A Review of Injectable Polymeric Hydrogel Systems for Application in Bone Tissue Engineering
Abstract
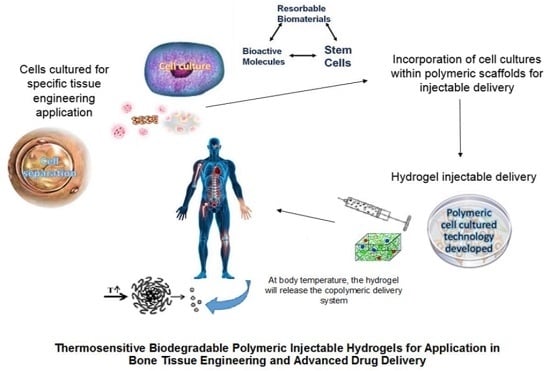
:1. Introduction
2. Approaches to the Synthetic Fabrication of Injectable Hydrogels for Application in Bone Tissue Engineering
2.1. Thermo-Induced Gelation: Classical Poly(NIPAAm)
2.2. Ionic-Mediated Gelation: The Stimulation of Alginate
2.3. Free Radical Polymerized Gelation: The Essential Vinyl Monomer
2.4. Self-Assembled Gelation: The Independent Amphiphiles
2.5. Chemically Crosslinked Gelation: The Sentimental Alkynes, Azides and Amines
3. Polymers Employed for the Synthesis of Stimuli-Responsive Hydrogel Systems
3.1. Natural Polymers
3.1.1. Hyaluronic Acid
3.1.2. Chitosan-Based Hydrogels
3.1.3. Cellulose-Derived Hydrogels
3.1.4. Other Natural Polymers
3.2. Synthetic Polymers
3.2.1. Polyacrylamide-Derived Thermo-Responsive Hydrogels
3.2.2. Poly(Oligo (Ethylene Glycol) Methacrylate)-Derived Thermo-Sensitive Hydrogels
3.2.3. Polyphosphazene-Derived Thermo-Sensitive Hydrogels
3.2.4. Pluronic-Derived Thermo-Sensitive Hydrogels
3.2.5. PEG-Polyester-Derived Thermo-Sensitive Hydrogels
4. Injectable/Implantable Research Technologies Employed in Bone Tissue Engineering Applications
4.1. Research Focused on Biomedical Applications
4.1.1. Bone Repair
4.1.2. Regeneration of Bone Tissue
5. Nano-Enabled Thermo-Responsive Hydrogel Systems
6. Advancing the Efficacy of Hydrogels in Drug Delivery Applications
6.1. Drug-Hydrogel Interactions
6.1.1. Physical Interactions
6.1.2. Covalent Bonding
7. Analogous Cell and Biomaterial Characterization for In Vitro Success and In Vivo Translation
8. Current Research Approaches to Date
9. Future Research Perspectives in the Field of Bone Tissue Engineering and Drug Delivery
10. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stolzing, A.; Jones, E.; McGonagle, D.; Scutt, A. Age-related changes in human bone marrow-derived mesenchymal stem cells: Consequences for cell therapies Mechanisms of Ageing and Development. Mech. Ageing Dev. 2008, 129, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Kofron, M.; Laurencin, C. Bone tissue engineering by gene delivery. Adv. Drug Deliv. Rev. 2006, 58, 555–576. [Google Scholar] [CrossRef] [PubMed]
- Arrington, E.; Smith, W.; Chambers, H.; Bucknell, A.; Davino, N. Complications of iliac crest bone graft harvesting. Clin. Orthop. Relat. Res. 1996, 329, 300–309. [Google Scholar] [CrossRef]
- Healy, K.E.; Guldberg, R.E. Bone tissue engineering. J. Musculoskelet. Neuronal Interact. 2007, 7, 328–330. [Google Scholar] [PubMed]
- Tran, R.T.; Gyawali, D.; Nair, P.; Yang, J. Synthesis, Degradation and Applications. Biodegradable Injectable Systems for Bone Tissue Engineering. Int. J. Polym. Sci. 2011, 14, 97–100. [Google Scholar]
- Ngiam, M.; Liao, S.; Patil, A.J.; Cheng, J.; Chan, C.K.; Ramakrishna, S. The fabrication of nano-hydroxyapatite on PLGA and PLGA/collagen nanofibrous composite scaffolds and their effects in osteoblastic behavior for bone tissue engineering. Bone 2009, 45, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Saravanan, S.; Pattnaik, S.; Moorthi, A.; Partridge, N.C.; Selvamurugan, N. Bio-composite scaffolds containing chitosan/nano-hydroxyapatite/nano-copper-zinc for bone tissue engineering. Int. J. Biol. Macromol. 2012, 50, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.C.D.; Mohamed, E.; Alberto, S.; Julie, E.G.; Aline, F.M. Osteogenic differentiation of human mesenchymal stem cells promotes mineralization within a biodegradable peptide hydrogel. J. Tissue Eng. 2016, 7, 1–15. [Google Scholar]
- Vacanti, J.P.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, S32–S34. [Google Scholar] [CrossRef]
- Khan, F.; Tanaka, M.; Ahmad, S.R. Fabrication of polymeric biomaterials: A strategy for tissue engineering and medical devices. J. Mater. Chem. B 2015, 3, 8224–8249. [Google Scholar] [CrossRef]
- Rabkin, E.; Schoen, E.F. Integration of concepts: Cardiac extracellular matrix remodeling after myocardial infarction. J. Card. Fail. 2002, 8, S344–S348. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue engineering: the challenges ahead. Sci. Am. 1999, 280, 62–65. [Google Scholar] [CrossRef]
- Hunt, N.C.; Grover, L.M. Cell encapsulation using biopolymer gels for regenerative medicine. Biotechnol. Lett. 2010, 32, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Kretlow, J.; Klouda, L.; Mikos, A. Injectable matrices and scaffolds for drug delivery in tissue engineering. Adv. Drug Deliv. Rev. 2007, 59, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Ifkovits, J.L.; Burdick, J.A. Photopolymerizable and Degradable Biomaterials for Tissue Engineering Applications. Tissue Eng. 2007, 13, 2369–2385. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Nuttelman, C.R.; Rice, M.A.; Rydholm, A.E.; Salinas, C.N.; Shah, D.N.; Anseth, K.S. Macromolecular monomers for the synthesis of hydrogel niches and their application in cell encapsulation and tissue engineering. Prog. Polym. Sci. 2008, 33, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue Engineering applications. Tissue Eng. 2008, 14, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Sivashanmugam, A.; Arun Kumar, R.; Vishnu Priya, M.; Nair, S.V.; Jayakumar, R. An overview of injectable polymeric hydrogels for tissue engineering. Eur. Polym. J. 2015, 72, 543–565. [Google Scholar] [CrossRef]
- Yu, F.; Cao, X.; Li, Y.; Zeng, L.; Yuan, B.; Chen, X. An injectable hyaluronic acid/PEG hydrogel for cartilage tissue engineering formed by integrating enzymatic crosslinking and Diels–Alder “click chemistry”. Polym. Chem. 2014, 5, 1082–1090. [Google Scholar] [CrossRef]
