Ring Contractions of Cycloalkanones
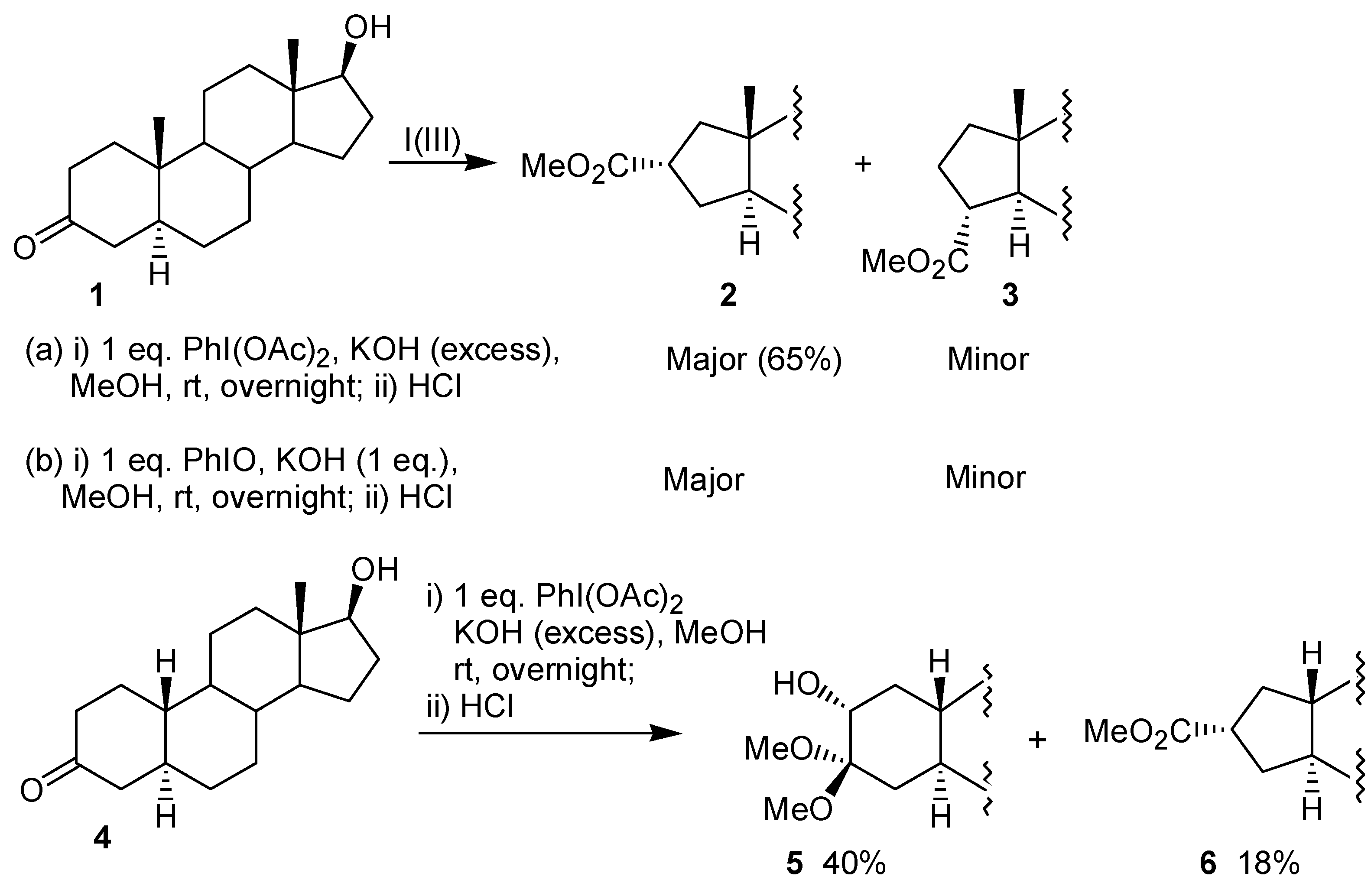
Two articles concerning the ring contraction of steroidal ketones mediated by iodine(III) were released nearly simultaneously in 1984. In the first paper, Daum reported that the reaction of the androstan-3-one
1 with iodobenzene diacetate leads to a mixture of the ring contraction products
2 and
3 [
17]. After recrystallization,
2 was obtained in 65% yield. A similar result was obtained when the reaction was performed with iodosobenzene, instead of iodobenzene diacetate. The configuration of the major product
2 has been explained by the formation of the adduct
7, which bears the iodine group at the equatorial position (
Figure 1). The behavior of the 19-norandrostan-3-one
4 toward the oxidation of iodobenzene diacetate was different from
1, because
4 gave as the major product the hydroxyketal
5. In this case, the ring contraction product
6 was obtained in poor yield (
Scheme 1). The author suggested that for the nor-androstanone
4 formation of the adduct
8 bearing the iodine(III) group in the axial position would occur, as shown in
Figure 1. Thus, for substrates where there are no steric restrictions, such as
4, the addition would lead to an intermediate with the iodine(III) group in the axial position, as in
8. On the other hand, for cycloalkanones where an axial iodine(III) atom would lead to strong 1,3-diaxial interactions, such as
1, the iodine(III) occupies the equatorial position, as in
7.
Figure 1.
Structure of the Intermediates proposed by Daum.
Figure 1.
Structure of the Intermediates proposed by Daum.
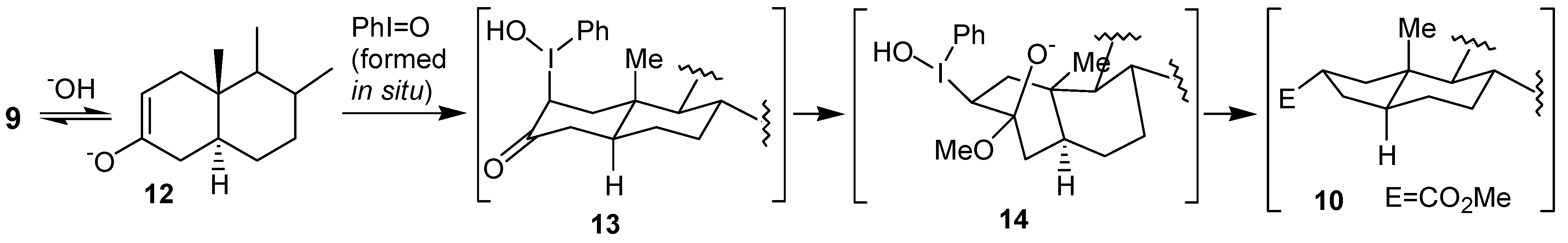
In the second paper Moriarty and co-workers [
18] described that the treatment of the 3-cholestanone
9 with trivalent iodine reagents, such as PhI(OAc)
2,
o-OIC
6H
4CO
2H or PhIO
2, led to an oxidative rearrangement, affording a 9:1 mixture of the ring contraction products
10 and
11, respectively (
Scheme 2). The formation of the major product has been explained by the initial hyperiodination of the enolate
12, giving the adduct
13, which bears the iodine group at the axial position, which contrasts with the intermediate
7, proposed by Daum [
17] (cf.
Figure 1). Then, the adduct
13 would be converted into the corresponding twist-boat form
14, on which the rearrangement would take place giving the ring contraction product
10 (
Scheme 3).
Under conditions similar to those used in the ring contraction of
9, the rearrangement of another cyclic ketone, namely 4-phenylcyclohexanone (
15), has been performed by the same group in the synthesis of 1-piperidinobenzobicyclo[2.2.1]heptene [
19]. In this example, however, the relative configuration of the ring contraction products
16 was not assigned (
Scheme 4). The oxidative rearrangement of cyclohexanones can also be promoted by thallium(III) [
20] and selenium(IV) [
21], in good yield and diastereoselectivity.
