Comparative Analysis of Polycyclic Aromatic Hydrocarbons and Halogenated Polycyclic Aromatic Hydrocarbons in Different Parts of Perilla frutescens (L.) Britt.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Method Optimization and Validation
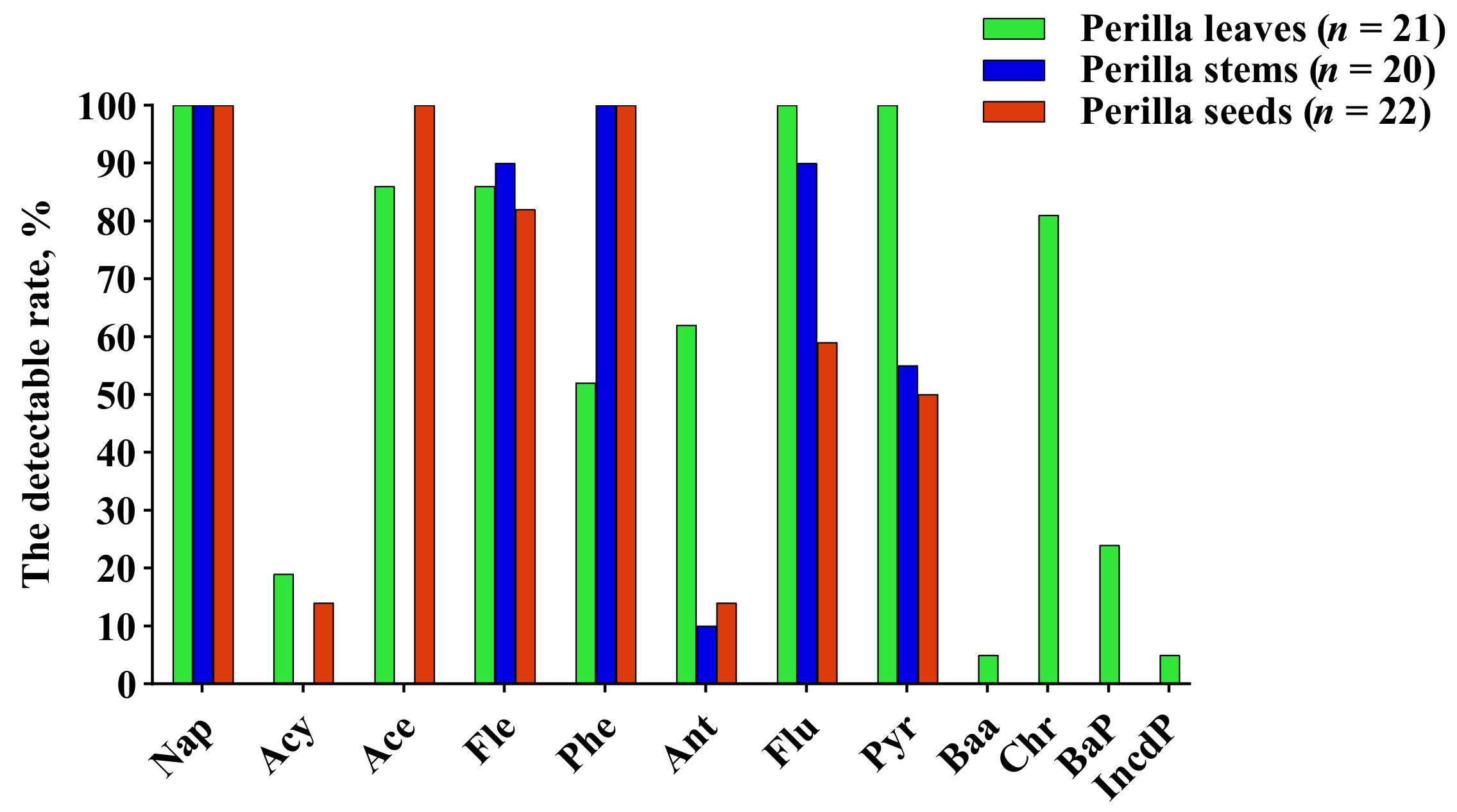
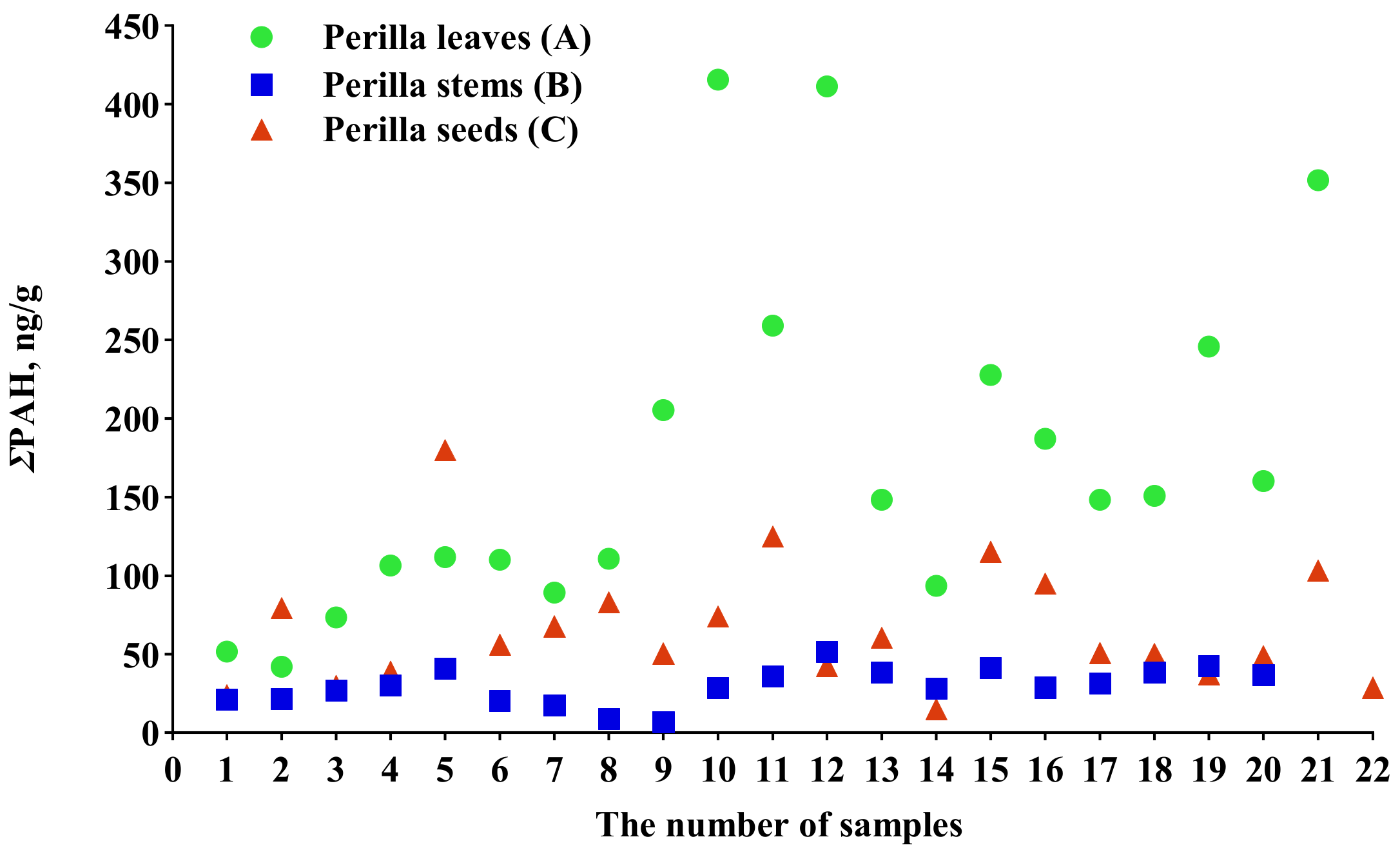
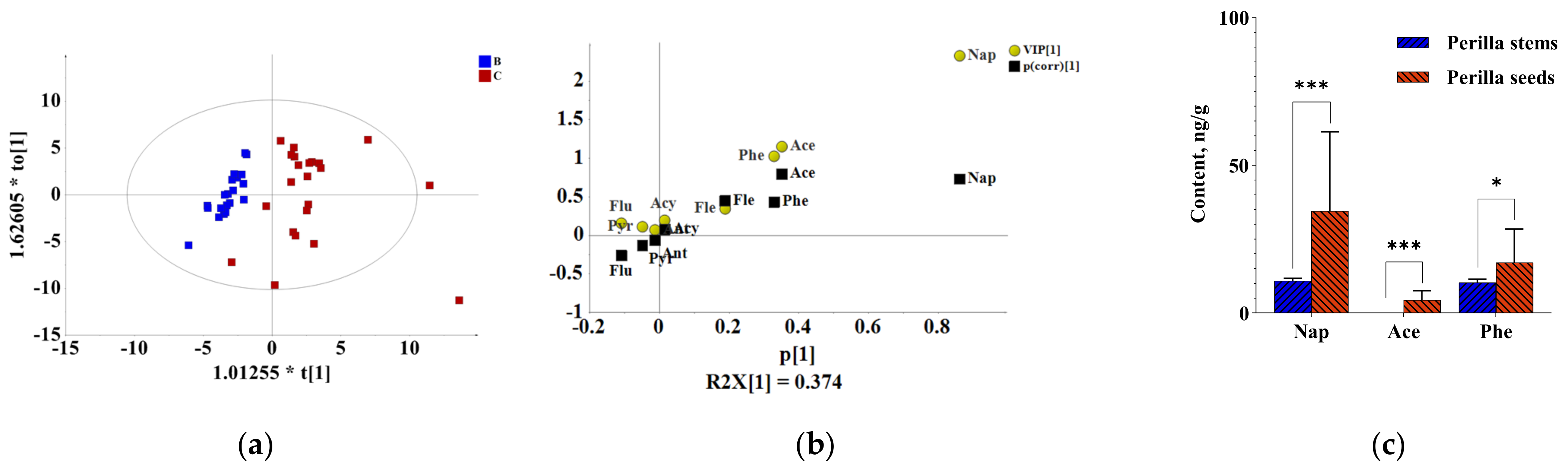
2.2. Comparative Analysis
2.3. Health Risk Assessment
3. Materials and Methods
3.1. Materials and Reagents
3.2. GC-MS Conditions
3.3. Sample Preparation
3.3.1. The Preparation of Perilla Leaves and Stems
3.3.2. The Preparation of Perilla Seeds
3.4. Method Verification
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Nitta, M.; Lee, J.K.; Ohnishi, O. Asian Perilla crops and their weedy forms: Their cultivation, utilization and genetic relationships. Econ. Bot. 2003, 57, 245–253. [Google Scholar] [CrossRef]
- Nitta, M.; Lee, J.K.; Kang, C.W.; Katsuta, M.; Yasumoto, S.; Liu, D.; Nagamine, T.; Ohnishi, O. The Distribution of Perilla Species. Genet. Resour. Crop Evol. 2005, 52, 797–804. [Google Scholar] [CrossRef]
- Ngan, T.T.K.; Toan, T.Q.; Cang, M.H. Evaluation of the Physical and Chemical Properties of Vietnamese Perilla frutescens L. Essential Oil. Asian J. Chem. 2020, 32, 1463–1466. [Google Scholar] [CrossRef]
- Kapoor, B.; Kapoor, D.; Gautam, S.; Singh, R.; Bhardwaj, S. Dietary Polyunsaturated Fatty Acids (PUFAs): Uses and Potential Health Benefits. Curr. Nutr. Rep. 2021, 10, 232–242. [Google Scholar] [CrossRef]
- Perić, A.; Čupić, M.B.; Vuković, M.K.; Dragović, V.G.; Nešić, V.; Lekić, N. Efficacy of a food supplement Lertal® as an adjuvant therapy of patients with moderate-to-severe seasonal allergic rhinoconjunctivitis. Egypt. J. Otolaryngol. 2021, 37, 54. [Google Scholar] [CrossRef]
