The Tissue Distribution and Urinary Excretion Study of Gallic Acid and Protocatechuic Acid after Oral Administration of Polygonum Capitatum Extract in Rats
Abstract
:1. Introduction
2. Results and Discussion
2.1. Method Development and Validation
2.1.1. Selectivity and Matrix Effects
2.1.2. Linearity and LLOQ
2.1.3. Precision, Accuracy and Extraction Recovery Tests
2.1.4. Stability
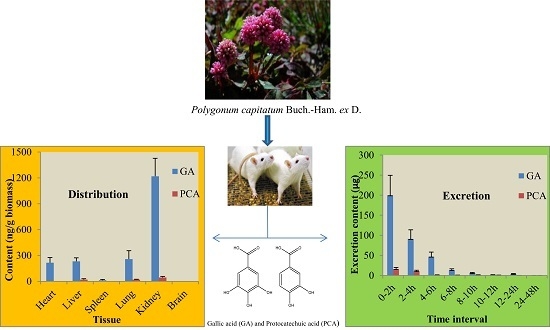
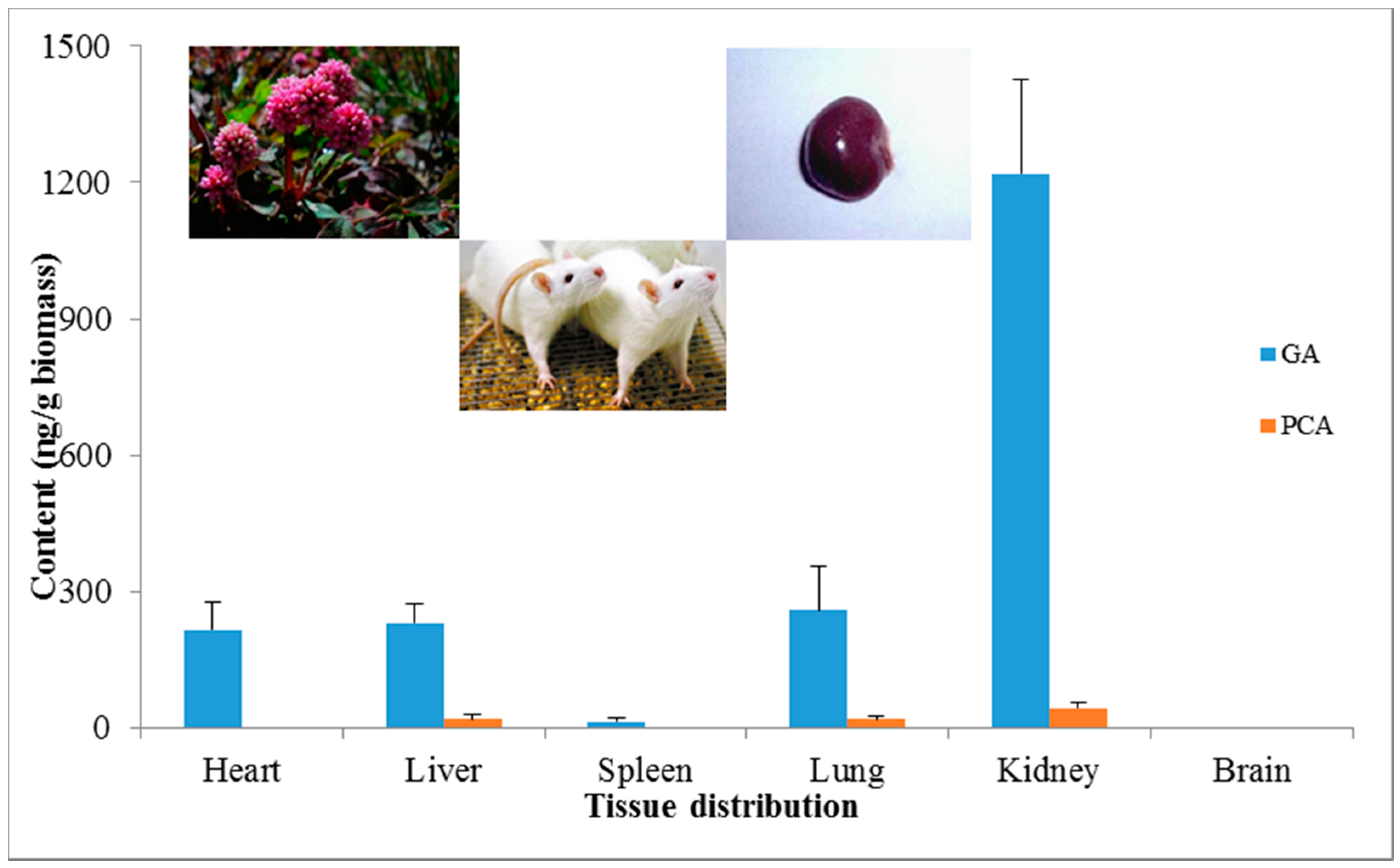
2.2. Tissue Distribution Study
2.3. Excretion Study
3. Experimental Section
3.1. Materials and Reagents
3.2. Experimental Animals
3.3. Analytical Method
3.4. Preparation of P. capitatum Extract
3.5. Method Validation
3.6. Distribution Studies
3.7. Metabolism and Excretion Studies
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- S.O.P. Commission. Pharmacopoeia of the People's Republic of China; Chinese Medical Science and Technology Press: Beijing, China, 2010. [Google Scholar]
- Ma, F.; Zhao, Y.; Gong, X.; Xie, Y.; Zhou, X. Optimization of quercitrin and total flavonoids extraction from Herba Polygoni Capitati by response surface methodology. Pharmacogn. Mag. 2014, 10 (Suppl. 1), S57–S64. [Google Scholar] [PubMed]
- Li, X.; Yu, M.; Meng, D.; Li, Z.; Zhang, L. A new chromone glycoside from Polygonum capitatum. Fitoterapia 2007, 78, 506–509. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Li, Z.-L.; Li, N.; Li, X. Chemical constituents of the aerial parts of Polygonum capitatum. J. Shenyang Pharm. Univ. 2008, 8, 633–635. [Google Scholar]
- Yongjun, L.; Hongfeng, L.; Yonglin, W. Studies on the chemical constituents of flavonoids from Polygonum capitatum. Chin. Pharm. J. 2000, 35, 300–302. [Google Scholar]
- Yan, X.-L.; Li, C.-Q.; Liu, Y.-X.; Chang, X.; Kang, W.-Y. Antioxidant Activity of Polygonum capitatum. China Pharm. 2010, 3659–3661. [Google Scholar]
- Wang, P.-Q.; Zhang, X.-N.; Liu, Y.-X.; Li, C.-Q.; Kang, W.-Y. Antibacterial Activities of Nine Polygonaceae Plants. Chin. J. Exp. Tradit. Med. Form. 2013, 19, 109–112. [Google Scholar]
- Liao, S.-G.; Zhang, L.-J.; Sun, F.; Zhang, J.-J.; Chen, A.Y.; Lan, Y.-Y.; Li, Y.-J.; Wang, A.-M.; He, X.; Xiong, Y.; et al. Antibacterial and anti-inflammatory effects of extracts and fractions from Polygonum capitatum. J. Ethnopharmacol. 2011, 134, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Roy, B.; Das, A.; Bhaumik, U.; Sarkar, A.K.; Bose, A.; Mukharjee, J.; Chakrabarty, U.S.; Das, A.K.; Pal, T.K. Determination of gemifloxacin in different tissues of rat after oral dosing of gemifloxacin mesylate by LC-MS/MS and its application in drug tissue distribution study. J. Pharm. Biomed. Anal. 2010, 52, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xie, L.; Zhang, J.; Weina, P.J. The