Reduced Levels of Selenium and Thioredoxin Reductase in the Thoracic Aorta Could Contribute to Aneurysm Formation in Patients with Marfan Syndrome
Abstract
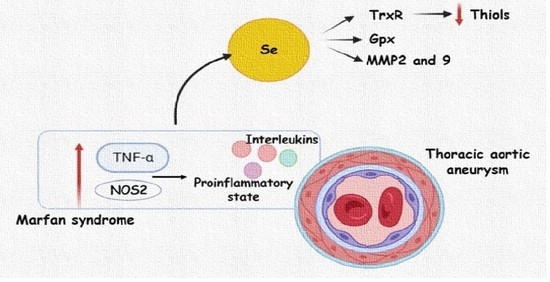
:1. Introduction
2. Results
2.1. Demographic Characteristics of the MFS Patients
2.2. Inflammatory Interleukins, Se, and Carbonylation Levels
2.3. Enzymatic Activities and Expressions
2.4. Immunohistochemistry
3. Discussion
4. Materials and Methods
4.1. Ethical Considerations
4.2. MFS Patients and Control Subjects
4.3. Histology and Immunohistochemistry
4.4. Thoracic Aortic Aneurysm Tissue Homogenization
4.5. TrxR and GPx Activities
4.6. Oxidative Stress Markers
4.7. Determinations of IL-1β, TNF, IL-6 and TGF-β1
4.8. Determinations of NOX1 and NOS2 by Western Blotting
4.9. Statistical Analysis
5. Conclusions
Study Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, K.L.; Yang, J.H.; Song, S.H.; Kim, J.Y.; Jang, S.Y.; Kim, J.M.; Kim, J.A.; Sung, K.I.; Kim, Y.W.; Suh, Y.L.; et al. Positive correlation between the dysregulation of transforming growth factor-β1 and aneurysmal pathological changes in patients with marfan syndrome. Circ. J. 2013, 77, 952–958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gambarin, F.I.; Favalli, V.; Serio, A.; Regazzi, M.; Pasotti, M.; Klersy, C.; Dore, R.; Savina, M.; Viganò, M.; Odero, A.; et al. Rationale and design of a trial evaluating the effects of losartan vs. nebivolol vs. the association of both on the progression of aortic root dilation in Marfan syndrome with FBN1 gene mutations. J. Cardiovasc. Med. 2009, 10, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Loeys, B.L.; Dietz, H.C.; Braverman, A.C.; Callewaert, B.L.; De Backer, J.; Devereux, R.B.; Hilhorst-Hofstee, Y.; Jondeau, G.; Faivre, L.; Milewicz, D.; et al. The revised Ghent nosology for the Marfan syndrome. J. Med. Genet. 2010, 47, 476–485. [Google Scholar] [CrossRef] [Green Version]
- Detaint, D.; Faivre, L.; Collod-Beroud, G.; Child, A.H.; Loeys, B.L.; Binquet, C.; Gautier, E.; Arbustini, E.; Mayer, K.; Mine Arslan-Kirchner, M.; et al. Cardiovascular manifestations in men and women carrying a FBN1 mutation. Eur. Heart J. 2010, 31, 2223–2229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neptune, E.R.; Frischmeyer, P.A.; Arking, D.E.; Myers, L.; Bunton, T.E.; Gayraud, B.; Ramirez, F.; Sakai, L.Y.; Dietz, H.C. Dysregulation of TGF-beta activation contributes to pathogenesis in Marfan syndrome. Nat. Genet. 2003, 33, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Ponticos, M.; Smith, B.D. Extracellular matrix synthesis in vascular disease: Hypertension, and atherosclerosis. J. Biomed. Res. 2014, 28, 25–39. [Google Scholar]
- Soto, M.E.; Soria-Castro, E.; Guarner-Lans, V.; Martínez-Guzmán, A.; Morales-Marín, C.; Martínez-Zavala, K.S.; Pérez-Torres, I. Preliminary analysis of the association of TRPV1 to the formation of Marfan syndrome aneurysms. Histol. Histopathol. 2019, 34, 1329–1343. [Google Scholar]
- Deleeuw, V.; De Clercq, A.; De Backer, J.; Sips, P. An overview of investigational and experimental drug treatment strategies for marfan syndrome. J. Exp. Pharmacol. 2021, 13, 755–779. [Google Scholar] [CrossRef]