- Ramesh, N.; Alka, A.M. Biodegradable and biocompatible temperature sensitive triblock copolymer hydrogels as draw agents for forward osmosis. Sep. Purif. Technol. 2016, 168, 83–92. [Google Scholar]
- Brandl, F.; Sommer, F.; Goepferich, A. Rational design of hydrogels for tissue engineering: Impact of physical factors on cell behaviour. Biomaterials 2007, 28, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Ding, J. Injectable hydrogels as unique biomedical materials. Chem. Soc. Rev. 2008, 37, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.S.; Andrews, G.P.; Caldwell, D.L.; Lorimer, C.; Gorman, S.P.; McCoy, C.P. Novel semi-interpenetrating hydrogel networks with enhanced mechanical properties and thermoresponsive engineered drug delivery, designed as bioactive endotracheal tube biomaterials. Eur. J. Pharm. Biopharm. 2012, 82, 563–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coughlan, D.C.; Corrigan, O.I. Release kinetics of benzoic acid and its sodium salt from a series of poly(N-isopropylacrylamide) matrices with various percentage crosslinking. J. Pharm. Sci. 2008, 97, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Jhon, Y.K.; Bhat, R.R.; Jeong, C.; Rojas, O.J.; Szleifer, I.; Genzer, J. Salt-induced depression of lower critical solution temperature in a surface-grafted neutral thermoresponsive polymer. Macromol. Rapid Commun. 2006, 27, 697–701. [Google Scholar] [CrossRef]
- Jones, D.S.; Lorimer, C.P.; McCoy, C.P.; Gorman, S.P. Characterization of the physicochemical, antimicrobial, and drug release properties of thermoresponsive hydrogel copolymers designed for medical device applications. J. Biomed. Mater. Res. Part B 2008, 85, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.S.; Lorimer, C.J.; Andrews, G.P.; McCoy, C.P.; Gorman, S.P. An examination of the thermorheological and drug release properties of zinc tetraphenylporphyrin-containing thermoresponsive hydrogels, designed as light activated antimicrobial implants. Chem. Eng. Sci. 2007, 62, 990–999. [Google Scholar] [CrossRef]
- Yoshida, T.; Aoyagi, T.; Kokufuta, E.; Okano, T. Newly designed hydrogel with both sensitive thermoresponse and biodegradability. J. Polym. Sci. Part A 2003, 41, 779–787. [Google Scholar] [CrossRef]
- Bessa, P.C.; Machado, R.; Nurnberger, S.; Dopler, D.; Banerjee, A.; Cunha, A.M.; Rodriguez-Cabello, J.C.; Redl, H.; van Griensven, M.; Reis, R.L.; et al. Thermoresponsive self-assembled elastin-based nanoparticles for delivery of bmps. J. Control. Release 2010, 142, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Rincon, A.C.; Molina-Martinez, I.T.; de Las Heras, B.; Alonso, M.; Bailez, C.; Rodriguez-Cabello, J.C.; Herrero-Vanrell, R. Biocompatibility of elastin-like polymer poly(vpavg) microparticles: In vitro and in vivo studies. J. Biomed. Mater. Res. Part A 2006, 78, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.Q.; Qiu, F.; Wang, T.; Jiang, X.J.; Zhang, X.Z.; Zhuo, R.X. Toward the development of partially biodegradable and injectable thermoresponsive hydrogels for potential biomedical applications. ACS Appl. Mater. Interfaces 2009, 1, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Meenach, S.A.; Anderson, K.W.; Hilt, J.Z. Synthesis and characterization of thermoresponsive poly(ethylene glycol)-based hydrogels and their magnetic nanocomposites. J. Polym. Sci. Part A 2010, 48, 3229–3235. [Google Scholar] [CrossRef]
- Papaphilippou, P.C.; Pourgouris, A.; Marinica, O.; Taculescu, A.; Athanasopoulos, G.I.; Vekas, L.; Krasia-Christoforou, T. Fabrication and characterization of superparamagnetic and thermoresponsive hydrogels based on oleic-acid-coated Fe3O4 nanoparticles, hexa(ethylene glycol) methyl ether methacrylate and 2-(acetoacetoxy)ethyl methacrylate. J. Magn. Magn. Mater. 2011, 323, 557–563. [Google Scholar] [CrossRef]
- Ma, Z.W.; Nelson, D.M.; Hong, Y.; Wagner, W.R. Thermally responsive injectable hydrogel incorporating methacrylate-polylactide for hydrolytic lability. Biomacromolecules 2010, 11, 1873–1881. [Google Scholar] [CrossRef] [PubMed]
- Stabenfeldt, S.E.; Garcia, A.J.; LaPlaca, M.C. Thermoreversible laminin-functionalized hydrogel for neural tissue engineering. J. Biomed. Mater. Res. Part A 2006, 77A, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Dong, A.; Radosz, M.; Shen, Y.Q. Thermoresponsive degradable poly(ethylene glycol) analogues. J. Biomed. Mater. Res. Part A 2008, 84A, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Garty, S.; Kimelman-Bleich, N.; Hayouka, Z.; Cohn, D.; Friedler, A.; Pelled, G.; Gazit, D. Peptide-modified “smart” hydrogels and genetically engineered stem cells for skeletal tissue engineering. Biomacromolecules 2010, 11, 1516–1526. [Google Scholar] [CrossRef] [PubMed]
- Kan, P.; Lin, X.Z.; Hsieh, M.F.; Chang, K.Y. Thermogelling emulsions for vascular embolization and sustained release of drugs. J. Biomed. Mater. Res. Part B 2005, 75, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Jha, A.K.; Jackson, W.M.; Healy, K.E. Controlling Osteogenic Stem Cell Differentiation via Soft Bioinspired Hydrogels. PLoS ONE 2014, 9, 98640. [Google Scholar] [CrossRef] [PubMed]
- Leea, K.Y.; Mooneya, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Place, E.S.; Rojo, L.; Gentleman, E.; Sardinha, J.P.; Stevens, M.M. Strontium- and Zinc-Alginate Hydrogels for Bone Tissue Engineering. Tissue Eng. Part A 2011, 17, 2713–2722. [Google Scholar] [CrossRef] [PubMed]