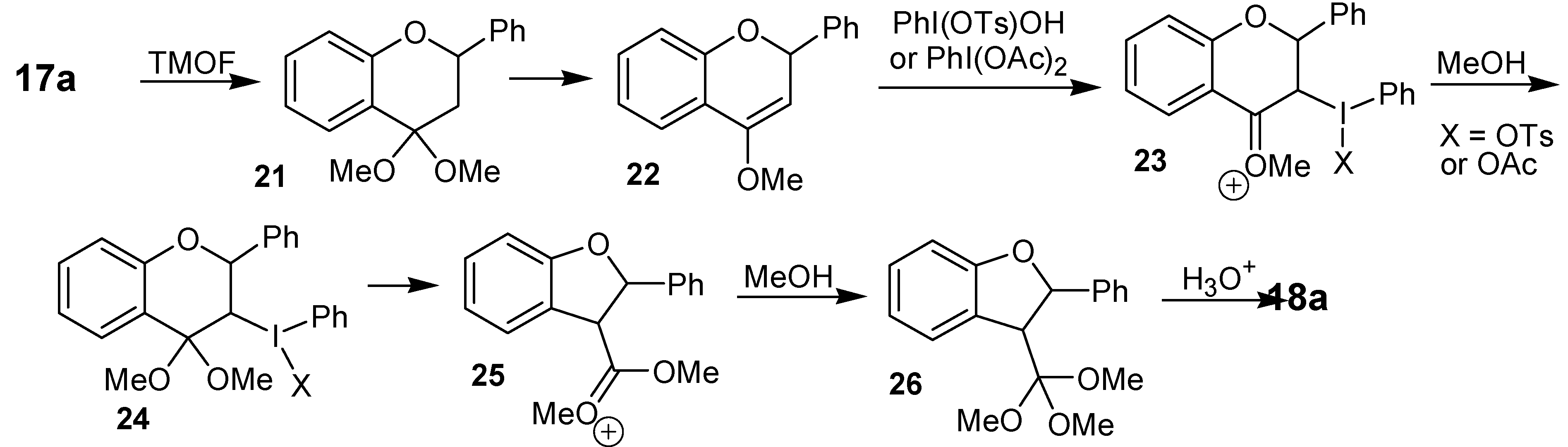
The ring contraction reaction of cyclic ketones with iodine(III) can also be performed with substrates other than the abovementioned cyclohexanone derivatives. One of such examples is the treatment of the flavanones
17a-h with iodobenzene diacetate or with [hydroxy(tosylosy)iodo]benzene (Koser´s reagent) in trimethyl orthoformate (TMOF), as solvent, which gave the dihydrobenzofuran derivatives
18a-g, in 35 to 75% yield [
22]. Although formed as a single diastereomer, the relative configuration of the ring contraction products was not assigned. Other products isolated in this study were the
cis-3-methoxyflavanones
19 and the flavones
20. The formation of the rearrangement products
18a-g has been rationalized by the mechanism shown in
Scheme 5, which is exemplified for
17a. A key feature of this mechanism is the electrophilic addition of the iodine(III) reagent in the enol ether moiety of
22 and the rearrangement of
24 in
25.
Table 1.
Reaction of Flavanones with Iodine(III)
a.
![Molecules 11 00421 i001]()
Table 1.
Reaction of Flavanones with Iodine(III)a. ![Molecules 11 00421 i001]()
| Entry | Substrate | Product (Yield)b |
| 1 | 17a (a: R=R1=H; Ar=Ph) | 18a (40%), 19a (27%), 20a (12%) |
| 2 | 17b (b: R=R1=H; Ar=p-ClC6H4) | 18b (60%), 20b (18%) |
| 3 | 17c (c: R=Cl; R1=H; Ar=Ph) | 18c (43%), 19c (36%), 20c (8%) |
| 4 | 17d (d: R=Cl; R1=H; Ar=p-ClC6H4) | 18d (35%), 19d (34%), 20d (16%) |
| 5 | 17e (e: R=Me; R1=H; Ar=Ph) | 18e (75%), 20e (16%) |
| 6 | 17f (f: R=Me; R1=H; Ar=p-ClC6H4) | 18f (75%), 20f (16%) |
| 7 | 17g (g: R=Cl; R1=Me; Ar=Ph) | 18g (47%), 20g (35%) |
| 8 | 17h (h: R=Cl; R1=H; Ar=p-OMeC6H4) | 20h (18%) |
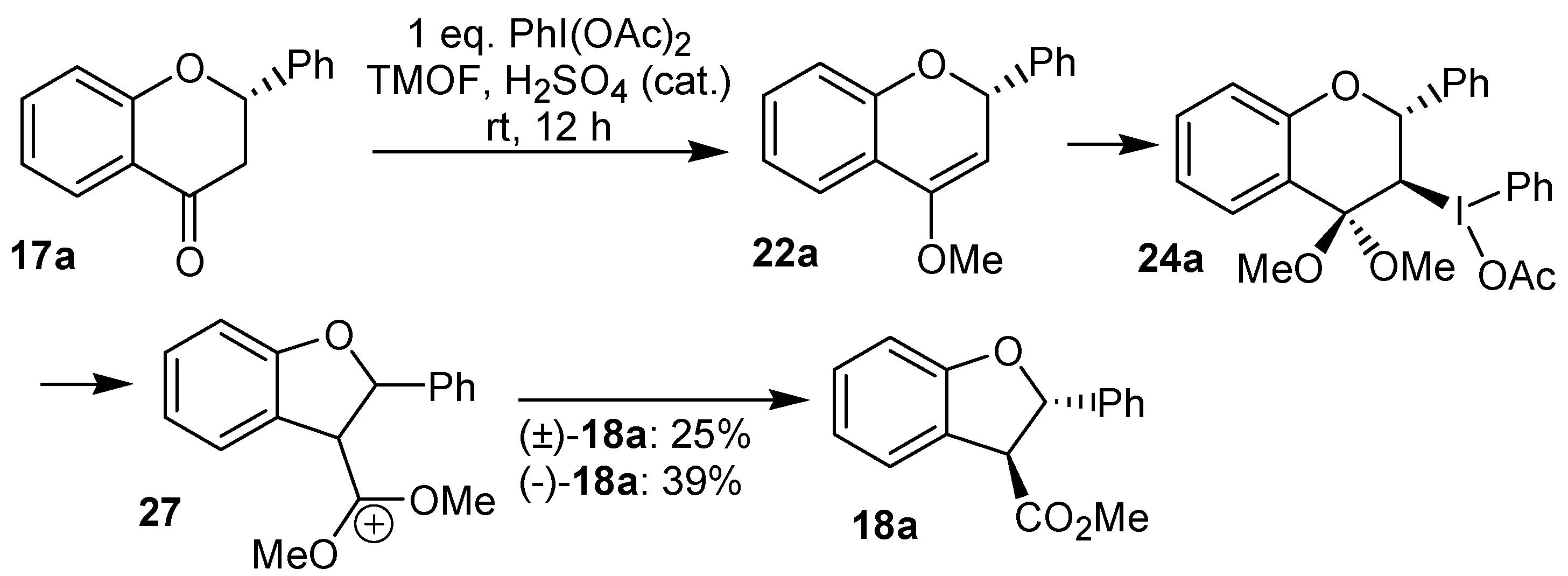
Some years later, the reaction of the flavanones
17a and (-)-
17a with iodobenzene diacetate was reinvestigated by Juhász
et al. [
23], which assigned a
trans relationship for the substituent at the benzofuran ring. Based on the formation of this product, the relative configuration of the intermediates
22a and
24a was also proposed (
Scheme 6).
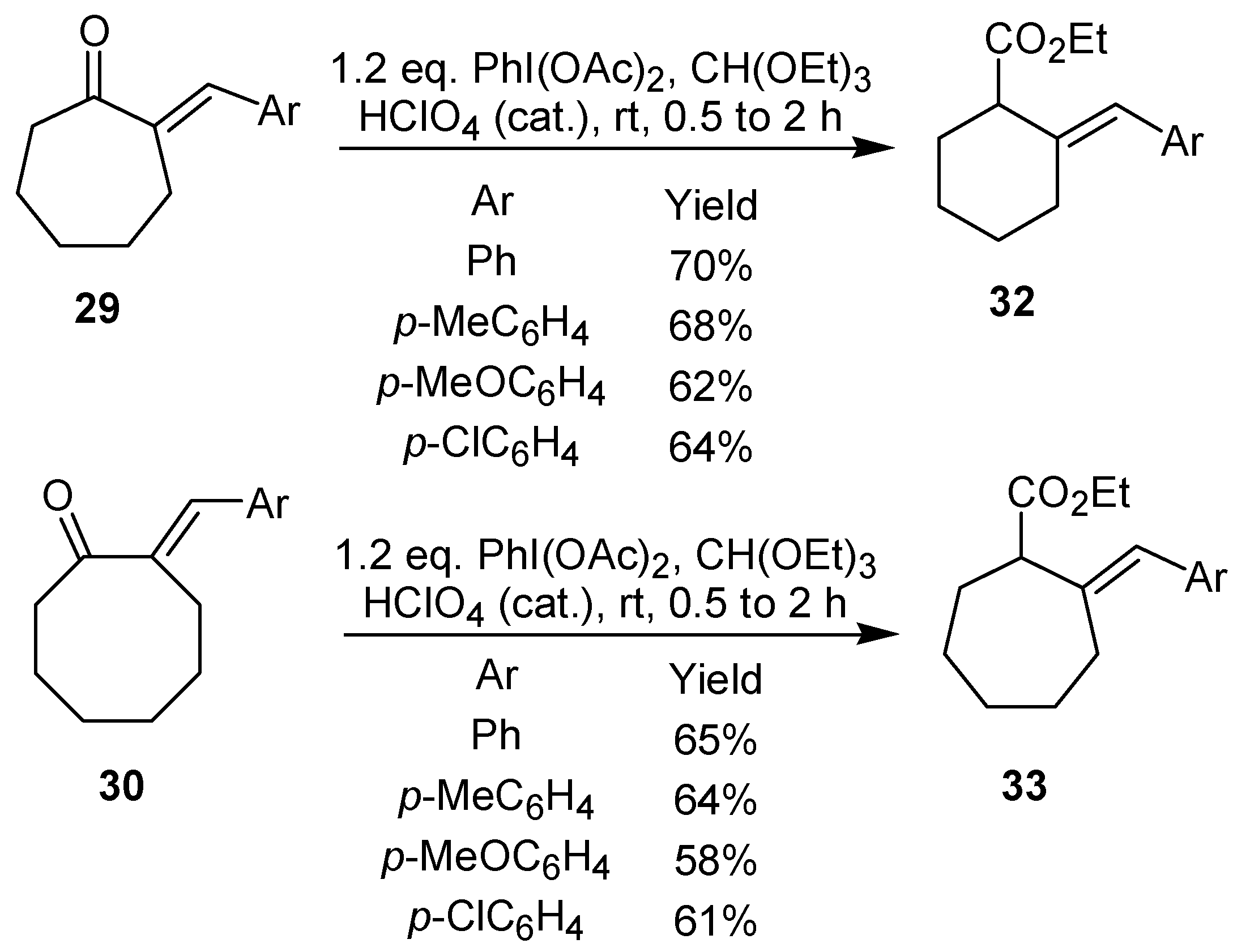
The treatment of 2-arylidenecycloalkanones, such as
28-30, with iodobenzene diacetate led to an oxidative rearrangement, which culminated with the formation of the 2-arylidenecycloalkane carboxylates
31-33 in good yield [
24]. It is worth mentioning that this approach can be used for the synthesis of five-, six-, and seven-membered ring derivatives, as shown in
Scheme 7 and
Scheme 8.
