Chemoresistance to Cancer Treatment: Benzo-α-Pyrene as Friend or Foe?
Abstract
:1. Introduction
2. Results
2.1. Benzo-α-Pyrene Reverse Individual Drug-Induced Cell Death and Aapoptosis in WHCO1 Cells
2.2. Cisplatin, 5-Fluorouracil, and Paclitaxel Differentially Affected the Expression of CYP1A1, CYP1A2, CYP1B1, and GSTP1 in WHCO1 Ccells
2.3. BaP Protects WHCO1 Cancer Cells from the Effects of Cisplatin, 5-fluorouracil, and Paclitaxel Combination Therapy
2.4. Effect of Combinations of Cisplatin, 5-Fluorouracil, Paclitaxel, and BaP on WHCO1 Cell Migration and Invasion
2.5. Chemotherapeutic Drugs Differentially Affect CYPs Expression and Survival Signaling Pathways
3. Discussion
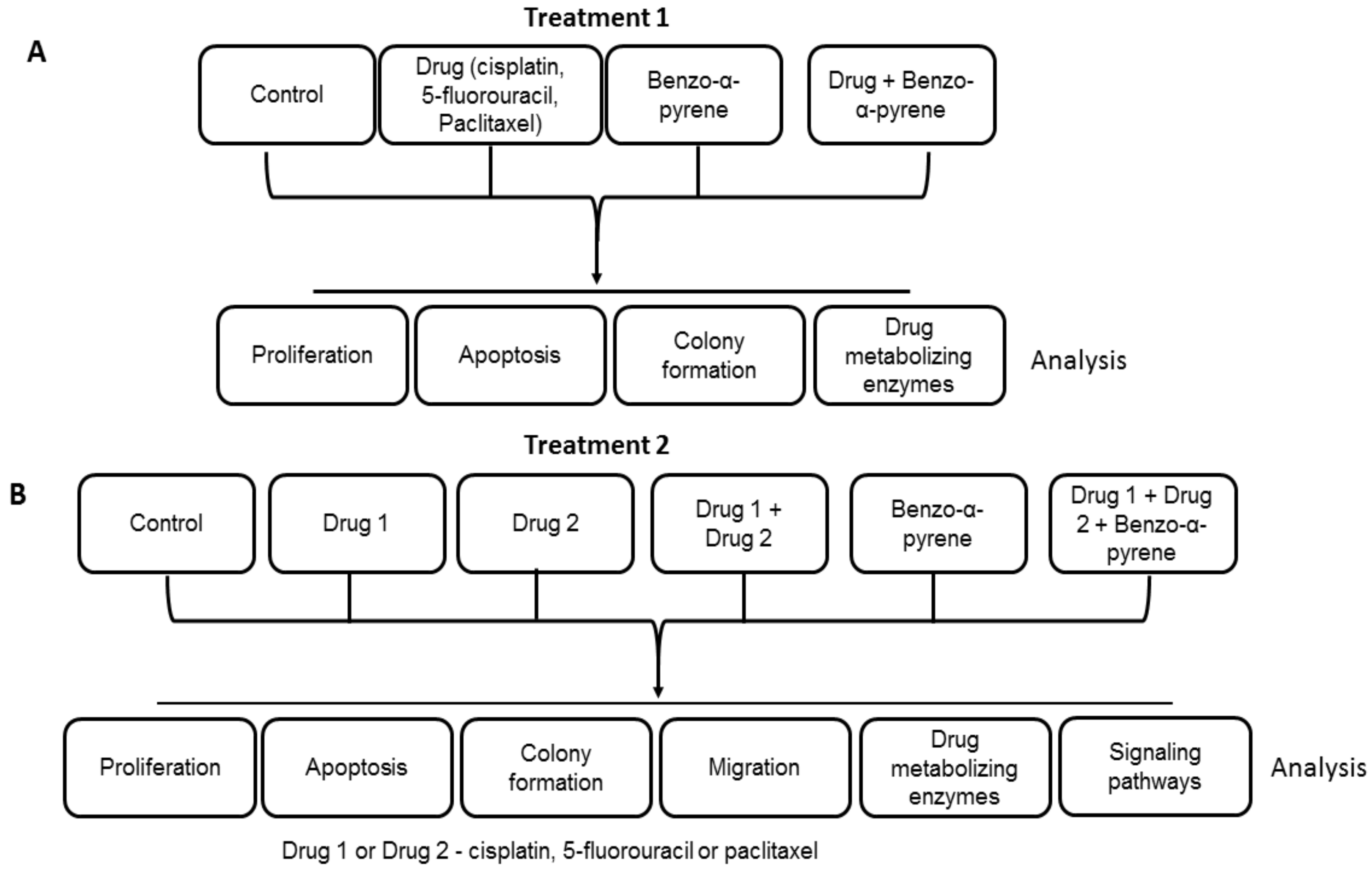
4. Materials and Methods
4.1. Cell Culture and Treatments
4.2. Cell Viability Assay
4.3. Microscopy
4.4. Annexin V Assay for Apoptosis
4.5. Scratch Wound Assay
4.6. Transwell Invasion Assay
4.7. Quantitative RT-PCR Analysis
4.8. Immunoblot Analysis
4.9. Colony Formation
4.10. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Karle, I.L.; Yagi, H.; Sayer, J.M.; Jerina, D.M. Crystal and molecular structure of a benzo[a]pyrene 7,8-diol 9,10-epoxide n2-deoxyguanosine adduct: Absolute configuration and conformation. Proc. Natl. Acad. Sci. USA 2004, 101, 1433–1438. [Google Scholar] [CrossRef] [PubMed]
- Levin, W.; Buening, M.K.; Wood, A.W.; Chang, R.L.; Kedzierski, B.; Thakker, D.R.; Boyd, D.R.; Gadaginamath, G.S.; Armstrong, R.N.; Yagi, H.; et al. An enantiomeric interaction in the metabolism and tumorigenicity of (+)- and (-)-benzo[a]pyrene 7,8-oxide. J. Biol. Chem. 1980, 255, 9067–9074. [Google Scholar] [PubMed]
- Courter, L.A.; Luch, A.; Musafia-Jeknic, T.; Arlt, V.M.; Fischer, K.; Bildfell, R.; Pereira, C.; Phillips, D.H.; Poirier, M.C.; Baird, W.M. The influence of diesel exhaust on polycyclic aromatic hydrocarbon-induced DNA damage, gene expression, and tumor initiation in sencar mice in vivo. Cancer Lett. 2008, 265, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Courter, L.A.; Musafia-Jeknic, T.; Fischer, K.; Bildfell, R.; Giovanini, J.; Pereira, C.; Baird, W.M. Urban dust particulate matter alters pah-induced carcinogenesis by inhibition of cyp1a1 and cyp1b1. Toxicol. Sci. 2007, 95, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Huzlik, J.; Bozek, F.; Pawelczyk, A.; Licbinsky, R.; Naplavova, M.; Pondelicek, M. Identifying risk sources of air contamination by polycyclic aromatic hydrocarbons. Chemosphere 2017, 183, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Jakovljevic, I.; Pehnec, G.; Sisovic, A.; Vadic, V.; Davila, S.; Godec, R. Concentrations of pahs and other gaseous pollutants in the atmosphere of a rural area. J. Environ. Sci. Health Part A 2016, 51, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, L.; Abuduwaili, J.; Li, Y. Distribution, source analysis, and ecological risk assessment of polycyclic aromatic hydrocarbons in the typical topsoil of the issyk-kul lake basin. Environ. Monit. Assess. 2017, 189. [Google Scholar] [CrossRef] [PubMed]
- Strandberg, B.; Julander, A.; Sjostrom, M.; Lewne, M.; Koca Akdeva, H.; Bigert, C. Evaluation of polyurethane foam passive air sampler (puf) as a tool for occupational pah measurements. Chemosphere 2018, 190, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Wolejko, E.; Wydro, U.; Jablonska-Trypuc, A.; Butarewicz, A.; Loboda, T. The effect of sewage sludge fertilization on the concentration of pahs in urban soils. Environ. Pollut. 2018, 232, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Bevan, D.R.; Ulman, M.R. Examination of factors that may influence disposition of benzo[a]pyrene in vivo: Vehicles and asbestos. Cancer Lett. 1991, 57, 173–179. [Google Scholar] [CrossRef]
- Gao, P.; Liu, S.; Feng, Y.; Lin, N.; Lu, B.; Zhang, Z.; Cui, F.; Xing, B.; Hammond, S.K. Concentrations of polycyclic aromatic hydrocarbons in resuspendable fraction of settled bus dust and its implications for human exposure. Environ. Pollut. 2015, 198, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.B.; Hashi, Y.; Liu, M.; Wei, Y.; Lin, J.M. Determination of particle-associated polycyclic aromatic hydrocarbons in urban air of beijing by gc/ms. Anal. Sci. 2007, 23, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Sera, N.; Fukuhara, K.; Miyata, N.; Tokiwa, H. Detection of nitro-azabenzo[a]pyrene derivatives in the semivolatile phase originating from airborne particulate matter, diesel and gasoline vehicles. Mutagenesis 1994, 9, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.P.; Post, G.B.; Lioy, P.J.; Waldman, J.M.; Greenberg, A. Assessment of carcinogenic risk from personal exposure to benzo(a)pyrene in the total human environmental exposure study (thees). Air Waste 1993, 43, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Lioy, P.J. Human exposure to benzo(a)pyrene:Some more of the story. Toxicol. Ind. Health 1992, 8, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Lioy, P.J.; Greenberg, A. Factors associated with human exposures to polycyclic aromatic hydrocarbons. Toxicol. Ind. Health 1990, 6, 209–223. [Google Scholar] [PubMed]
