Synthesis and Characterization of Bone Binding Antibiotic-1 (BBA-1), a Novel Antimicrobial for Orthopedic Applications
Abstract
:1. Introduction
2. Results
2.1. Identification of Synthesized BBA-1 Using HPLC
2.2. Characterization of BBA-1 Using Infra-Red Spectroscopy
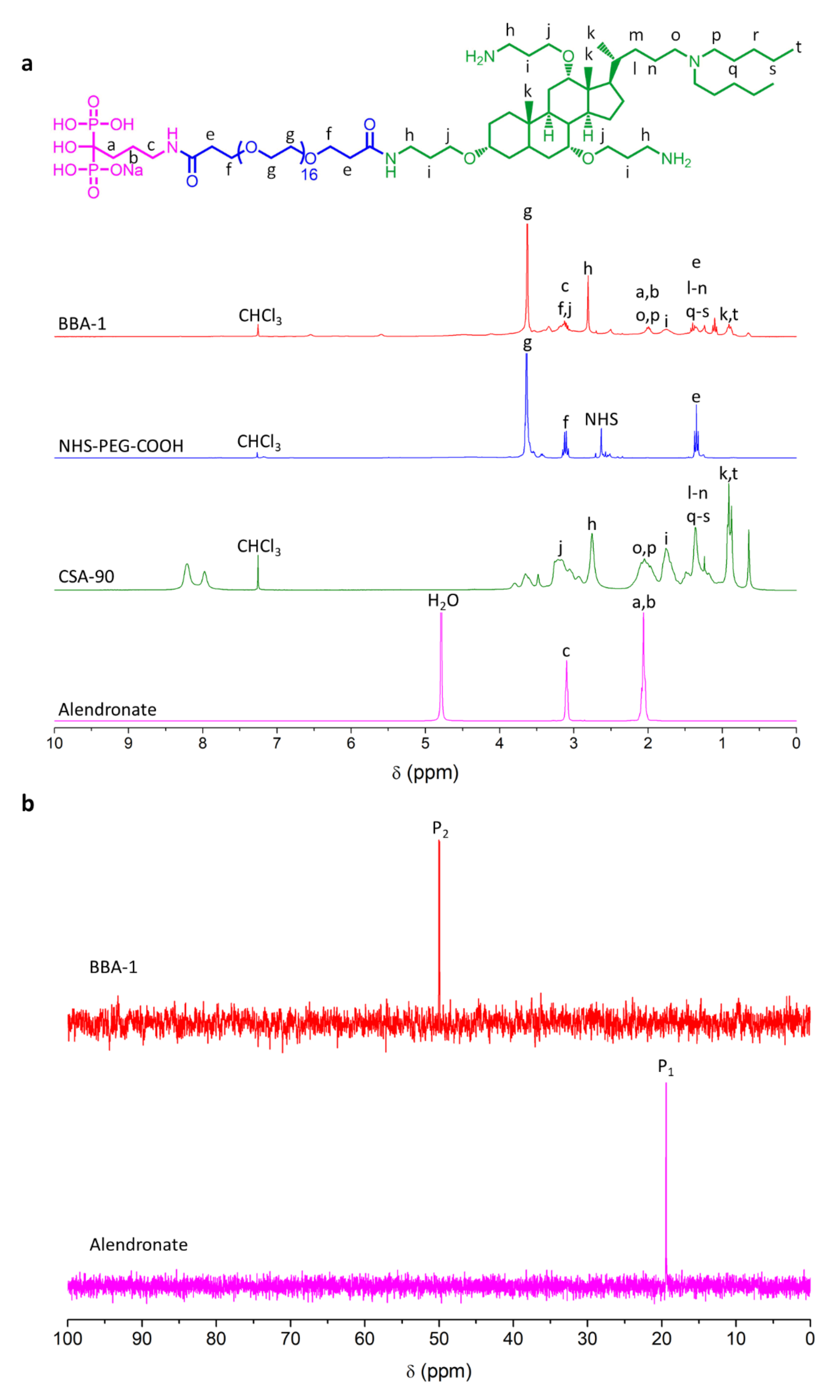
2.3. Characterization of BBA-1 by NMR Spectroscopy
2.4. Confirmation of Synthesized BBA-1 by Mass Spectrometry (MS)
2.5. BBA-1 Binding to Hydroxyapatite
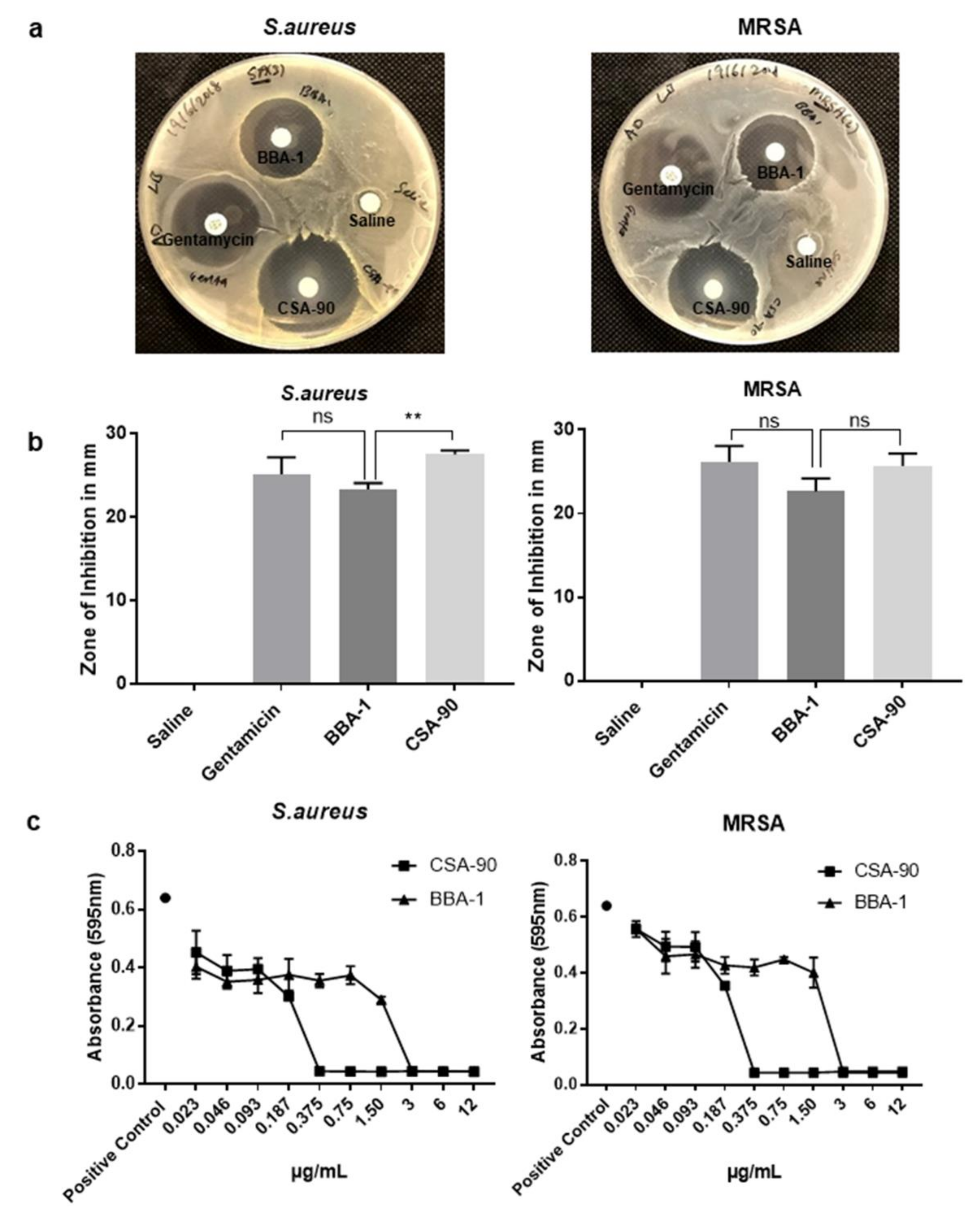
2.6. BBA-1 Has Antimicrobial Activity against Staphylococcus aureus
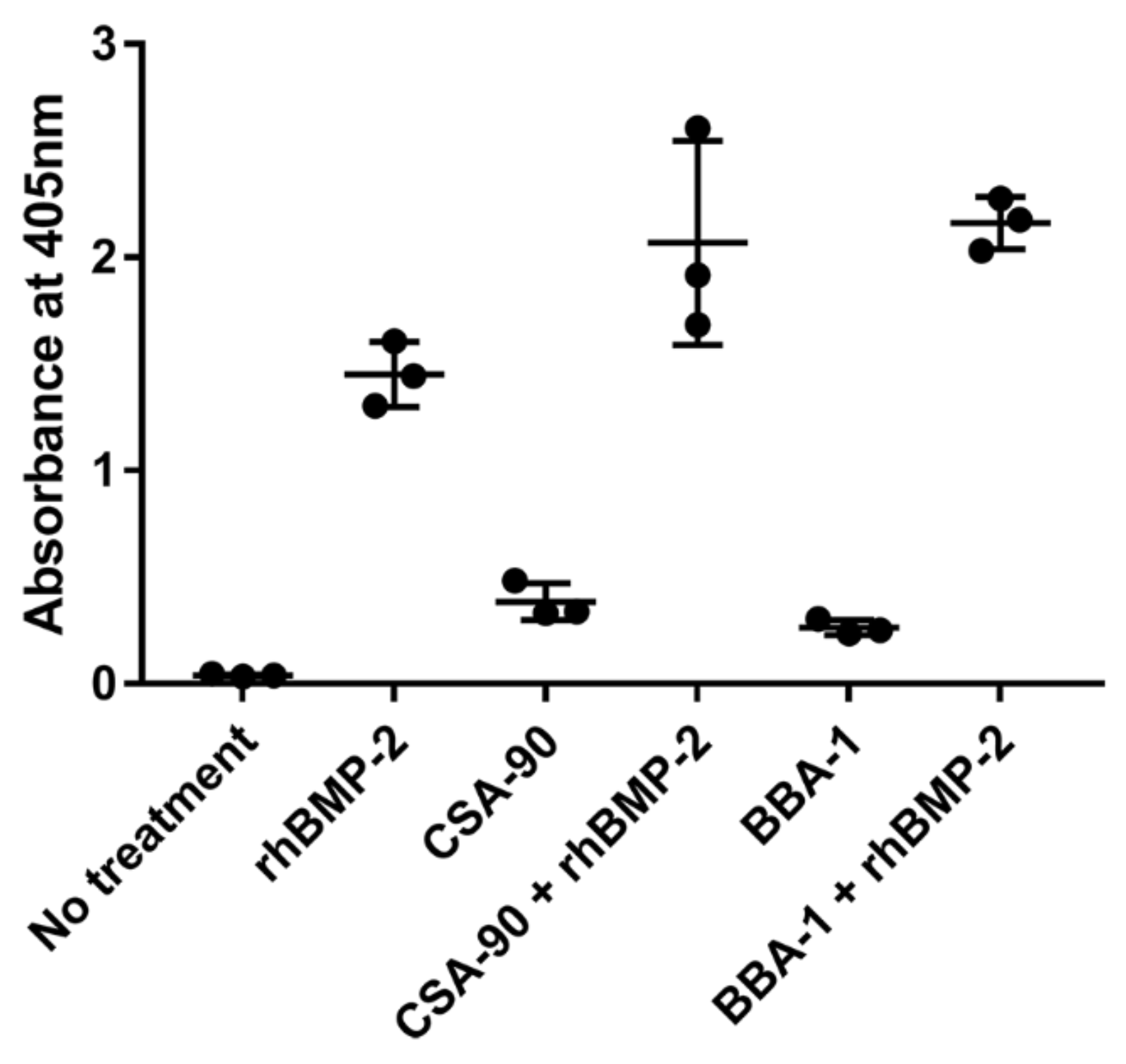
2.7. BBA-1 Promotes Osteogenic Differentiation in Cultured Osteoblast
2.8. BBA-1 Does Not Impair Protein Prenylation
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Synthesis of ALN-PEG-COOH
4.3. Conjugation of CSA-90 to the Carboxylic Terminal of ALN-PEG-COOH
4.4. Identification and Characterization of ALN-PEG-CSA-90 Conjugate (BBA-1)
4.5. In Vitro Bone Mineral Affinity Assay
4.6. Antibacterial Activity of BBA-1
4.7. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
4.8. Cell Culture and Osteogenesis Assays
4.9. Protein Prenylation Assays
4.10. Statistics
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Klenerman, L. A history of osteomyelitis from the Journal of Bone and Joint Surgery. J. Bone Jt. Surg. Br. 2007, 89-B, 667–670. [Google Scholar] [CrossRef] [Green Version]
- Conterno, L.O.; Turchi, M.D. Antibiotics for treating chronic osteomyelitis in adults. Cochrane Database Syst. Rev. 2013, Cd004439. [Google Scholar] [CrossRef]
- Mathews, J.A.; Ward, J.; Chapman, T.W.; Khan, U.M.; Kelly, M.B. Single-stage orthoplastic reconstruction of Gustilo-Anderson Grade III open tibial fractures greatly reduces infection rates. Injury 2015, 46, 2263–2266. [Google Scholar] [CrossRef]