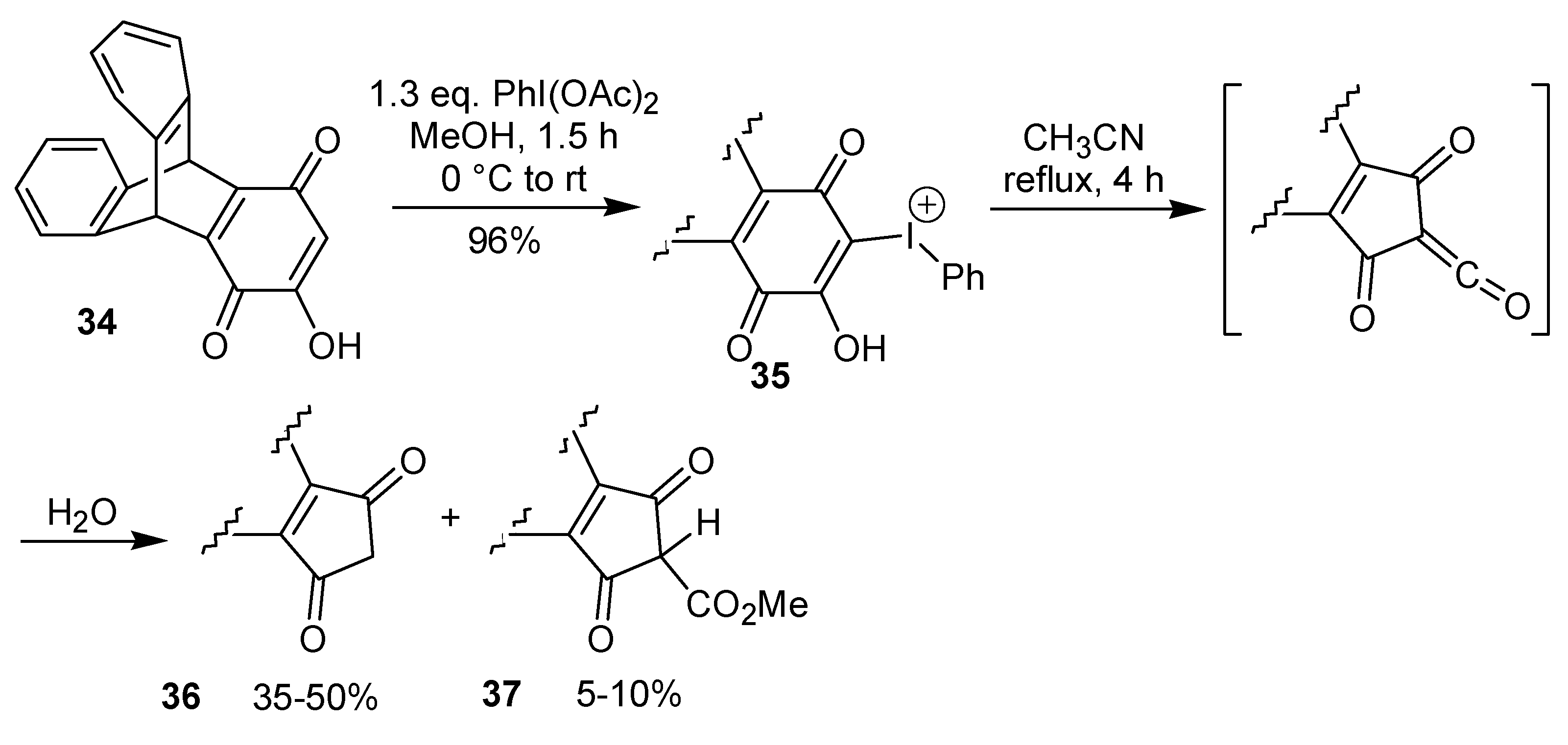
A two-step procedure to promote the ring contraction of 2-hydroxy-1,4-benzoquinones to 2-cyclo-pentene-1,4-diones has been developed by the group of Varvoglis and co-workers [
25,
26]. This approach is based on the thermolysis of a phenyliodonium species, which is obtained by reaction of a hydroxyquinone with iodobenzene diacetate. Representative examples of this transformation are shown in
Scheme 9. Recently, the preparation of the intermediate phenyliodonium ylide
35 by rearrangement of the triptycene quinone
34 utilizing iodobenzene diacetate has been described [
27]. The ring contraction step occurs through a sequence of events, initiated by a Wolff-type rearrangement of
35 to give a ketene. Reaction of the formed ketene with water gives a carboxylic acid that leads to
36 after a decarboxylation. The formation of the by-product
37 (
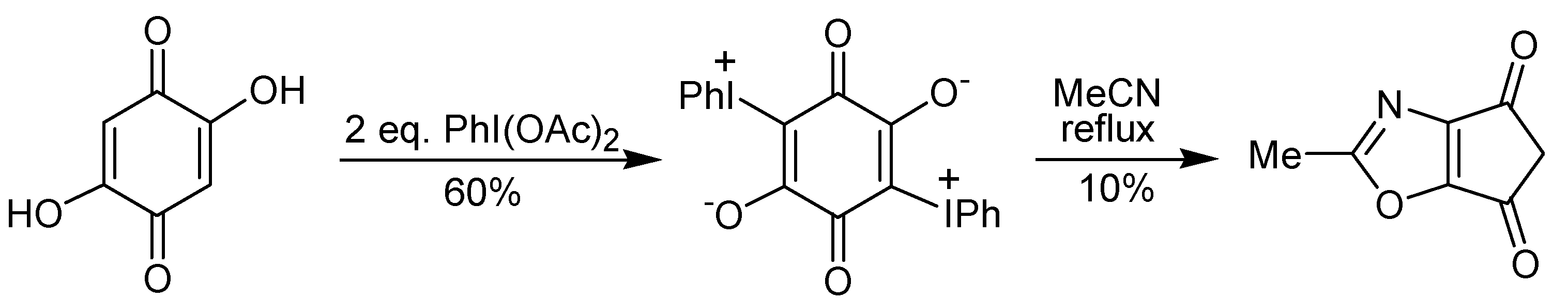
Scheme 10) is evidence for the proposed mechanism. The treatment of a 2,5-dihydroxy-1,4-benzoquinone with iodobenzene diacetate gave a double iodonium zwitterion, which when submitted to the thermolysis conditions led to the ring contraction, as well as to a formal [3+2] cycloaddition reaction, giving a isoxazole derivative, albeit in low yield, as exemplified in
Scheme 11 [
26].
