Iron Supply Affects Anthocyanin Content and Related Gene Expression in Berries of Vitis vinifera cv. Cabernet Sauvignon
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Different Iron Levels on Berry Physical and Chemical Characteristics
2.2. Effect of Different Iron Levels on Berry Anthocyanin Composition
2.3. Expression of the Flavonoid Pathway Genes
3. Materials and Methods
3.1. Plant Materials and Treatments
3.2. Reducing Sugar (RS) and Titratable Acid Content (TAC) Assay
3.3. Assay for Anthocyanins
3.3.1. Extraction of Anthocaynins
3.3.2. HPLC-MS Analyses of Anthocyanins
3.3.3. Qualitative and Quantitative Analysis of Anthocyanins
3.4. RNA Extraction and Gene Expression Analysis
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tato, L.; De Nisi, P.; Donnini, S.; Zocchi, G. Low iron availability and phenolic metabolism in a wild plant species (Parietaria judaica L.). Plant Physiol. Biochem. 2013, 72, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Bertamini, M.; Nedunchezhian, N. Grapevine growth and physiological responses to iron deficiency. J. Plant Nutr. 2005, 28, 737–749. [Google Scholar] [CrossRef]
- Legay, S.; Guignard, C.; Ziebel, J.; Evers, D. Iron uptake and homeostasis related genes in potato cultivated in vitro under iron deficiency and overload. Plant Physiol. Biochem. 2012, 60, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.S.; Smith, B.R.; Cheng, L.L. CO2 assimilation, photosynthetic enzymes, and carbohydrates of ‘concord’ grape leaves in response to iron supply. J. Am. Soc. Hort. Sci. 2004, 129, 738–744. [Google Scholar]
- Briat, J.F.; Fobis-Loisy, I.; Grignon, N.; Lobréaux, S.; Pascal, N.; Savino, G.; Thoiron, S.; von Wirén, N.; van Wuytswinkel, O. Cellular and molecular aspects of iron metabolism in plants. Biol. Cell 1995, 84, 69–81. [Google Scholar] [CrossRef]
- Buchanan, B.B. Regulation of CO2 assimilation in oxygenic photosynthesis: The ferredoxin/thioredoxin system. Arch. Biochem. Biophys. 1991, 288, 1–9. [Google Scholar] [CrossRef]
- Terry, N. Limiting factors in photosynthesis. Plant Physiol. 1980, 65, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Bertamini, M.; Nedunchezhian, N.; Borghi, B. Effect of iron deficiency induced changes on photosynthetic pigments, ribulose-1,5-bisphosphate carboxylase, and photosystem activities in field grown grapevine (Vitis vinifera L. cv. Pinot noir) leaves. Photosynth 2001, 39, 59–65. [Google Scholar] [CrossRef]
- Morales, F.; Abadía, A.; Belkhodja, R.; Abadía, J. Iron deficiency-induced changes in the photosynthetic pigment composition of field-grown pear (Pyrus communis L.) leaves. Plant Cell Environ. 1994, 17, 1153–1160. [Google Scholar] [CrossRef]
- Álvarez-Fernández, A.; Melgar, J.C.; Abadía, J.; Abadía, A. Effects of moderate and severe iron deficiency chlorosis on fruit yield, appearance and composition in pear (Pyrus communis L.) and peach (Prunus persica (L.) Batsch). Environ. Exp. Bot. 2011, 71, 280–286. [Google Scholar] [CrossRef]
- Álvarez-Fernández, A.; Paniagua, P.; Abadía, J.; Abadía, A. Effects of Fe deficiency chlorosis on yield and fruit quality in peach (Prunus persica L. Batsch). J. Agric. Food Chem. 2003, 51, 5738–5744. [Google Scholar] [CrossRef] [PubMed]
- Mellisho, C.D.; González-Barrio, R.; Ferreres, F.; Ortuño, M.F.; Conejero, W.; Torrecillas, A.; García-Mina, J.M.; Medina, S.; Gil-Izquierdo, A. Iron deficiency enhances bioactive phenolics in lemon juice. J. Sci. Food Agric. 2011, 91, 2132–2139. [Google Scholar] [CrossRef] [PubMed]
- Amiri, M.E.; Fallahi, E. Influence of mineral nutrients on growth, yield, berry quality, and petiole mineral nutrient concentrations of table grape. J. Plant Nutr. 2007, 30, 463–470. [Google Scholar] [CrossRef]
- Ahmed, F.F.; Akl, A.M.; El-Morsy, F.M. Yield and quality of ‘banaty’ grapes in response to spraying iron and zinc. Hortscience 1997, 32, 516. [Google Scholar]
