Why Do Ionic Surfactants Significantly Alter the Chemiluminogenic Properties of Acridinium Salt?
Abstract
1. Introduction
2. Results and Discussion
2.1. Critical Micellar Concentrations of Surfactants in CL Systems
2.2. Chemiluminogenic Properties of Acridinium Salt–Surfactant Systems
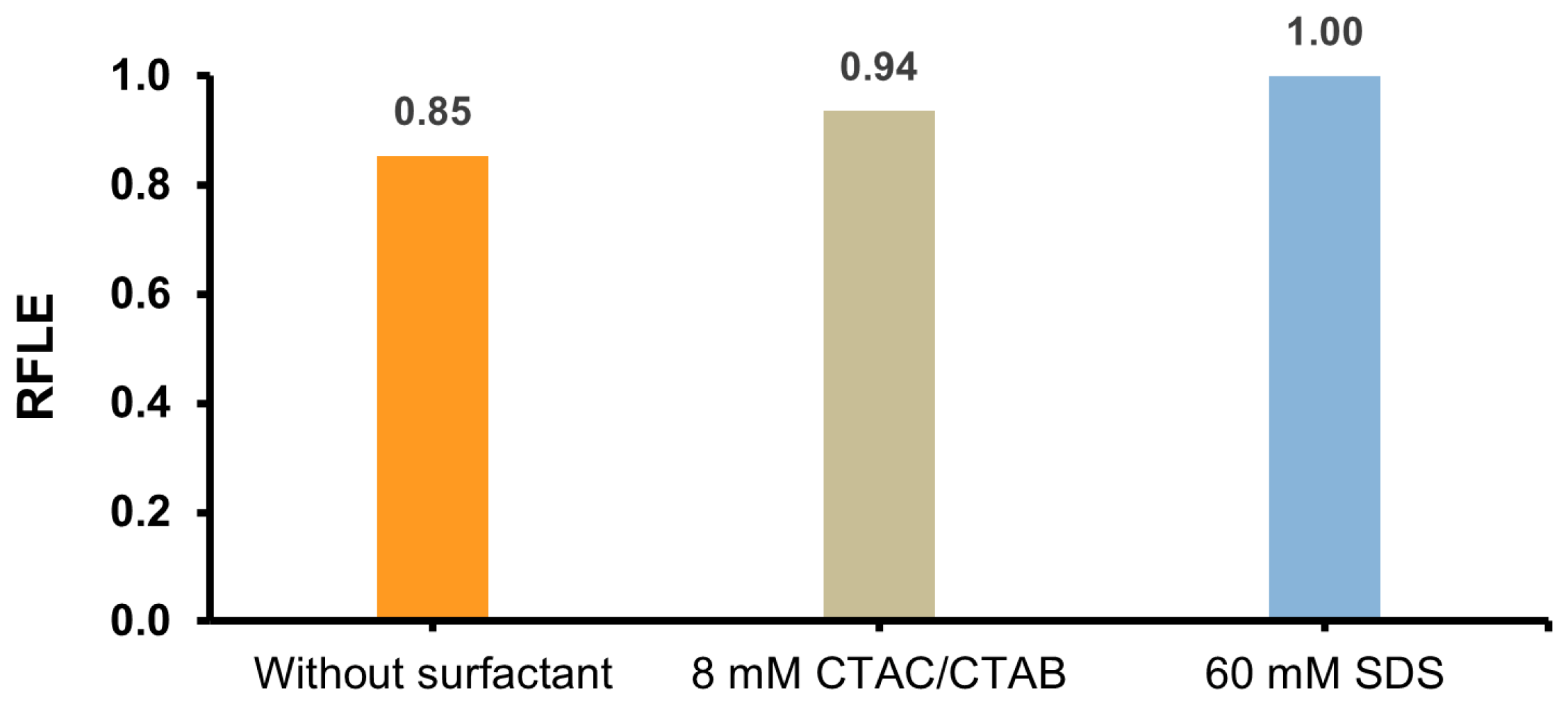
2.3. Ability to Fluorescence of Post-CL Mixtures Containing Surfactants
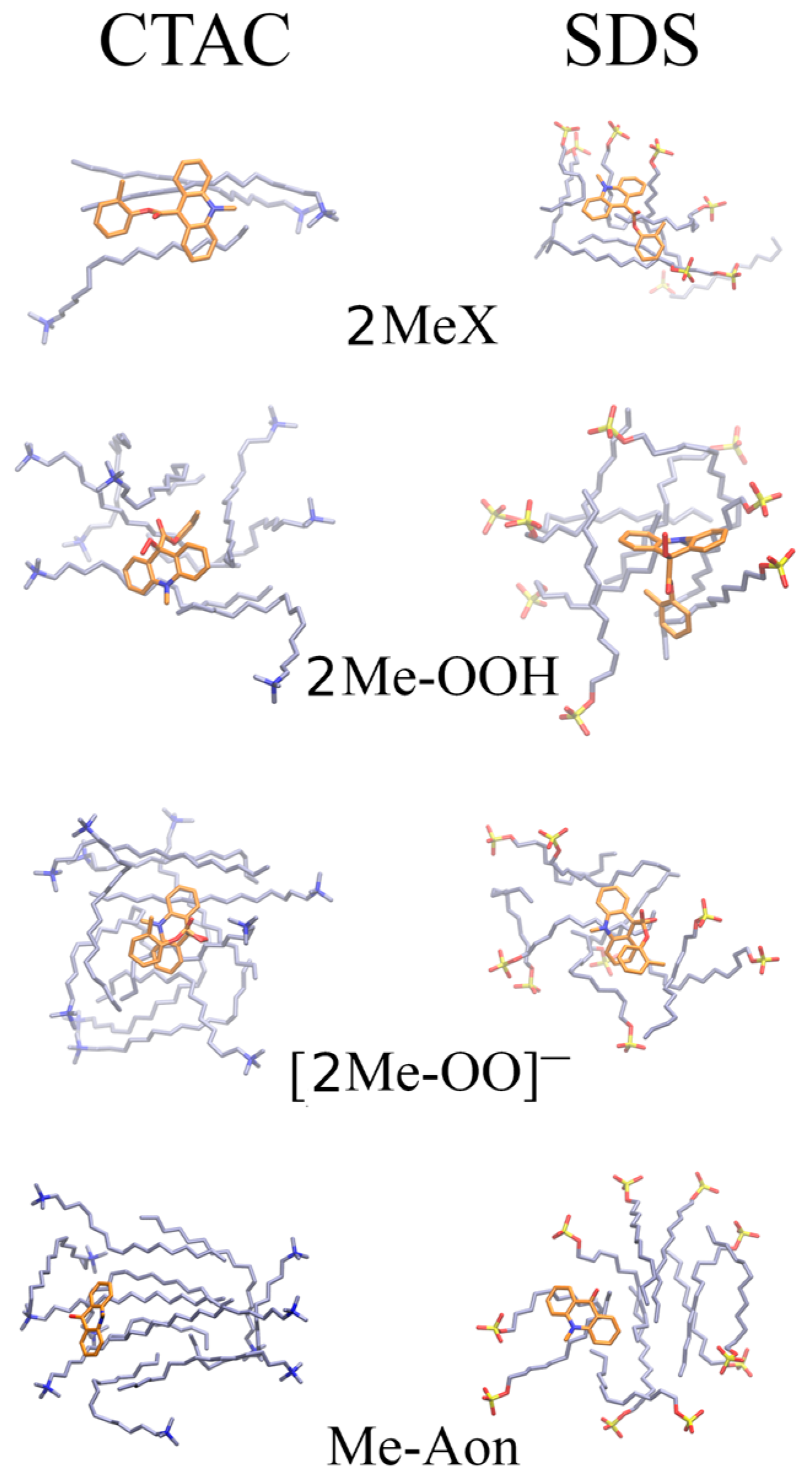
2.4. Molecular Dynamics-Based Analysis
2.5. Possible Side Reaction among Reagents
3. Materials and Methods
3.1. Reagents and Working Solutions
3.2. Conductometric Measurements
3.3. Measurements of Chemiluminescence
3.4. Measurements of Fluorescence
3.5. Molecular Dynamics-Based Analysis
3.6. The DFT Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gundermann, K.D.; McCapra, F. Chemiluminescence in organic chemistry. In Reactivity and Structure: Concepts in Organic Chemistry; Springer: Berlin/Heidelberg, Germany, 1987; pp. 109–118. [Google Scholar]
- Nakazono, M.; Oshikawa, Y.; Nakamura, M.; Kubota, H.; Nanbu, S. Strongly chemiluminescent acridinium esters under neutral conditions: Synthesis, properties, determination, and theoretical Study. J. Org. Chem. 2017, 82, 2450–2461. [Google Scholar] [CrossRef]
- Smith, K.; Mu, X.; Li, Z.; Holland, A.M.; Woodhead, J.S.; El-Hiti, G.A. Synthesis, structure elucidation, and chemiluminescent activity of new 9-substituted 10-(ω-(succinimidyloxycarbonyl)alkyl)acridinium esters. Luminescence 2023, 38, 487–496. [Google Scholar] [CrossRef]
- Krzymiński, K.; Roshal, A.D.; Rudnicki-Velasquez, P.B.; Żamojć, K. On the use of acridinium indicators for the chemiluminescent determination of the total antioxidant capacity of dietary supplements. Luminescence 2019, 34, 512–519. [Google Scholar] [CrossRef]
- Kricka, L.J. Clinical applications of chemiluminescence. Anal. Chim. Acta 2003, 500, 279–286. [Google Scholar] [CrossRef]
- Yang, M.; Huang, J.; Fan, J.; Du, J.; Pu, K.; Peng, X. Chemiluminescence for bioimaging and therapeutics: Recent advances and challenges. Chem. Soc. Rev. 2020, 49, 6800–6815. [Google Scholar] [CrossRef]
- Huertas-Pérez, J.F.; Moreno-González, D.; Airado-Rodríguez, D.; Lara, F.J.; García-Campaña, A.M. Advances in the application of chemiluminescence detection in liquid chromatography. Trends Anal. Chem. 2016, 75, 35–48. [Google Scholar] [CrossRef]