- Ji, W.-W.; Li, R.-P.; Li, M.; Wang, S.-Y.; Zhang, X.; Niu, X.-X.; Li, W.; Yan, L.; Wang, Y.; Fu, Q.; et al. Antidepressant-like effect of essential oil of Perilla frutescens in a chronic, unpredictable, mild stress-induced depression model mice. Chin. J. Nat. Med. 2014, 12, 753–759. [Google Scholar] [CrossRef]
- Banno, N.; Akihisa, T.; Tokuda, H.; Yasukawa, K.; Higashihara, H.; Ukiya, M.; Watanabe, K.; Kimura, Y.; Hasegawa, J.; Nishino, H. Triterpene acids from the leaves of Perilla frutescens and their anti-inflammatory and antitumor-promoting effects. Biosci. Biotechnol. Biochem. 2004, 68, 85–90. [Google Scholar] [CrossRef] [Green Version]
- Feng, L.-J.; Yu, C.-H.; Ying, K.-J.; Hua, J.; Dai, X.-Y. Hypolipidemic and antioxidant effects of total flavonoids of Perilla Frutescens leaves in hyperlipidemia rats induced by high-fat diet. Food Res. Int. 2011, 44, 404–409. [Google Scholar] [CrossRef]
- Liu, J.; Wan, Y.; Zhao, Z.; Chen, H. Determination of the content of rosmarinic acid by HPLC and analytical comparison of volatile constituents by GC-MS in different parts of Perilla frutescens (L.) Britt. Chem. Cent. J. 2013, 7, 61. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.; Qiu, J.-F.; Ma, L.-J.; Hu, Y.-J.; Li, P.; Wan, J.-B. Phytochemical and phytopharmacological review of Perilla frutescens L. (Labiatae), a traditional edible-medicinal herb in China. Food Chem. Toxicol. 2017, 108, 375–391. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2010; Volume 92. [Google Scholar]
- Liu, Q.; Xu, X.; Wang, L.; Wang, D. Transformation reactivity of parent polycyclic aromatic hydrocarbons and the formation trend of halogenated polycyclic aromatic hydrocarbons in the presence of bromide ion during chlorination. Chem. Eng. J. 2020, 400, 125901. [Google Scholar] [CrossRef]