The ring contraction reaction is not the only reaction pathway that may take place in the oxidation of cyclic ketones with hypervalent iodine. A highly explored reaction is the α-oxidation of ketones, which occurs under different reaction conditions, including with
o-iodosylbenzoic acid [
28] (
Table 2, entry 1) and with iodobenzene diacetate [
29] (entry 2), both under basic conditions. It is interesting to note that the latter condition is similar to that employed in the ring contraction of cyclohexanone derivatives (see
Scheme 2 and
Scheme 4). Thus, the course of the oxidation of cyclic ketones with iodobenzene diacetate/KOH is determined by the structure of the substrate. Other important protocols developed for the α-oxidation are: i) the one-pot oxidation of alcohols to the corresponding carbonyl compound followed by α-tosyloxylation (entry 3) [
30]; ii) the solvent-free reaction of ketones with PhI(OAc)
2/
p-TsOH (entry 4) [
31]; and iii) the proline-catalyzed asymmetric α-oxidation of ketones (entry 5) [
32].
Table 2.
Representative examples of the α-oxidation of cyclic ketones.
Table 2.
Representative examples of the α-oxidation of cyclic ketones.
| Entry | Substrate | Conditions | Product (Yield) | Reference |
| 1 | ![Molecules 11 00421 i002]() | 1.1 eq. o-H2OCC6H4IO, 3 eq. KOH, MeOH, rt, overnight | ![Molecules 11 00421 i003]() | Moriarty et al. [28] |
| 2 | ![Molecules 11 00421 i004]() | 1.1 eq. PhI(OAc)2, 3 eq. KOH, MeOH, 0-5 °C for 1 h and 23-25 °C for 20 h | ![Molecules 11 00421 i005]() | Moriarty et al. [29] |
| 3 | ![Molecules 11 00421 i006]() | 3.0 eq. PhIO, 2.5 eq. p-TsOH.H2O, CH3CN, 60 °C, 3 h | ![Molecules 11 00421 i007]() | Ueno et al. [30] |
| 4 | ![Molecules 11 00421 i002]() | PhI(OAc)2, p-TsOH
grinding, 20 min | ![Molecules 11 00421 i008]() | Yusubov and Wirth [31] |
| 5 | ![Molecules 11 00421 i002]() | i) 0.33 eq. PhIO, L-proline (10-30 mol %), DMF, rt, 16-24 h;
ii) NaBH4, MeOH, 0 °C | ![Molecules 11 00421 i009]() | Engqvist et al. [32] |
Ring Contractions of Cycloalkenes
The oxidation of cycloalkenes with hypervalent iodine has been reported by several authors. The analysis of these papers reveals that products formed by an addition to the double bond is the most often observed reaction pathway. Selected examples of this transformation are shown in
Table 3. However, treatment of cyclic olefins with iodine(III) reagents may also lead to ring contraction products, as discussed in the following paragraphs.
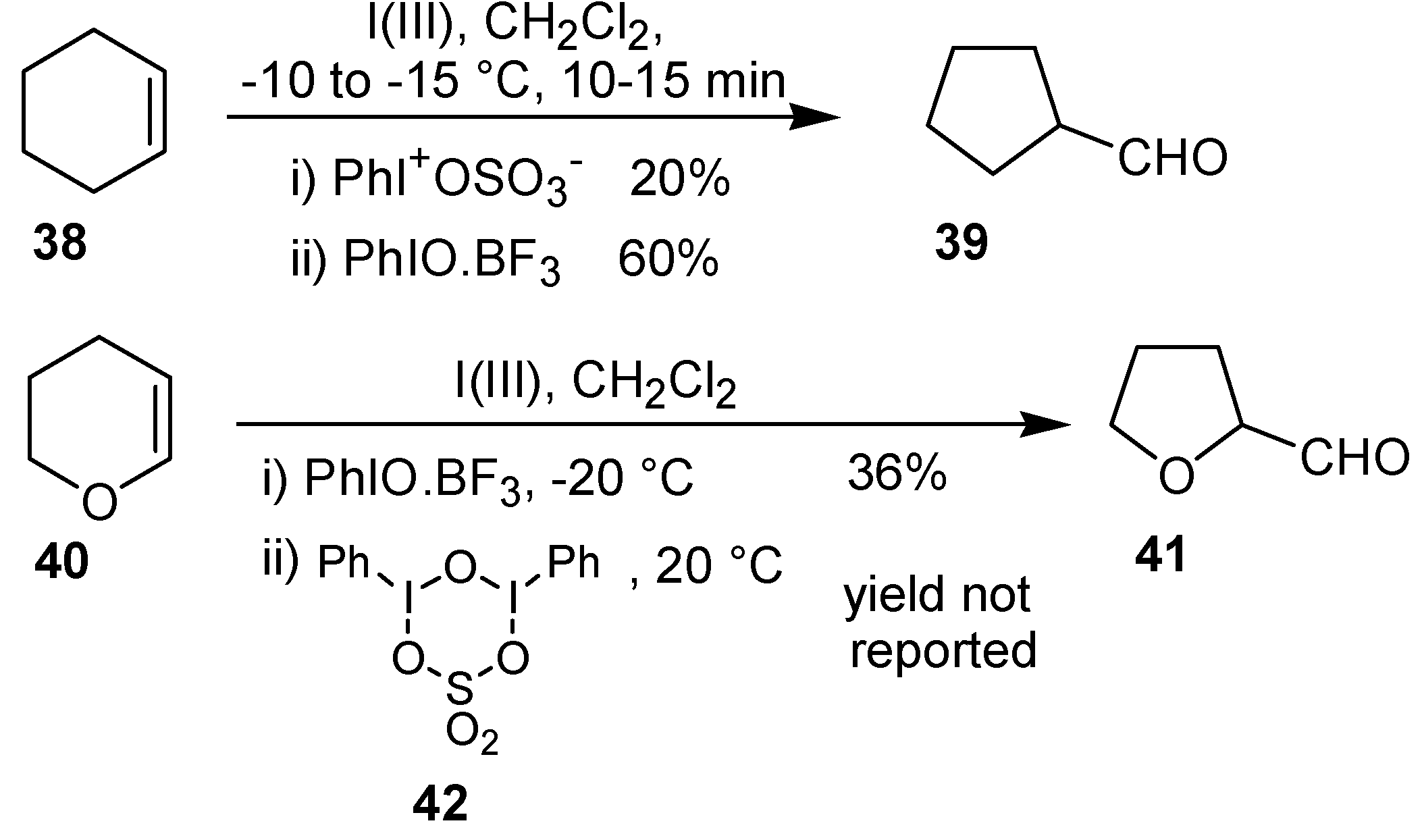
Zefirov
et al. [
38] investigated the oxidation of cyclohexene (
38) and 2,3-dihydropyran (
40) with trivalent iodine under different conditions. Treatment of the olefin
38 with phenyl iodosulfate or with iodosobenzene led to the oxidative rearrangement product
39, in 20 and 60% yield, respectively. Furthermore, the complex iodosobenzene/BF
3 and the sulfate bearing an iodine(III) atom
42 were utilized to carry out the transformation of the substrate
40 into the corresponding tetrahydrofuran
41 (
Scheme 12).
Table 3.
Representative examples of I(III)-promoted addition reactions of cyclohexene.
Table 3.
Representative examples of I(III)-promoted addition reactions of cyclohexene.