- Giokas, D.L.; Vlessidis, A.G.; Tsogas, G.Z.; Evmiridis, N.P. Nanoparticle-assisted chemiluminescence and its applications in analytical chemistry. Trends Anal. Chem. 2010, 29, 1113–1126. [Google Scholar] [CrossRef]
- Gámiz-Gracia, L.; García-Campaña, A.M.; Huertas-Pérez, J.F.; Lara, F.J. Chemiluminescence detection in liquid chromatography: Applications to clinical, pharmaceutical, environmental and food analysis—A review. Anal. Chim. Acta 2009, 640, 7–28. [Google Scholar] [CrossRef]
- Arakawa, H.; Tsuruoka, K.; Ohno, K.I.; Tajima, N.; Nagano, H. Development of a highly sensitive chemiluminescent assay for hydrogen peroxide under neutral conditions using acridinium ester and its application to an enzyme immunoassay. Luminescence 2014, 29, 374–377. [Google Scholar] [CrossRef]
- Roda, A.; Guardigli, M. Analytical chemiluminescence and bioluminescence: Latest achievements and new horizons. Anal. Bioanal. Chem. 2012, 402, 69–76. [Google Scholar] [CrossRef]
- Richardson, A.P.; Kim, J.B.; Barnard, G.J.; Collins, W.P.; McCapra, F. Chemiluminescence immunoassay of plasma progesterone, with progesterone-acridinium ester used as the labelled antigen. Clin. Chem. 1985, 31, 1664–1668. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Sun, L.; Chu, X. Chemiluminescence immunoassay. Trends Anal. Chem. 2009, 28, 404–415. [Google Scholar] [CrossRef]
- Goryacheva, I.Y.; Lenain, P.; De Saeger, S. Nanosized labels for rapid immunotests. Trends Anal. Chem. 2013, 46, 30–43. [Google Scholar] [CrossRef]
- Zhang, J.; Cheng, P.; Pu, K. Recent advances of molecular optical probes in imaging of β-galactosidase. Bioconjug. Chem. 2019, 30, 2089–2101. [Google Scholar] [CrossRef]
- Ogawa, A.; Arai, H.; Tanizawa, H.; Miyahara, T.; Toyo’oka, T. On-line screening method for antioxidants by liquid chromatography with chemiluminescence detection. Anal. Chim. Acta 1999, 383, 221–230. [Google Scholar] [CrossRef]
- Brown, R.C.; Li, Z.; Rutter, A.J.; Mu, X.; Weeks, O.H.; Smith, K.; Weeks, I. Development and application of a novel acridinium ester for use as a chemiluminescent emitter in nucleic acid hybridisation assays using chemiluminescence quenching. Org. Biomol. Chem. 2009, 7, 386–394. [Google Scholar] [CrossRef]
