Synthesis and Characterization of Bioactive Glass and Zinc Oxide Nanoparticles with Enamel Remineralization and Antimicrobial Capabilities
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Observation of Particle Morphology under Scanning and Transmission Electron Microscopy
2.3. Measurement of Specific Surface Area Using the Gas Absorption Method and Evaluation of Surface Electrical Properties through Zeta Potential Measurement
2.4. Crystal Structure Analysis by X-ray Diffraction
2.5. Evaluation of Acid Buffering Capacity
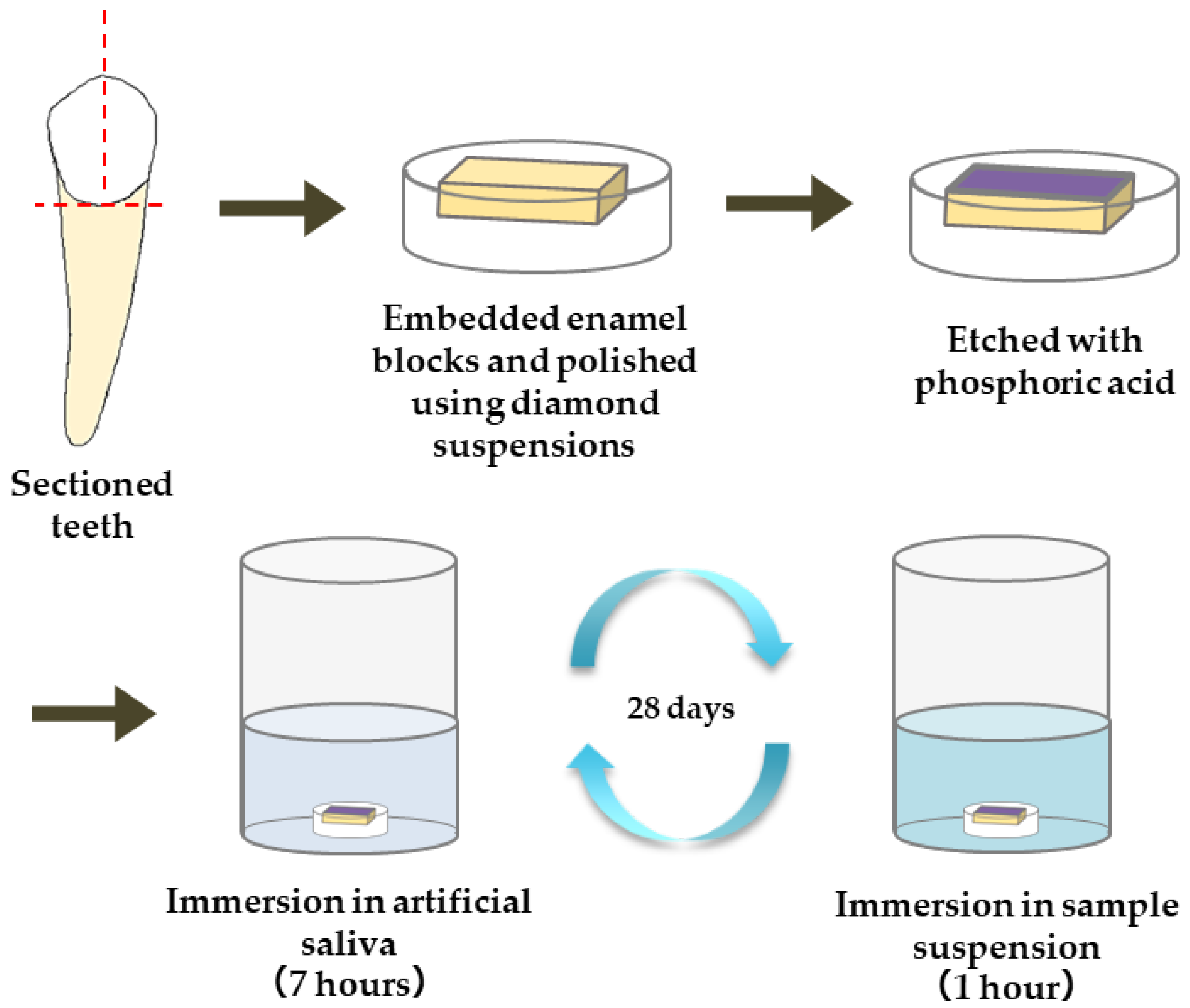
2.6. Evaluating the Capacity of the Particles to Induce Enamel Remineralization
2.7. Evaluation of Antimicrobial Properties
2.8. Statistical Analysis
3. Results
3.1. Morphological Observation of Particle Samples and Identification of Their Crystal Structures
3.2. Measurement of the Specific Surface Area of Each Particle Sample and Evaluation of Surface Charge Properties by Zeta Potential
3.3. Evaluation of Acid Buffering Capacity
3.4. Evaluation of Enamel Remineralization Capacity
3.5. Evaluation of Antimicrobial Properties
4. Discussion
4.1. Properties of Bioactive Glass Nanoparticles
4.2. Evaluation of Remineralization by Changes in Mechanical Properties of the Surface of Enamel Samples
4.3. Antimicrobial Effect of Nanoparticle Suspensions
4.4. Future Clinical Application of Nanoparticle Materials
5. Conclusions
- The particle diameter of bioactive glass microparticles was uneven, ranging from several micrometers to several tens of micrometers. On the other hand, the bioactive nanoparticles were agglomerated, but had individual particle diameters of approximately 20 nm.