- Lioy, P.L.; Waldman, J.M.; Greenberg, A.; Harkov, R.; Pietarinen, C. The total human environmental exposure study (thees) to benzo(a)pyrene: Comparison of the inhalation and food pathways. Arch. Environ. Health 1988, 43, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Hattemer-Frey, H.A.; Travis, C.C. Benzo-a-pyrene: Environmental partitioning and human exposure. Toxicol. Ind. Health 1991, 7, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.M.; Schell, T.D.; Richie, J.P., Jr.; Sun, Y.W.; Zhang, S.M.; Calcagnotto, A.; Aliaga, C.; Gowda, K.; Amin, S.; El-Bayoumy, K. Effects of chronic alcohol consumption on DNA damage and immune regulation induced by the environmental pollutant dibenzo[a,l]pyrene in oral tissues of mice. J. Environ. Sci. Health Part C 2017, 35, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Kasala, E.R.; Bodduluru, L.N.; Barua, C.C.; Gogoi, R. Antioxidant and antitumor efficacy of luteolin, a dietary flavone on benzo(a)pyrene-induced experimental lung carcinogenesis. Biomed. Pharmacother. 2016, 82, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Masumura, K.; Toyoda-Hokaiwado, N.; Niimi, N.; Gruz, P.; Wada, N.A.; Takeiri, A.; Jishage, K.I.; Mishima, M.; Nohmi, T. Limited ability of DNA polymerase kappa to suppress benzo[a]pyrene-induced genotoxicity in vivo. Environ. Mol. Mutagen. 2017, 58, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Godschalk, R.W.L.; van Schooten, F.J. Inflammation and the chemical carcinogen benzo[a]pyrene: Partners in crime. Mutat. Res. 2017, 774, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.Y.; Hu, W.; Downward, G.S.; Seow, W.J.; Bassig, B.A.; Ji, B.T.; Wei, F.; Wu, G.; Li, J.; He, J.; et al. Personal exposure to fine particulate matter and benzo[a]pyrene from indoor air pollution and leukocyte mitochondrial DNA copy number in rural china. Carcinogenesis 2017, 38, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Gelboin, H.V. Benzo[alpha]pyrene metabolism, activation and carcinogenesis: Role and regulation of mixed-function oxidases and related enzymes. Physiol. Rev. 1980, 60, 1107–1166. [Google Scholar] [CrossRef] [PubMed]
- Greb, W.; Strobel, R.; Rohrborn, G. Transformation of bhk 21/cl 13 cells by various polycyclic aromatic hydrocarbons using the method of styles. Toxicol. Lett. 1980, 7, 143–148. [Google Scholar] [CrossRef]
- Rivedal, E.; Sanner, T. Potentiating effect of cigarette smoke extract on morphological transformation of hamster embryo cells by benzo[alpha]pyrene. Cancer Lett. 1980, 10, 193–198. [Google Scholar] [CrossRef]
- Chakradeo, P.P.; Kayal, J.J.; Bhide, S.V. Effect of benzo(a)pyrene and methyl(acetoxymethyl) nitrosamine on thymidine uptake and induction of aryl hydrocarbon hydroxylase activity in human fetal oesophageal cells in culture. Cell Biol. Int. 1993, 17, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Heimann, R.; Rice, R.H. Polycyclic aromatic hydrocarbon toxicity and induction of metabolism in cultivated esophageal and epidermal keratinocytes. Cancer Res. 1983, 43, 4856–4862. [Google Scholar] [PubMed]
- Port, J.L.; Yamaguchi, K.; Du, B.; De Lorenzo, M.; Chang, M.; Heerdt, P.M.; Kopelovich, L.; Marcus, C.B.; Altorki, N.K.; Subbaramaiah, K.; et al. Tobacco smoke induces cyp1b1 in the aerodigestive tract. Carcinogenesis 2004, 25, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Uno, S.; Dragin, N.; Miller, M.L.; Dalton, T.P.; Gonzalez, F.J.; Nebert, D.W. Basal and inducible cyp1 mrna quantitation and protein localization throughout the mouse gastrointestinal tract. Free Radic. Biol. Med. 2008, 44, 570–583. [Google Scholar] [CrossRef] [PubMed]
- Grzywacz, V.; Balcueva, E. Undiagnosed esophageal adenocarcinoma presenting as multiple brain metastases. Case Rep. Oncol. 2017, 10, 938–944. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Z.; Che, Y.; Yuan, Z.; Lu, Z.; Li, Y.; Wan, J.; Sun, H.; Chen, Z.; Pu, J.; et al. Rabdocoestin b exhibits antitumor activity by inducing g2/m phase arrest and apoptosis in esophageal squamous cell carcinoma. Cancer Chemother. Pharmacol. 2018, 81, 469–481. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Ma, L.; Li, J.; Bai, H.; Wang, D. Elevated hbxip expression is associated with aggressive phenotype and poor prognosis in esophageal squamous cell carcinoma. Am. J. Cancer Res. 2017, 7, 2190–2198. [Google Scholar] [PubMed]
- Xu, Y.; Feingold, P.L.; Surman, D.R.; Brown, K.; Xi, S.; Davis, J.L.; Hernandez, J.; Schrump, D.S.; Ripley, R.T. Bile acid and cigarette smoke enhance the aggressive phenotype of esophageal adenocarcinoma cells by downregulation of the mitochondrial uncoupling protein-2. Oncotarget 2017, 8, 101057–101071. [Google Scholar] [CrossRef] [PubMed]
- Pennathur, A.; Gibson, M.K.; Jobe, B.A.; Luketich, J.D. Oesophageal carcinoma. Lancet 2013, 381, 400–412. [Google Scholar] [CrossRef]
- Pennathur, A.; Xi, L.; Litle, V.R.; Gooding, W.E.; Krasinskas, A.; Landreneau, R.J.; Godfrey, T.E.; Luketich, J.D. Gene expression profiles in esophageal adenocarcinoma predict survival after resection. J. Thorac. Cardiovasc. Surg. 2013, 145, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Adorno-Cruz, V.; Kibria, G.; Liu, X.; Doherty, M.; Junk, D.J.; Guan, D.; Hubert, C.; Venere, M.; Mulkearns-Hubert, E.; Sinyuk, M.; et al. Cancer stem cells: Targeting the roots of cancer, seeds of metastasis, and sources of therapy resistance. Cancer Res. 2015, 75, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Eun, K.; Ham, S.W.; Kim, H. Cancer stem cell heterogeneity: Origin and new perspectives on csc targeting. BMB Rep. 2017, 50, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Gomez, N.C.; Adam, R.C.; Nikolova, M.; Yang, H.; Verma, A.; Lu, C.P.; Polak, L.; Yuan, S.; Elemento, O.; et al. Stem cell lineage infidelity drives wound repair and cancer. Cell 2017, 169, 636–650. [Google Scholar] [CrossRef] [PubMed]
