Zingiber officinale Roscoe Rhizomes Attenuate Oxaliplatin-Induced Neuropathic Pain in Mice
Abstract
1. Introduction
2. Results
2.1. Oxaliplatin Administration Induces Cold and Mechanical Allodynia in Mice
2.2. Anti-Allodynic Effect of Z. officinale on Oxaliplatin-Induced Neuropathic Pain in Mice
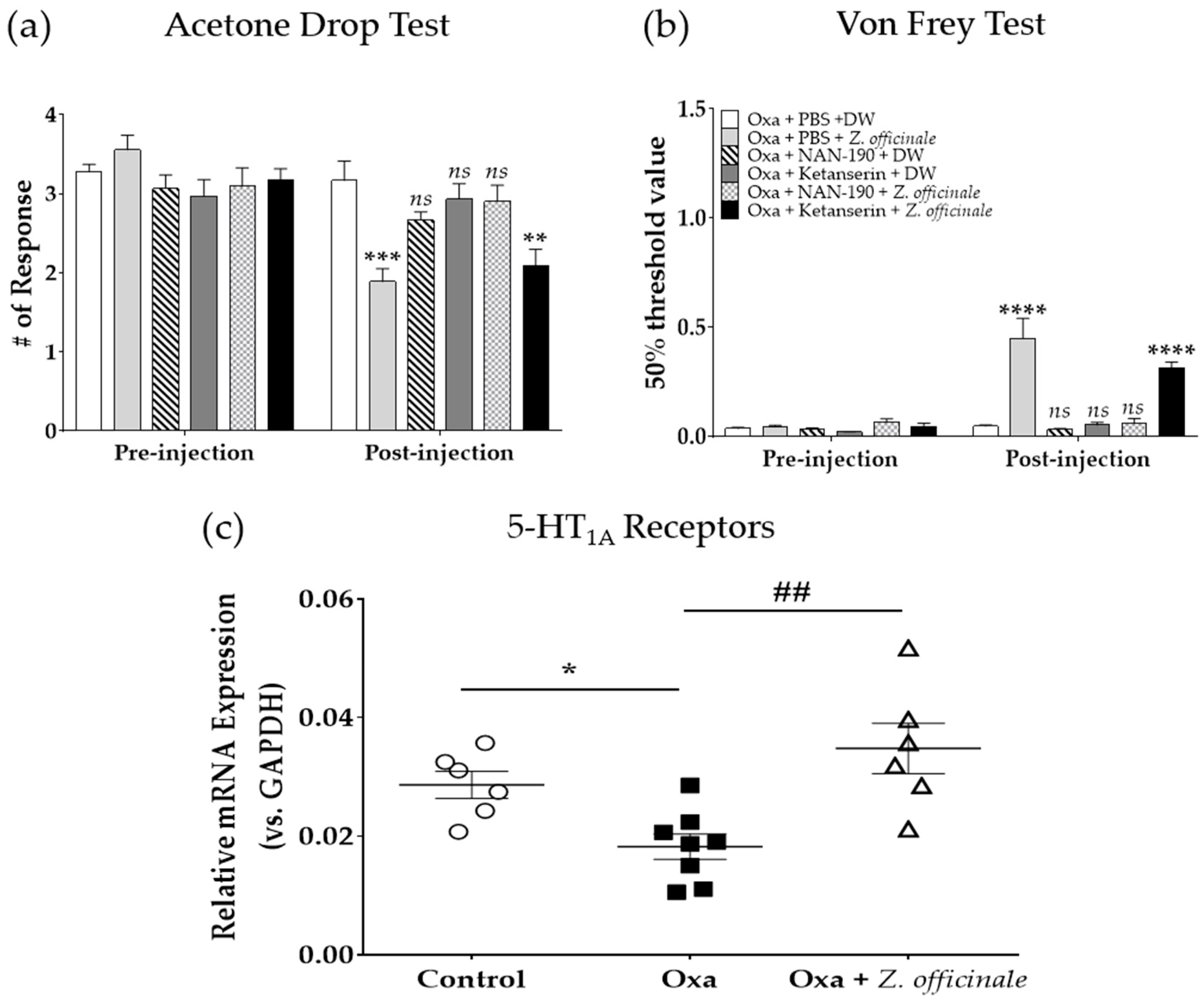
2.3. Spinal 5-HT Receptors Are Involved in the Neuropathic Pain-Alleviating Effect of Z. officinale
2.4. Spinal 5-HT1A but Not 5-HT2A is Involved in the Anti-Allodynic Effect of Z. officinale
2.5. Identification of Active Components in Z. officinale by Using HPLC
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Oxaliplatin Administration
4.3. Preparation of and Treatment with Z. Officinale
4.4. Treatment with Serotonergic Antagonists
4.5. Behavioral Assessments
4.6. Behavioral Tests Schedules
4.7. High-Performance Liquid Chromatography (HPLC) Analysis of the Z. officinale Extract
4.8. RNA Extraction and Real-Time PCR
4.9. Statistical Analysis
5. Conclusions
6. Patent
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Graham, J.; Muhsin, M.; Kirkpatrick, P. Oxaliplatin; Nature Publishing Group: Berlin, Germany, 2004. [Google Scholar]
- Di Francesco, A.; Ruggiero, A.; Riccardi, R. Cellular and molecular aspects of drugs of the future: Oxaliplatin. Cell. Mol. Life Sci. CMLS 2002, 59, 1914–1927. [Google Scholar] [CrossRef] [PubMed]
- Ta, L.E.; Low, P.A.; Windebank, A.J. Mice with cisplatin and oxaliplatin-induced painful neuropathy develop distinct early responses to thermal stimuli. Mol. Pain 2009, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Lehky, T.; Leonard, G.; Wilson, R.; Grem, J.; Floeter, M. Oxaliplatin-induced neurotoxicity: Acute hyperexcitability and chronic neuropathy. Muscle Nerve 2004, 29, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Pachman, D.R.; Qin, R.; Seisler, D.K.; Smith, E.M.L.; Beutler, A.S.; Ta, L.E.; Lafky, J.M.; Wagner-Johnston, N.; Ruddy, K.J.; Dakhil, S.R. Clinical course of patients with oxaliplatin-associated neuropathy: N08cb (alliance). Am. Soc. Clin. Oncol. 2014, 32, 3595. [Google Scholar] [CrossRef]
- Kim, W. Effect of oxaliplatin on voltage-gated sodium channels in peripheral neuropathic pain. Processes 2020, 8, 680. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, W. The role of satellite glial cells, astrocytes, and microglia in oxaliplatin-induced neuropathic pain. Biomedicines 2020, 8, 324. [Google Scholar] [CrossRef]