- Bioactive glass nanoparticles and microparticles possess rapid acid buffering capacity.
- The enamel samples immersed in the materials containing bioactive glass nanoparticles (nB and nBZ) exhibit superior recovery of mechanical properties compared with those immersed in other materials, and apatite-like structures are deposited on the surface of these enamel samples.
- Materials containing zinc oxide nanoparticles (nBZ) have a broad antimicrobial spectrum.
- Bioactive glass and zinc oxide nanoparticles can suppress demineralization through acid buffering and antimicrobial effects on cariogenic bacteria while promoting remineralization.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Angle, E.H. The latest and best in orthodontic mechanism. Dent. Cosm. 1928, 70, 1143–1158. [Google Scholar]
- Buonocore, M.G. A simple method of increasing the adhesion of acrylic filling materials to enamel surface. J. Dent. Res. 1955, 34, 849–850. [Google Scholar] [CrossRef]
- Newman, G.V. Epoxy adhesives for orthodontic attachments. Am. J. Orthod. 1965, 51, 901–912. [Google Scholar] [CrossRef] [PubMed]
- Sudjalim, T.R.; Woods, M.G.; Manton, D.J.; Reynolds, E.C. Prevention of demineralization around orthodontic brackets in vitro. Am. J. Orthod. Dentofac. Orthop. 2007, 131, e1–e9. [Google Scholar] [CrossRef]
- Kohda, N.; Iijima, M.; Brantley, W.A.; Muguruma, T.; Yuasa, T.; Nakagaki, S.; Mizoguchi, I. Effects of bonding materials on the mechanical properties of enamel around orthodontic brackets. Angle. Orthod. 2012, 82, 187–195. [Google Scholar] [CrossRef]
- Wennstromn, J.L.; Lindhe, J. Some periodontal tissue reactions to orthodontic tooth movement in monkeys. J. Clin. Periodontol. 1987, 14, 121–129. [Google Scholar] [CrossRef]
- Ten Cate, J.M. Contemporary perspective on the use of fluoride products in caries prevention. Br. Dent. J. 2013, 214, 161–167. [Google Scholar] [CrossRef]
- Yolken, R.; Konecuy, P.; McCarthy, P. Acute fluoride poisoning. Paediatrics 1976, 58, 90–93. [Google Scholar] [CrossRef]
- Den Besten, P.; Li, W. Chronic fluoride toxicity: Dental fluorosis. Monogr. Oral. Sci. 2011, 22, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 5, 117–141. [Google Scholar] [CrossRef]
- Kokubo, T. Bioactive glass ceramics: Properties and applications. Biomaterials 1991, 12, 155–163. [Google Scholar] [CrossRef]
- Yli-Urpo, H.; Vallittu, P.K.; Narhi, T.O.; Forsback, A.P.; Vakiparta, M. Release of silica, calcium, phosphorus, and fluoride fromglass ionomer cement containing bioactive glass. J. Biomater. Appl. 2004, 19, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Iijima, M.; Hashimoto, M.; Kohda, N.; Nakagaki, S.; Muguruma, T.; Endo, K.; Mizoguchi, I. Crystal growth on bioactive glass sputter-coated alumina in artificial saliva. Dent. Mater. J. 2013, 32, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Oliva, A.; Salerno, A.; Locardi, B.; Riccio, V.; Della Ragione, F.; Iardino, P.; Zappia, V. Behaviour of human osteoblasts cultured on bioactive glass coatings. Biomaterials 1998, 19, 1019–1025. [Google Scholar] [CrossRef]
- Peitl, O.; Zanotto, E.D.; Hench, L.L. Highly bioactive P2O5-Na2O-CaO-SiO2 glass-ceramics. J. Non-Cryst. Solids. 2001, 292, 115–126. [Google Scholar] [CrossRef]
- Lopez-Esteban, S.; Saiz, E.; Fujino, S.; Oku, T.; Suganuma, K.; Tomsia, A.P. Bioactive glass coatings for orthopedic metallic implants. J. Eur. Ceram. Soc. 2003, 23, 2921–2930. [Google Scholar] [CrossRef]