- Jorge, L.S.; Chueire, A.G.; Rossit, A.R.B. Osteomyelitis: A current challenge. Braz. J. Infect. Dis. 2010, 14, 310–315. [Google Scholar] [CrossRef] [Green Version]
- Malizos, K.N. Global Forum: The Burden of Bone and Joint Infections: A Growing Demand for More Resources. J. Bone Jt. Surg. Am. 2017, 99, e20. [Google Scholar] [CrossRef]
- Birt, M.C.; Anderson, D.W.; Bruce Toby, E.; Wang, J. Osteomyelitis: Recent advances in pathophysiology and therapeutic strategies. J. Orthop. 2017, 14, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Waldvogel, F.A.; Papageorgiou, P.S. Osteomyelitis: The past decade. N. Engl. J. Med. 1980, 303, 360–370. [Google Scholar] [CrossRef] [PubMed]
- De Graeff, J.J.; Paulino Pereira, N.R.; van Wulfften Palthe, O.D.; Nelson, S.B.; Schwab, J.H. Prognostic Factors for Failure of Antibiotic Treatment in Patients with Osteomyelitis of the Spine. Spine 2017, 42, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.Z.; Feng, Y.; Pollard, J.; Chin, J.N.; Rybak, M.J.; Bucki, R.; Epand, R.F.; Epand, R.M.; Savage, P.B. Ceragenins: Cholic acid-based mimics of antimicrobial peptides. Acc. Chem. Res. 2008, 41, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Savage, P.B.; Li, C.; Taotafa, U.; Ding, B.; Guan, Q. Antibacterial properties of cationic steroid antibiotics. FEMS Microbiol. Lett. 2002, 217, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mills, R.; Cheng, T.L.; Mikulec, K.; Peacock, L.; Isaacs, D.; Genberg, C.; Savage, P.B.; Little, D.G.; Schindeler, A. CSA-90 Promotes Bone Formation and Mitigates Methicillin-resistant Staphylococcus aureus Infection in a Rat Open Fracture Model. Clin. Orthop. Relat. Res. 2018, 476, 1311–1323. [Google Scholar] [CrossRef]