- Ohura, T.; Morita, M.; Kuruto-Niwa, R.; Amagai, T.; Sakakibara, H.; Shimoi, K. Differential action of chlorinated polycyclic aromatic hydrocarbons on aryl hydrocarbon receptor-mediated signaling in breast cancer cells. Environ. Toxicol. 2010, 25, 180–187. [Google Scholar] [CrossRef]
- Huang, C.; Xu, X.; Wang, D.; Ma, M.; Rao, K.; Wang, Z. The aryl hydrocarbon receptor (AhR) activity and DNA-damaging effects of chlorinated polycyclic aromatic hydrocarbons (Cl-PAHs). Chemosphere 2018, 211, 640–647. [Google Scholar] [CrossRef]
- Yuan, K.; Qing, Q.; Wang, Y.; Lin, F.; Chen, B.; Luan, T.; Wang, X. Characteristics of chlorinated and brominated polycyclic aromatic hydrocarbons in the Pearl River Estuary. Sci. Total Environ. 2020, 739, 139774. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, X.; Wang, L.; Lin, L.; Wang, D. Simultaneous determination of forty-two parent and halogenated polycyclic aromatic hydrocarbons using solid-phase extraction combined with gas chromatography-mass spectrometry in drinking water. Ecotoxicol. Environ. Saf. 2019, 181, 241–247. [Google Scholar] [CrossRef]
- Kamiya, Y.; Ikemori, F.; Ohura, T. Optimisation of pre-treatment and ionisation for GC/MS analysis for the determination of chlorinated PAHs in atmospheric particulate samples. Int. J. Environ. Anal. Chem. 2015, 95, 1157–1168. [Google Scholar] [CrossRef]
- Saitta, M.; Di Bella, G.; Fede, M.R.; Lo Turco, V.; Potorti, A.G.; Rando, R.; Russo, M.T.; Dugo, G. Gas chromatography-tandem mass spectrometry multi-residual analysis of contaminants in Italian honey samples. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2017, 34, 800–808. [Google Scholar] [CrossRef]
- Yan, K.; Wu, S.; Gong, G.; Xin, L.; Ge, Y. Simultaneous Determination of Typical Chlorinated, Oxygenated, and European Union Priority Polycyclic Aromatic Hydrocarbons in Milk Samples and Milk Powders. J. Agric. Food Chem. 2021, 69, 3923–3931. [Google Scholar] [CrossRef]