- Russo, R.; Malinconico, M.; Santagata, G. Effect of Cross-Linking with Calcium Ions on the Physical Properties of Alginate Films. Biomacromolecules 2007, 8, 3193–3197. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.A.; Kim, M.; Kim, T.; Kim, J.; Lee, L.H.; Park, J.; Jonathan, C.; Kim, K.H. Utilizing Core–Shell Fibrous Collagen-Alginate Hydrogel Cell Delivery System for Bone Tissue Engineering. Tissue Eng. Part A 2014, 20, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Bulmus, V. RAFT polymerization mediated bioconjugation strategies. Polym. Chem. 2011, 2, 1463–1472. [Google Scholar] [CrossRef]
- Davis, K.A.; Burdick, J.A.; Anseth, K.S. Photoinitiated crosslinked degradable copolymer networks for tissue engineering applications. Biomaterials 2003, 24, 2485–2495. [Google Scholar] [CrossRef]
- Mahdieh, Z.; Bagheri, R.; Eslami, M.; Mohammad Amiri, M.; Shokrgozar, M.A.; Mehrjoo, M. Thermoplastic starch/ethylene vinyl alcohol/forsterite nanocomposite as a candidate material for bone tissue engineering. Mater. Sci. Eng. 2016, 69, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Tu, R.S.; Tirrell, M. Bottom-up design of biomimetic assemblies. Adv. Drug Deliv. Rev. 2004, 56, 1537–1563. [Google Scholar] [CrossRef] [PubMed]
- Wong Po Foo, C.T.; Lee, J.S.; Mulyasasmita, W.; Parisi-Amon, A.; Heilshorn, S.C. Two-component protein-engineered physical hydrogels for cell encapsulation. Proc. Natl. Acad. Sci. USA 2009, 106, 22067–22072. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A.; Kobayashi, H.J. Bone regeneration through controlled release of bone morphogenetic protein-2 from 3-D tissue engineered nano-scaffold. J. Control. Release 2007, 117, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A.; Kobayashi, H.; Tabata, Y. Enhanced angiogenesis through controlled release of basic fibroblast growth factor from peptide amphiphile for tissue regeneration. Biomaterials 2006, 27, 5836–5844. [Google Scholar] [CrossRef] [PubMed]
- Royals, M.A.; Fujita, S.M.; Yewey, G.L.; Rodriguez, J.; Schultheiss, P.C.; Dunn, R.L. Biocompatibility of a biodegradable in situ forming implant system in rhesus monkeys. J. Biomed. Mater. Res. 1999, 45, 231–239. [Google Scholar] [CrossRef]
- Strehin, I.; Nahas, Z.; Arora, K.; Nguyen, T.; Elisseeff, J. A versatile pH sensitive chondroitin sulfate–PEG tissue adhesive and hydrogel. Biomaterials 2010, 31, 2788–2797. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Jiang, D.; Yan, L.; Wu, J. In vitro and in vivo osteogenic activity of the novel vancomycin-loaded bone-like hydroxyapatite/poly(amino acid) scaffold. J. Biomater. Appl. 2016, 30, 1566–1577. [Google Scholar] [CrossRef] [PubMed]
- Sakai, S.; Hirose, K.; Taguchi, K.; Ogushi, Y.; Kawakami, K. An injectable, in situ enzymatically gellable, gelatin derivative for drug delivery and tissue engineering. Biomaterials 2009, 30, 3371–3377. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Hiemstra, C.; Zhong, Z.; Feijen, J. Enzyme-mediated fast in situ formation of hydrogels from dextran–tyramine conjugates. Biomaterials 2007, 28, 2791–2800. [Google Scholar] [CrossRef] [PubMed]
- Sperinde, J.; Griffith, L. Synthesis and Characterization of Enzymatically-Cross-Linked Poly(ethylene glycol) Hydrogels. Macromolecules 1997, 30, 5255–5264. [Google Scholar] [CrossRef]
- Goa, K.L.; Benfield, P. Hyaluronic acid. A review of its pharmacology and use as a surgical aid in ophthalmology, and its therapeutic potential in joint disease and wound healing. Drugs 1994, 47, 536–566. [Google Scholar] [CrossRef] [PubMed]
- Ohya, S.; Nakayama, Y.; Matsuda, T. Thermoresponsive artificial extracellular matrix for tissue engineering: Hyaluronic acid bioconjugated with poly(N-isopropylacrylamide) grafts. Biomacromolecules 2001, 2, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Mayol, L.; Quaglia, F.; Borzacchiello, A.; Ambrosio, L.; La Rotonda, M.I. A novel poloxamers/hyaluronic acid in situ forming hydrogel for drug delivery: Rheological, mucoadhesive and in vitro release properties. Eur. J. Pharm. Biopharm. 2008, 70, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.N.; Shah, S.R.; Lu, S.; Tatara, A.M.; Lee, E.J.; Roh, T.T.; Tabata, Y.; Mikos, A.G. Injectable dual-gelling cell-laden composite hydrogels for bone tissue engineering. Biomaterials 2016, 83, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yan, Y.; Wang, X.; Xiong, Z.; Lin, F.; Wu, R.; Zhang, R. Three-dimensional Gelatin and Gelatin/ Hyaluronan Hydrogel Structures for Traumatic Brain Injury. J. Bioact. Compat. Polym. 2007, 22, 19–29. [Google Scholar] [CrossRef]
- Hsu, S.H.; Leu, Y.L.; Hu, J.W.; Feng, J.Y. Physicochemical characterization and drug release of thermosensitive hydrogels composed of a hyaluronic acid/pluronic f127 graft. Chem. Pharm. Bull. 2009, 57, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Babo, P.S.; Santo, V.E.; Gomes, M.E.; Reis, R.L. Development of an Injectable Calcium Phosphate/Hyaluronic Acid Microparticles System for Platelet Lysate Sustained Delivery Aiming Bone Regeneration. Macromol. Biosci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Chenite, A.; Chaput, C.; Wang, D.; Combes, C.; Buschmann, M.D.; Hoemann, C.D.; Leroux, J.C.; Atkinson, B.L.; Binette, F.; Selmani, A. Novel injectable neutral solutions of chitosan form biodegradable gels in situ. Biomaterials 2000, 21, 2155–2161. [Google Scholar] [CrossRef]
- Shi, W.; Ji, Y.; Zhang, X.; Shu, S.; Wu, Z. Characterization of pH- and thermosensitive hydrogel as a vehicle for controlled protein delivery. J. Pharm. Sci. 2011, 100, 886–895. [Google Scholar] [CrossRef] [PubMed]
