidenced by attached pollen. J. Econ. Entomol. 1993, 86, 1429–1439. [Google Scholar] [CrossRef]
- Liu, Y.; Fu, X.; Mao, L.; Xing, Z.; Wu, K. Host plants identification for adult Agrotis ipsilon, a long-distance migratory insect. Int. J. Mol. Sci. 2016, 17, 851. [Google Scholar] [CrossRef]
- Chang, H.; Guo, J.; Fu, X.; Liu, Y.; Wyckhuys, K.A.; Hou, Y.; Wu, K. Molecular-assisted pollen grain analysis reveals spatiotemporal origin of long-distance migrants of a noctuid moth. Int. J. Mol. Sci. 2018, 19, 567. [Google Scholar] [CrossRef]
- Spence, K.O.; Rosenheim, J.A. Isotopic enrichment in herbivorous insects: A comparative field-based study of variation. Oecologia 2005, 146, 89–97. [Google Scholar] [CrossRef]
- Wanner, H.; Gu, H.; Günther, D.; Hein, S.; Dorn, S. Tracing spatial distribution of parasitism in fields with flowering plant strips using stable isotope marking. Biol. Control 2006, 39, 240–247. [Google Scholar] [CrossRef]
- Traugott, M.; Kamenova, S.; Ruess, L.; Seeber, J.; Plantegenest, M. Empirically characterising trophic networks: What emerging DNA-based methods, stable isotope and fatty acid analyses can offer. Adv. Ecol. Res. 2013, 49, 177–224. [Google Scholar]
- Jia, H.; Liu, Y.; Li, X.; Li, H.; Pan, Y.; Hu, C.; Zhou, X.; Wyckhuys, K.A.; Wu, K. Windborne migration amplifies insect-mediated pollination services. Elife 2022, 11, e76230. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhao, S.Y.; Wang, M.L.; Yu, W.H.; Wyckhuys, K.A.G.; Wu, K.M. Floral visitation can enhance fitness of Helicoverpa armigera (Lepidoptera: Noctuidae) long-distance migrants. J. Econ. Entomol. 2019, 112, 2655–2662. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fu, X.; Mao, L.; Xing, Z.; Wu, K. Identification of host plant use of adults of a long-distance migratory insect, Mythimna separata. PLoS ONE 2017, 12, e0184116. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.; Sayyed, A.H.; Ahmad, I. Effect of host plants on life-history traits of Spodoptera exigua (Lepidoptera: Noctuidae). J. Pest Sci. 2010, 83, 165–172. [Google Scholar] [CrossRef]
- Zheng, X.L.; Cong, X.P.; Wang, X.P.; Lei, C.L. A review of geographic distribution, overwintering and migration in Spodoptera exigua (Lepidoptera: Noctuidae). J. Entomol. Res. Soc. 2011, 13, 39–48. (In Chinese) [Google Scholar]
- Wang, X.; Yang, X.; Zhou, L.; Wyckhuys, K.A.; Jiang, S.; Van Liem, N.; Vi, L.X.; Ali, A.; Wu, K. Population genetics unveils large-scale migration dynamics and population turnover of Spodoptera exigua. Pest Manag. Sci. 2022, 78, 612–625. [Google Scholar] [CrossRef]
- Fu, X.; Feng, H.; Liu, Z.; Wu, K. Transregional migration of the beet armyworm, Spodoptera exigua (Lepidoptera: Noctuidae), in North-East Asia. PLoS ONE 2017, 12, e0183582. [Google Scholar] [CrossRef]
- Jones, G.D. Pollen analyses for pollination research, acetolysis. J. Pollinat. Ecol. 2014, 13, 203–217. [Google Scholar] [CrossRef]
- Jones, G.D.; Jones, S.D. The uses of pollen and its implication for entomology. Neotrop. Entomol. 2001, 30, 341–350. [Google Scholar] [CrossRef]
- Guo, J.; Fu, X.; Zhao, S.; Shen, X.; Wyckhuys, K.A.; Wu, K. Long-term shifts in abundance of (migratory) crop-feeding and beneficial insect species in northeastern Asia. J. Pest Sci. 2020, 93, 583–594. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, H.; Liu, D.; Khashaveh, A.; Li, Q.; Wyckhuys, K.A.; Wu, K. Long-term insect censuses capture progressive loss of ecosystem functioning in East Asia. Sci. Adv. 2023, 9, eade9341. [Google Scholar] [CrossRef]
- Guo, J.; Liu, Y.; Jia, H.; Chang, H.; Wu, K. Visiting Plants of Mamestra brassicae (Lepidoptera: Noctuidae) Inferred from identification of adhering pollen grains. Environ. Entomol. 2022, 51, 505–512. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liu, Y.; Guo, J.; Chang, H.; Wu, K. Host plants and pollination regions for the long-distance migratory noctuid moth, Hadula trifolii Hufnagel in China. Ecol. Evol. 2022, 12, e8819. [Google Scholar] [CrossRef]