- Dzobo, K.; Senthebane, D.A.; Rowe, A.; Thomford, N.E.; Mwapagha, L.M.; Al-Awwad, N.; Dandara, C.; Parker, M.I. Cancer stem cell hypothesis for therapeutic innovation in clinical oncology? Taking the root out, not chopping the leaf. Omics 2016, 20, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Senthebane, D.A.; Rowe, A.; Thomford, N.E.; Shipanga, H.; Munro, D.; Mazeedi, M.; Almazyadi, H.A.M.; Kallmeyer, K.; Dandara, C.; Pepper, M.S.; et al. The role of tumor microenvironment in chemoresistance: To survive, keep your enemies closer. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Oguri, T.; Achiwa, H.; Bessho, Y.; Muramatsu, H.; Maeda, H.; Niimi, T.; Sato, S.; Ueda, R. The role of thymidylate synthase and dihydropyrimidine dehydrogenase in resistance to 5-fluorouracil in human lung cancer cells. Lung Cancer 2005, 49, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Poorter, R.L.; Bakker, P.J.; Huizing, M.T.; Taat, C.W.; Rietbroek, R.C.; Gouma, D.J.; Rauws, E.A.; Veenhof, C.H. Intermittent continuous infusion of ifosfamide and 5-fluorouracil in patients with advanced adenocarcinoma of the pancreas. Ann. Oncol. 1995, 6, 1048–1049. [Google Scholar] [CrossRef] [PubMed]
- Ofverholm, A.; Arkblad, E.; Skrtic, S.; Albertsson, P.; Shubbar, E.; Enerback, C. Two cases of 5-fluorouracil toxicity linked with gene variants in the dpyd gene. Clin. Biochem. 2010, 43, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Gallego, J.; Cervantes, A.; Pericay, C.; Isla, D. Seom clinical guidelines for the treatment of oesophageal cancer. Clin. Transl. Oncol. 2011, 13, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Martin-Richard, M.; Diaz Beveridge, R.; Arrazubi, V.; Alsina, M.; Galan Guzman, M.; Custodio, A.B.; Gomez, C.; Munoz, F.L.; Pazo, R.; Rivera, F. Seom clinical guideline for the diagnosis and treatment of esophageal cancer (2016). Clin. Transl. Oncol. 2016, 18, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Ando, T.; Ishiguro, H.; Kuwabara, Y.; Kimura, M.; Mitsui, A.; Sugito, N.; Mori, R.; Ogawa, R.; Katada, T.; Fujii, Y. Relationship between expression of 5-fluorouracil metabolic enzymes and 5-fluorouracil sensitivity in esophageal carcinoma cell lines. Dis. Esophagus 2008, 21, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Due, S.L.; Watson, D.I.; Bastian, I.; Ding, G.Q.; Sukocheva, O.A.; Astill, D.S.; Vat, L.; Hussey, D.J. Tamoxifen enhances the cytotoxicity of conventional chemotherapy in esophageal adenocarcinoma cells. Surg. Oncol. 2016, 25, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, D.; Meade, G. Chemical modulation of chemotherapy resistance in cultured oesophageal carcinoma cells. Biochem. Soc. Trans. 2000, 28, 27–32. [Google Scholar] [PubMed]
- Tsunoo, H.; Komura, S.; Ohishi, N.; Akiyama, S.; Kasai, Y.; Ito, K.; Nakao, A.; Yagi, K. Effects of interferon-beta in combination with 5-fluorouracil on the growth of esophageal cancer cells in vitro. Anticancer Res. 2001, 21, 3301–3306. [Google Scholar] [PubMed]
- Su, X.Y.; Yin, H.T.; Li, S.Y.; Huang, X.E.; Tan, H.Y.; Dai, H.Y.; Shi, F.F. Intervention effects of nedaplatin and cisplatin on proliferation and apoptosis of human tumour cells in vitro. Asian Pac. J. Cancer Prev. 2012, 13, 4531–4536. [Google Scholar] [CrossRef] [PubMed]
- Takashima, N.; Ishiguro, H.; Kuwabara, Y.; Kimura, M.; Mitui, A.; Mori, Y.; Mori, R.; Tomoda, K.; Hamaguchi, M.; Ogawa, R.; et al. Gene expression profiling of the response of esophageal carcinoma cells to cisplatin. Dis. Esophagus 2008, 21, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Kaschula, C.H.; Hunter, R.; Stellenboom, N.; Caira, M.R.; Winks, S.; Ogunleye, T.; Richards, P.; Cotton, J.; Zilbeyaz, K.; Wang, Y.; et al. Structure-activity studies on the anti-proliferation activity of ajoene analogues in whco1 oesophageal cancer cells. Eur. J. Med. Chem. 2012, 50, 236–254. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Tanaka, M.; Inagaki, A.; Wanibuchi, H.; Izumi, Y.; Miura, K.; Nagayama, K.; Shiota, M.; Iwao, H. Establishment of a 5-fluorouracil-resistant triple-negative breast cancer cell line. Int. J. Oncol. 2013, 43, 1985–1991. [Google Scholar] [CrossRef] [PubMed]
- Gamarra-Luques, C.D.; Hapon, M.B.; Goyeneche, A.A.; Telleria, C.M. Resistance to cisplatin and paclitaxel does not affect the sensitivity of human ovarian cancer cells to antiprogestin-induced cytotoxicity. J. Ovarian Res. 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Kurtyka, C.A.; Chen, L.; Cress, W.D. E2f inhibition synergizes with paclitaxel in lung cancer cell lines. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Sprouse, A.A.; Herbert, B.S. Resveratrol augments paclitaxel treatment in mda-mb-231 and paclitaxel-resistant mda-mb-231 breast cancer cells. Anticancer Res. 2014, 34, 5363–5374. [Google Scholar] [PubMed]