| Entry | Conditions | Product (Yield) | Reference |
| 1 | PhI(OTs)OH, rt, 1.5 h | ![Molecules 11 00421 i010]() | Koser et al. [33] |
| 2 | ![Molecules 11 00421 i011]() , ClCH2CH2Cl, 25 °C, 15-20 h , ClCH2CH2Cl, 25 °C, 15-20 h | ![Molecules 11 00421 i012]() | Zefirov et al. [34] |
| 3 | ![Molecules 11 00421 i013]() , AcOEt, LiClO4, rt, 2 h. , AcOEt, LiClO4, rt, 2 h. | ![Molecules 11 00421 i014]() | Zhdankin et al. [35] |
| 4 | CF3CH2I(OTs)OH, CH2Cl2, 0 °C, 3-4 h | ![Molecules 11 00421 i015]() | Zhdankin et al. [36] |
| 5 | PhI(O2PPh2)OH, I2, ClCH2CH2Cl, rt, 1 d | ![Molecules 11 00421 i016]() | Togo et al. [37] |
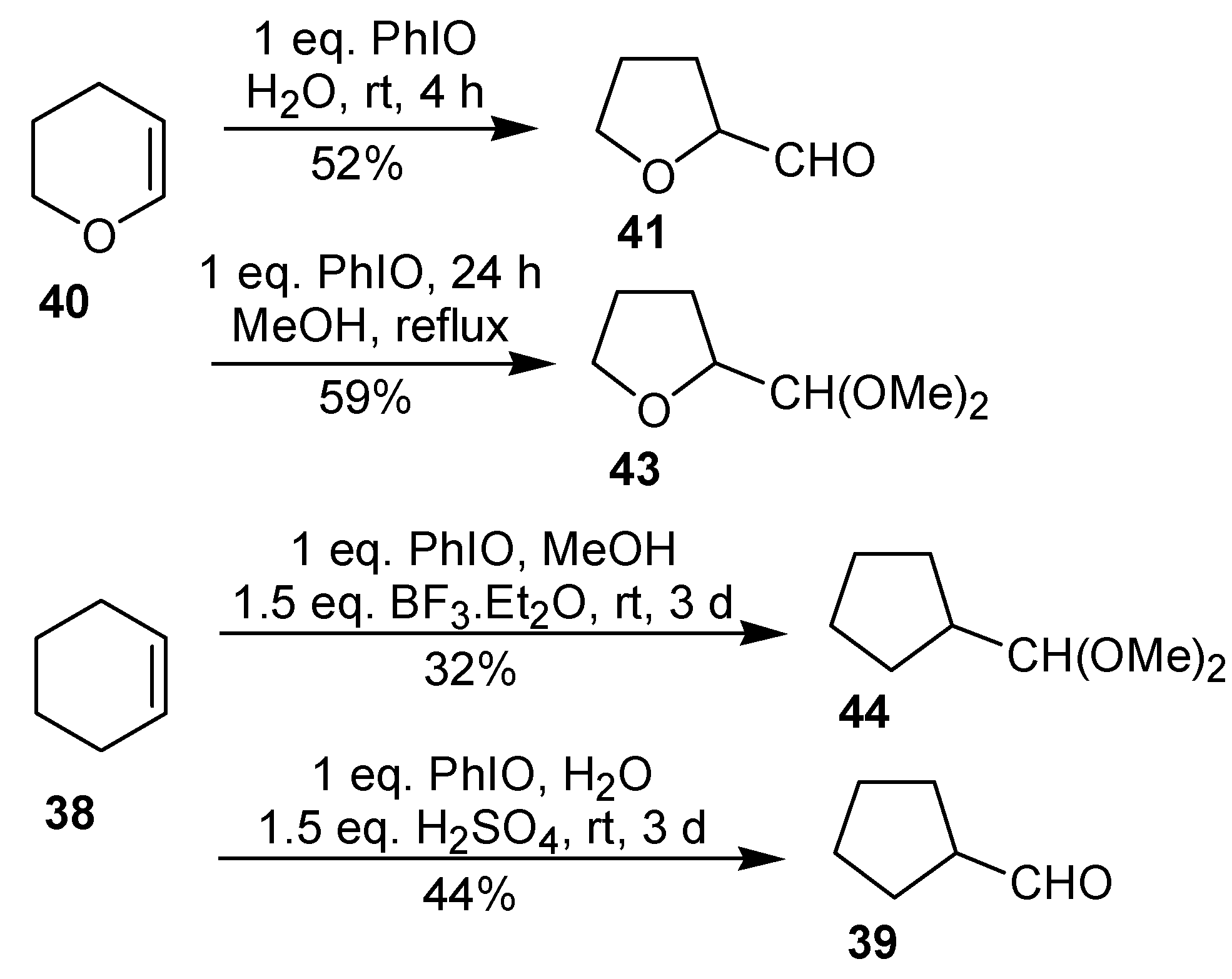
In 1996, Moriarty
et al. [
39] described that the reaction of dihydropyran (
40) with iodosobenzene in H
2O or in MeOH, which gives the ring contraction product in moderate yield. Under similar conditions, no reaction between cyclohexene and PhIO has been observed. However, the addition of an acid catalyst (BF
3.Et
2O or H
2SO
4) led to the ring contraction product, albeit in low yield (
Scheme 13).
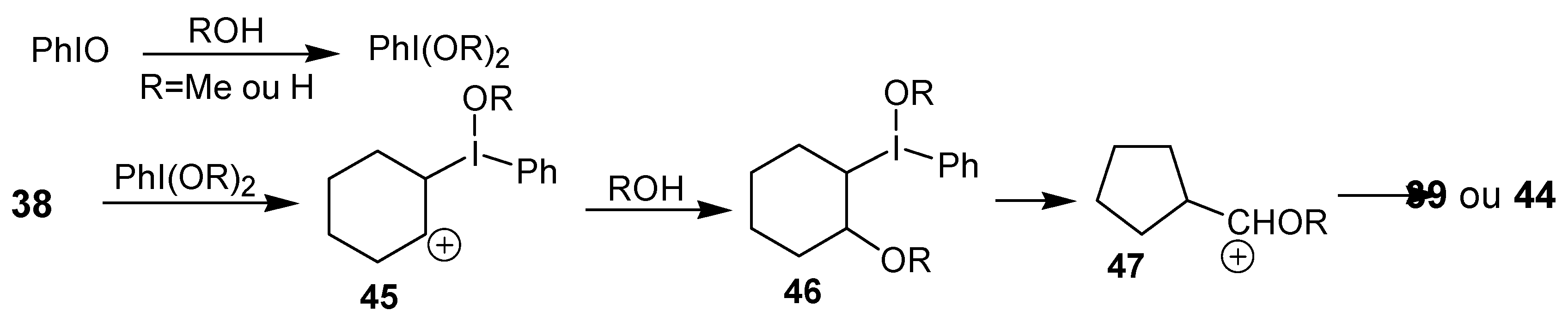
The authors proposed that the reactive species in this reaction is PhI(OMe)
2 or PhI(OH)
2, which would be generated by the reaction of iodosobenzene with MeOH or H
2O, respectively. Then, an electrophilic addition of iodine(III) to the double bond would give the carbocation
45, that would lead to the adduct
46 after addition of a molecule of solvent. The rearrangement would occur at this intermediate, producing the observed products, as exemplified for the cyclohexene in
Scheme 14.
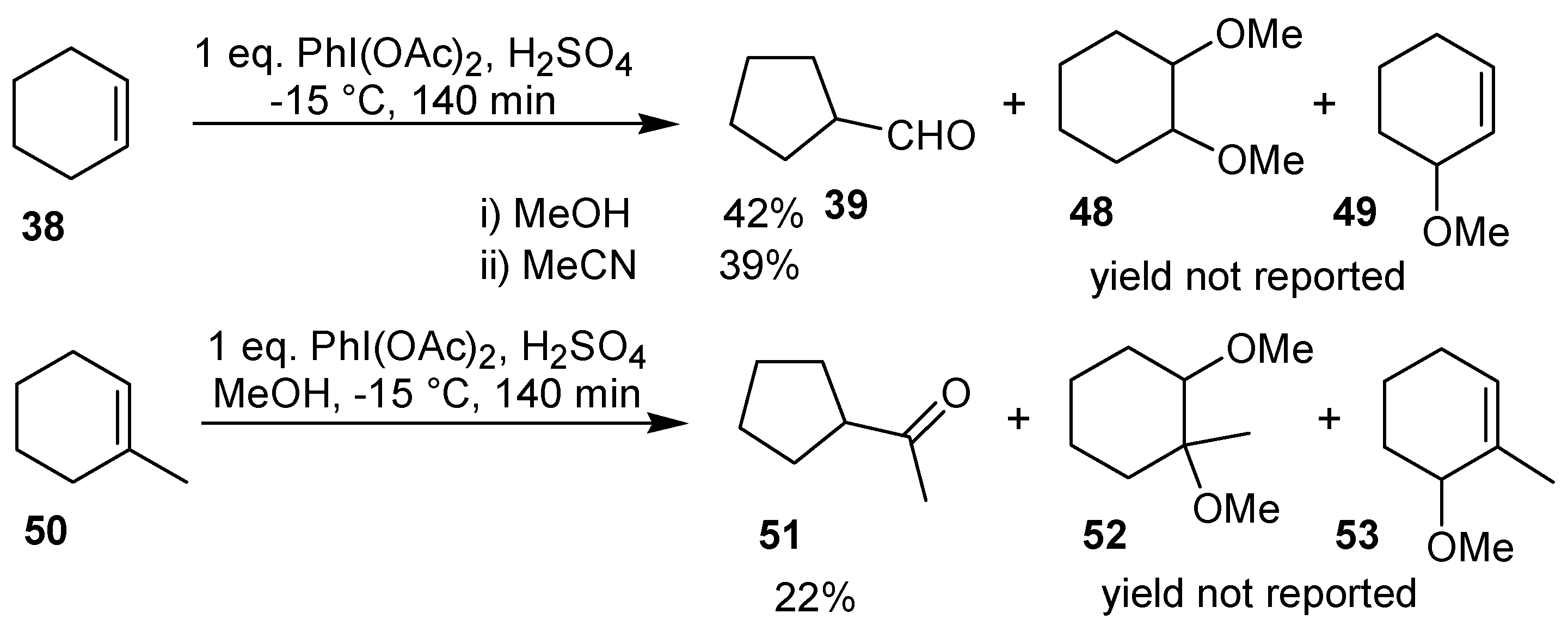
The ring contraction of cyclohexene can also be performed using the commercially available iodobenzene diacetate in the presence of an acid catalyst, giving
39, in 39-42% yield [
40,
41]. Under analogous conditions, the oxidation of 1-methylcyclohexene gave the cyclopentane derivative
51 in 22% yield. In both cases, the aldehyde was isolated as the corresponding 2,4-dinitrophenylhydrazone. In these reactions, the formation of products of addition of two molecules of solvent (
48 and
52) and of allylic oxidation (
49 and
53) was also observed (
Scheme 15) [
41].