- Tudor, O.; Dennis, R.L.H.; Greatorex-Davies, J.N.; Sparks, T.H. Flower preferences of woodland butterflies in the UK: Nectaring specialists are species of conservation concern. Biol. Conserv. 2004, 119, 397–403. [Google Scholar] [CrossRef]
- Krenn, H.W. Feeding mechanisms of adult Lepidoptera: Structure, function, and evolution of the mouthparts. Annu. Rev. Entomol. 2010, 55, 307–327. [Google Scholar] [CrossRef] [PubMed]
- Forup, M.L.; Memmott, J. The relationship between the availability of nest sites and pollen limitation in the hoverfly Eristalis tenax. Ecol. Entomol. 2005, 30, 492–496. [Google Scholar]
- Gibson, R.H.; Nelson, I.L.; Hopkins, G.W.; Hamlett, B.J. Pollen transfer by the thrips Frankliniella intonsa (Thysanoptera: Thripidae) promotes self-fertilisation in plants of strawberry Fragaria x ananassa (Rosaceae). Ann. Appl. Biol. 2006, 148, 63–68. [Google Scholar]
- Turnock, A.C.; James, J.W.; Dickson, J.G. A simple and versatile method for studying the surface morphology of pollen grains using scanning electron microscopy. New Phytol. 1978, 81, 631–637. [Google Scholar]
- Salmaki, Y.; Jamzad, Z.; Zarre, S.; Bräuchler, C. Pollen morphology of Stachys (Lamiaceae) in Iran and its systematic implication. Flora 2008, 203, 627–639. [Google Scholar] [CrossRef]
- Khansari, E.; Zarre, S.; Alizadeh, K.; Attar, F.; Aghabeigi, F.; Salmaki, Y. Pollen morphology of Campanula (Campanulaceae) and allied genera in Iran with special focus on its systematic implication. Flora 2012, 207, 203–211. [Google Scholar] [CrossRef]
- García-Robledo, C.; Erickson, D.L.; Staines, C.L.; Erwin, T.L.; Kress, W.J. Tropical Plant-Herbivore Networks: Reconstructing Species Interactions Using DNA Barcodes. PLoS ONE 2013, 8, e52967. [Google Scholar] [CrossRef]
- Ghaffar, A.; Attique, M.R.; Naveed, M.R.; Jan, M.T. Effect of different hosts on the development and survival of Spodoptera exigua (Hubner) (Noctuidae: Lepidoptera). Pak. J. Zool. 2002, 34, 229–231. [Google Scholar]
- Scheirs, J.; Bruyn, L.D. Integrating optimal foraging and optimal oviposition theory in plant-insect research. Oikos 2002, 96, 187–191. [Google Scholar] [CrossRef]
- Buxton, V.L.; Enos, J.K.; Sperry, J.H.; Ward, M.P. A review of conspecific attraction for habitat selection across taxa. Ecol. Evol. 2020, 10, 12690–12699. [Google Scholar] [CrossRef] [PubMed]
- Schäpers, A.; Brockmoeller, T.; von Wehrden, H. Plant density, synchrony, and quality affect flower visitation and interplant movement of pollinators in experimental plant populations. Ecol. Evol. 2019, 9, 2415–2426. [Google Scholar]
- Guo, M.; Du, L.; Chen, Q.; Feng, Y.; Zhang, J.; Zhang, X.; Liu, Y. Odorant receptors for detecting flowering plant cues are functionally conserved across moths and butterflies. Mol. Biol. Evol. 2021, 38, 1413–1427. [Google Scholar] [CrossRef] [PubMed]
- Hagler, J.R.; Jackson, C.G. Methods for marking insects: Current techniques and future prospects. Annu. Rev. Entomol. 2001, 46, 511–543. [Google Scholar] [CrossRef]
- Hendrix, W.H., III; Mueller, T.F.; Phillips, J.R.; Davis, O.K. Pollen as an indicator of long-distance movement of Heliothis zea (Lepidoptera: Noctuidae). Environ. Entomol. 1987, 16, 1148–1151. [Google Scholar] [CrossRef]
- Hendrix, W.H.; Showers, W.B. Tracing black cutworm and armyworm (Lepidoptera: Noctuidae) northward migration using pithecellobium and calliandra pollen. Environ. Entomol. 1992, 21, 1092–1096. [Google Scholar] [CrossRef]
- Suchan, T.; Talavera, G.; Sáez, L.; Ronikier, M.; Vila, R. Pollen metabarcoding as a tool for tracking long-distance insect migrations. Mol. Ecol. Resour. 2019, 19, 149–162. [Google Scholar] [CrossRef]