- Ruel-Gariepy, E.; Leclair, G.; Hildgen, P.; Gupta, A.; Leroux, J.C. Thermosensitive chitosan-based hydrogel containing liposomes for the delivery of hydrophilic molecules. J. Control. Release 2002, 82, 373–383. [Google Scholar] [CrossRef]
- Gordon, S.; Teichmann, E.; Young, K.; Finnie, K.; Rades, T.; Hook, S. In vitro and in vivo investigation of thermosensitive chitosan hydrogels containing silica nanoparticles for vaccine delivery. Eur. J. Pharm. Sci. 2010, 41, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, G.; Leroux, J.C.; Damas, J.; Adam, A. Biocompatibility of thermosensitive chitosan-based hydrogels: An in vivo experimental approach to injectable biomaterials. Biomaterials 2002, 23, 2717–2722. [Google Scholar] [PubMed]
- LogithKumar, R.; KeshavNarayan, A.; Dhivya, S.; Chawla, A.; Saravanan, S.; Selvamurugan, N. A review of chitosan and its derivatives in bone tissue engineering. Carbohydr. Polym. 2016, 151, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Yang, J.; Wu, H.; Hu, Z.; Yi, J.; Tong, J.; Zhu, X. Preparation and characterization of quaternary ammonium chitosan hydrogel with significant antibacterial activity. Int. J. Biol. Macromol. 2015, 79, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, C.; Qiao, C.; Mu, X.; Li, T.; Xu, J.; Shi, L.; Zhang, D. A simple and convenient method to synthesize N-[(2-hydroxyl)-propyl-3-trimethylammonium] chitosan chloride in an ionic liquid. Carbohydr. Polym. 2015, 130, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Teeri, T.T.; Brumer, H.; Daniel, G.; Gatenholm, P. Biomimetic engineering of cellulose-based materials. Trends Biotechnol. 2007, 25, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C. Control of thermogelation properties of hydrophobically-modified methylcellulose. J. Bioact. Compat. Polym. 2005, 20, 5–13. [Google Scholar] [CrossRef]
- Chen, H.; Fan, M. Novel thermally sensitive pH-dependent chitosan/ carboxymethyl cellulose hydrogels. J. Bioact. Compat. Polym. 2008, 23, 38–48. [Google Scholar] [CrossRef]
- Karewicz, A.; Zasada, K.; Szczubiałka, K.; Zapotoczny, S.; Lach, R.; Nowakowska, M. ‘Smart’ alginate-hydroxypropylcellulose microbeads for controlled release of heparin. Int. J. Pharm. 2010, 385, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Liu, M.; Ni, B.; Gao, C. A novel pH- and thermo-sensitive PVP/CMC semi-IPN hydrogel: Swelling, phase behavior, and drug release study. J. Polym. Sci. B Polym. Phys. 2010, 48, 1749–1756. [Google Scholar] [CrossRef]
- Atila, D.; Keskin, D.; Tezcaner, A. Crosslinked pullulan/cellulose acetate fibrous scaffolds for bone tissue engineering. Mater. Sci. Eng. 2016, 69, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Kleinman, H.K.; Martin, G.R. Matrigel: Basement membrane matrix with biological activity. Semin. Cancer Biol. 2005, 15, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Tai, K.F.; Chen, P.J.; Chen, D.S.; Hwang, L.H. Concurrent delivery of GM-CSF and endostatin genes by a single adenoviral vector provides a synergistic effect on the treatment of orthotopic liver tumors. J. Gene Med. 2003, 5, 386–398. [Google Scholar] [CrossRef] [PubMed]
- Le, U.M.; Shaker, D.S.; Sloat, B.R.; Cui, Z. A thermo-sensitive polymeric gel containing a gadolinium (Gd) compound encapsulated into liposomes significantly extended the retention of the Gd in tumors. Drug Dev. Ind. Pharm. 2008, 34, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Compte, M.; Alonso-Camino, V.; Santos-Valle, P.; Cuesta, A.M.; Sánchez-Martín, D.; López, M.R.; Vicario, J.L.; Salas, C.; Sanz, L.; Alvarez-Vallina, L. Factory neovessels: Engineered human blood vessels secreting therapeutic proteins as a new drug delivery system. Gene Ther. 2010, 17, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Velasco, M.A.; Narváez-Tovar, C.A.; Garzón-Alvarado, D.A. Design, Materials, and Mechanobiology of Biodegradable Scaffolds for Bone Tissue Engineering. BioMed Res. Int. 2015, 2015, 729076. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Bhunia, B.; Banerjee, I.; Datta, P.; Dhara, S.; Maiti, T.K. Enzymatically crosslinked carboxymethyl–chitosan/gelatin/nano-hydroxyapatite injectable gels for in situ bone tissue engineering application. Mater. Sci. Eng. 2011, 31, 1295–1304. [Google Scholar] [CrossRef]
- Puértolas, J.A.; Vadillo, J.L.; Sánchez-Salcedo, S.; Nieto, A.; Gómez-Barrena, E.; Vallet-Regí, M. Compression behavior of biphasic calcium phosphate and biphasic calcium phosphate–agarose scaffolds for bone regeneration. Acta Biomater. 2011, 7, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Vihola, H.; Laukkanen, A.; Valtola, L.; Tenhu, H.; Hirvonen, J. Cytotoxicity of thermosensitive polymers poly(N-isopropylacrylamide), poly(Nvinylcaprolactam) and amphiphilically modified poly(N-vinylcaprolactam). Biomaterials 2005, 26, 3055–3064. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, J.; Nakayama, M.; Sakai, K.; Okano, T. Temperature-induced intracellular uptake of thermoresponsive polymeric micelles. Biomacromolecules 2009, 10, 1331–1336. [Google Scholar] [CrossRef] [PubMed]
- Alzari, V.; Monticelli, O.; Nuvoli, D.; Kenny, J.M.; Mariani, A. Stimuli responsive hydrogels prepared by frontal polymerization. Biomacromolecules 2009, 10, 2672–2677. [Google Scholar] [CrossRef] [PubMed]