- Ni, H.G.; Guo, J.Y. Parent and halogenated polycyclic aromatic hydrocarbons in seafood from South China and implications for human exposure. J. Agric. Food Chem. 2013, 61, 2013–2018. [Google Scholar] [CrossRef]
- Ishizaki, A.; Sito, K.; Kataoka, H. Analysis of contaminant polycyclic aromatic hydrocarbons in tea products and crude drugs. Anal. Methods 2011, 3, 299–305. [Google Scholar] [CrossRef]
- Yan, Z.; Yuan, J.; Zhu, G.; Zou, Y.; Chen, C.; Yang, S.; Yao, S. A new strategy based on cholesterol-functionalized iron oxide magnetic nanoparticles for determination of polycyclic aromatic hydrocarbons by high-performance liquid chromatography with cholesterol column. Anal. Chim. Acta 2013, 780, 28–35. [Google Scholar] [CrossRef]
- Yu, B.; Zhang, D.; Tan, L.-H.; Zhao, S.-P.; Wang, J.-W.; Yao, L.; Cao, W.-G. Polycyclic aromatic hydrocarbons in traditional Chinese medicines: An analytical method based on different medicinal parts, levels, distribution, and sources. RSC Adv. 2017, 7, 4671–4680. [Google Scholar] [CrossRef] [Green Version]
- Kipopoulou, A.M.; Manoli, E.; Samara, C. Bioconcentration of polycyclic aromatic hydrocarbons in vegetables grown in an industrial area. Environ. Pollut. 1999, 106, 369–380. [Google Scholar] [CrossRef]
- Lin, D.; Zhu, L.; He, W.; Tu, Y. Tea plant uptake and translocation of polycyclic aromatic hydrocarbons from water and around air. J. Agric. Food Chem. 2006, 54, 3658–3662. [Google Scholar] [CrossRef]
- Fismes, J.; Perrin-Ganier, C.; Empereur-Bissonnet, P.; Morel, J.L. Soil-to-root transfer and translocation of polycyclic aromatic hydrocarbons by vegetables grown on industrial contaminated soils. J. Environ. Qual. 2002, 31, 1649–1656. [Google Scholar] [CrossRef]