- Zúñiga-Muñoz, A.M.; Pérez-Torres, I.; Guarner-Lans, V.; Núñez-Garrido, E.; Velázquez-Espejel, R.; Huesca-Gómez, C.; Gamboa-Ávila, R.; Soto, M.E. Glutathione system participation in thoracic aneurysms from patients with Marfan syndrome. Vasa 2017, 46, 177–186. [Google Scholar] [CrossRef]
- Soto, M.E.; Manzano-Pech, L.G.; Guarner-Lans, V.; Díaz-Galindo, J.A.; Vásquez, X.; Castrejón-Tellez, V.; Gamboa, R.; Huesca, C.; Fuentevilla-Alvárez, G.; Pérez-Torres, I. Oxidant/antioxidant profile in the thoracic aneurysm of patients with the Loeys-Dietz syndrome. Oxid. Med. Cell. Longev. 2020, 2020, 5392454. [Google Scholar] [CrossRef]
- Risbano, M.G.; Gladwin, M.T. Therapeutics targeting of dysregulated redox equilibrium and endothelial dysfunction. Handb. Exp. Pharmacol. 2013, 218, 315–349. [Google Scholar]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Schmidt, R.L.; Simonović, M. Synthesis and decoding of selenocysteine and human health. Croat. Med. J. 2012, 53, 535–550. [Google Scholar] [CrossRef] [PubMed]
- Zwolak, I.; Zaporowska, H. Selenium interactions and toxicity: A review. Selenium interactions and toxicity. Cell. Biol. Toxicol. 2012, 28, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Shimada, B.K.; Alfulaij, N.; Seale, L.A. The Impact of selenium deficiency on cardiovascular function. Int. J. Mol. Sci. 2021, 22, 10713. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, L. Selenoprotein P—Selenium transport protein, enzyme and biomarker of selenium status. Free Radic. Biol. Med. 2022, 191, 150–163. [Google Scholar] [CrossRef]
- Burk, R.F.; Hill, K.E. Selenoprotein P—Expression, functions, and roles in mammals. Biochim. Biophys. Acta 2009, 1790, 1441–1447. [Google Scholar] [CrossRef] [Green Version]
- Kieliszek, M. Selenium fascinating microelement, properties and sources in food. Molecules 2019, 24, E1298. [Google Scholar] [CrossRef] [Green Version]
- Socha, K.; Borawska, M.H.; Gacko, M.; Guzowski, A. Diet and the content of selenium and lead in patients with abdominal aortic aneurysm. Vasa 2011, 40, 381–389. [Google Scholar] [CrossRef]
- Witkowska, A.M.; Borawska, M.H.; Gacko, M. Relationship among TNF-alpha, sICAM-1, and selenium in presurgical patients with abdominal aortic aneurysms. Biol. Trace Elem. Res. 2006, 114, 31–40. [Google Scholar] [CrossRef]
- Socha, K.; Karwowska, A.; Kurianiuk, A.; Markiewicz-Żukowska, R.; Guzowski, A.; Gacko, M.; Hirnle, T.; Borawska, M.H. Estimation of selected minerals in aortic aneurysms-impaired ratio of zinc to lead may predispose? Biol. Trace Elem. Res. 2021, 199, 2811–2818. [Google Scholar] [CrossRef] [PubMed]
- Strauss, E.; Oszkinis, G.; Staniszewski, R. SEPP1 gene variants and abdominal aortic aneurysm: Gene association in relation to metabolic risk factors and peripheral arterial disease coexistence. Sci. Rep. 2014, 4, 7061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Torres, I.; Soto, M.E.; Manzano-Pech, L.; Díaz-Diaz, E.; Soria-Castro, E.; Rubio-Ruíz, M.E.; Guarner-Lans, V. Oxidative stress in plasma from patients with Marfan syndrome is modulated by deodorized garlic preliminary findings. Oxid. Med. Cell. Longev. 2022, 2022, 5492127. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Flohé, L. Selenium and redox signaling. Arch. Biochem. Biophys. 2017, 617, 48–59. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Guarner-Lans, V.; Rubio-Ruiz, M.E.; Reductive stress in inflammation-associated diseases and the pro-oxidant. Effect of antioxidant agents. Int. J. Mol. Sci. 2017, 18, 2098. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef]