- Chalker-Scott, L. Environmental significance of anthocyanins in plant stress responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Takahama, U. Oxidation of vacuolar and apoplastic phenolic substrates by peroxidase: Physiological significance of the oxidation reactions. Phytochem. Rev. 2004, 207–219. [Google Scholar] [CrossRef]
- Soubeyrand, E.; Basteau, C.; Hilbert, G.; van Leeuwen, C.; Delrot, S.; Gomès, E. Nitrogen supply affects anthocyanin biosynthetic and regulatory genes in grapevine cv. Cabernet-sauvignon berries. Phytochemistry 2014, 103, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Tian, L.; Liu, H.; Pan, Q.; Zhan, J.; Huang, W. Sugars induce anthocyanin accumulation and flavanone 3-hydroxylase expression in grape berries. Plant Growth Regul. 2009, 58, 251–260. [Google Scholar] [CrossRef]
- Ryan, J.-M.; Revilla, E. Anthocyanin composition of Cabernet Sauvignon and Tempranillo grapes at different stages of ripening. J. Agric. Food Chem. 2003, 51, 3372–3378. [Google Scholar] [CrossRef] [PubMed]
- Hilbert, G.; Soyer, J.P.; Molot, C.; Giraudon, J.; Milin, S.; Gaudillere, J. Effects of nitrogen supply on must quality and anthocyanin accumulation in berries of cv. Merlot. Vitis 2003, 42, 69–76. [Google Scholar]
- Delgado, R.; Martín, P.; del Álamo, M.; González, M.-R. Changes in the phenolic composition of grape berries during ripening in relation to vineyard nitrogen and potassium fertilization rates. J. Sci. Food Agric. 2004, 84, 623–630. [Google Scholar] [CrossRef]
- Yin, Y.; Borges, G.; Sakuta, M.; Crozier, A.; Ashihara, H. Effect of phosphate deficiency on the content and biosynthesis of anthocyanins and the expression of related genes in suspension-cultured grape (Vitis sp.) cells. Plant Physiol. Biochem. 2012, 55, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Song, C.Z.; Liu, M.Y.; Meng, J.F.; Chi, M.; Xi, Z.M.; Zhang, Z.W. Promoting effect of foliage sprayed zinc sulfate on accumulation of sugar and phenolics in berries of Vitis vinifera cv. Merlot growing on zinc deficient soil. Molecules 2015, 20, 2536–2554. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Sun, H.; Sun, T.; Wang, Q.; Yao, Y. Modifications of ‘Gold Finger’ grape berry quality as affected by the different rootstocks. J. Agric. Food Chem. 2016, 64, 4189–4197. [Google Scholar] [CrossRef] [PubMed]
- Pestana, M.; Correia, P.J.; de Varennes, A.; Abadía, J.; Faria, E.A. Effectiveness of different foliar iron applications to control iron chlorosis in orange trees grown on a calcareous soil. J. Plant Nutr. 2001, 24, 613–622. [Google Scholar] [CrossRef]
- Mark kliewer, W.; Howarth, L.; Omori, M. Concentrations of tartaric acid and malic acids and their salts in Vitis vinifera grapes. Am. J. Enol. Vitic. 1967, 18, 42–54. [Google Scholar]
- García-Beneytez, E.; Cabello, F.; Revilla, E. Analysis of grape and wine anthocyanins by HPLC-MS. J. Agric. Food Chem. 2003, 51, 5622–5629. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; He, Y.N.; Yue, T.X.; Wang, J.; Zhang, Z.W. Effects of climatic conditions and soil properties on Cabernet Sauvignon berry growth and anthocyanin profiles. Molecules 2014, 19, 13683–13703. [Google Scholar] [CrossRef] [PubMed]
- Downey, M.O.; Rochfort, S. Simultaneous separation by reversed-phase high-performance liquid chromatography and mass spectral identification of anthocyanins and flavonols in Shiraz grape skin. J. Chromatogr. A 2008, 1201, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Pan, Q.; Jin, Z.; Mu, L.; Duan, C. Comparison on phenolic compounds in Vitis vinifera cv. Cabernet Sauvignon wines from five wine-growing regions in china. Food Chem. 2011, 125, 77–83. [Google Scholar] [CrossRef]
- Liang, N.-N.; Zhu, B.-Q.; Han, S.; Wang, J.-H.; Pan, Q.-H.; Reeves, M.J.; Duan, C.-Q.; He, F. Regional characteristics of anthocyanin and flavonol compounds from grapes of four Vitis vinifera varieties in five wine regions of china. Food Res. Int. 2014, 64, 264–274. [Google Scholar] [CrossRef]