- Schindeler, A.; Yu, N.Y.C.; Cheng, T.L.; Sullivan, K.; Mikulec, K.; Peacock, L.; Matthews, R.; Little, D.G. Local Delivery of the Cationic Steroid Antibiotic CSA-90 Enables Osseous Union in a Rat Open Fracture Model of Staphylococcus aureus Infection. J. Bone Jt. Surg. Am. 2015, 97A, 302–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reyes, C.; Hitz, M.; Prieto-Alhambra, D.; Abrahamsen, B. Risks and Benefits of Bisphosphonate Therapies. J. Cell. Biochem. 2016, 117, 20–28. [Google Scholar] [CrossRef]
- Young, R.N.; Grynpas, M.D. Targeting therapeutics to bone by conjugation with bisphosphonates. Curr. Opin. Pharmacol. 2018, 40, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Russell, R.G.G.; Watts, N.B.; Ebetino, F.H.; Rogers, M.J. Mechanisms of action of bisphosphonates: Similarities and differences and their potential influence on clinical efficacy. Osteoporos. Int. 2008, 19, 733–759. [Google Scholar] [CrossRef]
- Roelofs, A.J.; Thompson, K.; Gordon, S.; Rogers, M.J. Molecular mechanisms of action of bisphosphonates: Current status. Clin. Cancer Res. 2006, 12, 6222S–6230S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farrell, K.B.; Karpeisky, A.; Thamm, D.H.; Zinnen, S. Bisphosphonate conjugation for bone specific drug targeting. Bone Rep. 2018, 9, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.S.; Aher, N.; Patil, R.; Khandare, J. Poly(ethylene glycol)-Prodrug Conjugates: Concept, Design, and Applications. J. Drug Deliv. 2012, 2012, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, N.; Jurczyluk, J.; Shay, G.; Tnimov, Z.; Alexandrov, K.; Munoz, M.A.; Skinner, O.P.; Pavlos, N.J.; Rogers, M.J. A highly sensitive prenylation assay reveals in vivo effects of bisphosphonate drug on the Rab prenylome of macrophages outside the skeleton. Small GTPases 2015, 6, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.L.; Zhao, Y.-P.; Li, H.-Q.; Na, R.; Li, F.; Mei, Q.-B.; Zhao, M.-G.; Zhou, S.-Y. Doxorubicin-poly (ethylene glycol)-alendronate self-assembled micelles for targeted therapy of bone metastatic cancer. Sci. Rep. 2015, 5, 14614. [Google Scholar] [CrossRef] [PubMed]