- Plant, A.L.; Benson, D.M.; Smith, L.C. Cellular uptake and intracellular localization of benzo(a)pyrene by digital fluorescence imaging microscopy. J. Cell Biol. 1985, 100, 1295–1308. [Google Scholar] [CrossRef] [PubMed]
- Barhoumi, R.; Mouneimne, Y.; Ramos, K.S.; Safe, S.H.; Phillips, T.D.; Centonze, V.E.; Ainley, C.; Gupta, M.S.; Burghardt, R.C. Analysis of benzo[a]pyrene partitioning and cellular homeostasis in a rat liver cell line. Toxicol. Sci. 2000, 53, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Chary, P.; Harris, C.M.; Harris, T.M.; Lloyd, R.S. Differential tolerance to DNA polymerization by hiv-1 reverse transcriptase on n6 adenine c10r and c10s benzo[a]pyrene-7,8-dihydrodiol 9,10-epoxide-adducted templates. J. Biol. Chem. 1997, 272, 5805–5813. [Google Scholar] [CrossRef] [PubMed]
- Lavrukhin, O.V.; Lloyd, R.S. Mutagenic replication in a human cell extract of dnas containing site-specific and stereospecific benzo(a)pyrene-7,8-diol-9,10-epoxide DNA adducts placed on the leading and lagging strands. Cancer Res. 1998, 58, 887–891. [Google Scholar] [PubMed]
- Lloyd, D.R.; Hanawalt, P.C. P53-dependent global genomic repair of benzo[a]pyrene-7,8-diol-9,10-epoxide adducts in human cells. Cancer Res. 2000, 60, 517–521. [Google Scholar] [PubMed]
- Omidian, K.; Rafiei, H.; Bandy, B. Polyphenol inhibition of benzo[a]pyrene-induced oxidative stress and neoplastic transformation in an in vitro model of carcinogenesis. Food Chem. Toxicol. 2017, 106, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.Y.; Wang, J.D.; Lu, Y.J.; Shen, Z.Y. Study to elucidate molecular mechanism behind zinc chemo-preventive role during lung carcinogenesis. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 3457–3464. [Google Scholar] [PubMed]
- Dixon, R.L.; Lee, I.P. Metabolism of benzo(a)pyrene by isolated perfused testis and testicular homogenate. Life Sci. 1980, 27, 2439–2444. [Google Scholar] [CrossRef]
- Lubawy, W.C.; Isaac, R.S. Influence of pretreatment with tobacco smoke condensate or 3-methylcholanthrene on [14c] benzo (a) pyrene metabolism in the isolated perfused rabbit lung. Toxicol. Lett. 1980, 7, 153–159. [Google Scholar] [CrossRef]
- Paulson, B.K.; Porter, T.J.; Kent, C. The effect of polycyclic aromatic hydrocarbons on choline kinase activity in mouse hepatoma cells. Biochim. Biophys. Acta 1989, 1004, 274–277. [Google Scholar] [CrossRef]
- Wrighton, S.A.; Fahl, W.E.; Shinnick, F.L., Jr.; Jefcoate, C.R. Characteristics of microsomal reduction of benzo[a]pyrene 4,5-oxide. Chem. Biol. Interact. 1982, 40, 345–356. [Google Scholar] [CrossRef]
- Alomirah, H.; Al-Zenki, S.; Husain, A.; Sawaya, W.; Ahmed, N.; Gevao, B.; Kannan, K. Benzo[a]pyrene and total polycyclic aromatic hydrocarbons (pahs) levels in vegetable oils and fats do not reflect the occurrence of the eight genotoxic pahs. Food Addit. Contam. Part A 2010, 27, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Diggs, D.L.; Huderson, A.C.; Harris, K.L.; Myers, J.N.; Banks, L.D.; Rekhadevi, P.V.; Niaz, M.S.; Ramesh, A. Polycyclic aromatic hydrocarbons and digestive tract cancers: A perspective. J. Environ. Sci. Health Part C 2011, 29, 324–357. [Google Scholar] [CrossRef] [PubMed]
- Wormley, D.D.; Chirwa, S.; Nayyar, T.; Wu, J.; Johnson, S.; Brown, L.A.; Harris, E.; Hood, D.B. Inhaled benzo(a)pyrene impairs long-term potentiation in the f1 generation rat dentate gyrus. Cell. Mol. Biol. 2004, 50, 715–721. [Google Scholar] [PubMed]
- Lee, B.M.; Shim, G.A. Dietary exposure estimation of benzo[a]pyrene and cancer risk assessment. J. Toxicol. Environ. Health Part A 2007, 70, 1391–1394. [Google Scholar] [CrossRef] [PubMed]
- Merlo, F.; Bolognesi, C.; Peluso, M.; Valerio, F.; Abbondandolo, A.; Puntoni, R. Airborne levels of polycyclic aromatic hydrocarbons: 32p-postlabeling DNA adducts and micronuclei in white blood cells from traffic police workers and urban residents. J. Environ. Pathol. Toxicol. Oncol. 1997, 16, 157–162. [Google Scholar] [PubMed]
- Bolognesi, C.; Merlo, F.; Rabboni, R.; Valerio, F.; Abbondandolo, A. Cytogenetic biomonitoring in traffic police workers: Micronucleus test in peripheral blood lymphocytes. Environ. Mol. Mutagen. 1997, 30, 396–402. [Google Scholar] [CrossRef]
- Lindstedt, G.; Sollenberg, J. Polycyclic aromatic hydrocarbons in the occupational environment: With special reference to benzo[a]pyrene measurements in swedish industry. Scand. J. Work Environ. Health 1982, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S.; Brown, R.J. Correlations in polycyclic aromatic hydrocarbon (pah) concentrations in uk ambient air and implications for source apportionment. J. Environ. Monit. 2012, 14, 2072–2082. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Xue, Z.; Zhu, X.; Jia, C. Long-term trends (1990–2014), health risks, and sources of atmospheric polycyclic aromatic hydrocarbons (pahs) in the U.S. Environ. Pollut. 2017, 220, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Polynuclear aromatic compounds, part 4, bitumens, coal-tars and derived products, shale-oils and soots. IARC Monogr. Eval. Carcinog. Risk Chem. Hum. 1985, 35, 1–247.
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Some non-heterocyclic polycyclic aromatic hydrocarbons and some related exposures. IARC Monogr. Eval. Carcinog. Risk Chem. Hum. 2010, 92, 1–853. [Google Scholar]