- Roda, A.; Pasini, P.; Mirasoli, M.; Michelini, E.; Guardigli, M. Biotechnological applications of bioluminescence and chemiluminescence. Trends Biotechnol. 2004, 22, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Krzymiński, K.; Ożóg, A.; Malecha, P.; Roshal, A.D.; Wróblewska, A.; Zadykowicz, B.; Błażejowski, J. Chemiluminogenic features of 10-methyl-9-(phenoxycarbonyl)acridinium trifluoromethanesulfonates alkyl substituted at the benzene ring in aqueous media. J. Org. Chem. 2011, 76, 1072–1085. [Google Scholar] [CrossRef]
- Zadykowicz, B.; Czechowska, J.; Ożóg, A.; Renkevich, A.; Krzymiński, K. Effective chemiluminogenic systems based on acridinium esters bearing substituents of various electronic and steric properties. Org. Biomol. Chem. 2016, 14, 652–668. [Google Scholar] [CrossRef]
- Santana Rodríguez, J.J. The role of organised media in chemiluminescence reactions. In Chemiluminescence in Analytical Chemistry, 1st ed.; García-Campaña, A.M., Baeyens, W.R.G., Eds.; Marcel Dekker: New York, NY, USA, 2001; pp. 285–319. [Google Scholar]
- Holmberg, K. Surfactants. In Ullmann’s Encyclopaedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2019. [Google Scholar]
- Hansson, P.; Jönsson, B.; Ström, C.; Söderman, O. Determination of micellar aggregation numbers in dilute surfactant systems with the fluorescence quenching method. J. Phys. Chem. B 2000, 104, 3496–3506. [Google Scholar] [CrossRef]
- Patist, A.; Oh, S.G.; Leung, R.; Shah, D.O. Kinetics of micellisation: Its significance to technological processes. Colloids Surf. A Physicochem. Eng. Asp. 2001, 176, 3–16. [Google Scholar] [CrossRef]
- Missel, P.J.; Mazer, N.A.; Benedek, G.B.; Young, C.Y. Thermodynamic analysis of the growth of sodium dodecyl sulfate micelles. J. Phys. Chem. 1980, 84, 1044–1057. [Google Scholar] [CrossRef]
- Chatterjee, A.; Moulik, S.P.; Sanyal, S.K.; Mishra, B.K.; Puri, P.M. Thermodynamics of micelle formation of ionic surfactants: A critical assessment for sodium dodecyl sulfate, cetyl pyridinium chloride and dioctyl sulfosuccinate (Na salt) by microcalorimetric, conductometric, and tensiometric measurements. J. Phys. Chem. B 2001, 105, 12823–12831. [Google Scholar] [CrossRef]
- Rahman, A.; Brown, C.W. Effect of pH on the critical micelle concentration of sodium dodecyl sulphate. J. Appl. Polym. Sci. 1983, 28, 1331–1334. [Google Scholar] [CrossRef]
- Palladino, P.; Ragone, R. Ionic strength effects on the critical micellar concentration of ionic and nonionic surfactants: The binding model. Langmuir 2011, 27, 14065–14070. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Ismail, K. Aggregation and adsorption properties of sodium dodecyl sulfate in water–acetamide mixtures. J. Colloid Interface Sci. 2008, 327, 198–203. [Google Scholar] [CrossRef]