- Tinkov, A.A.; Bjørklund, G.; Skalny, A.V.; Arne Holmgren, A.; Skalnaya, M.G.; Chirumbolo, S.; Aaseth, J. The role of the thioredoxin/thioredoxin reductase system in the metabolic syndrome: Towards a possible prognostic marker? Cell. Mol. Life Sci. 2018, 75, 1567–1586. [Google Scholar] [CrossRef]
- Martinez-Pinna, R.; Lindholt, J.S.; Blanco-Colio, L.M.; Dejouvencel, T.; Madrigal-Matute, J.; Ramos-Mozo, P.; Martin-Ventura, J.L. Increased levels of thioredoxin in patients with abdominal aortic aneurysms (AAAs). A potential link of oxidative stress with AAA evolution. Atherosclerosis 2010, 212, 333–338. [Google Scholar] [CrossRef]
- Soto, M.E.; Manzano-Pech, L.; Palacios-Chavarría, A.; Valdez-Vázquez, R.R.; Guarner-Lans, V.; Pérez-Torres, I. N-Acetyl cysteine restores the diminished activity of the antioxidant enzymatic system caused by SARS-CoV-2 infection: Preliminary findings. Pharmaceuticals 2023, 16, 591. [Google Scholar] [CrossRef]
- Kirsch, J.; Schneider, H.; Pagel, J.I.; Rehberg, M.; Singer, M.; Hellfritsch, J.; Chillo, O.; Schubert, K.M.; Qiu, J.; Pogoda, k.; et al. Endothelial dysfunction, and a prothrombotic, proinflammatory phenotype is caused by loss of mitochondrial thioredoxin reductase in endothelium. Arterioscler. Throm. Vasc. Biol. 2016, 36, 1891–1899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chupakhin, E.; Krasavin, M. Thioredoxin reductase inhibitors: Updated patent review (2017–present). Expert Opin. Ther. Pat. 2021, 31, 745–775. [Google Scholar] [CrossRef] [PubMed]
- Schrenk, S.; Cenzi, C.; Bertalot, T.; Conconi, M.T.; Di Liddo, R. Structural and functional failure of fibrillin-1 in human diseases (review). Int. J. Mol. Med. 2018, 41, 1213–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pincemail, J.; Tchana-Sato, V.; Courtois, A.; Musumeci, L.; Cheramy-Bien, J.P.; Munten, J.; Labropoulos, N.; Defraigne, J.O.; Sakalihasan, N. Alteration of blood oxidative stress status in patients with thoracic aortic dissection: A pilot study. Antioxidants 2023, 12, 1106. [Google Scholar] [CrossRef] [PubMed]
- Akkuş, O.; Kaypaklı, O.; Koca, H.; Topuz, M.; Kaplan, M.; Baykan, A.O.; Samsa, M.Z.; Quisi, A.; Erel, Ö.; Neşelioglu, S.; et al. Thiol/disulphide homeostasis in thoracic aortic aneurysm and acute aortic syndrome. Biomark. Med. 2018, 12, 349–358. [Google Scholar] [CrossRef]
- Farina, A.R.; Tacconelli, A.; Cappabianca, L.; Masciulli, M.P.; Holmgren, A.; Beckett, G.J.; Gulino, A.; Mackay, A.R. Thioredoxin alters the matrix metalloproteinase/tissue inhibitors of metalloproteinase balance and stimulates human SK-N-SH neuroblastoma cell invasion. Eur. J. Biochem. 2001, 268, 405–413. [Google Scholar] [CrossRef]