- Censi, R.; Vermonden, T.; Deschout, H.; Braeckmans, K.; di Martino, P.; De Smedt, S.C.; van Nostrum, C.F.; Hennink, W.E. Photopolymerized thermosensitive poly(HPMAlactate)-PEG-based hydrogels: Effect of network design on mechanical properties, degradation, and release behavior. Biomacromolecules 2010, 11, 2143–2151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misra, G.P.; Singh, R.S.J.; Aleman, T.S.; Jacobson, S.G.; Gardner, T.W.; Lowe, T.L. Subconjunctivally implantable hydrogels with degradable and thermoresponsive properties for sustained release of insulin to the retina. Biomaterials 2009, 30, 6541–6547. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.T.; Keller, T.F.; Bhat, R.; Garipcan, B.; Jandt, K.D. A novel two-level microstructured poly(N-isopropylacrylamide) hydrogel for controlled release. Acta Biomater. 2010, 6, 3890–3898. [Google Scholar] [CrossRef] [PubMed]
- Kabanov, A.V.; Batrakova, E.V.; Alakhov, V.Y. Pluronic block copolymers as novel polymer therapeutics for drug and gene delivery. J. Control. Release 2002, 82, 189–212. [Google Scholar] [CrossRef]
- Hsiue, G.H.; Chang, R.W.; Wang, C.H.; Lee, S.H. Development of in situ thermosensitive drug vehicles for glaucoma therapy. Biomaterials 2003, 24, 2423–2430. [Google Scholar] [CrossRef]
- Kim, Y.S.; Gil, E.S.; Lowe, T.L. Synthesis and characterization of thermoresponsiveco-biodegradable linear-dendritic copolymers. Macromolecules 2006, 39, 7805–7811. [Google Scholar] [CrossRef]
- Yu, J.; Yang, L.; Liang, X.; Dong, T.; Qu, H.; Rong, M.; Liu, H. Aptamer and PNIPAAm co-conjugated nanoparticles regulate activity of enzyme with different temperature. Talanta 2016, 159, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.C.; Kim, J.W.; Hyun, D.C.; Jeong, U.; Weitz, D.A. Regulating volume transitions of highly responsive hydrogel scaffolds by adjusting the network properties of microgel building block colloids. Langmuir 2010, 26, 3854–3859. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhuang, X.; He, P.; Xiao, C.; He, C.; Sun, J.; Chen, X.; Jing, X. Synthesis of biodegradable thermo- and pH-responsive hydrogels for controlled drug release. Polymer 2009, 50, 4308–4316. [Google Scholar] [CrossRef]
- Ankareddi, I.; Bailey, M.M.; Brazel, C.S.; Rasco, J.F.; Hood, R.D. Developmental toxicity assessment of thermoresponsive poly(Nisopropylacrylamide-co-acrylamide) oligomers in CD-1 mice. Birth Defects Res. B Dev. Reprod. Toxicol. 2008, 83, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Hong, Y.; Ma, Z.; Wagner, W.R. Protein-reactive, thermoresponsive copolymers with high flexibility and biodegradability. Biomacromolecules 2008, 9, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Wu, W.; Zhou, S. Engineering oligo(ethylene glycol)-based thermosensitive microgels for drug delivery applications. Polymer 2010, 51, 3926–3933. [Google Scholar] [CrossRef]
- Ahn, S.; Monge, E.C.; Song, S.C. Ion and pH effect on the lower critical solution temperature phase behavior in neutral and acidic poly(organophosphazene) counterparts. Langmuir 2009, 25, 2407–2418. [Google Scholar] [CrossRef] [PubMed]
- Al-Abd, A.M.; Hong, K.Y.; Song, S.C.; Kuh, H.J. Pharmacokinetics of doxorubicin after intratumoral injection using a thermosensitive hydrogel in tumor-bearing mice. J. Control. Release 2010, 142, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kumbar, S.G.; Bhattacharyya, S.; Nukavarapu, S.P.; Khan, Y.M.; Nair, L.S.; Laurencin, C.T. In vitro and in vivo characterization of biodegradable poly(organophosphazenes) for biomedical applications. J. Inorg. Organomet. Plym. Mater. 2006, 16, 365–385. [Google Scholar] [CrossRef]
- Teasdale, I.; Brüggemann, O. Polyphosphazenes: Multifunctional, Biodegradable Vehicles for Drug and Gene Delivery. Polymers 2013, 5, 161–187. [Google Scholar] [CrossRef] [PubMed]
- Allcock, H.R.; Morozowich, N.L. Investigation of Apatite Mineralization on Antioxidant Polyphosphazenes for Bone Tissue Engineering. Polym. Chem. 2012, 3, 578–590. [Google Scholar] [CrossRef]
- Muhammad, A.; Haojie, Y.; Li, W.; Hamad, K.; Nasir, M.A.; Zain-ul-Abdin; Yongsheng, C.; Fujie, Ren.; Muhammad, S. Sustained release of hydrophilic drug from polyphosphazenes/poly(methyl methacrylate) based microspheres and their degradation study. Mater. Sci. Eng. 2016, 58, 169–179. [Google Scholar]
- Choi, W.I.; Yoon, K.C.; Im, S.K.; Kim, Y.H.; Yuk, S.H.; Tae, G. Remarkably enhanced stability and function of core/shell nanoparticles composed of a lecithin core and a pluronic shell layer by photo-crosslinking the shell layer: In vitro and in vivo study. Acta Biomater. 2010, 6, 2666–2673. [Google Scholar] [CrossRef] [PubMed]
- Gil, E.; Hudson, S. Stimuli-reponsive polymers and their bioconjugates. Polymer 2004, 29, 1173–1222. [Google Scholar] [CrossRef]
- Niu, G.; Du, F.; Song, L.; Zhang, H.; Yang, J.; Cao, H.; Zheng, Y.; Yang, Z.; Wang, G.; Yang, H.; et al. Synthesis and characterization of reactive poloxamer 407s for biomedical applications. J. Control. Release 2009, 138, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Clapper, J.; Skeie, J.; Mullins, R.; Guymon, C.A. Development and characterization of photopolymerizable biodegradable materials from PEG--PLA--PEG block macromonomers. Polymer 2007, 48, 6554–6564. [Google Scholar] [CrossRef]
- Yang, Z.; Ding, J. A thermosensitive and biodegradable physical gel with chemically crosslinked nanogels as the building block. Macromol. Rapid. Commun. 2008, 29, 751–756. [Google Scholar] [CrossRef]