- Kim, W.; Kim, M.J.; Go, D.; Min, B.-I.; Na, H.S.; Kim, S.K. Combined effects of bee venom acupuncture and morphine on oxaliplatin-induced neuropathic pain in mice. Toxins 2016, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Chung, Y.; Choi, S.; Min, B.-I.; Kim, S.K. Duloxetine protects against oxaliplatin-induced neuropathic pain and spinal neuron hyperexcitability in rodents. Int. J. Mol. Sci. 2017, 18, 2626. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Gang, J.; Yang, E.; Kim, W.; Jin, Y.-H. Bee venom acupuncture attenuates oxaliplatin-induced neuropathic pain by modulating action potential threshold in a-fiber dorsal root ganglia neurons. Toxins 2020, 12, 737. [Google Scholar] [CrossRef]
- Sepahvand, R.; Esmaeili-Mahani, S.; Arzi, A.; Rasoulian, B.; Abbasnejad, M. Ginger (Zingiber officinale roscoe) elicits antinociceptive properties and potentiates morphine-induced analgesia in the rat radiant heat tail-flick test. J. Med. Food 2010, 13, 1397–1401. [Google Scholar] [CrossRef] [PubMed]
- White, B. Ginger: An overview. Am. Fam. Phys. 2007, 75, 1689–1691. [Google Scholar]
- Langner, E.; Greifenberg, S.; Gruenwald, J. Ginger: History and use. Adv. Ther. 1998, 15, 25–44. [Google Scholar] [PubMed]
- Prasad, S.; Tyagi, A.K. Ginger and its constituents: Role in prevention and treatment of gastrointestinal cancer. Gastroenterol. Res. Pract. 2015, 2015, 142979. [Google Scholar] [CrossRef] [PubMed]
- Habib, S.H.M.; Makpol, S.; Hamid, N.A.A.; Das, S.; Ngah, W.Z.W.; Yusof, Y.A.M. Ginger extract (Zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics 2008, 63, 807–813. [Google Scholar] [CrossRef]
- Ojewole, J.A. Analgesic, antiinflammatory and hypoglycaemic effects of ethanol extract of Zingiber officinale (roscoe) rhizomes (zingiberaceae) in mice and rats. Phytother. Res. 2006, 20, 764–772. [Google Scholar] [CrossRef]
- Haniadka, R.; Rajeev, A.G.; Palatty, P.L.; Arora, R.; Baliga, M.S. Zingiber officinale (ginger) as an anti-emetic in cancer chemotherapy: A review. J. Altern. Complement. Med. 2012, 18, 440–444. [Google Scholar] [CrossRef]
- Black, C.D.; Herring, M.P.; Hurley, D.J.; O’Connor, P.J. Ginger (Zingiber officinale) reduces muscle pain caused by eccentric exercise. J. Pain 2010, 11, 894–903. [Google Scholar] [CrossRef]
- Fajrin, F.A.; Nurrochmad, A.; Nugroho, A.E.; Susilowati, R. The improvement of pain behavior and sciatic nerves morphology in mice model of painful diabetic neuropathy upon administration of ginger (Zingiber officinale roscoe.) extract and its pungent compound, 6-shogaol. J. Nat. Sci. Biol. Med. 2019, 10, 149. [Google Scholar]
- Borgonetti, V.; Governa, P.; Biagi, M.; Pellati, F.; Galeotti, N. Zingiber officinale roscoe rhizome extract alleviates neuropathic pain by inhibiting neuroinflammation in mice. Phytomedicine 2020, 78, 153307. [Google Scholar] [CrossRef]