- Scholz, N.; Behnke, T.; Resch-Genger, U. Determination of the critical micelle concentration of neutral and ionic surfactants with fluorometry, conductometry, and surface tension—A method comparison. J. Fluoresc. 2018, 28, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Grayeski, M.L.; Woolf, E. Enhancement of lucigenin chemiluminescence with cyclodextrin. In Analytical Applications of Bioluminescence and Chemiluminescence; Kricka, L.J., Stanley, P.E., Thorpe, G.H.G., Whitehead, T.P., Eds.; Academic Press: New York, NY, USA, 1984; pp. 565–569. [Google Scholar]
- Howie, C.L.; Grayeski, M.L. Effect of micelles and cyclodextrin solutions on acridinium chemiluminescence. In Bioluminescence and Chemiluminescence. New Perspectives; Scholmerich, J., Andreesen, R., Kapp, A., Ernest, M., Woods, W.G., Eds.; John Wiley: New York, NY, USA, 1987; pp. 415–418. [Google Scholar]
- Natrajan, A.; Sharpe, D.; Wen, D. Effect of surfactants on the chemiluminescence of acridinium dimethylphenyl ester labels and their conjugates. Org. Biomol. Chem. 2011, 9, 5092–5103. [Google Scholar] [CrossRef]
- Natrajan, A.; Sharpe, D.; Wen, D. Chemiluminescence from alkoxy-substituted acridinium dimethylphenyl ester labels. Org. Biomol. Chem. 2012, 10, 3432–3447. [Google Scholar] [CrossRef]
- Nakazono, M.; Nanbu, S. Enhancement effect on the chemiluminescence of acridinium esters under neutral conditions. Luminescence 2017, 33, 345–348. [Google Scholar] [CrossRef]
- Natrajan, A.; Wen, D.; Sharpe, D. Synthesis and properties of chemiluminescent acridinium ester labels with fluorous tags. Org. Biomol. Chem. 2014, 12, 3887–3901. [Google Scholar] [CrossRef] [PubMed]
- Natrajan, A.; Wen, D. Use of degradable cationic surfactants with cleavable linkages for enhancing the chemiluminescence of acridinium ester labels. RSC Adv. 2013, 3, 21398–21404. [Google Scholar] [CrossRef]
- Ando, Y.; Niwa, K.; Yamada, N.; Irie, T.; Enomoto, T.; Kubota, H.; Ohmiya, Y.; Akiyama, H. Development of a quantitative bio/chemiluminescence spectrometer determining quantum yields: Reexamination of the aqueous luminol chemiluminescence standard. Photochem. Photobiol. 2007, 83, 1205–1210. [Google Scholar] [CrossRef] [PubMed]
- Atkins, P.W.; De Paula, J.; Keeler, J. Atkins’ Physical Chemistry; Oxford University Press: Oxford, UK, 2017. [Google Scholar]