distribution pattern of intravenous [14C] artesunate in rat tissues by quantitative whole-body autoradiography and tissue dissection techniques. J. Pharm. Biomed. Anal. 2008, 48, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Bent, S.; Ko, R. Commonly used herbal medicines in the United States: A review. Am. J. Med. 2004, 116, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Yang, J.; Gao, X.; Chen, P.; Du, F.; Sun, Y.; Wang, F.; Xu, F.; Shang, H.; Huang, Y. Plasma and urinary tanshinol from Salvia miltiorrhiza (Danshen) can be used as pharmacokinetic markers for cardiotonic pills, a cardiovascular herbal medicine. Drug Metab. Dispos. 2008, 36, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Lazarowych, N.J.; Pekos, P. Use of fingerprinting and marker compounds for identification and standardization of botanical drugs: Strategies for applying pharmaceutical HPLC analysis to herbal products. Drug Inf. J. 1998, 32, 497–512. [Google Scholar] [CrossRef]
- Puchert, T.; Lochmann, D.; Menezes, J.C.; Reich, G. Near-infrared chemical imaging (NIR-CI) for counterfeit drug identification—A four-stage concept with a novel approach of data processing (Linear Image Signature). J. Pharm. Biomed. Anal. 2010, 51, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Sheng, Y.-X.; Li, L.; Wang, Q.; Guo, H.-Z.; Guo, D.-A. Simultaneous determination of gallic acid, albiflorin, paeoniflorin, ferulic acid and benzoic acid in Si–Wu decoction by high-performance liquid chromatography DAD method. J. Pharm. Biomed. Anal. 2005, 37, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.-H.; Hsu, J.-D.; Lo, M.-H.; Chu, C.-Y.; Chou, F.-P.; Huang, C.-L.; Wang, C.-J. Inhibitory effect of Hibiscus protocatechuic acid on tumor promotion in mouse skin. Cancer Lett. 1998, 126, 199–207. [Google Scholar] [CrossRef]
- Aruoma, O.I.; Murcia, A.; Butler, J.; Halliwell, B. Evaluation of the antioxidant and prooxidant actions of gallic acid and its derivatives. J. Agric. Food Chem. 1993, 41, 1880–1885. [Google Scholar] [CrossRef]
- Wang, H.; Provan, G.J.; Helliwell, K. Determination of hamamelitannin, catechins and gallic acid in witch hazel bark, twig and leaf by HPLC. J. Pharm. And Biomed. Anal. 2003, 33, 539–544. [Google Scholar] [CrossRef]
- Tseng, T.-H.; Wang, C.-J.; Kao, E.-S.; Chu, H.-Y. Hibiscus protocatechuic acid protects against oxidative damage induced by tert-butylhydroperoxide in rat primary hepatocytes. Chem. Biol. Int. 1996, 101, 137–148. [Google Scholar] [CrossRef]
- Liu, C.-L.; Wang, J.-M.; Chu, C.-Y.; Cheng, M.-T.; Tseng, T.-H. In vivo protective effect of protocatechuic acid on tert-butyl hydroperoxide-induced rat hepatotoxicity. Food Chem. Toxicol. 2002, 40, 635–641. [Google Scholar] [CrossRef]