- Gryszczyńska, B.; Budzyń, M.; Formanowicz, D.; Formanowicz, P.; Krasiński, Z.; Majewska, N.; Iskra, M.; Kasprzak, M.P. Advanced oxidation protein products and carbonylated proteins levels in endovascular and open repair of an abdominal aortic aneurysm: The effect of pre-, intra-, and postoperative treatment. Biomed. Res. Int. 2019, 2019, 7976043. [Google Scholar] [CrossRef] [Green Version]
- Pisano, C.; Balistreri, C.R.; Ricasoli, A.; Ruvolo, G. Cardiovascular disease in ageing: An overview on thoracic aortic aneurysm as an emerging inflammatory disease. Mediat. Inflamm. 2017, 2017, 1274034. [Google Scholar] [CrossRef]
- Li, T.; Zhang, X.; Sang, L.; Li, X.T.; Sun, H.Y.; Yang, J.; Yuan, Y. The interaction effects between TLR4 and MMP9 gene polymorphisms contribute to aortic aneurysm risk in a Chinese Han population. BMC Cardiovasc. Disord. 2019, 19, 72. [Google Scholar] [CrossRef] [Green Version]
- Singh, K.K.; Rommel, K.; Mishra, A.; Karck, M.; Haverich, A.; Schmidtke, J.; Arslan-Kirchner, M. TGFBR1 and TGFBR2 mutations in patients with features of Marfan syndrome and Loeys-Dietz syndrome. Hum. Mutat. 2006, 27, 770–777. [Google Scholar]
- Soto, M.E.; Iturriaga-Hernández, A.V.; Guarner-Lans, V.; Zuñiga-Muñoz, A.; Aranda Fraustro, A.; Velázquez Espejel, E.; Pérez-Torres, I. Participation of oleic acid in the formation of the aortic aneurysm in Marfan syndrome patients. Prostaglandins Other Lipid Mediat. 2016, 123, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cheng, X.; Li, H.; Qiu, F.; Yang, N.; Wang, B.; Lu, H.; Wu, H.; Shen, Y.; Wang, Y.; et al. Quercetin reduces oxidative stress and inhibits activation of c-Jun N-terminal kinase/activator protein-1 signaling in an experimental mouse model of abdominal aortic aneurysm. Mol. Med. Rep. 2014, 9, 435–442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soto, M.E.; Guarner-Lans, V.; Herrera-Morales, K.Y.; Pérez-Torres, I. Participation of arachidonic acid metabolism in the Aortic aneurysm formation in patients with Marfan syndrome. Front. Physiol. 2018, 9, 77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ndebele, P. The declaration of Helsinki, 50 years later. JAMA 2013, 310, 2145–2146. [Google Scholar] [CrossRef] [Green Version]
- Bentall, H.; De Bono, A. A technique for complete replacement of the ascending aorta. Thorax 1968, 23, 338–339. [Google Scholar] [CrossRef] [Green Version]






| Patients | Age | Sex | HFH | AD | EL | SS | Total Systemic Score | Gen FBN1 | Total Ghent Criteria |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 38 | M | + | + | 10 | + | 3 | ||
| 2 | 29 | M | + | + | + | + | 14 | 4 | |
| 3 | 33 | M | + | + | + | + | 16 | 4 | |
| 4 | 56 | F | + | + | 12 | 2 | |||
| 5 | 20 | M | + | + | + | 18 | 3 | ||
| 6 | 25 | F | + | + | 7 | 2 | |||
| 7 | 27 | F | + | + | + | 8 | + | 4 | |
| 8 | 36 | F | + | + | + | 12 | 3 | ||
| 9 | 41 | F | + | + | + | 12 | 3 | ||
| 10 | 37 | F | + | + | + | 12 | 3 | ||
| 11 | 27 | M | + | + | + | 9 | 3 | ||
| 12 | 51 | M | + | + | 12 | + | 3 | ||
| 13 | 61 | F | + | + | + | 13 | + | 4 | |
| 14 | 29 | F | + | + | + | + | 12 | 4 | |
| 15 | 17 | M | + | + | + | 12 | 3 | ||
| 16 | 31 | M | + | + | + | 14 | 3 | ||
| 17 | 60 | M | + | + | + | + | 11 | 4 | |
| 18 | 45 | M | + | + | 10 | + | 3 | ||