- Avogadro: An Open-Source Molecular Builder and Visualization Tool. Version 1.2.0. Available online: http://avogadro.cc/ (accessed on 17 May 2024).
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision C. 01; Gaussian, Inc.: Wallingford, CT, USA, 2019. [Google Scholar]
- Labanowski, J.K.; Andzelm, J.W. Density Functional Methods in Chemistry; Springer: New York, NY, USA, 1991. [Google Scholar]
- Becke, A.D. Density-Functional Exchange-Energy Approximation with Correct Asymptotic Behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density-Functional Thermochemistry. III. The Role of Exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Hehre, W.J.; Ditchfield, R.; Pople, J.A. Self-Consistent Molecular Orbital Methods. XII. Further Extensions of Gaussian-Type Basis Sets for Use in Molecular Orbital Studies of Organic Molecules. J. Chem. Phys. 1972, 56, 2257–2261. [Google Scholar] [CrossRef]
- Bayly, C.I.; Cieplak, P.; Cornell, W.D.; Kollman, P.A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: The RESP model. J. Phys. Chem. 1993, 97, 10269–10280. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and testing of a general Amber force field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- Case, D.A.; Belfon, K.; Ben-Shalom, I.Y.; Brozell, S.R.; Cerutti, D.S.; Cheatham, T.E., III; Cruzeiro, V.W.D.; Darden, T.A.; Duke, R.E.; Giambasu, G.; et al. AMBER 2020; University of California: San Francisco, CA, USA, 2020. [Google Scholar]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 4, 33–38. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing, R Core Team: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 17 May 2024).
- Hariharan, P.C.; Pople, J.A. The influence of polarization functions on molecular orbital hydrogenation energies. Theor. Chim. Acta 1973, 28, 213–222. [Google Scholar] [CrossRef]
- Tomasi, J.; Persico, M. Molecular interactions in solution: An overview of methods based on continuous distributions of the solvent. Chem. Rev. 1994, 94, 2027–2094. [Google Scholar] [CrossRef]
- Barone, V.; Cossi, M.; Tomasi, J. A new definition of cavities for the computation of solvation free energies by the polarisable continuum model. J. Chem. Phys. 1997, 107, 3210–3221. [Google Scholar] [CrossRef]
- Chemcraft—Graphical Software for Visualization of Quantum Chemistry Computations. Available online: https://www.chemcraftprog.com (accessed on 17 May 2024).