A recent publication by Justik and Koser described the rearrangement of arylalkenes with [hydroxy(tosyloxy)iodo]benzene in methanol giving α-aryl ketones as products [
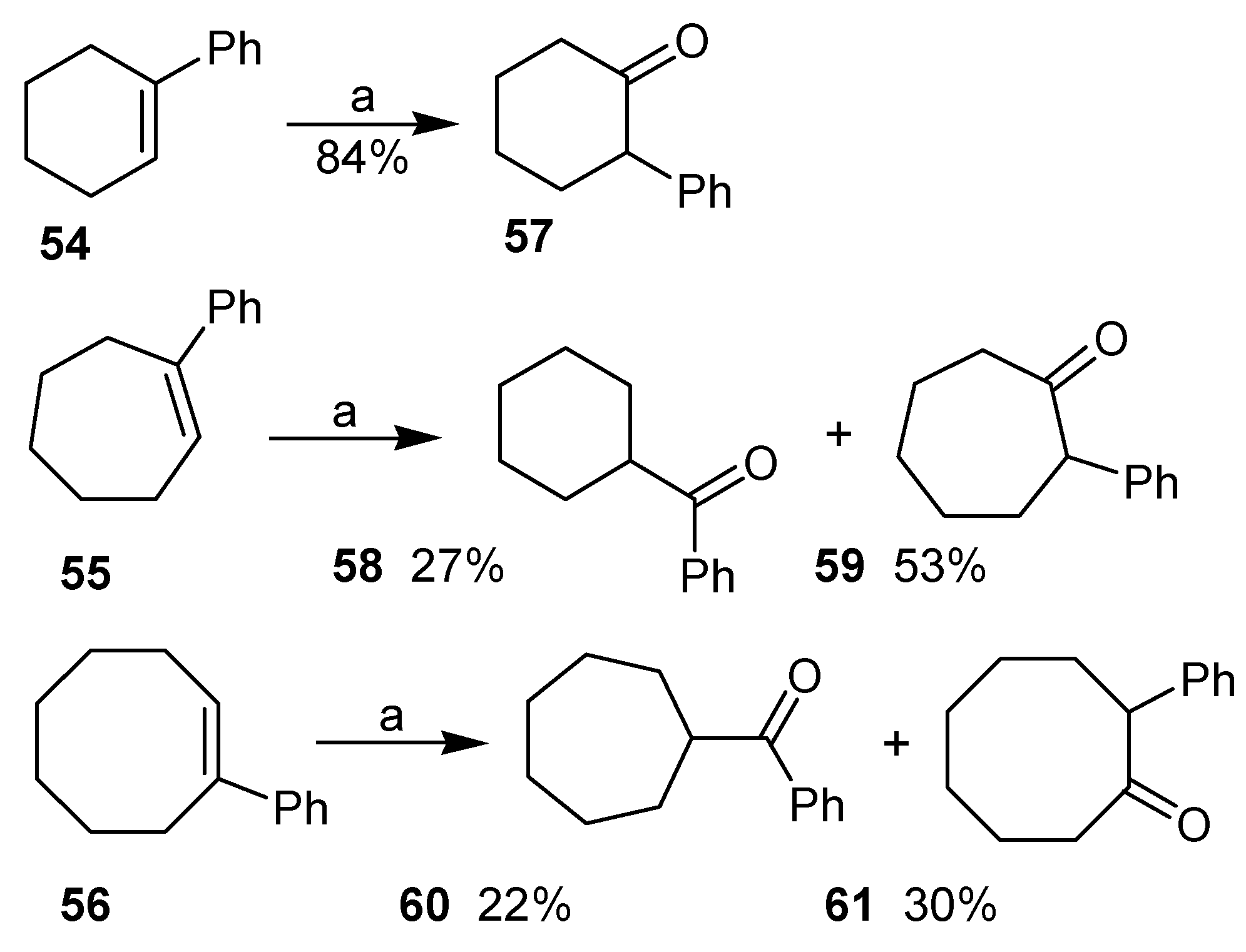
42]. In the case of 1‑phenylcycloalkenes, the formation of two different rearrangement products was observed. The first is that originated by the migration of the phenyl group, as exemplified by the oxidation of 1-phenyl-cyclohexene (
54). The second possibility would be the ring contraction reaction, which took place with 1-phenylcycloheptene (
55) and with 1-phenylcyclooctene (
56), although in these cases products of phenyl migration (
59 and
61, respectively) were also isolated, as the major product (
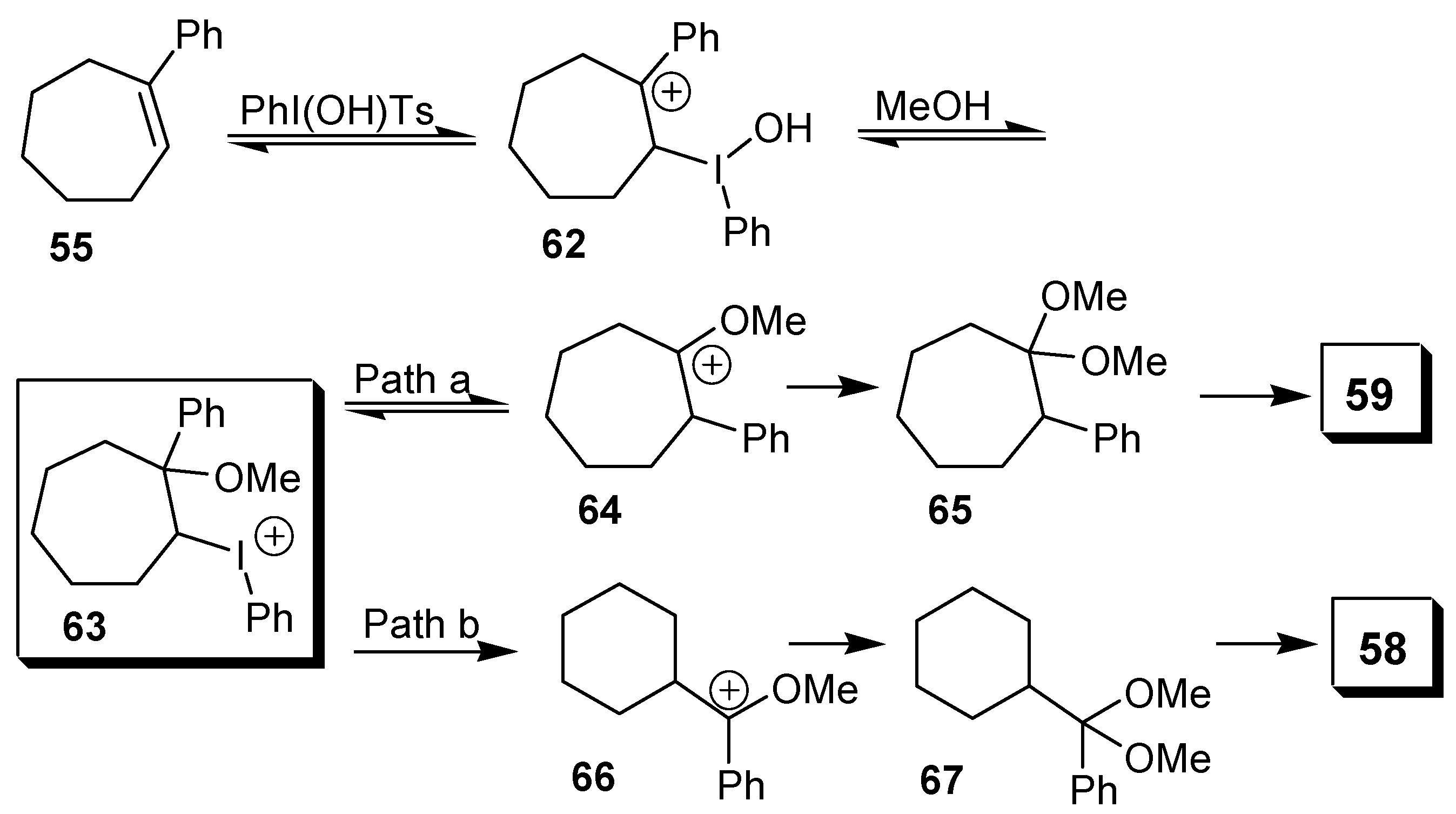
Scheme 16). The mechanism proposed by the authors shows the electrophilic addition of the [hydroxy(tosyloxy)iodo]benzene to the double bond, giving the adduct
63, after addition of a molecule of solvent to the carbocation
62. This adduct would then originate either the product of phenyl migration (Path a) or the ring contraction product (Path b), as exemplified for 1-phenylcycloheptene in
Scheme 17.