- Zhao, B.; Fu, S.; Li, H.; Chen, Z. Chemical Characterization of Chinese Perilla Seed Oil. J. Oleo Sci. 2021, 70, 1575–1583. [Google Scholar] [CrossRef]
- Khodabakhshian, R.; Bayati, M.R.; Emadi, B. An evaluation of IR spectroscopy for authentication of adulterated turmeric powder using pattern recognition. Food Chem. 2021, 364, 130406. [Google Scholar] [CrossRef]
- Worley, B.; Powers, R. PCA as a practical indicator of OPLS-DA model reliability. Curr. Metab. 2016, 4, 97–103. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Song, W.; Tao, W.; Zhang, J.; Zhang, X.; Zhao, J.; Yong, J.; Gao, X.; Guo, L. Identification wild and cultivated licorice by multidimensional analysis. Food Chem. 2021, 339, 128111. [Google Scholar] [CrossRef]
- Nisbet, I.C.T.; Lagoy, P.K. Toxic equivalence factors (TEFs) for polycyclic aromatic hydrocarbons (PAHs). Regul. Toxicol. Pharmacol. 1992, 16, 290–300. [Google Scholar] [CrossRef]
- Phan Thi, L.A.; Ngoc, N.T.; Quynh, N.T.; Thanh, N.V.; Kim, T.T.; Anh, D.H.; Viet, P.H. Polycyclic aromatic hydrocarbons (PAHs) in dry tea leaves and tea infusions in Vietnam: Contamination levels and dietary risk assessment. Environ. Geochem. Health 2020, 42, 2853–2863. [Google Scholar] [CrossRef] [PubMed]
- US EPA. Risk Assessment Guidance for Superfund: Volume III—Part A, Process for Conducting Probabilistic Risk Assessment. 2001. Available online: https://www.epa.gov/risk/risk-assessment-guidance-superfund-rags-volume-iii-part (accessed on 6 April 2022).
- US EPA. Integrated Risk Information System: Benzo[a]pyrene (BaP) (CASRN 50-32-8); Environmental Protection Agency: Washington, DC, USA, 2001. Available online: https://iris.epa.gov/ChemicalLanding/&substance_nmbr=136 (accessed on 6 April 2022).