- Sommer, C. Serotonin in pain and pain control. In Handbook of Behavioral Neuroscience; Elsevier: Amsterdam, The Netherlands, 2010; Volume 21, pp. 457–471. [Google Scholar]
- Millan, M.J. Descending control of pain. Prog. Neurobiol. 2002, 66, 355–474. [Google Scholar] [CrossRef]
- Barnes, N.M.; Sharp, T. A review of central 5-ht receptors and their function. Neuropharmacology 1999, 38, 1083–1152. [Google Scholar] [CrossRef]
- Bardin, L. The complex role of serotonin and 5-ht receptors in chronic pain. Behav. Pharmacol. 2011, 22, 390–404. [Google Scholar] [CrossRef] [PubMed]
- Bardin, L.; Lavarenne, J.; Eschalier, A. Serotonin receptor subtypes involved in the spinal antinociceptive effect of 5-ht in rats. Pain 2000, 86, 11–18. [Google Scholar] [CrossRef]
- Li, D.; Lee, J.H.; Choi, C.W.; Kim, J.; Kim, S.K.; Kim, W. The analgesic effect of venlafaxine and its mechanism on oxaliplatin-induced neuropathic pain in mice. Int. J. Mol. Sci. 2019, 20, 1652. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Li, D.X.; Yoon, H.; Go, D.; Quan, F.S.; Min, B.-I.; Kim, S.K. Serotonergic mechanism of the relieving effect of bee venom acupuncture on oxaliplatin-induced neuropathic cold allodynia in rats. BMC Complement. Altern. Med. 2014, 14, 471. [Google Scholar] [CrossRef]
- Hershman, D.L.; Lacchetti, C.; Dworkin, R.H.; Lavoie Smith, E.M.; Bleeker, J.; Cavaletti, G.; Chauhan, C.; Gavin, P.; Lavino, A.; Lustberg, M.B. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American society of clinical oncology clinical practice guideline. J. Clin. Oncol. 2014, 32, 1941–1967. [Google Scholar] [CrossRef]
- Stahl, S.M.; Grady, M.M.; Moret, C.; Briley, M. Snris: The pharmacology, clinical efficacy, and tolerability in comparison with other classes of antidepressants. CNS Spectr. 2005, 10, 732–747. [Google Scholar] [CrossRef]
- Zimmerman, C.; Atherton, P.J.; Pachman, D.; Seisler, D.; Wagner-Johnston, N.; Dakhil, S.; Lafky, J.M.; Qin, R.; Grothey, A.; Loprinzi, C.L. Mc11c4: A pilot randomized, placebo-controlled, double-blind study of venlafaxine to prevent oxaliplatin-induced neuropathy. Support. Care Cancer 2016, 24, 1071–1078. [Google Scholar] [CrossRef]
- Kagiava, A.; Tsingotjidou, A.; Emmanouilides, C.; Theophilidis, G. The effects of oxaliplatin, an anticancer drug, on potassium channels of the peripheral myelinated nerve fibres of the adult rat. Neurotoxicology 2008, 29, 1100–1106. [Google Scholar] [CrossRef]
- Zheng, H.; Xiao, W.H.; Bennett, G.J. Functional deficits in peripheral nerve mitochondria in rats with paclitaxel-and oxaliplatin-evoked painful peripheral neuropathy. Exp. Neurol. 2011, 232, 154–161. [Google Scholar] [CrossRef]