- Lu, Z.; Nie, G.; Belton, P.S.; Tang, H.; Zhao, B. Structure–activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem. Int. 2006, 48, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.-C.; Hsieh, C.-L.; Wang, H.-E.; Chung, J.-Y.; Chen, K.-C.; Peng, R.Y. Ferulic acid is nephrodamaging while gallic acid is renal protective in long term treatment of chronic kidney disease. Clin. Nutr. 2012, 31, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Xiao, C.; Wang, Y.; Tang, H. Gallic acid intake induces alterations to systems metabolism in rats. J. Proteome Res. 2012, 12, 991–1006. [Google Scholar] [CrossRef] [PubMed]
- Margalef, M.; Pons, Z.; Bravo, F.I.; Muguerza, B.; Arola-Arnal, A. Tissue distribution of rat flavanol metabolites at different doses. J. Nutr. Biochem. 2015, 26, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Dok-Go, H.; Lee, K.H.; Kim, H.J.; Lee, E.H.; Lee, J.; Song, Y.S.; Lee, Y.-H.; Jin, C.; Lee, Y.S.; Cho, J. Neuroprotective effects of antioxidative flavonoids, quercetin,(+)-dihydroquercetin and quercetin 3-methyl ether, isolated from Opuntia ficus-indica var. Saboten. Brain Res. 2003, 965, 130–136. [Google Scholar] [CrossRef]
- Manach, C.; Donovan, J.L. Pharmacokinetics and metabolism of dietary flavonoids in humans. Free Radic. Res. 2004, 38, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P. E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Masucci, J.A.; Mahan, A.D.; Kwasnoski, J.D.; Caldwell, G.W. A Novel Method for Determination of Drug Distribution in Rat Brain Tissue Sections by LC/MS/MS: Functional Tissue Microanalysis. Curr. Top. Med. Chem. 2012, 12, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Q.; Hop, C.E.C.A. Strategies for characterization of drug metabolites using liquid chromatography-tandem mass spectrometry in conjunction with chemical derivatization and on-line H/D exchange approaches. J. Pharm. Biomed. Anal. 2005, 37, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Cooks, R.G.; Ouyang, Z.; Takats, Z.; Wiseman, J.M. Ambient Mass Spectrometry. Science 2006, 311, 1566–1570. [Google Scholar] [CrossRef] [PubMed]
- Volpi, N.; Bergonzini, G. Analysis of flavonoids from propolis by on-line HPLC-electrospray mass spectrometry. J. Pharm. Biomed. Anal. 2006, 42, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Gong, X.; Zhou, X.; Zhao, Y.; Li, M. An UHPLC-MS/MS method for simultaneous quantification of gallic acid and protocatechuic acid in rat plasma after oral administration of Polygonum capitatum extract and its application to pharmacokinetics. J. Ethnopharmacol. 2015, 162, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Jun, C.-D.; Suk, K.; Choi, B.