- World Health Organization (WHO). World Health Statistics 2021. Available online: https://apps.who.int/iris/bitstream/handle/10665/342703/9789240027053-eng.pdf (accessed on 6 April 2022).
- Means, B. Risk-Assessment Guidance for Superfund. Volume 1. Human Health Evaluation Manual. Part A. Interim Report (Final); Environmental Protection Agency: Washington, DC, USA, 1989.
- Zachara, A.; Galkowska, D.; Juszczak, L. Contamination of Tea and Tea Infusion with Polycyclic Aromatic Hydrocarbons. Int. J. Environ. Res. Public Health 2017, 15, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Name | Abbreviation | Rt (min) | Quantitative Ion | Qualitative Ion | Quantitative Internal Standard |
|---|---|---|---|---|---|
| Naphthalene-d8 | Nap-d8 | 12.4 | 136.1 | 137.1, 108.0 | \ |
| Naphthalene | Nap | 12.5 | 128.0 | 127.0, 101.9 | Nap-d8 |
| Phenanthrene-d10 | Ace-d10 | 20.3 | 164.1 | 162.1, 160.1 | \ |
| Acenaphthylene | Acy | 19.6 | 152.0 | 151.0, 76.0, 63.0 | Ace-d10 |
| Acenaphthene | Ace | 20.5 | 154.0 | 152.0, 76.0 | Ace-d10 |
| Fluorene | Fle | 22.8 | 166.0 | 165.0, 139.0 | Ace-d10 |
| Acenaphthene-d10 | Phe-d10 | 26.0 | 188.1 | 160.1, 94.1 | \ |
| Phenanthrene | Phe | 26.1 | 178.1 | 89.1, 152.0 | Phe-d10 |
| Anthracene | Ant | 26.3 | 178.1 | 89.1, 152.0 | Phe-d10 |
| 2-Bromofluorene | 2-BrFle | 27.8 | 165.0 | 244.0, 245.9, 162.9 | Phe-d10 |
| 2,7-Dichlorofluorene | 2,7-Cl2Fle | 29.5 | 199.0 | 234.0, 162.9, 200.9 | Phe-d10 |
| Fluoranthene | Flu | 30.1 | 202.0 | 200.0, 100.9, 88.0 | Phe-d10 |
| Pyrene | Pyr | 31.0 | 202.0 | 100.9, 200.0 | Phe-d10 |
| 9-Bromoanthracene | 9-BrAnt | 31.1 | 258.0 | 177.0, 176.0, 88.0 | Phe-d10 |
| Chrysene-d12 | Chr-d12 | 37.9 | 240.1 | 236.1, 120.1 | \ |
| 9,10-Dibromoanthracene | 9,10-Br2Ant | 37.7 | 336.0 | 176.0, 88.1 | Chr-d12 |
| Benzo(a)anthracene | Baa | 37.9 | 228.0 | 225.9, 114.1 | Chr-d12 |
| Chrysene | Chr | 38.1 | 228.0 | 225.9, 114.1 | Chr-d12 |
| Perylene-d12 | Per-d12 | 48.9 | 264.1 | 260.1, 265.1 | \ |
| Benzo(b)fluoranthene | BbF | 46.0 | 252.0 | 250.1, 126.0, 112.8 | Per-d12 |
| Benzo(k)fluoranthene | BkF | 46.2 | 252.0 | 250.1, 126.0, 112.8 | Per-d12 |
| Benzo(a)pyrene | BaP | 48.4 | 252.0 | 250.0, 126.0 | Per-d12 |
| Indeno(1,2,3-c,d)pyrene | IncdP | 56.0 | 276.0 | 138.0, 277.0 | Per-d12 |
| Dibenzo(a,h)anthracene | DahA | 56.3 | 278.0 | 279.0, 138.8 | Per-d12 |
| Benzo(g,h,i)perylene | BghiP | 57.2 | 276.0 | 137.1, 138.0, 274.0 | Per-d12 |
| NO. | Perilla Leaves (A) | Perilla Stems (B) | Perilla Seeds (C) | |||