Scheme 16.
Reagents and Conditions: 0.92 eq. PhI(OH)Ts; 95% MeOH, rt. Reaction times: 54: 20 min; 55: 1 h and; 56: 8h.
Scheme 16.
Reagents and Conditions: 0.92 eq. PhI(OH)Ts; 95% MeOH, rt. Reaction times: 54: 20 min; 55: 1 h and; 56: 8h.
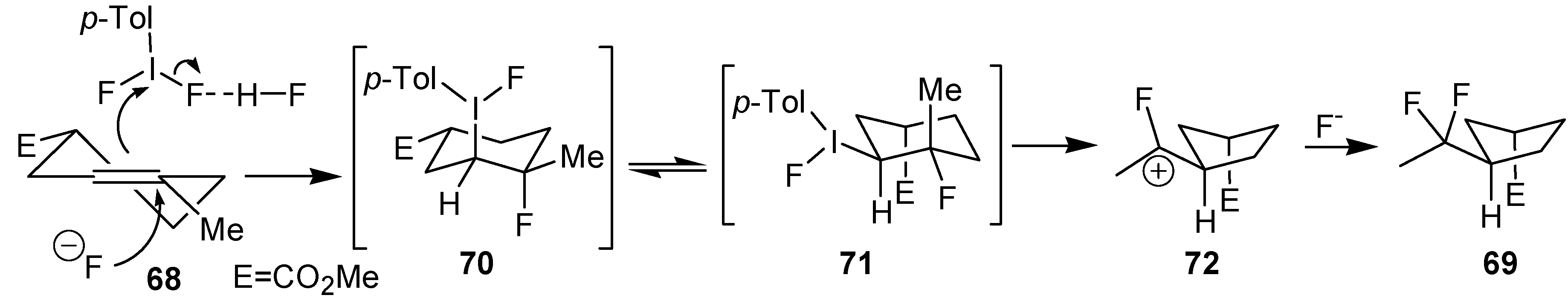
An efficient method to perform the rearrangement of cycloalkenes has been developed by Hara and co-workers [
43,
44]. This approach consists in the treatment of the olefin with iodotoluene difluoride in the presence of Et
3N-5HF, leading to difluoride ring contraction products in poor to very good yield. This reaction occurs with high degree of diastereoselectivity, giving exclusively
trans diastereomers (
Table 4, entry 1). Moreover, such a protocol can be used for the ring contraction of cyclohexene (entries 1-3), cycloheptene (entry 4) and cyclooctene derivatives (entry 5). The formation of these compounds can be explained by a mechanism where the first step is the electrophilic addition of the hypervalent reagent, giving the fluoride adduct
70, which leads to the carbocation
72 after a ring inversion to reach the required anti-periplanarity for the rearrangement. Reaction of
72 with a fluoride anion gives the observed product
69 (
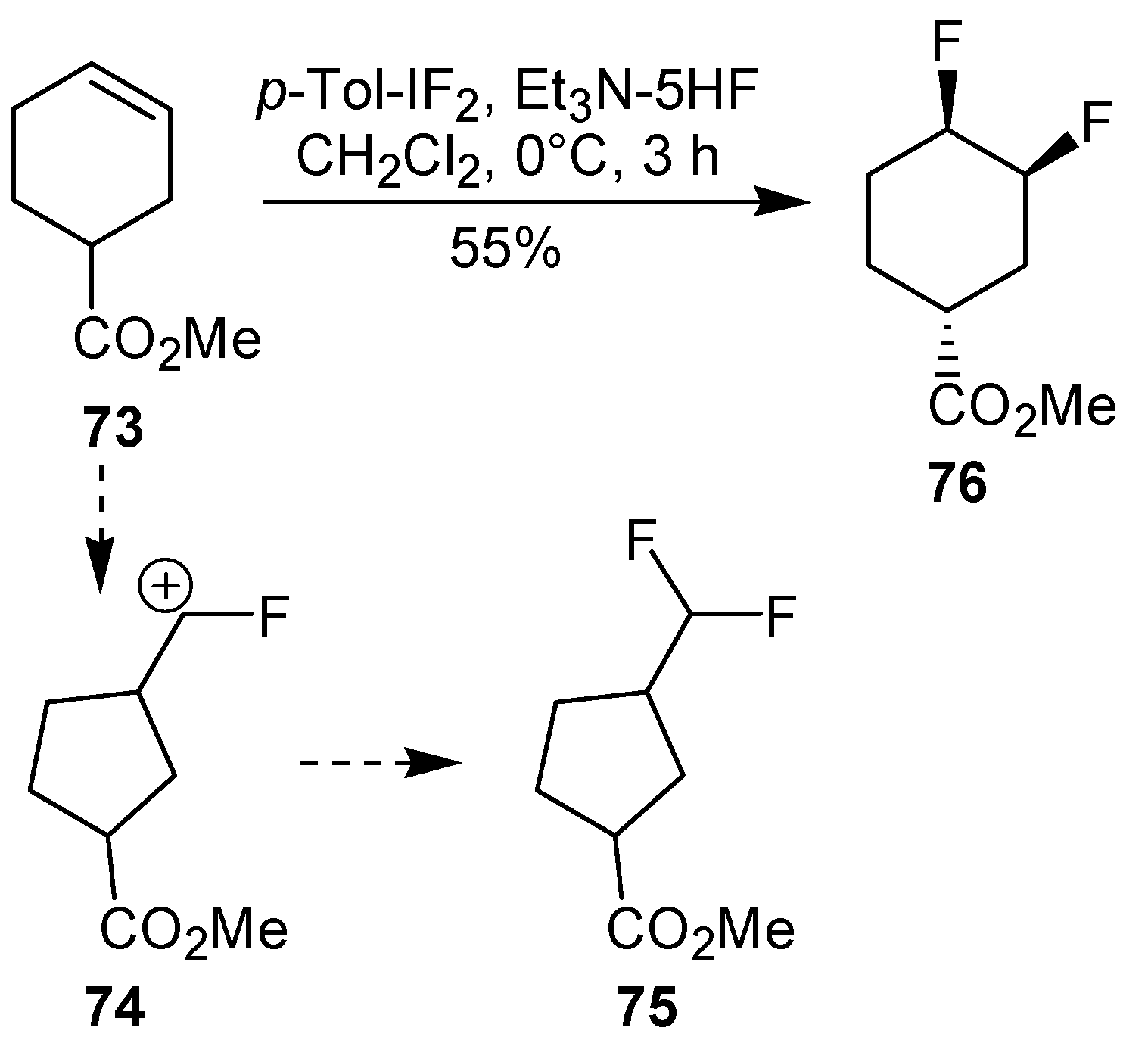
Scheme 18). Contrasting with the above mentioned examples, the reaction of 4-substituted-cyclohexenes without substituents in the double bond, such as
73, with iodotoluene difluoride gives exclusively a
cis-difluoro compound, such as
76, in good yield, as shown in
Scheme 19 (compare to
Table 4, entry 1) [
45]. This different behavior has been explained considering the stability of the possible carbocation intermediates. The ring contraction of
68 would occur through the secondary carbocation
72, whereas the formation of the hypothetically product
75 would take place through the primary cation
74.
Table 4.