- Ide, F.; Iida, N.; Nakatsuru, Y.; Oda, H.; Tanaka, K.; Ishikawa, T. Mice deficient in the nucleotide excision repair gene xpa have elevated sensitivity to benzo[a]pyrene induction of lung tumors. Carcinogenesis 2000, 21, 1263–1265. [Google Scholar] [PubMed]
- Iwagawa, M.; Maeda, T.; Izumi, K.; Otsuka, H.; Nishifuji, K.; Ohnishi, Y.; Aoki, S. Comparative dose-response study on the pulmonary carcinogenicity of 1,6-dinitropyrene and benzo[a]pyrene in f344 rats. Carcinogenesis 1989, 10, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, A.M.; Weinstein, I.B.; Jennette, K.W.; Grzeskowiak, K.; Nakanishi, K.; Harvey, R.G.; Autrup, H.; Harris, C. Structures of benzo(a)pyrene--nucleic acid adducts formed in human and bovine bronchial explants. Nature 1977, 269, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Davis, M.E.; Hart, J.E.; Blicharz, A.; Laden, F.; Garshick, E. Potential air toxics hot spots in truck terminals and cabs. Res. Rep. 2012, 5–82. [Google Scholar]
- Chemical agents and Related occupations. IARC Monogr. Eval. Carcinog. Risk Chem. Hum. 2012, 100F, 1–599.
- Matsumoto, R.M.; Jusko, W.J.; Corcoran, G.B. Hepatic cytochrome p-450 and in vitro drug metabolism in an overfed rat model of obesity. Drug Nutr. Interact. 1988, 5, 236–247. [Google Scholar] [PubMed]
- Nimura, Y.; Yokoyama, S.; Fujimori, M.; Aoki, T.; Adachi, W.; Nasu, T.; He, M.; Ping, Y.M.; Iida, F. Genotyping of the cyp1a1 and gstm1 genes in esophageal carcinoma patients with special reference to smoking. Cancer 1997, 80, 852–857. [Google Scholar] [CrossRef]
- Paken, J.; Govender, C.D.; Sewram, V. Research protocol: Cisplatin-associated ototoxicity amongst patients receiving cancer chemotherapy and the feasibility of an audiological monitoring program. BMC Women's Health 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Kurmasheva, R.T.; Kurmashev, D.; Reynolds, C.P.; Kang, M.; Wu, J.; Houghton, P.J.; Smith, M.A. Initial testing (stage 1) of m6620 (formerly vx-970), a novel atr inhibitor, alone and combined with cisplatin and melphalan, by the pediatric preclinical testing program. Pediatr. Blood Cancer 2018, 65. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Duan, W.; Zhang, L.; Li, X.; Fu, X.; Wang, X.; Wu, J.; Sun, Z.; Zhang, X.; Chang, Y.; et al. The efficacy and safety of gemcitabine, cisplatin, prednisone, thalidomide versus chop in patients with newly diagnosed peripheral t-cell lymphoma with analysis of biomarkers. Br. J. Haematol. 2017, 178, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Dollner, R.; Granzow, C.; Neudert, M.; Dietz, A. Ex vivo chemosensitivity of head and neck carcinoma to cytostatic drug combinations. Anticancer Res. 2006, 26, 1651–1655. [Google Scholar] [PubMed]
- Erlichman, C.; Wu, A. Effects of 5-fluorouracil and leucovorin in spheroids: A model for solid tumours. Anticancer Res. 1991, 11, 671–675. [Google Scholar] [PubMed]
- Howat, S.; Park, B.; Oh, I.S.; Jin, Y.W.; Lee, E.K.; Loake, G.J. Paclitaxel: Biosynthesis, production and future prospects. New Biotechnol. 2014, 31, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Frederiks, C.N.; Lam, S.W.; Guchelaar, H.J.; Boven, E. Genetic polymorphisms and paclitaxel- or docetaxel-induced toxicities: A systematic review. Cancer Treat. Rev. 2015, 41, 935–950. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.W.; Frederiks, C.N.; van der Straaten, T.; Honkoop, A.H.; Guchelaar, H.J.; Boven, E. Genotypes of cyp2c8 and fgd4 and their association with peripheral neuropathy or early dose reduction in paclitaxel-treated breast cancer patients. Br. J. Cancer 2016, 115, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Kaschula, C.H.; Hunter, R.; Cotton, J.; Tuveri, R.; Ngarande, E.; Dzobo, K.; Schafer, G.; Siyo, V.; Lang, D.; Kusza, D.A.; et al. The garlic compound ajoene targets protein folding in the endoplasmic reticulum of cancer cells. Mol. Carcinog. 2016, 55, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Shen, L.; Yu, M.; Ni, J.; Dong, X.; Zhou, Y.; Wu, S. Colon cancer cells treated with 5fluorouracil exhibit changes in polylactosaminetype nglycans. Mol. Med. Rep. 2014, 9, 1697–1702. [Google Scholar] [CrossRef] [PubMed]
- Saczko, J.; Kaminska, I.; Kotulska, M.; Bar, J.; Choromanska, A.; Rembialkowska, N.; Biezunska-Kusiak, K.; Rossowska, J.; Nowakowska, D.; Kulbacka, J. Combination of therapy with 5-fluorouracil and cisplatin with electroporation in human ovarian carcinoma model in vitro. Biomed. Pharmacother. 2014, 68, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, P.; Chen, L.; Gao, W.; Zeng, F.; Kong, L.X. Targeted delivery of 5-fluorouracil to ht-29 cells using high efficient folic acid-conjugated nanoparticles. Drug Deliv. 2015, 22, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Yerlikaya, A.; Altikat, S.; Irmak, R.; Cavga, F.Z.; Kocacan, S.A.; Boyaci, I. Effect of bortezomib in combination with cisplatin and 5fluorouracil on 4t1 breast cancer cells. Mol. Med. Rep. 2013, 8, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.M.; Zhang, L.M.; Liu, J.J.; Wan, L.J.; Chen, Y.Q.; Zhang, S.Q.; Yan, Z.W.; Jiang, J.H. Synthesis and antitumor activity of conjugates of 5-fluorouracil and emodin. Eur. J. Med. Chem. 2012, 47, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Andreadis, C.; Vahtsevanos, K.; Sidiras, T.; Thomaidis, I.; Antoniadis, K.; Mouratidou, D. 5-fluorouracil and cisplatin in the treatment of advanced oral cancer. Oral Oncol. 2003, 39, 380–385. [Google Scholar] [CrossRef]