- Guo, D.D.; Xu, C.X.; Quan, J.S.; Song, C.K.; Jin, H.; Kim, D.D.; Choi, Y.J.; Cho, M.H.; Cho, C.S. Synergistic anti-tumor activity of paclitaxel-incorporated conjugated linoleic acid-coupled poloxamer thermosensitive hydrogel in vitro and in vivo. Biomaterials 2009, 30, 4777–4785. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lu, W.L.; Wang, J.C.; Zhang, X.; Zhang, H.; Wang, X.Q.; Zhou, T.Y.; Zhang, Q. Controlled delivery of recombinant hirudin based on thermo-sensitive Pluronic F127 hydrogel for subcutaneous administration: In vitro and in vivo characterization. J. Control. Release 2007, 117, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Kondiah, P.P.D.; Choonara, Y.E.; Tomar, L.K.; Tyagi, C.; du Toit, L.S.; Kumar, P.; Modi, G.; Pillay, V. Novel pH sensitive oral Interferon-β delivery system for application in Multiple Sclerosis. Int. J. Pharm. 2013, 456, 459–472. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, J.; Oh, S.; Kim, S.; Hah, Y.; Park, B.; Deok Ryong Kim, D.; Gyu-Jin Rho, G.; Maeng, G.; Jeon, G.; et al. Tissue-engineered bone formation using periosteal-derived cells and polydioxanone/pluronic F127 scaffold with pre-seeded adipose tissue-derived CD146 positive endothelial-like cells. Biomaterials 2011, 32, 5033–5045. [Google Scholar] [CrossRef] [PubMed]
- Buwalda, S.J.; Dijkstra, P.J.; Calucci, L.; Forte, C.; Feijen, J. Influence of amide versus ester linkages on the properties of eight-armed PEGPLA star block copolymer hydrogels. Biomacromolecules 2010, 11, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Toyoda, H.; Namikawa, T.; Hoshino, M.; Terai, H.; Miyamoto, S.; Takaoka, K. Optimized use of a biodegradable polymer as a carrier material for the local delivery of recombinant human bone morphogenetic protein-2 (rhBMP-2). Biomaterials 2006, 27, 2035–2041. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.S.; Kim, J.H.; Park, H.; Kim, K.; Chan Kwon, I.; Lee, D.S. Biodegradability and biocompatibility of a pH- and thermosensitive hydrogel formed from a sulfonamide-modified poly(epsilon-caprolactone-co-lactide)-poly(ethylene glycol)-poly(epsiloncaprolactone-co-lactide) block copolymer. Biomaterials 2006, 27, 5178–5185. [Google Scholar] [CrossRef] [PubMed]
- Changjiang, F.; Dong-An, W. A biodegradable PEG-based micro-cavitary hydrogel as scaffold for cartilage tissue engineering. Eur. Polym. J. 2015, 72, 651–660. [Google Scholar]
- Jiang, W.W.; Su, S.H.; Eberhart, R.C.; Tang, L. Phagocyte responses to degradable polymers. J. Biomed. Mater. Res. A 2007, 82, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Klouda, L. Thermoresponsive hydrogels in biomedical applications. A seven-year update. Eur. J. Pharm. Biopharm. 2015, 97, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Petit, A.; Sandker, M.; Muller, B.; Meyboom, R.; van Midwoud, P.; Bruin, P.; Redout, E.M.; Versluijs-Helder, M.; van der Lest, C.H.; Buwalda, S.J.; et al. Release behavior and intraarticular biocompatibility of celecoxib-loaded acetyl-capped PCLA-PEG-PCLA thermogels. Biomaterials 2014, 35, 7919–7928. [Google Scholar] [CrossRef] [PubMed]
- Sandker, M.J.; Petit, A.; Redout, E.M.; Siebelt, M.; Muller, B.; Bruin, P.; Meyboom, R.; Vermonden, T.; Hennink, W.E.; Weinans, H. In situ forming acyl-capped PCLA-PEG- PCLA triblock copolymer based hydrogels. Biomaterials 2013, 34, 8002–8011. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lai, Y.; Yu, L.; Ding, J. Effects of immobilizing sites of RGD peptides in amphiphilic block copolymers on efficacy of cell adhesion. Biomaterials 2010, 31, 7873–7882. [Google Scholar] [CrossRef] [PubMed]
- Karam, J.P.; Muscari, C.; Sindji, L.; Bastiat, G.; Bonafe, F.; Venier-Julienne, M.C.; Montero-Menei, N.C. Pharmacologically active microcarriers associated with thermosensitive hydrogel as a growth factor releasing biomimetic 3D scaffold for cardiac tissue-engineering. J. Control. Release 2014, 192, 82–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, C.; Su, P.; Chen, X.; Meng, Y.; Yu, W.; Xiang, A.P.; Wang, Y. Biocompatibility and osteogenesis of biomimetic bioglass-collagen-phosphatidylserine composite scaffolds for bone tissue engineering. Biomaterials 2011, 32, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Marelli, B.; Ghezzi, C.E.; Mohn, D.; Stark, W.J.; Barralet, J.E.; Boccaccini, A.R.; Nazhat, S.N. Accelerated mineralization of dense collagen-nano bioactive glass hybrid gels increases scaffold stiffness and regulates osteoblastic function. Biomaterials 2011, 32, 8915–8926. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, E.; Partap, S.; Azevedo, M.M.; Jell, G.; Stevens, M.M.; O’Brien, F.J. Hypoxia-mimicking bioactive glass/collagen glycosaminoglycan composite scaffolds to enhance angiogenesis and bone repair. Biomaterials 2015, 52, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Moreira, C.D.; Carvalho, S.M.; Mansur, H.S.; Pereira, M.M. Thermogelling chitosan–collagen–bioactive glass nanoparticle hybrids as potential injectable systems for tissue engineering. Mater. Sci. Eng. 2016, 58, 1207–1216. [Google Scholar] [CrossRef] [PubMed]
- Kuttappan, S.; Mathew, D.; Nair, M.B. Biomimetic composite scaffolds containing bioceramics and collagen/gelatin for bone tissue engineering—A mini review. Int. J. Biol. Macromol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Niranjan, R.; Koushik, C.; Saravanan, S.; Moorthi, A.; Vairamani, M.; Selvamurugan, N. A novel injectable temperature-sensitive zinc doped chitosan/β-glycerophosphate hydrogel for bone tissue engineering. Int. J. Biol. Macromol. 2013, 54, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Lü, S.; Cao, Z.; Gao, C.; Duan, H.; Xu, X.; Sun, L.; Gao, N.; Feng, C.; Liu, M. Self-reinforcing injectable hydrogel with both high water content and mechanical strength for bone repair. Chem. Eng. J. 2016, 288, 546–556. [Google Scholar] [CrossRef]