- Hitomi, S.; Ono, K.; Terawaki, K.; Matsumoto, C.; Mizuno, K.; Yamaguchi, K.; Imai, R.; Omiya, Y.; Hattori, T.; Kase, Y. [6]-gingerol and [6]-shogaol, active ingredients of the traditional japanese medicine hangeshashinto, relief oral ulcerative mucositis-induced pain via action on Na+ channels. Pharmacol. Res. 2017, 117, 288–302. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, M.L.; Beaudry, F.; Vachon, P. Intrathecal [6]-gingerol administration alleviates peripherally induced neuropathic pain in male sprague–dawley rats. Phytother. Res. 2013, 27, 1251–1254. [Google Scholar] [CrossRef] [PubMed]
- Fajrin, F.A.; Nugroho, A.E.; Nurrochmad, A.; Susilowati, R. Ginger extract and its compound, 6-shogaol, attenuates painful diabetic neuropathy in mice via reducing trpv1 and nmdar2b expressions in the spinal cord. J. Ethnopharmacol. 2020, 249, 112396. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.; Darcsi, A.; Kéry, Á.; Riethmüller, E. Blood-brain barrier permeability study of ginger constituents. J. Pharm. Biomed. Anal. 2020, 177, 112820. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Wang, N.S.; Mi, S.Q. Plasma pharmacokinetics and tissue distribution of [6]-gingerol in rats. Biopharm. Drug Dispos. 2008, 29, 529–537. [Google Scholar] [CrossRef]
- Kia, H.K.; Miquel, M.-C.; McKernan, R.M.; Laporte, A.-M.; Lombard, M.-C.; Bourgoin, S.; Hamon, M.; Vergé, D. Localization of 5-ht3 receptors in the rat spinal cord: Immunohistochemistry and in situ hybridization. Neuroreport 1995, 6, 257–261. [Google Scholar] [CrossRef]
- Serra, J.; Solà, R.; Quiles, C.; Casanova-Molla, J.; Pascual, V.; Bostock, H.; Valls-Solé, J. C-nociceptors sensitized to cold in a patient with small-fiber neuropathy and cold allodynia. Pain 2009, 147, 46–53. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Wei, Y.-Y.; Huang, J.; Wang, W.; Tamamaki, N.; Li, Y.-Q.; Wu, S.-X. Expression patterns of 5-ht receptor subtypes 1a and 2a on gabaergic neurons within the spinal dorsal horn of gad67-gfp knock-in mice. J. Chem. Neuroanat. 2009, 38, 75–81. [Google Scholar] [CrossRef]
- Marlier, L.; Teilhac, J.-R.; Cerruti, C.; Privat, A. Autoradiographic mapping of 5-ht1, 5-ht1a, 5-ht1b and 5-ht2 receptors in the rat spinal cord. Brain Res. 1991, 550, 15–23. [Google Scholar] [CrossRef]
- Nadeson, R.; Goodchild, C. Antinociceptive role of 5-ht1a receptors in rat spinal cord. Br. J. Anaesth. 2002, 88, 679–684. [Google Scholar] [CrossRef]
- Bardin, L.; Tarayre, J.-P.; Koek, W.; Colpaert, F.C. In the formalin model of tonic nociceptive pain, 8-oh-dpat produces 5-ht1a receptor-mediated, behaviorally specific analgesia. Eur. J. Pharmacol. 2001, 421, 109–114. [Google Scholar] [CrossRef]