| 19 | 43 | M | + | + | + | 7 | 3 |
| Total n = 38 (100%) | MFS n = 19 (50%) | CS n = 19 (50%) | p | |
|---|---|---|---|---|
| Demographic variables in mean ± SE | ||||
| Age | 39 ± 15 | 35 ± 12 | 43 ± 17 | NS |
| Weight | 75 ± 17 | 75 ± 13 | 75 ± 20 | NS |
| Size | 1.70 ± 0.13 | 1.77 ± 0.09 | 1.63 ± 0.11 | 0.0001 |
| BMI | 25.6 ± 5.1 | 23.7 ± 3.4 | 27.5 ± 5.9 | 0.01 |
| Cholesterol | 150.2 ± 48.9 | 148.7 ± 45.2 | 151.7 ± 53.3 | NS |
| C-HDL | 39.2 ± 11.6 | 37.3 ± 13.2 | 41.2 ± 9.6 | NS |
| C-LDL | 97.7 ± 33.3 | 97.7 ± 30.5 | 97.8 ± 43.8 | NS |
| Triglycerides | 146 ± 82 | 146 ± 79 | 145 ± 86 | NS |
| Glucose | 102 ± 26 | 100 ± 28 | 104 ± 25 | NS |
| Creatinine | 0.88 ± 0.44 | 0.93 ± 0.56 | 0.83 ± 0.29 | NS |
| Comorbidities (%) | ||||
| Diabetes Mellitus | 3 (7) | 0 | 3 (13) | 0.07 |
| SAH | 16 (33) | 5 (21) | 11 (46) | 0.06 |
| Hypothyroidism | 1 (2) | 0 | 1 (4) | NS |
| Alcoholism | 13 (27) | 7 (29) | 6 (25) | NS |
| Smoking | 19 (40) | 10 (42) | 9 (38) | NS |
| Echocardiography evaluation (% or median with the Min–Max or mean ± SE) | ||||
| LVEF | 54 ± 11 | 50 ± 11 | 57 ± 11 | 0.02 |
| Surgical | ||||
| Aneurysm | 22 (49) | 11 (46) | 21 (88) | 0.002 |
| Dissection | 16 (67) | 13 (54) | 3 (13) | 0.002 |
| Clamp-Time | 143 (46–253) | 154 (46–253) | 128 (48–250) | NS |
| Extracorporeal circulation | 187 (93–440) | 197 (148–336) | 177 (93–440) | NS |
| EURO-SCOREII | 3 (4–17) | 3 (4–12) | 2 (5–17) | NS |
| Variables (mg of Protein) | CS Median (Min–Max) | MFS Median (Min–Max) |
|---|---|---|
| IL-6 (pg) | 227.1 (0.0–888.96 | 409.1 (111.3–1137.1) *** |
| TGF-β1 (pg) | 570.3 (21.7–1076.3) | 964.6 (384.2–3086.6) ** |
| Carbonylation (nmol carbonyls) | 2.3 (0.8–3.6) | 2.5 (1.7–7.5) † |
| Se (pg) | 224.2 (183.2–322.4) | 205.5 (24.4–243.9) * |
| Thiols (µM) | 196.4 (119.1–286.5) | 164.8 (58.2–246.7) * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soto, M.E.; Pérez-Torres, I.; Manzano-Pech, L.; Soria-Castro, E.; Morales-Marín, A.; Ramírez-Marroquín, E.S.; Martínez-Hernández, H.; Herrera-Alarcón, V.; Guarner-Lans, V. Reduced Levels of Selenium and Thioredoxin Reductase in the Thoracic Aorta Could Contribute to Aneurysm Formation in Patients with Marfan Syndrome. Int. J. Mol. Sci. 2023, 24, 10429. https://doi.org/10.3390/ijms241310429
Soto ME, Pérez-Torres I, Manzano-Pech L, Soria-Castro E, Morales-Marín A, Ramírez-Marroquín ES, Martínez-Hernández H, Herrera-Alarcón V, Guarner-Lans V. Reduced Levels of Selenium and Thioredoxin Reductase in the Thoracic Aorta Could Contribute to Aneurysm Formation in Patients with Marfan Syndrome. International Journal of Molecular Sciences. 2023; 24(13):10429. https://doi.org/10.3390/ijms241310429
Chicago/Turabian StyleSoto, María Elena, Israel Pérez-Torres, Linaloe Manzano-Pech, Elizabeth Soria-Castro, Almilcar Morales-Marín, Edgar Samuel Ramírez-Marroquín, Humberto Martínez-Hernández, Valentín Herrera-Alarcón, and Verónica Guarner-Lans. 2023. "Reduced Levels of Selenium and Thioredoxin Reductase in the Thoracic Aorta Could Contribute to Aneurysm Formation in Patients with Marfan Syndrome" International Journal of Molecular Sciences 24, no. 13: 10429. https://doi.org/10.3390/ijms241310429