- Vo, T.N.; Ekenseair, A.K.; Spicer, P.P.; Watson, B.M.; Tzouanas, S.N.; Roh, T.T.; Mikos, A.G. In vitro and in vivo evaluation of self-mineralization and biocompatibility of injectable, dual-gelling hydrogels for bone tissue engineering. J. Control. Release 2015, 205, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Ahlin Grabnar, P.; Kristl, J. The manufacturing techniques of drug-loaded polymeric nanoparticles from preformed polymers. J. Microencapsul. 2011, 28, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Barichello, J.M.; Morishita, M.; Takayama, K.; Nagai, T. Absorption of insulin from Pluronic F-127 gels following subcutaneous administration in rats. Int. J. Pharm. 1999, 184, 189–198. [Google Scholar] [CrossRef]
- Peng, Q.; Sun, X.; Gong, T.; Wu, C.Y.; Zhang, T.; Tan, J.; Zhang, Z.R. Injectable and biodegradable thermosensitive hydrogels loaded with PHBHHx nanoparticles for the sustained and controlled release of insulin. Acta Biomater. 2013, 9, 5063–5069. [Google Scholar] [CrossRef] [PubMed]
- Gou, M.; Li, X.; Dai, M.; Gong, C.; Wang, X.; Xie, Y.; Deng, H.; Chen, L.; Zhao, X.; Qian, Z.; et al. A novel injectable local hydrophobic drug delivery system: Biodegradable nanoparticles in thermo-sensitive hydrogel. Int. J. Pharm. 2008, 359, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Licciardi, M.; Amatoa, G.; Cappellic, A.; Paolinoc, M.; Giulianic, G.; Belmonted, B.; Guarnottad, C.; Pitarresi, G.; Giammona, G. Evaluation of thermoresponsive properties and biocompatibility of polybenzofulvene aggregates for leuprolide delivery. Int. J. Pharm. 2012, 438, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Lien, Y.H.; Wu, J.H.; Liao, J.W.; Wu, T.M. In vitro evaluation of the thermosensitive and magnetic nanoparticles for the controlled drug delivery of vitamin D3. Macromol. Res. 2013, 21, 511–518. [Google Scholar] [CrossRef]
- Lien, Y.H.; Wu, T.M. Preparation and characterization of thermosensitive polymers grafted onto silica-coated iron oxide nanoparticles. J. Colloid Interface Sci. 2008, 326, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Lien, Y.H.; Wu, T.M.; Wu, J.H.; Liao, J.W. Cytotoxicity and drug release behaviour of PNIPAM grafted on silica-coated iron oxide nanoparticles. J. Nanopart. Res. 2011, 13, 5065–5075. [Google Scholar] [CrossRef]
- Li, Y.; Pan, S.; Zhang, W.; Du, Z. Novel thermo-sensitive core-shell nanoparticles for targeted paclitaxel delivery. Nanotechnology 2009, 20, 065104. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Uchida, R.; Tanigawa, H.; Uno, K.; Murakami, A. Application of polymer gels containing side-chain phosphate groups to drug-delivery contact lenses. J. Appl. Polym. Sci. 2005, 98, 731–735. [Google Scholar] [CrossRef]
- Rodriguez, R.; Alvarez-Lorenzo, C.; Concheiro, A. Interactions of ibuprofen with cationic polysaccharides in aqueous dispersions and hydrogels: Rheological and diffusional implications. Eur. J. Pharm. Sci. 2003, 20, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.G.; Akerman, B.; Renck, H.; Lindberg, B. Effects of macromolecular adjuvants on the duration of prilocaine. Acta Anaesthesiol. Scand. 1988, 32, 355–364. [Google Scholar]
- Doherty, M.M.; Hughes, P.J.; Korszniak, N.V.; Charman, W.N. Prolongation of Lidocaine-Induced Epidural Anesthesia by Medium Molecular Weight Hyaluronic Acid Formulations: Pharmacodynamic and Pharmacokinetic Studies in the Rabbit. Anesth. Analg. 1995, 80, 740–746. [Google Scholar] [PubMed]
- Johansson, A.; Hassan, H.; Renck, H. Effects of Adjuvants to Local Anaesthetics on Their Duration. Acta Anaesthesiol. Scand. 1985, 29, 735–379. [Google Scholar] [CrossRef]
- Zumbuehl, A.; Ferreira, L.; Kuhn, D.; Astashkina, A.; Long, L.; Yeo, Y.; Iaconis, T.; Ghannoum, M.; Fink, G.R.; Langer, R.; et al. Antifungal hydrogels. Proc. Natl. Acad. Sci. USA 2007, 104, 12994–12998. [Google Scholar] [CrossRef] [PubMed]
- Nuttelman, C.R.; Tripodi, M.C.; Anseth, K.S. Dexamethasone-functionalized gels induce osteogenic differentiation of encapsulated hMSCs. J. Biomed. Mater. Res. Part A 2006, 76, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Feeney, M.; Giannuzzo, M.; Paolicelli, P.; Casadei, M.A. Hydrogels of Dextran Containing Nonsteroidal Anti-Inflammatory Drugs as Pendant Agents. Drug Deliv. 2007, 14, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Schoenmakers, R.G.; van de Wetering, P.; Elbert, D.L.; Hubbell, J.A. The effect of the linker on the hydrolysis rate of drug-linked ester bonds. J. Control. Release 2004, 95, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Sims, D.; Butler, P.E.; Casanova, R.; Lee, B.T.; Randolph, M.A.; Lee, W.P.; Vacanti, C.A.; Yaremchuk, M.J. Injectable cartilage using polyethylene oxide polymer substrates. Plast. Reconstr. Surg. 1996, 98, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Bryant, S.J.; Nuttelman, C.R.; Anseth, K.S. The effects of crosslinking density on cartilage formation in photocrosslinkable hydrogels. Biomed. Sci. Instrum. 1999, 35, 309–314. [Google Scholar] [PubMed]
- Bryant, S.J.; Anseth, K.S. Controlling the spatial distribution of ECM components in degradable PEG hydrogels for tissue engineering cartilage. J. Biomed. Mater. Res. 2003, 64, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Ronga, M.; Grassi, F.A.; Bulgheroni, P. Arthroscopic autologous chondrocyte implantation for the treatment of a chondral defect in the tibial plateau of the knee. Arthroscopy 2004, 20, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Silverman, R.; Passaretti, D.; Haung, W.; Randolph, M.A.; Yaremchuk, M.J. Injectable tissue-engineered cartilage using a fibrin glue polymer. Plast. Reconstr. Surg. 1999, 103, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Ting, V.; Sims, C.D.; Brecht, L.E.; McCarthy, J.G.; Kasabian, A.K.; Connelly, P.R.; Elisseeff, J.; Gittes, G.K.; Longaker, M.T. In vitro prefabrication of human cartilage shapes using fibrin glue and human chondrocytes. Ann. Plast. Surg. 1998, 40, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Isogai, N.; Landis, W.J.; Mori, R.; Gotoh, Y.; Gerstenfeld, L.C.; Upton, J.; Vacanti, J.P. Experimental use of fibrin glue to induce site-directed osteogenesis from cultured periosteal cells. Plast. Reconstr. Surg. 2000, 105, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Thirion, S.; Berenbaum, F. Culture and phenotyping of chondrocytes in primary culture. Methods Mol. Med. 2004, 100, 1–14. [Google Scholar] [PubMed]