- Van Steenwinckel, J.; Noghero, A.; Thibault, K.; Brisorgueil, M.-J.; Fischer, J.; Conrath, M. The 5-ht2a receptor is mainly expressed in nociceptive sensory neurons in rat lumbar dorsal root ganglia. Neuroscience 2009, 161, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Thibault, K.; Van Steenwinckel, J.; Brisorgueil, M.-J.; Fischer, J.; Hamon, M.; Calvino, B.; Conrath, M. Serotonin 5-ht2a receptor involvement and fos expression at the spinal level in vincristine-induced neuropathy in the rat. Pain 2008, 140, 305–322. [Google Scholar] [CrossRef] [PubMed]
- Nievergelt, A.; Huonker, P.; Schoop, R.; Altmann, K.-H.; Gertsch, J. Identification of serotonin 5-ht1a receptor partial agonists in ginger. Bioorg. Med. Chem. 2010, 18, 3345–3351. [Google Scholar] [CrossRef]
- Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar] [CrossRef]
- Dixon, W.J. Efficient analysis of experimental observations. Annu. Rev. Pharmacol. Toxicol. 1980, 20, 441–462. [Google Scholar] [CrossRef]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1994, 53, 55–63. [Google Scholar] [CrossRef]
- You, H.; Ireland, B.; Moeszinger, M.; Zhang, H.; Snow, L.; Krepich, S.; Takagawa, V. Determination of bioactive nonvolatile ginger constituents in dietary supplements by a rapid and economic hplc method: Analytical method development and single-laboratory validation. Talanta 2019, 194, 795–802. [Google Scholar] [CrossRef]




| Type | Sequence |
|---|---|
| GAPDH (Forward) | 5′-GGAGGTAGCTCCTGATTCGC-3′ |
| GAPDH (Reverse) | 5′-CACATTGGGGGTAGGAACAC-3′ |
| HTR1A (Forward) | 5′-TACTCCACTTTCGGCGCTTT-3′ |
| HTR1A (Reverse) | 5′-GGAGGTAGCTCCTGATTCGC-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.H.; Min, D.; Lee, D.; Kim, W. Zingiber officinale Roscoe Rhizomes Attenuate Oxaliplatin-Induced Neuropathic Pain in Mice. Molecules 2021, 26, 548. https://doi.org/10.3390/molecules26030548
Lee JH, Min D, Lee D, Kim W. Zingiber officinale Roscoe Rhizomes Attenuate Oxaliplatin-Induced Neuropathic Pain in Mice. Molecules. 2021; 26(3):548. https://doi.org/10.3390/molecules26030548
Chicago/Turabian StyleLee, Ji Hwan, Daeun Min, Donghun Lee, and Woojin Kim. 2021. "Zingiber officinale Roscoe Rhizomes Attenuate Oxaliplatin-Induced Neuropathic Pain in Mice" Molecules 26, no. 3: 548. https://doi.org/10.3390/molecules26030548
APA StyleLee, J. H., Min, D., Lee, D., & Kim, W. (2021). Zingiber officinale Roscoe Rhizomes Attenuate Oxaliplatin-Induced Neuropathic Pain in Mice. Molecules, 26(3), 548. https://doi.org/10.3390/molecules26030548