- Hata, K.; Tokuhiro, H.; Nakatsuka, K.; Miki, T.; Nishizawa, Y.; Morii, H.; Miura, M. Measurement of bone-specific alkaline phosphatase by an immunoselective enzyme assay method. Ann. Clin. Biochem. 1996, 33 Pt 2, 127–131. [Google Scholar] [CrossRef]
- Dunford, J.E.; Thompson, K.; Coxon, F.P.; Luckman, S.P.; Hahn, F.M.; Poulter, C.D.; Ebetino, F.H.; Rogers, M.J. Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J. Pharmacol. Exp. Ther. 2001, 296, 235–242. [Google Scholar]
- Epand, R.F.; Pollard, J.E.; Wright, J.O.; Savage, P.B.; Epand, R.M. Depolarization, Bacterial Membrane Composition, and the Antimicrobial Action of Ceragenins. Antimicrob. Agents Chemother. 2010, 54, 3708–3713. [Google Scholar] [CrossRef] [Green Version]
- Niemirowicz, K.; Surel, U.; Wilczewska, A.Z.; Mystkowska, J.; Piktel, E.; Gu, X.; Namiot, Z.; Akowska, A.K.L.; Savage, P.B.; Bucki, R. Bactericidal activity and biocompatibility of ceragenin-coated magnetic nanoparticles. J. Nanobiotechnol. 2015, 13, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gdowski, A.S.; Ranjan, A.; Sarker, M.R.; Vishwanatha, J.K. Bone-targeted cabazitaxel nanoparticles for metastatic prostate cancer skeletal lesions and pain. Nanomedicine 2017, 12, 2083–2095. [Google Scholar] [CrossRef]
- Li, B.; Webster, T.J. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2018, 36, 22–32. [Google Scholar] [CrossRef] [Green Version]