-J.; Lim, H.; Park, S.; Lee, S.H.; Shin, H.-Y.; Kim, D.-K.; Shin, T.-Y. Gallic acid inhibits histamine release and pro-inflammatory cytokine production in mast cells. Toxicol. Sci. 2006, 91, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Soleas, G.J.; Grass, L.; Josephy, P.D.; Goldberg, D.M.; Diamandis, E.P. A comparison of the anticarcinogenic properties of four red wine polyphenols. Clin. Biochem. 2002, 35, 119–124. [Google Scholar] [CrossRef]
- Jang, A.; Srinivasan, P.; Lee, N.Y.; Song, H.P.; Lee, J.W.; Lee, M.; Jo, C. Comparison of hypolipidemic activity of synthetic gallic acid–linoleic acid ester with mixture of gallic acid and linoleic acid, gallic acid, and linoleic acid on high-fat diet induced obesity in C57BL/6 Cr Slc mice. Chem. Biol. Interact. 2008, 174, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-L.; Yen, G.-C. Effect of gallic acid on high fat diet-induced dyslipidaemia, hepatosteatosis and oxidative stress in rats. Br. J. Nutr. 2007, 98, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Priscilla, D.H.; Prince, P.S. M. Cardioprotective effect of gallic acid on cardiac troponin-T, cardiac marker enzymes, lipid peroxidation products and antioxidants in experimentally induced myocardial infarction in Wistar rats. Chem. Biol. Interact. 2009, 179, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Sabu, M.C.; Kuttan, R. Anti-diabetic activity of medicinal plants and its relationship with their antioxidant property. J. Ethnopharmacol. 2002, 81, 155–160. [Google Scholar] [CrossRef]
- Shahrzad, S.; Bitsch, I. Determination of gallic acid and its metabolites in human plasma and urine by high-performance liquid chromatography. J. Chromatogr. B: Biomed. Sci. Appl. 1998, 705, 87–95. [Google Scholar] [CrossRef]
- Booth, A.N.; Masri, M.S.; Robbins, D.J.; Emerson, O.H.; Jones, F.T.; DeEds, F. The metabolic fate of gallic acid and related compounds. J. Biol. Chem. 1959, 234, 3014–3016. [Google Scholar] [PubMed]
- Shahrzad, S.; Aoyagi, K.; Winter, A.; Koyama, A.; Bitsch, I. Pharmacokinetics of gallic acid and its relative bioavailability from tea in healthy humans. J. Nutr. 2001, 131, 1207–1210. [Google Scholar] [PubMed]
- Hodgson, J.M.; Morton, L.W.; Puddey, I.B.; Beilin, L.J.; Croft, K.D. Gallic acid metabolites are markers of black tea intake in humans. J. Agric. Food Chem. 2000, 48, 2276–2280. [Google Scholar] [CrossRef] [PubMed]
- Zong, L.; Inoue, M.; Nose, M.; Kojima, K.; Sakaguchi, N.; Isuzugawa, K.; Takeda, T.; Ogihara, Y. Metabolic fate of gallic acid orally administered to rats. Biol. Pharm. Bull. 1999, 22, 326–329. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds gallic acid (GA) and protocatechuic acid (PCA) are available from the authors.