- Hu, G.; Lim, K.S.; Horvitz, N.; Clark, S.J.; Reynolds, D.R.; Sapir, N.; Chapman, J.W. Mass seasonal bioflows of high-flying insect migrants. Science 2016, 354, 1584–1587. [Google Scholar] [CrossRef]
- Hallworth, M.T.; Marra, P.P.; McFarland, K.P.; Zahendra, S.; Studds, C.E. Tracking dragons: Stable isotopes reveal the annual cycle of a long-distance migratory insect. Biol. Lett. 2018, 14, 20180741. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Fu, X.; Wu, K. Seasonal migration of white-backed planthopper Sogatella furcifera Horváth (Hemiptera: Delphacidae) over the Bohai Sea in northern China. J. Asia-Pac. Entomol. 2017, 20, 1358–1363. [Google Scholar] [CrossRef]
- Kim, K.S.; Sappington, T.W. Population genetics strategies to characterize long-distance dispersal of insects. J. Asia-Pac. Entomol. 2013, 16, 87–97. [Google Scholar] [CrossRef]
- Feng, H.Q.; Wu, K.M.; Cheng, D.F.; Guo, Y.Y. Radar observations of the autumn migration of the beet armyworm Spodoptera exigua (Lepidoptera: Noctuidae) and other moths in northern China. Bull. Entomol. Res. 2003, 93, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.Q.; Wu, K.M. Vertical-Pointing Searchlight Trap. China Patent CN201020229775.3, 2010. [Google Scholar]
- Chen, P.H.; Pan, Y.B.; Chen, R.K. High-throughput procedure for single pollen grain collection and polymerase chain reaction in plants. J. Integr. Plant Biol. 2008, 50, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Taberlet, P.; Coissac, E.; Pompanon, F.; Gielly, L.; Miquel, C.; Valentini, A.; Vermat, T.; Corthier, G.; Brochmann, C.; Willerslev, E. Power and limitations of the chloroplast trnL (UAA) intron for plant DNA barcoding. Nucleic Acids Res. 2007, 35, e14. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Xu, C.; Lei, L.; Li, C.; Zhang, Y.; Zhou, S. Barcoding the kingdom Plantae: New PCR primers for ITS regions of plants with improved universality and specificity. Mol. Ecol. Resour. 2016, 16, 138–149. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. 1990, 18, 315–322. [Google Scholar]
- Hawkins, J.; De Vere, N.; Griffith, A.; Ford, C.R.; Allainguillaume, J.; Hegarty, M.J.; Adams-Groom, B. Using DNA Metabarcoding to Identify the Floral Composition of Honey: A New Tool for Investigating Honey Bee Foraging Preferences. PLoS ONE 2015, 10, e0134735. [Google Scholar] [CrossRef]
- Ma, D.W.; Zhang, C.H.; Gao, S.Z.; Ma, N.; Liu, H.H.; Zhang, Y.P.; Sun, L. Pollen Flora of China Vegetables by SEM; China Agriculture Press: Beijing, China, 1999. [Google Scholar]
- Li, T.Q.; Cao, H.J.; Kang, M.S.; Zhang, Z.X.; Zhao, N.; Zhang, H. Pollen Flora of China Woody Plants by SEM; Science Press: Beijing, China, 2010; 1233p. [Google Scholar]
- SPSS Incorporation. SPSS 13.0 for the Windows, version 13.0; SPSS Inc.: Chicago, IL, USA, 2006.






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, H.; Wang, T.; Li, X.; Zhao, S.; Guo, J.; Liu, D.; Liu, Y.; Wu, K. Pollen Molecular Identification from a Long-Distance Migratory Insect, Spodoptera exigua, as Evidenced for Its Regional Pollination in Eastern Asia. Int. J. Mol. Sci. 2023, 24, 7588. https://doi.org/10.3390/ijms24087588
Jia H, Wang T, Li X, Zhao S, Guo J, Liu D, Liu Y, Wu K. Pollen Molecular Identification from a Long-Distance Migratory Insect, Spodoptera exigua, as Evidenced for Its Regional Pollination in Eastern Asia. International Journal of Molecular Sciences. 2023; 24(8):7588. https://doi.org/10.3390/ijms24087588
Chicago/Turabian StyleJia, Huiru, Tengli Wang, Xiaokang Li, Shengyuan Zhao, Jianglong Guo, Dazhong Liu, Yongqiang Liu, and Kongming Wu. 2023. "Pollen Molecular Identification from a Long-Distance Migratory Insect, Spodoptera exigua, as Evidenced for Its Regional Pollination in Eastern Asia" International Journal of Molecular Sciences 24, no. 8: 7588. https://doi.org/10.3390/ijms24087588