- Konigsberg, B.S.; Valle, C.J.D.; Ting, N.T.; Qiu, F.; Sporer, S.M. Acute Hematogenous Infection Following Total Hip and Knee Arthroplasty. J. Arthroplast. 2014, 29, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Sedghizadeh, P.P.; Sun, S.; Junka, A.F.; Richard, E.; Sadrerafi, K.; Mahabady, S.; Bakhshalian, N.; Tjokro, N.; Bartoszewicz, M.; Oleksy, M.; et al. Design, Synthesis, and Antimicrobial Evaluation of a Novel Bone-Targeting Bisphosphonate-Ciprofloxacin Conjugate for the Treatment of Osteomyelitis Biofilms. J. Med. Chem. 2017, 60, 2326–2343. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.S.E.; Houghton, T.J.; Kang, T.; Dietrich, E.; Delorme, D.; Ferreira, S.S.; Caron, L.; Viens, F.; Arhin, F.F.; Sarmiento, I.; et al. Bisphosphonated fluoroquinolone esters as osteotropic prodrugs for the prevention of osteomyelitis. Bioorg. Med. Chem. 2008, 16, 9217–9229. [Google Scholar] [CrossRef]
- Lewis Phillips, G.D.; Li, G.; Dugger, D.L.; Crocker, L.M.; Parsons, K.L.; Mai, E.; Blattler, W.A.; Lambert, J.M.; Chari, R.V.J.; Lutz, R.J.; et al. Targeting HER2-Positive Breast Cancer with Trastuzumab-DM1, an Antibody-Cytotoxic Drug Conjugate. Cancer Res. 2008, 68, 9280–9290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCombs, J.R.; Owen, S.C. Antibody Drug Conjugates: Design and Selection of Linker, Payload and Conjugation Chemistry. AAPS J. 2015, 17, 339–351. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, M.; Monteiro, F.J.; Ferraz, M.P. Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomatter 2012, 2, 176–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houghton, T.J.; Tanaka, K.S.E.; Kang, T.; Dietrich, E.; Lafontaine, Y.; Delorme, D.; Ferreira, S.S.; Viens, F.; Arhin, F.F.; Sarmiento, I.; et al. Linking Bisphosphonates to the Free Amino Groups in Fluoroquinolones: Preparation of Osteotropic Prodrugs for the Prevention of Osteomyelitis. J. Med. Chem. 2008, 51, 6955–6969. [Google Scholar] [CrossRef]