| Sample | Analyte | Linearity Range (ng/mL) | Regression Equation | γ2 | LLOQ (ng/mL) |
|---|---|---|---|---|---|
| Kidney | GA | 30 ~ 3000 | Y = 0.003733X + 0.017920 | 0.9976 | 30 |
| PCA | 10 ~ 1000 | Y = 0.015356X − 0.043218 | 0.9907 | 10 | |
| Lung | GA | 30 ~ 3000 | Y = 0.002384X − 0.031599 | 0.9973 | 30 |
| PCA | 10 ~ 1000 | Y = 0.015622X − 0.026021 | 0.9968 | 10 | |
| Heart | GA | 30 ~ 3000 | Y = 0.002114X − 0.003614 | 0.9934 | 30 |
| PCA | 10 ~ 1000 | Y = 0.009321X + 0.036632 | 0.9938 | 10 | |
| Liver | GA | 30 ~ 3000 | Y = 0.002397X − 0.042016 | 0.9955 | 30 |
| PCA | 10 ~ 1000 | Y = 0.011005X + 0.009947 | 0.9999 | 10 | |
| Spleen | GA | 30 ~ 3000 | Y = 0.006551X + 0.023125 | 0.9941 | 30 |
| PCA | 10 ~ 1000 | Y = 0.015537X − 0.035119 | 0.9953 | 10 | |
| Brain | GA | 30 ~ 3000 | Y = 0.006548X + 0.053760 | 0.9976 | 30 |
| PCA | 10 ~ 1000 | Y = 0.018007X − 0.039536 | 0.9904 | 10 | |
| Urine | GA | 30 ~ 6000 | Y = 0.007068X − 0.021609 | 0.9925 | 30 |
| PCA | 10 ~ 2000 | Y = 0.016433X + 0.235975 | 0.9907 | 10 |
| Time Interval | GA Urinary Excretion (μg) | PCA Urinary Excretion (μg) |
|---|---|---|
| 0–2 h | 199.56 ± 49.89 | 15.92 ± 3.98 |
| 2–4 h | 90.96 ± 22.74 | 10.48 ± 2.62 |
| 4–6 h | 46.64 ± 11.66 | 1.08 ± 0.27 |
| 6–8 h | 13.16 ± 3.29 | 0.64 ± 0.16 |
| 8–10 h | 6.00 ± 1.50 | 1.04 ± 0.26 |
| 10–12 h | 1.52 ± 0.38 | 0.39 ± 0.39 |
| 12–24 h | 3.60 ± 0.90 | ND |
| 24–48 h | ND | ND |
| Total | 361.44 ± 90.69 | 29.55 ± 7.68 |
| Excretion rate | 14.60% ± 3.66% | 15.72% ± 4.09% |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, F.-W.; Deng, Q.-F.; Zhou, X.; Gong, X.-J.; Zhao, Y.; Chen, H.-G.; Zhao, C. The Tissue Distribution and Urinary Excretion Study of Gallic Acid and Protocatechuic Acid after Oral Administration of Polygonum Capitatum Extract in Rats. Molecules 2016, 21, 399. https://doi.org/10.3390/molecules21040399
Ma F-W, Deng Q-F, Zhou X, Gong X-J, Zhao Y, Chen H-G, Zhao C. The Tissue Distribution and Urinary Excretion Study of Gallic Acid and Protocatechuic Acid after Oral Administration of Polygonum Capitatum Extract in Rats. Molecules. 2016; 21(4):399. https://doi.org/10.3390/molecules21040399
Chicago/Turabian StyleMa, Feng-Wei, Qing-Fang Deng, Xin Zhou, Xiao-Jian Gong, Yang Zhao, Hua-Guo Chen, and Chao Zhao. 2016. "The Tissue Distribution and Urinary Excretion Study of Gallic Acid and Protocatechuic Acid after Oral Administration of Polygonum Capitatum Extract in Rats" Molecules 21, no. 4: 399. https://doi.org/10.3390/molecules21040399