- Wang, M. Developing bioactive composite materials for tissue replacement. Biomaterials 2003, 24, 2133–2151. [Google Scholar] [CrossRef]
- Jones, J.R. Review of bioactive glass: From Hench to hybrids. Acta. Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef]
- Burwell, A.K.; Litkowski, L.J.; Greenspan, D.C. Calcium sodium phosphosilicate (NovaMin®): Remineralization potential. Adv. Dent. Res. 2009, 21, 35–39. [Google Scholar] [CrossRef]
- Wang, X.; Megert, B.; Hellwig, E.; Neuhaus, K.W.; Lussi, A. Preventing erosion with novel agents. J. Dent. 2011, 39, 163–170. [Google Scholar] [CrossRef]
- Hosokawa, H.; Murakoshi, K.; Wada, Y.; Yanagida, S.; Satoh, M. Extended X-ray absorption fine structure analysis of ZnS nanocrystallites in N,N-Dimethylformamide. An effect of counteranions on the microscopic structure of a solvated surface. Langmuir 1996, 12, 3598–3603. [Google Scholar] [CrossRef]
- Mogyorosi, K.; Dekany, I.; Fendler, J.H. Preparation and characterization of clay mineral intercalated titanium dioxide nanoparticles. Langmuir 2003, 19, 2938–2946. [Google Scholar] [CrossRef]
- Brus, L.E. Electron-electron and electron-hole interactions in small semiconductor crystallites: The size dependence of the lowest excited electronic state. J. Chem. Phys. 1984, 80, 4403–4409. [Google Scholar] [CrossRef]
- Spanhel, L.; Weller, H.; Henglein, A. Photochemistry of semiconductor colloids. 22. electron ejection from illuminated cadmium sulfide into attached titanium and zinc oxide particles. J. Am. Chem. Soc. 1987, 109, 6632–6635. [Google Scholar] [CrossRef]
- Alli, Y.A.; Adewuyi, S.; Bada, B.S.; Thomas, S.; Anuar, H. Quaternary Trimethyl Chitosan Chloride Capped Bismuth Nanoparticles with Positive Surface Charges: Catalytic and Antibacterial Activities. J. Cluster Sci. 2021, 8, 2311–2324. [Google Scholar] [CrossRef]
- Alli, Y.A.; Ejeromedoghene, O.; Oladipo, A.; Adewuyi, S.; Amolegbe, S.A.; Anuar, H.; Thomas, S. Compressed hydrogen-induced synthesis of quaternary trimethyl chitosan-silver nanoparticles with dual antibacterial and antifungal activities. ACS Appl. Bio Mater. 2022, 5, 5240–5254. [Google Scholar] [CrossRef]
- Sawai, J.; Yoshikawa, T. Quantitative evaluation of antifungal activity of metallic oxide powders (MgO, CaO and ZnO) by an indirect conductimetric assay. J. Appl. Microbiol. 2004, 96, 803–809. [Google Scholar] [CrossRef]
- Perez Espitia, P.J.; Ferreira Soares, N.F.; dos Reis Coimbra, J.S.; de Andrade, N.J.; Cruz, R.S.; Alves Medeiros, E.A. Zinc oxide nanoparticles: Synthesis, antimicrobial activity and food packaging applications. Food Bioproc. Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Zhou, J.; Xu, N.; Wang, Z.L. Dissolving behaviour and stability of ZnO wires in biofluids: A study on biodegradability and biocompatibility of ZnO nano-structures. Adv. Mater. 2006, 18, 2432–2435. [Google Scholar] [CrossRef]
- Li, Z.; Yang, R.; Yu, M.; Bai, F.; Li, C.; Wang, Z.L. Cellular level biocompatibility and biosafety of ZnO nanowires. J. Phys. Chem. C 2008, 112, 20114–20117. [Google Scholar] [CrossRef]
- Kawaguchi, K.; Iijima, M.; Endo, K.; Mizoguchi, I. Electrophoretic deposition as a new bioactive glass coating process for orthodontic stainless steel. Coatings 2017, 7, 199. [Google Scholar] [CrossRef]