- Kuroda, K.; Fukuda, T.; Okumura, K.; Yoneyama, H.; Isogai, H.; Savage, P.B.; Isogai, E. Ceragenin CSA-13 induces cell cycle arrest and antiproliferative effects in wild-type and p53 null mutant HCT116 colon cancer cells. Anticancer Drugs 2013, 24, 826–834. [Google Scholar] [CrossRef] [PubMed]
- Van Bambeke, F.; Mingeot-Leclercq, M.P.; Struelens, M.J.; Tulkens, P.M. The bacterial envelope as a target for novel anti-MRSA antibiotics. Trends Pharmacol. Sci. 2008, 29, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Schindeler, A.; Morse, A.; Peacock, L.; Mikulec, K.; Yu, N.Y.C.; Liu, R.; Kijumnuayporn, S.; McDonald, M.M.; Baldock, P.A.; Ruys, A.J.; et al. Rapid cell culture and pre-clinical screening of a transforming growth factor-beta (TGF-beta) inhibitor for orthopaedics. BMC Musculoskelet. Disord. 2010, 11, 105. [Google Scholar] [CrossRef] [PubMed] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamble, S.; Valtchev, P.; Dao, A.; Pelras, T.; Rogers, M.J.; Savage, P.B.; Dehghani, F.; Schindeler, A. Synthesis and Characterization of Bone Binding Antibiotic-1 (BBA-1), a Novel Antimicrobial for Orthopedic Applications. Molecules 2021, 26, 1541. https://doi.org/10.3390/molecules26061541
Kamble S, Valtchev P, Dao A, Pelras T, Rogers MJ, Savage PB, Dehghani F, Schindeler A. Synthesis and Characterization of Bone Binding Antibiotic-1 (BBA-1), a Novel Antimicrobial for Orthopedic Applications. Molecules. 2021; 26(6):1541. https://doi.org/10.3390/molecules26061541
Chicago/Turabian StyleKamble, Sumedh, Peter Valtchev, Aiken Dao, Théophile Pelras, Michael J Rogers, Paul B. Savage, Fariba Dehghani, and Aaron Schindeler. 2021. "Synthesis and Characterization of Bone Binding Antibiotic-1 (BBA-1), a Novel Antimicrobial for Orthopedic Applications" Molecules 26, no. 6: 1541. https://doi.org/10.3390/molecules26061541