- Iop, A.; Cartei, G.; Vigevani, E.; Clocchiatti, L.; Mansutti, M.; Sibau, A.M. Mitomycin c, cisplatin, and 5-fluorouracil for advanced and/or recurrent head and neck squamous cell carcinomas. Am. J. Clin. Oncol. 1997, 20, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Alba, E.; Sanchez-Chaparro, M.A.; Alonso, L.; Ribelles, N.; Ramon Delgado, J.; Rueda, A.; Pablo Tenllado, P.; Solano, J.; Urquiza, R. Cisplatin, 5-fluorouracil, and high-dose folinic acid in patients with advanced unresectable head and neck cancer. Am. J. Clin. Oncol. 1996, 19, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.A.; Turner, J.; McIlwrath, A.J.; Brown, R.; Dive, C. Cisplatin- and paclitaxel-induced apoptosis of ovarian carcinoma cells and the relationship between bax and bak up-regulation and the functional status of p53. Mol. Pharmacol. 1998, 53, 819–826. [Google Scholar] [PubMed]
- Kobayashi, Y.; Seino, K.; Hosonuma, S.; Ohara, T.; Itamochi, H.; Isonishi, S.; Kita, T.; Wada, H.; Kojo, S.; Kiguchi, K. Side population is increased in paclitaxel-resistant ovarian cancer cell lines regardless of resistance to cisplatin. Gynecol. Oncol. 2011, 121, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.J.; Neuwelt, A.J.; Muldoon, L.L.; Neuwelt, E.A. Acetaminophen enhances cisplatin- and paclitaxel-mediated cytotoxicity to skov3 human ovarian carcinoma. Anticancer Res. 2013, 33, 2391–2400. [Google Scholar] [PubMed]
- Xia, Y.; Li, Y.H.; Chen, Y.; Zhang, J.H.; Liu, Q.; Deng, J.Y.; Ai, T.S.; Zhu, H.T.; Fan, J.H.; Badakhshi, H.; et al. A phase ii study of concurrent chemoradiotherapy combined with a weekly paclitaxel and 5-fluorouracil regimen to treat patients with advanced oesophageal carcinoma. Radiat. Oncol. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Monjanel-Mouterde, S.; Frenay, C.; Catalin, J.; Boutin, C.; Durand, A.; Astoul, P. Pharmacokinetics of intrapleural cisplatin for the treatment of malignant pleural effusions. Oncol. Rep. 2000, 7, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Giannakakou, P.; Robey, R.; Fojo, T.; Blagosklonny, M.V. Low concentrations of paclitaxel induce cell type-dependent p53, p21 and g1/g2 arrest instead of mitotic arrest: Molecular determinants of paclitaxel-induced cytotoxicity. Oncogene 2001, 20, 3806–3813. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Gupta, K.; Yao, J.; Ye, K.; Panda, D.; Giannakakou, P.; Joshi, H.C. Paclitaxel-resistant human ovarian cancer cells undergo c-jun nh2-terminal kinase-mediated apoptosis in response to noscapine. J. Biol. Chem. 2002, 277, 39777–39785. [Google Scholar] [CrossRef] [PubMed]
- Dutta, K.; Ghosh, D.; Nazmi, A.; Kumawat, K.L.; Basu, A. A common carcinogen benzo[a]pyrene causes neuronal death in mouse via microglial activation. PLoS ONE 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Buschini, A.; Villarini, M.; Feretti, D.; Mussi, F.; Dominici, L.; Zerbini, I.; Moretti, M.; Ceretti, E.; Bonfiglioli, R.; Carrieri, M.; et al. Multicentre study for the evaluation of mutagenic/carcinogenic risk in nurses exposed to antineoplastic drugs: Assessment of DNA damage. Occup. Environ. Med. 2013, 70, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Gajski, G.; Ladeira, C.; Geric, M.; Garaj-Vrhovac, V.; Viegas, S. Genotoxicity assessment of a selected cytostatic drug mixture in human lymphocytes: A study based on concentrations relevant for occupational exposure. Environ. Res. 2018, 161, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Das, D.N.; Panda, P.K.; Mukhopadhyay, S.; Sinha, N.; Mallick, B.; Behera, B.; Maiti, T.K.; Bhutia, S.K. Prediction and validation of apoptosis through cytochrome p450 activation by benzo[a]pyrene. Chem. Biol. Interact. 2014, 208, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Jinzhu, Y.; Qinli, Z.; Jin, Y.; Pan, K.; Jianjun, H.; Qiao, N. Aluminum and benzo[a]pyrene co-operate to induce neuronal apoptosis in vitro. J. Toxicol. Sci. 2015, 40, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Michurina, S.V.; Arkhipov, S.A.; Kolesnikov, S.I. Hepatocyte apoptosis in rats exposed to benzo(a)pyrene. Bull. Exp. Biol. Med. 2014, 158, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.S.; Zhang, H.M.; Zhao, J.; Liu, H.J.; Niu, Q. Involvement of mitochondrial pathway in benzo[a]pyrene-induced neuron apoptosis. Hum. Exp. Toxicol. 2014, 33, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Haisa, M.; Naomoto, Y.; Kawashima, R.; Satomoto, K.; Yamatuji, T.; Tanaka, N. Effect of 5-fluorouracil on cell cycle regulatory proteins in human colon cancer cell line. Jpn. J. Cancer res. 1999, 90, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, R.; Kusunoki, M.; Yanagi, H.; Noda, M.; Furuyama, J.I.; Yamamura, T.; Hashimoto-Tamaoki, T. Dual antitumor effects of 5-fluorouracil on the cell cycle in colorectal carcinoma cells: A novel target mechanism concept for pharmacokinetic modulating chemotherapy. Cancer Res. 2001, 61, 1029–1037. [Google Scholar] [PubMed]