- Hong, Z.; Reis, R.L.; Mano, J.F. Preparation and in vitro characterization of novel bioactive glass ceramic nanoparticles. J. Biomed. Mater. Res. A 2009, 88, 304–313. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Oppermann, D.A.; Crimp, M.J.; Bement, D.M. In vitro stability predictions for the bone/hydroxyapatite composite system. J. Biomed. Mater. Res. 1998, 42, 412–416. [Google Scholar] [CrossRef]
- Almoudi, M.M.; Hussein, A.S.; Abu Hassan, M.I.; Mohamad Zain, N. A systematic review on antibacterial activity of zinc against Streptococcus mutans. Saudi Dent. J. 2018, 30, 283–291. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquérol, J.; Siemieniewska, T. Reporting physisorption data for gas/ solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Stöber, W.; Fink, A.; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Sepulveda, P.; Jones, J.R.; Hench, L.L. Characterization of melt-derived 45S5 and sol-gel-derived 58S bioactive glasses. J. Biomed. Mater. Res. 2001, 58, 734–740. [Google Scholar] [CrossRef]
- Lin, S.; Ionescu, C.; Pike, K.J.; Smith, M.E.; Jones, J.R. Nanostructure evolution and calcium distribution in sol-gel derived bioactive glass. J. Mater. Chem. 2009, 19, 1276–1282. [Google Scholar] [CrossRef]
- Gumustas, M.; Sengel-Turk, C.T.; Gumustas, A.; Ozkan, S.A.; Uslu, B. Chapter 5—Effect of polymer-based nanoparticles on the assay of antimicrobial drug delivery systems. In Multifunctional Systems for Combined Delivery, Biosensing and Diagnostics; Grumezescu, A.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 67–108. [Google Scholar] [CrossRef]
- Gjorgievska, E.S.; Nicholson, J.W. A preliminary study of enamel remineralization by dentifrices based on Recalden (CPP-ACP) and Novamin (calcium-sodium-phosphosilicate). Acta Odontol. Latinoam. 2010, 23, 234–239. [Google Scholar]
- Legler, L.R.; Retief, D.H.; Bradley, E.L. Effects of phosphoric acid concentration and etch duration on enamel depth of etch: An in vitro study. Am. J. Orthod. Dentofac. Orthop. 1990, 98, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Cury, J.A.; Simoes, G.S.; Del Bel Cury, A.A.; Goncalves, N.C.; Tabchoury, C.P.M. Effect of a calcium carbonate-based dentifrice on in situ enamel remineralization. Caries Res. 2005, 39, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Lippert, F.; Lynch, R.J. Comparison of knoop and vickers surface microhardness and transverse microradiography for the study of early caries lesion formation in human and bovine enamel. Arch. Oral. Biol. 2014, 59, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Jiang, W.; Li, Y.; Chen, X.; Liu, J.; Chen, T.; Miao, G. The effects of morphology on physicochemical properties, bioactivity and biocompatibility of micro-/nano-bioactive glasses. Adv. Powder Technol. 2018, 29, 1812–1819. [Google Scholar] [CrossRef]
- Jiang, W.; Mashayekhi, H.; Xing, B. Bacterial toxicity comparison between nano- and micro-scaled oxide particles. Environ. Pollut. 2009, 157, 1619–1625. [Google Scholar] [CrossRef]
- Waltimo, T.; Brunner, T.J.; Vollenweider, M.; Stark, W.J.; Zehnder, M. Antimicrobial effect of nanometric bioactive glass 45S5. J. Dent. Res. 2007, 86, 754–757. [Google Scholar] [CrossRef]
- Jalal, R.; Goharshadi, E.K.; Abareshi, M.; Moosavi, M.; Yousefi, A.; Nancarrow, P. ZnO nanofluids: Green synthesis, characterization, and antibacterial activity. Mater. Chem. Phys. 2010, 121, 198–201. [Google Scholar] [CrossRef]