|---|---|---|---|---|---|---|
| TEQ | ILCR | TEQ | ILCR | TEQ | ILCR | |
| 1 | 0.05 | 4.05 × 10−8 | 0.02 | 1.66 × 10−8 | 0.02 | 1.88 × 10−8 |
| 2 | 0.04 | 3.30 × 10−8 | 0.02 | 1.68 × 10−8 | 0.08 | 6.24 × 10−8 |
| 3 | 0.17 | 1.32 × 10−7 | 0.03 | 2.13 × 10−8 | 0.03 | 2.34 × 10−8 |
| 4 | 0.18 | 1.45 × 10−7 | 0.03 | 2.36 × 10−8 | 0.04 | 3.06 × 10−8 |
| 5 | 0.22 | 1.70 × 10−7 | 0.04 | 3.21 × 10−8 | 0.18 | 1.41 × 10−7 |
| 6 | 0.19 | 1.53 × 10−7 | 0.02 | 1.60 × 10−8 | 0.06 | 4.42 × 10−8 |
| 7 | 0.09 | 7.01 × 10−8 | 0.02 | 1.36 × 10−8 | 0.07 | 5.31 × 10−8 |
| 8 | 0.30 | 2.38 × 10−7 | 0.01 | 6.82 × 10−9 | 0.08 | 6.51 × 10−8 |
| 9 | 0.38 | 2.97 × 10−7 | 0.01 | 5.52 × 10−9 | 0.05 | 3.97 × 10−8 |
| 10 | 0.67 | 5.29 × 10−7 | 0.03 | 2.23 × 10−8 | 0.07 | 5.81 × 10−8 |
| 11 | 2.28 | 1.79 × 10−6 | 0.04 | 2.80 × 10−8 | 0.12 | 9.81 × 10−8 |
| 12 | 0.64 | 5.02 × 10−7 | 0.07 | 5.50 × 10−8 | 0.04 | 3.35 × 10−8 |
| 13 | 0.25 | 2.00 × 10−7 | 0.04 | 3.02 × 10−8 | 0.06 | 4.75 × 10−8 |
| 14 | 0.09 | 7.35 × 10−8 | 0.03 | 2.21 × 10−8 | 0.02 | 1.20 × 10−8 |
| 15 | 0.36 | 2.84 × 10−7 | 0.04 | 3.24 × 10−8 | 0.14 | 1.11 × 10−7 |
| 16 | 18.18 | 1.43 × 10−5 | 0.03 | 2.25 × 10−8 | 0.12 | 9.15 × 10−8 |
| 17 | 19.22 | 1.51 × 10−5 | 0.03 | 2.47 × 10−8 | 0.05 | 3.98 × 10−8 |
| 18 | 19.15 | 1.50 × 10−5 | 0.06 | 4.46 × 10−8 | 0.05 | 3.94 × 10−8 |
| 19 | 20.37 | 1.60 × 10−5 | 0.04 | 3.32 × 10−8 | 0.04 | 2.93 × 10−8 |
| 20 | 0.29 | 2.29 × 10−7 | 0.04 | 2.90 × 10−8 | 0.05 | 3.83 × 10−8 |
| 21 | 26.89 | 2.11 × 10−5 | - | - | 0.12 | 9.65 × 10−8 |
| 22 | - | - | - | - | 0.03 | 2.25 × 10−8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, P.; Jin, B.; Lian, C.; Guo, K.; Ma, C. Comparative Analysis of Polycyclic Aromatic Hydrocarbons and Halogenated Polycyclic Aromatic Hydrocarbons in Different Parts of Perilla frutescens (L.) Britt. Molecules 2022, 27, 3133. https://doi.org/10.3390/molecules27103133
Wang P, Jin B, Lian C, Guo K, Ma C. Comparative Analysis of Polycyclic Aromatic Hydrocarbons and Halogenated Polycyclic Aromatic Hydrocarbons in Different Parts of Perilla frutescens (L.) Britt. Molecules. 2022; 27(10):3133. https://doi.org/10.3390/molecules27103133
Chicago/Turabian StyleWang, Pengfei, Bo Jin, Chaojie Lian, Kaijing Guo, and Chen Ma. 2022. "Comparative Analysis of Polycyclic Aromatic Hydrocarbons and Halogenated Polycyclic Aromatic Hydrocarbons in Different Parts of Perilla frutescens (L.) Britt." Molecules 27, no. 10: 3133. https://doi.org/10.3390/molecules27103133