Ring Contraction of Cicloalkenes with 1.3 eq. p-Tol-IF2.a
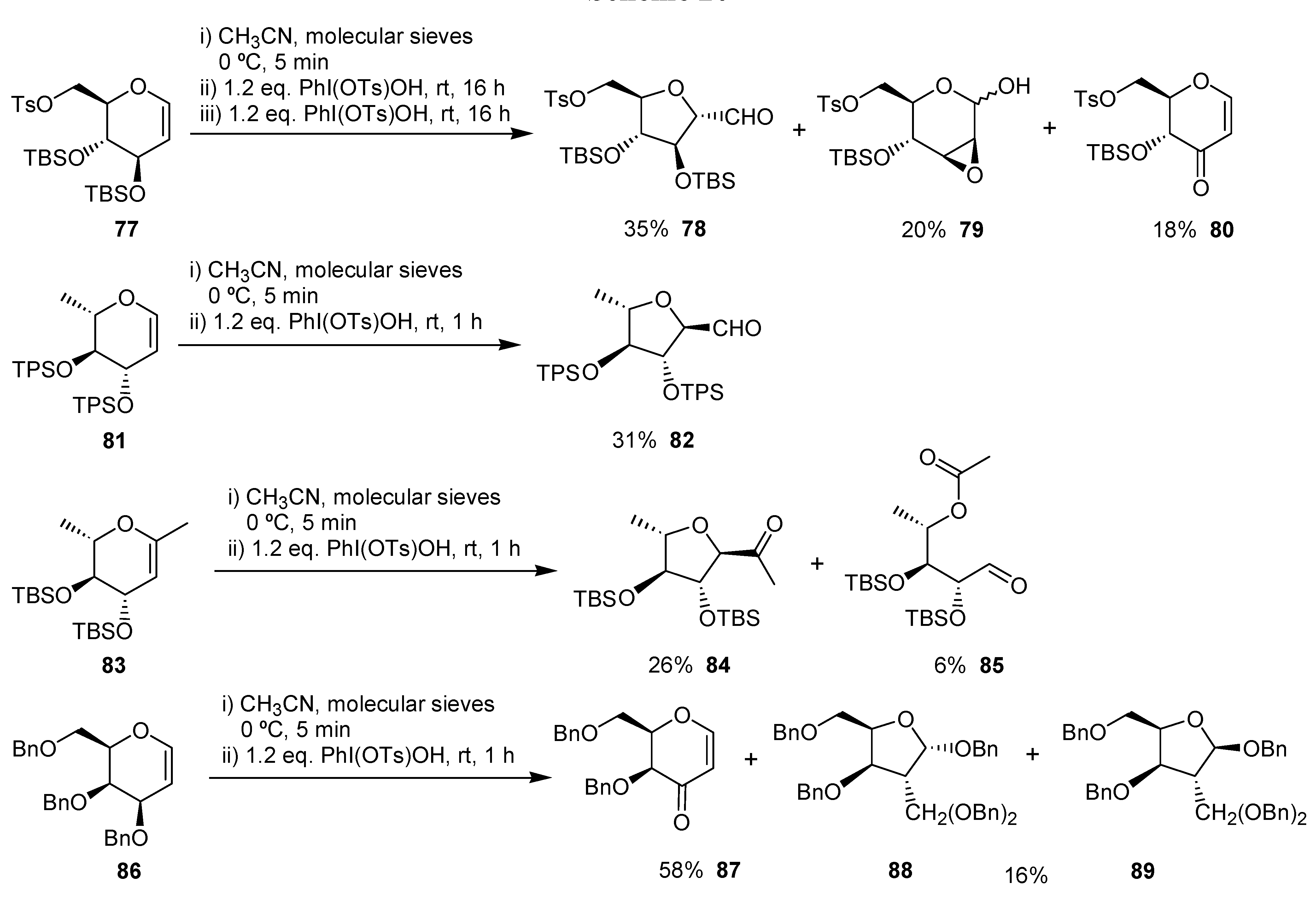
The ring contraction of carbohydrate derivatives has been used to obtain functionalized tetrahydrofurans by Kirsching and co-workers. [
46,
47,
48] This transformation was carried out using PhI(Ots)OH in acetonitrile in the presence of molecular sieves and gave the corresponding ring contraction products from 16 to 35% yield (
Scheme 20).
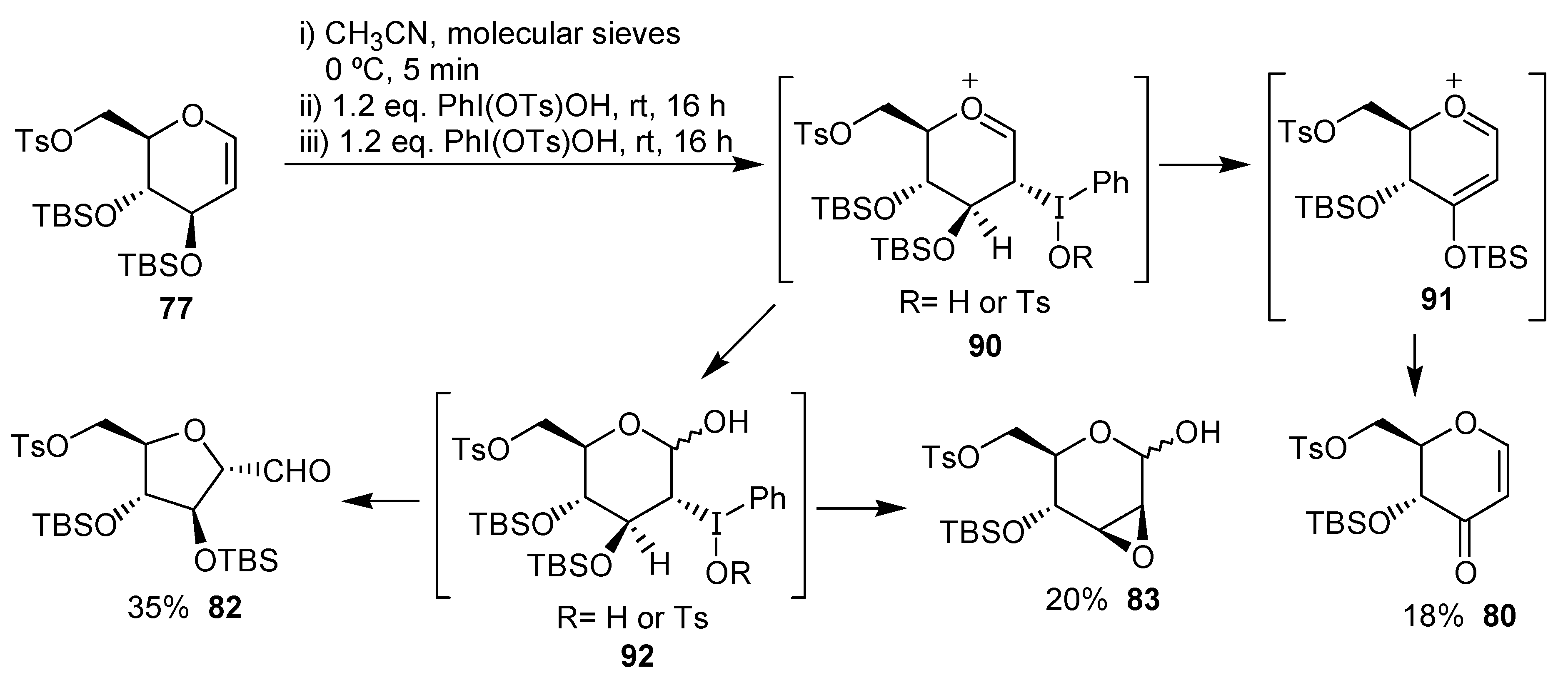
The mechanism proposed by the authors for the ring contraction of the carbohydrates derivatives is exemplified for
77 in
Scheme 21.
The first step is the electrophilic addition of the iodine(III) reagent to the unsaturation, giving the oxonium ion 90. This intermediate would give the enone 80 through 91, by a reductive elimination. Alternatively, the oxonium 90 may give, after addition of water, the adduct 92, on which would occur the rearrangement leading to the tetrahydrofuran 82 or an intramolecular reductive displacement giving 83.


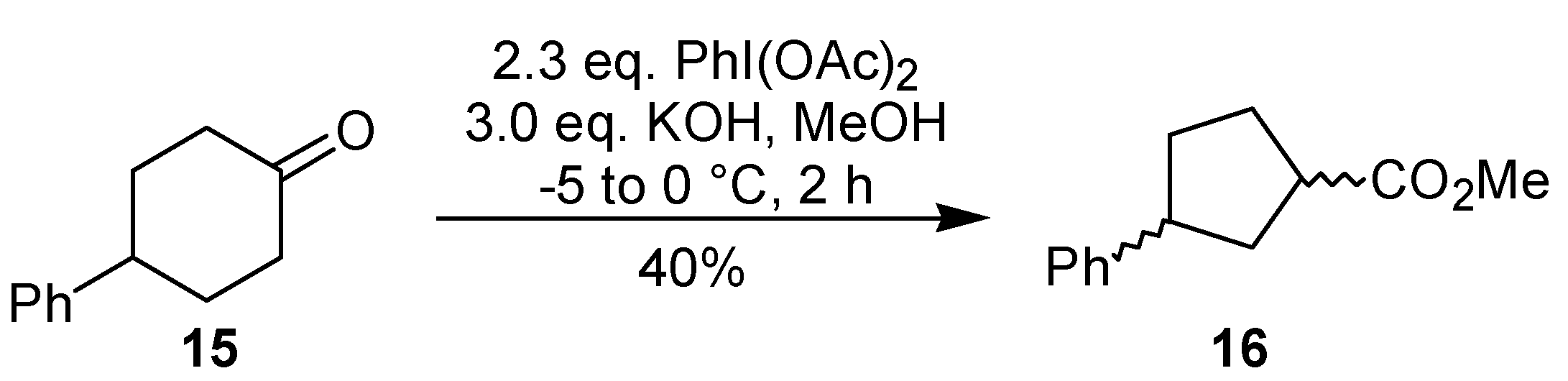













 , ClCH2CH2Cl, 25 °C, 15-20 h
, ClCH2CH2Cl, 25 °C, 15-20 h
 , AcOEt, LiClO4, rt, 2 h.
, AcOEt, LiClO4, rt, 2 h.