| Surfactant | CMC [mM] | |
|---|---|---|
| Experimental Conditions | ||
| 0.25 mM HCl + 0.50 mM HNO3 | H2O [21] | |
| CTAC | 0.86 | 1.3 |
| CTAB | 0.68 | 0.9 |
| SDS | 7.34 | 8.1 |
| System | Maximal ICL [RLU] | kCL [s−1] | RCLE [RLU2] | |
|---|---|---|---|---|
| No surfactant | 379 | 0.179 | 2341 | |
| CTAC | 0.4 mM | 571 | 0.251 | 2427 |
| 0.8 mM | 1080 | 0.514 | 2520 | |
| 4.0 mM | 4256 | 3.214 | 1433 | |
| 8.0 mM | 4848 | 3.206 | 1615 | |
| CTAB | 0.4 mM | 653 | 0.277 | 2416 |
| 0.8 mM | 1691 | 0.897 | 2305 | |
| 4.0 mM | 4732 | 3.422 | 1510 | |
| 8.0 mM | 4838 | 3.405 | 1599 | |
| SDS | 10 mM | 205 | 0.130 | 2350 |
| 20 mM | 104 | 0.030 | 3320 | |
| 40 mM | 91 | 0.015 | 6076 | |
| 60 mM | 68 | 0.010 | 6398 | |
| Emission Process | Difference in CL Efficiency (%) Change in AUC Value (below/above CMC) | ||
| CTAC | CTAB | SDS | |
| CL | +6/−35 | +1/−34 | +21/+166 |
| FL (>CMC) | +9 | +9 | +15 |
| Emission Process | The Difference in CL Dynamics (%) Change in kCL Value (below/above CMC) | ||
| CTAC | CTAB | SDS | |
| CL | +114/+1697 | +228/+1807 | −55/−93 |
| Relative Solvent-Accessible Surface Area | ||||
|---|---|---|---|---|
| Surfactant | 2MeX | 2Me-OOH | [2Me-OO]− | Me-Aon |
| CTAC | 0.71 ± 0.12 | 0.65 ± 0.14 | 0.60 ± 0.13 | 0.54 ± 0.13 |
| SDS | 0.65 ± 0.14 | 0.63 ± 0.13 | 0.73 ± 0.13 | 0.59 ± 0.15 |
| Surfactant | 2MeX | 2Me-OOH | [2Me-OO]− | Me-Aon | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ele | VDW | Total | Ele | VDW | Total | Ele | VDW | Total | Ele | VDW | Total | |
| CTAC | 0.02 | −1.76 | −1.74 | 0.00 | −1.97 | −1.97 | −0.06 | −2.01 | −2.07 | 0.00 | −1.69 | −1.69 |
| SDS | −0.06 | −1.98 | −2.04 | −0.01 | −1.99 | −2.00 | 0.02 | −1.62 | −1.60 | 0.00 | −1.58 | −1.58 |
| Gas Phase | Aqueous Phase | ||
|---|---|---|---|
| Δr,298H0 | Δr,298G0 | Δr,298H0 | Δr,298G0 |
| reaction between SDS and OOH− | |||
| 55.1 | 57.6 | 49.9 | 52.4 |
| reaction between SDS and H2O2 | |||
| 28.3 | 29.7 | 30.4 | 31.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mańkowska, M.; Krzymiński, K.; Wyrzykowski, D.; Zadykowicz, B.; Samsonov, S.A. Why Do Ionic Surfactants Significantly Alter the Chemiluminogenic Properties of Acridinium Salt? Molecules 2024, 29, 3736. https://doi.org/10.3390/molecules29163736
Mańkowska M, Krzymiński K, Wyrzykowski D, Zadykowicz B, Samsonov SA. Why Do Ionic Surfactants Significantly Alter the Chemiluminogenic Properties of Acridinium Salt? Molecules. 2024; 29(16):3736. https://doi.org/10.3390/molecules29163736
Chicago/Turabian StyleMańkowska, Magdalena, Karol Krzymiński, Dariusz Wyrzykowski, Beata Zadykowicz, and Sergey A. Samsonov. 2024. "Why Do Ionic Surfactants Significantly Alter the Chemiluminogenic Properties of Acridinium Salt?" Molecules 29, no. 16: 3736. https://doi.org/10.3390/molecules29163736
APA StyleMańkowska, M., Krzymiński, K., Wyrzykowski, D., Zadykowicz, B., & Samsonov, S. A. (2024). Why Do Ionic Surfactants Significantly Alter the Chemiluminogenic Properties of Acridinium Salt? Molecules, 29(16), 3736. https://doi.org/10.3390/molecules29163736