- Kasemets, K.; Ivask, A.; Dubourguier, H.C.; Kahru, A. Toxicity of nanoparticles of ZnO, CuO and TiO2 to yeast Saccharomyces cerevisiae. Toxicol. In Vitro 2009, 23, 1116–1122. [Google Scholar] [CrossRef]
- Stoimenov, P.K.; Klinger, R.L.; Marchin, G.L.; Klabunde, K.J. Metal oxide nanoparticles as bactericidal agents. Langmuir 2002, 18, 6679–6686. [Google Scholar] [CrossRef]
- Lynch, R.J.M. Zinc in the mouth, its interactions with dental enamel and possible effects on caries; a review of the literature. Int. Dent. J. 2011, 61, 46–54. [Google Scholar] [CrossRef]
- Singh, P.; Garg, A.; Pandit, S.; Mokkapati, V.; Mijakovic, I. Antimicrobial effects of biogenic nanoparticles. Nanomaterials 2018, 8, 1009. [Google Scholar] [CrossRef]
- Englander, H.R.; Keyes, P.H.; Gestwicki, M.; Sulit, H.A. Clinical anticaries effect of repeated topical sodium fluoride applications by mouthpieces. J. Am. Dent. Assoc. 1967, 75, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Sundararaj, D.; Venkatachalapathy, S.; Tandon, A.; Pereira, A. Critical evaluation of incidence and prevalence of white spot lesions during fixed orthodontic appliance treatment: A meta-analysis. J. Int. Soc. Prev. Commun. Dent. 2015, 5, 433–439. [Google Scholar] [CrossRef]
- Moshiri, M.; Eckhart, J.E.; McShane, P.; German, D.S. Consequence of poor oral hygiene during aligner therapy. J. Clin. Orthod. 2013, 47, 494–498. [Google Scholar]
- Alli, Y.A.; Ogunleye, M.T.; Ejeromedoghene, O.; Adewuyi, S.; Bodunde, J.G.; Balla, F.; Akiode, O.K.; Oladoye, P.O.; Oluwole, K.S.; Thomas, S. Hydrogen-assisted facile synthesis of N, N, N-trimethyl chitosan stabilized zinc oxide nanoparticles and its influence on the quality of Solanum lycopersicum L. Food Hydrocoll. Health 2023, 3, 100124. [Google Scholar] [CrossRef]













| Group | Samples |
|---|---|
| Con | only artificial saliva |
| F | fluoride + cetylpyridinium chloride |
| nB | bioactive glass nanoparticle |
| nBZ | bioactive glass nanoparticle + zinc oxide nanoparticle |
| μB | bioactive glass microparticle |
| Species | Strain | |
|---|---|---|
| Gram positive | Streptococcus mutans | NTCT 10499 |
| Streptococcus sanguinis | JICC 136 | |
| Streptococcus salivarius | HHT | |
| Streptococcus gordonii | DL-1 | |
| Staphylococcus aureus | JCM 20624 | |
| Gram negative | Aggregatibacter actinomycetemcomitans | Y4 |
| Porphyromonas gingivalis | ATCC 33277 | |
| Fusobacterium nucleatum | ATCC 25586 | |
| Prevotella intermedia | JCM 8353 | |
| Fungus | Candida albicancs | FC18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagasaki, R.; Nagano, K.; Nezu, T.; Iijima, M. Synthesis and Characterization of Bioactive Glass and Zinc Oxide Nanoparticles with Enamel Remineralization and Antimicrobial Capabilities. Materials 2023, 16, 6878. https://doi.org/10.3390/ma16216878
Nagasaki R, Nagano K, Nezu T, Iijima M. Synthesis and Characterization of Bioactive Glass and Zinc Oxide Nanoparticles with Enamel Remineralization and Antimicrobial Capabilities. Materials. 2023; 16(21):6878. https://doi.org/10.3390/ma16216878
Chicago/Turabian StyleNagasaki, Ryota, Keiji Nagano, Takashi Nezu, and Masahiro Iijima. 2023. "Synthesis and Characterization of Bioactive Glass and Zinc Oxide Nanoparticles with Enamel Remineralization and Antimicrobial Capabilities" Materials 16, no. 21: 6878. https://doi.org/10.3390/ma16216878