- Kafienah, W.; Jakob, M.; Demarteau, O.; Frazer, A.; Barker, M.D.; Martin, I.; Hollander, A.P. Three-dimensional tissue engineering of hyaline cartilage: Comparison of adult nasal and articular chondrocytes. Tissue Eng. 2002, 8, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.H.; Schumacher, B.L.; Klein, T.J.; Thonar, E.J.; Masuda, K.; Sah, R.L.; Watson, D. Tissue-engineered human nasal septal cartilage using the alginate-recovered-chondrocyte method. Laryngoscope 2004, 114, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Saadeh, P.B.; Brent, B.; Mehrara, B.J.; Steinbrech, D.S.; Ting, V.; Gittes, G.K.; Longaker, M.T. Human cartilage engineering: Chondrocyte extraction, proliferation, and characterization for construct development. Ann. Plast. Surg. 1999, 42, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Fussenegger, M.; Meinhart, J.; Hobling, W.; Kullich, W.; Funk, S.; Bernatzky, G. Stabilized autologous fibrin-chondrocyte constructs for cartilage repair in vivo. Ann. Plast. Surg. 2003, 51, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.W.; Zaporojan, V.; Peretti, G.M.; Roses, R.E.; Morse, K.B.; Roy, A.K.; Mesa, J.M.; Randolph, M.A.; Bonassar, L.J.; Yaremchuk, M.J. Injectable tissue-engineered cartilage with different chondrocyte sources. Plast. Reconstr. Surg. 2004, 113, 1361–1371. [Google Scholar] [CrossRef] [PubMed]
- Mercier, N.R.; Costantino, H.R.; Tracy, M.A.; Bonassar, L.J. A novel injectable approach for cartilage formation in vivo using PLG microspheres. Ann. Biomed. Eng. 2004, 32, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Watson, B.M.; Vo, T.N.; Tatara, A.M.; Shah, S.R.; Scott, D.W.; Engel, P.S.; Mikos, A.G. Biodegradable, phosphate-containing, dual-gelling macromers for cellular delivery in bone tissue engineering. Biomaterials 2015, 67, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; An, G.; Liang, M.; Liu, Y.; Zhang, B.; Wang, Y. A composited PEG-silk hydrogel combining with polymeric particles delivering rhBMP-2 for bone regeneration. Mater. Sci. Eng. 2016, 65, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Kawataa, M.; Azumab, K.; Izawaa, H.; Morimotoa, M.; Saimotoa, H.; Ifukua, S. Biomineralization of calcium phosphate crystals on chitin nanofiber hydrogel for bone regeneration material. Carbohydr. Polym. 2016, 136, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Cui, N.; Qian, J.; Liu, T.; Zhao, N.; Wang, H. Hyaluronic acid hydrogel scaffolds with a triple degradation behavior for bone tissue engineering. Carbohydr. Polym. 2015, 126, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.D.; Jang, H.L.; Ahn, H.; Lee, H.K.; Park, J.; Lee, E.; Lee, E.A.; Jeong, Y.; Kim, D.; Nam, K.T.; et al. Biomimetic whitlockite inorganic nanoparticles-mediated in situ remodeling and rapid bone regeneration. Biomaterials 2017, 112, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Shankhwar, N.; Kumar, M.; Mandal, B.B.; Srinivasan, A. Novel polyvinyl alcohol-bioglass 45S5 based composite nanofibrous membranes as bone scaffolds. Mater. Sci. Eng. 2016, 69, 1167–1174. [Google Scholar] [CrossRef] [PubMed]





| Hydrogel Source | Polymer/s Used | Gelation Mechanism | References |
|---|---|---|---|
| Natural | Hyaluronic acid, chitosan | Thermal/chemical/free radical crosslinking | [59,65,66,68] |
| Cellulose, Agarose, Matrigel | Thermal crosslinking | [75,76,78] | |
| Gelatin, Collagen | Thermal/chemical crosslinking | [81,82,83] | |
| Synthetic | PDMA, PHPMA, PNVCL | ||
| PNIPAAm, Fibroin, PLA | |||
| NIPAAm/MBAAm | |||
| Oligolactide-(2-HEMA) | Thermal crosslinking | [87,88,91,92], | |
| OligoLA-HEMA, L-Lysine | [97,98] | ||
| PNIPAAm-Co-HEMA) | |||
| P(L-glutamic acid) | |||
| POEGMA, OEGMA, MEO2MA | ATRP crosslinking | [101,102,103] | |
| Poly(organophosphazene) | |||
| a-amino-w-methoxypoly(ethylene glycol), lleoEt | Chemical crosslinking | [104,106,108] | |
| PEO-PPO-PEO, mPEG-PCL | |||
| PEG, PGA, PLA, PCL, PLGA | Thermal crosslinking | [111,112,114], | |
| PVL, DX, SMO, PCLLA | [117] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kondiah, P.J.; Choonara, Y.E.; Kondiah, P.P.D.; Marimuthu, T.; Kumar, P.; Du Toit, L.C.; Pillay, V. A Review of Injectable Polymeric Hydrogel Systems for Application in Bone Tissue Engineering. Molecules 2016, 21, 1580. https://doi.org/10.3390/molecules21111580
Kondiah PJ, Choonara YE, Kondiah PPD, Marimuthu T, Kumar P, Du Toit LC, Pillay V. A Review of Injectable Polymeric Hydrogel Systems for Application in Bone Tissue Engineering. Molecules. 2016; 21(11):1580. https://doi.org/10.3390/molecules21111580
Chicago/Turabian StyleKondiah, Pariksha J., Yahya E. Choonara, Pierre P. D. Kondiah, Thashree Marimuthu, Pradeep Kumar, Lisa C. Du Toit, and Viness Pillay. 2016. "A Review of Injectable Polymeric Hydrogel Systems for Application in Bone Tissue Engineering" Molecules 21, no. 11: 1580. https://doi.org/10.3390/molecules21111580