- Kalmes, M.; Hennen, J.; Clemens, J.; Blomeke, B. Impact of aryl hydrocarbon receptor (ahr) knockdown on cell cycle progression in human hacat keratinocytes. Biol. Chem. 2011, 392, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahim, M.; Smith, R., 3rd.; Safe, S. Aryl hydrocarbon receptor gene silencing with small inhibitory rna differentially modulates ah-responsiveness in mcf-7 and hepg2 cancer cells. Mol. Pharmacol. 2003, 63, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Korzeniewski, N.; Wheeler, S.; Chatterjee, P.; Duensing, A.; Duensing, S. A novel role of the aryl hydrocarbon receptor (ahr) in centrosome amplification—Implications for chemoprevention. Mol. Cancer 2010, 9. [Google Scholar] [CrossRef] [PubMed]
- Kohle, C.; Bock, K.W. Coordinate regulation of phase I and II xenobiotic metabolisms by the ah receptor and nrf2. Biochem. Pharmacol. 2007, 73, 1853–1862. [Google Scholar] [CrossRef] [PubMed]
- Miao, W.; Hu, L.; Scrivens, P.J.; Batist, G. Transcriptional regulation of nf-e2 p45-related factor (nrf2) expression by the aryl hydrocarbon receptor-xenobiotic response element signaling pathway: Direct cross-talk between phase I and II drug-metabolizing enzymes. J. Biol. Chem. 2005, 280, 20340–20348. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; McMahon, M. Nrf2 and keap1 mutations: Permanent activation of an adaptive response in cancer. Trends Biochem. Sci. 2009, 34, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Ba, Q.; Li, J.; Huang, C.; Qiu, H.; Li, J.; Chu, R.; Zhang, W.; Xie, D.; Wu, Y.; Wang, H. Effects of benzo[a]pyrene exposure on human hepatocellular carcinoma cell angiogenesis, metastasis, and nf-kappab signaling. Environ. Health Perspect. 2015, 123, 246–254. [Google Scholar] [PubMed]
- Castillo-Sanchez, R.; Villegas-Comonfort, S.; Galindo-Hernandez, O.; Gomez, R.; Salazar, E.P. Benzo-[a]-pyrene induces fak activation and cell migration in mda-mb-231 breast cancer cells. Cell Biol. Toxicol. 2013, 29, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Palatnik, A.; Xin, H.; Su, E.J. Dichotomous effects of aryl hydrocarbon receptor (ahr) activation on human fetoplacental endothelial cell function. Placenta 2016, 44, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhai, W.; Wang, H.; Xia, X.; Zhang, C. Benzo(a)pyrene promotes a549 cell migration and invasion through up-regulating twist. Arch. Toxicol. 2015, 89, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhao, L.; He, W.; Yang, J.; Geng, C.; Chen, Y.; Liu, T.; Chen, H.; Li, Y. Benzo[a]pyrene promotes gastric cancer cell proliferation and metastasis likely through the aryl hydrocarbon receptor and erk-dependent induction of mmp9 and c-myc. Int. J. Oncol. 2016, 49, 2055–2063. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chang, L.; Jin, H.; Xia, Y.; Wang, L.; He, W.; Li, W.; Chen, H. Benzopyrene promotes lung cancer a549 cell migration and invasion through up-regulating cytokine il8 and chemokines ccl2 and ccl3 expression. Exp. Biol. Med. 2016, 241, 1516–1523. [Google Scholar] [CrossRef] [PubMed]
- Verhofstad, N.; Pennings, J.L.; van Oostrom, C.T.; van Benthem, J.; van Schooten, F.J.; van Steeg, H.; Godschalk, R.W. Benzo(a)pyrene induces similar gene expression changes in testis of DNA repair proficient and deficient mice. BMC Genom. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- Verhofstad, N.; van Oostrom, C.T.; Zwart, E.; Maas, L.M.; van Benthem, J.; van Schooten, F.J.; van Steeg, H.; Godschalk, R.W. Evaluation of benzo(a)pyrene-induced gene mutations in male germ cells. Toxicol. Sci. 2011, 119, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Steelman, L.S.; Abrams, S.L.; Ruvolo, P.; Ruvolo, V.; Cocco, L.; Ratti, S.; Martelli, A.M.; Neri, L.M.; Candido, S.; Libra, M.; et al. Drug-resistance in doxorubicin-resistant fl5.12 hematopoietic cells: Elevated mdr1, drug efflux and side-population positive and decreased bcl2-family member expression. Oncotarget 2017, 8, 113013–113033. [Google Scholar] [CrossRef] [PubMed]
- Booher, R.N.; Hatch, H.; Dolinski, B.M.; Nguyen, T.; Harmonay, L.; Al-Assaad, A.S.; Ayers, M.; Nebozhyn, M.; Loboda, A.; Hirsch, H.A.; et al. Mcl1 and bcl-xl levels in solid tumors are predictive of dinaciclib-induced apoptosis. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.M.; Lee, L.; Hicks, D.J.; Joly, M.M.; Elion, D.; Rahman, B.; McKernan, C.; Sanchez, V.; Balko, J.M.; Stricker, T.; et al. Key survival factor, mcl-1, correlates with sensitivity to combined bcl-2/bcl-xl blockade. Mol. Cancer Res. 2017, 15, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Flores, M.L.; Castilla, C.; Avila, R.; Ruiz-Borrego, M.; Saez, C.; Japon, M.A. Paclitaxel sensitivity of breast cancer cells requires efficient mitotic arrest and disruption of bcl-xl/bak interaction. Breast Cancer Res. Treat. 2012, 133, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Kasai, S.; Sasaki, T.; Watanabe, A.; Nishiya, M.; Yasuhira, S.; Shibazaki, M.; Maesawa, C. Bcl-2/bcl-xl inhibitor abt-737 sensitizes pancreatic ductal adenocarcinoma to paclitaxel-induced cell death. Oncol. Lett. 2017, 14, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Lieber, J.; Kirchner, B.; Eicher, C.; Warmann, S.W.; Seitz, G.; Fuchs, J.; Armeanu-Ebinger, S. Inhibition of bcl-2 and bcl-x enhances chemotherapy sensitivity in hepatoblastoma cells. Pediatr. Blood Cancer 2010, 55, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.; Briasoulis, E.; Linardou, H.; Bafaloukos, D.; Papadimitriou, C. Taxane resistance in breast cancer: Mechanisms, predictive biomarkers and circumvention strategies. Cancer Treat. Rev. 2012, 38, 890–903. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Dou, Y.; Zhou, K.; Huo, J.; Yang, T.; Qin, T.; Liu, W.; Wang, S.; Yang, D.; Chang, L.; et al. Targeting the bcl-2 family and p-glycoprotein reverses paclitaxel resistance in human esophageal carcinoma cell line. Biomed. Pharmacother. 2017, 90, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Chun, E.; Lee, K.Y. Bcl-2 and bcl-xl are important for the induction of paclitaxel resistance in human hepatocellular carcinoma cells. Biochem. Biophys. Res. Commun. 2004, 315, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, T.; Nagaoka, T.; Omae, T.; Ono, S.; Otani, S.; Yoshida, A. Benzo(e)pyrene inhibits endothelium-dependent no-mediated dilation of retinal arterioles via superoxide production and endoplasmic reticulum stress. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5978–5984. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, R.; Das, S.K.; Patri, M. Modulation of benzo[a]pyrene induced anxiolytic-like behavior by retinoic acid in zebrafish: Involvement of oxidative stress and antioxidant defense system. Neurotox. Res. 2017, 31, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Sarma, S.N.; Blais, J.M.; Chan, H.M. Neurotoxicity of alkylated polycyclic aromatic compounds in human neuroblastoma cells. J. Toxicol. Environ. Health Part A 2017, 80, 285–300. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Padilla, M.T.; Xu, X.; Desai, D.; Krzeminski, J.; Amin, S.; Lin, Y. Quercetin inhibits multiple pathways involved in interleukin 6 secretion from human lung fibroblasts and activity in bronchial epithelial cell transformation induced by benzo[a]pyrene diol epoxide. Mol. Carcinog. 2016, 55, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Xu, Z.; Zhao, H.; Sun, Z.; Wang, L.; Guo, Z.; Zhao, Y.; Gao, Z.; Wang, Q. Macrophages promote benzopyrene-induced tumor transformation of human bronchial epithelial cells by activation of nf-kappab and stat3 signaling in a bionic airway chip culture and in animal models. Oncotarget 2015, 6, 8900–8913. [Google Scholar] [PubMed]
- Parsa, M.; Ostad, S.N.; Moogahi, S.M.; Bayat, M.; Ghahremani, M.H. The effect of benzo[alpha]pyrene on expression and signaling cross talk of aryl hydrocarbon receptor and nfatc1 in mouse lung tissue. Toxicol. Ind. Health 2016, 32, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Lv, B.; Zha, J.; Wang, Z. Benzo[a]pyrene induced p53-mediated cell cycle arrest, DNA repair, and apoptosis pathways in chinese rare minnow (gobiocypris rarus). Environ. Toxicol. 2017, 32, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.Z.; Chan, J.; Wang, Q.; Zhang, W.; Sun, C.D.; Wang, L.H. Twist transcriptionally up-regulates akt2 in breast cancer cells leading to increased migration, invasion, and resistance to paclitaxel. Cancer Res. 2007, 67, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, S.; Hosseini, M.; Shahidsales, S.; Maftouh, M.; Ferns, G.A.; Ghayour-Mobarhan, M.; Hassanian, S.M.; Avan, A. Targeting the akt/pi3k signaling pathway as a potential therapeutic strategy for the treatment of pancreatic cancer. Curr. Med. Chem. 2017, 24, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Serebriiskii, I.; Castello-Cros, R.; Lamb, A.; Golemis, E.A.; Cukierman, E. Fibroblast-derived 3d matrix differentially regulates the growth and drug-responsiveness of human cancer cells. Matrix. Biol. 2008, 27, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Veale, R.B.; Thornley, A.L. Increased single class low affinity egf receptors expressed by human oesophageal squamous carcinoma cell lines. South Afr. J. Sci. 1989, 85, 375–379. [Google Scholar]
- Dzobo, K.; Vogelsang, M.; Thomford, N.E.; Dandara, C.; Kallmeyer, K.; Pepper, M.S.; Parker, M.I. Wharton’s jelly-derived mesenchymal stromal cells and fibroblast-derived extracellular matrix synergistically activate apoptosis in a p21-dependent mechanism in whco1 and mda mb 231 cancer cells in vitro. Stem. Cells Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |







© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dzobo, K.; Hassen, N.; Senthebane, D.A.; Thomford, N.E.; Rowe, A.; Shipanga, H.; Wonkam, A.; Parker, M.I.; Mowla, S.; Dandara, C. Chemoresistance to Cancer Treatment: Benzo-α-Pyrene as Friend or Foe? Molecules 2018, 23, 930. https://doi.org/10.3390/molecules23040930
Dzobo K, Hassen N, Senthebane DA, Thomford NE, Rowe A, Shipanga H, Wonkam A, Parker MI, Mowla S, Dandara C. Chemoresistance to Cancer Treatment: Benzo-α-Pyrene as Friend or Foe? Molecules. 2018; 23(4):930. https://doi.org/10.3390/molecules23040930
Chicago/Turabian StyleDzobo, Kevin, Naseeha Hassen, Dimakatso Alice Senthebane, Nicholas Ekow Thomford, Arielle Rowe, Hendrina Shipanga, Ambroise Wonkam, M. Iqbal Parker, Shaheen Mowla, and Collet Dandara. 2018. "Chemoresistance to Cancer Treatment: Benzo-α-Pyrene as Friend or Foe?" Molecules 23, no. 4: 930. https://doi.org/10.3390/molecules23040930