- He, J.J.; Liu, Y.X.; Pan, Q.H.; Cui, X.Y.; Duan, C.Q. Different anthocyanin profiles of the skin and the pulp of Yan73 (Muscat Hamburg × Alicante Bouschet) grape berries. Molecules 2010, 15, 1141–1153. [Google Scholar] [CrossRef] [PubMed]
- Woznicki, T.L.; Aaby, K.; Sønsteby, A.; Heide, O.M.; Wold, A.B.; Remberg, S.F. Influence of controlled postflowering temperature and daylength on individual phenolic compounds in four black currant cultivars. J. Agric. Food Chem. 2016, 64, 752–761. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Mu, L.; Yan, G.L.; Liang, N.N.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010, 15, 9057–9091. [Google Scholar] [CrossRef] [PubMed]
- Arozarena, I.; Ayestarán, B.; Cantalejo, M.J.; Navarro, M.; Vera, M.; Abril, I.; Casp, A. Anthocyanin composition of Tempranillo, Garnacha and Cabernet Sauvignon grapes from high- and low-quality vineyards over two years. Eur. Food Res. Technol. 2002, 214, 303–309. [Google Scholar] [CrossRef]
- He, F.; Liang, N.N.; Mu, L.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Anthocyanins and their variation in red wines I. Monomeric anthocyanins and their color expression. Molecules 2012, 17, 1571–1601. [Google Scholar] [CrossRef] [PubMed]
- Roggero, J.P.; Coen, S.; Ragonnet, B. High performance liquid chromatography survey on changes in pigment content in ripening grapes of Syrah. An approach to anthocyanin metabolism. Am. J. Enol. Vitic. 1986, 37, 77–83. [Google Scholar]
- Jackson, R.S. Wine Science: Principle and Applications, 3rd ed.; Elsevier-Academic Press: Oxford, UK, 2008; pp. 287–295. [Google Scholar]
- Valentinuzzi, F.; Mason, M.; Scampicchio, M.; Andreotti, C.; Cesco, S.; Mimmo, T. Enhancement of the bioactive compound content in strawberry fruits grown under iron and phosphorus deficiency. J. Sci. Food Agric. 2015, 95, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil; Calif. Agric. Exp. Station Circular 347; College of Agriculture, University of California: Berkeley, CA, USA, 1950; pp. 2–32. [Google Scholar]
- Brandon, R.S.; Cheng, L. Iron assimilation and carbon metabolism in “Concord” grapevines grown at different pHs. J. Am. Soc. Hortic. Sci. 2007, 132, 473–483. [Google Scholar]
- Administration, Center for Nanoscale Systems. Analytical Methods of Wine and Fruit Wine; China Standards Press: Beijing, China, 2006; pp. 2–23. [Google Scholar]
- Liang, N.N.; Pan, Q.H.; He, F.; Wang, J.; Reeves, M.J.; Duan, C.Q. Phenolic profiles of Vitis davidii and Vitis quinquangularis species native to china. J. Agric. Food Chem. 2013, 61, 6016–6027. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.T.; Goto-Yamamoto, N.; Kobayashi, S.; Esaka, M. Effects of plant hormones and shading on the accumulation of anthocyanins and the expression of anthocyanin biosynthetic genes in grape berry skins. Plant Sci. 2004, 167, 247–252. [Google Scholar] [CrossRef]
- Liu, M.-Y.; Song, C.-Z.; Chi, M.; Wang, T.-M.; Zuo, L.-L.; Li, X.-L.; Zhang, Z.-W.; Xi, Z.-M. The effects of light and ethylene and their interaction on the regulation of proanthocyanidin and anthocyanin synthesis in the skins of Vitis vinifera berries. Plant Growth Regul. 2015, 79, 377–390. [Google Scholar] [CrossRef]
- Bogs, J.; Downey, M.O.; Harvey, J.S.; Ashton, A.R.; Tanner, G.J.; Robinson, S.P. Proanthocyanidin synthesis and expression of genes encoding leucoanthocyanidin reductase and anthocyanidin reductase in developing grape berries and grapevine leaves. Plant Physiol. 2005, 139, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; He, F.; Wang, J.; Li, Z.; Pan, Q.H. Simple rain-shelter cultivation prolongs accumulation period of anthocyanins in wine grape berries. Molecules 2014, 19, 14843–14861. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
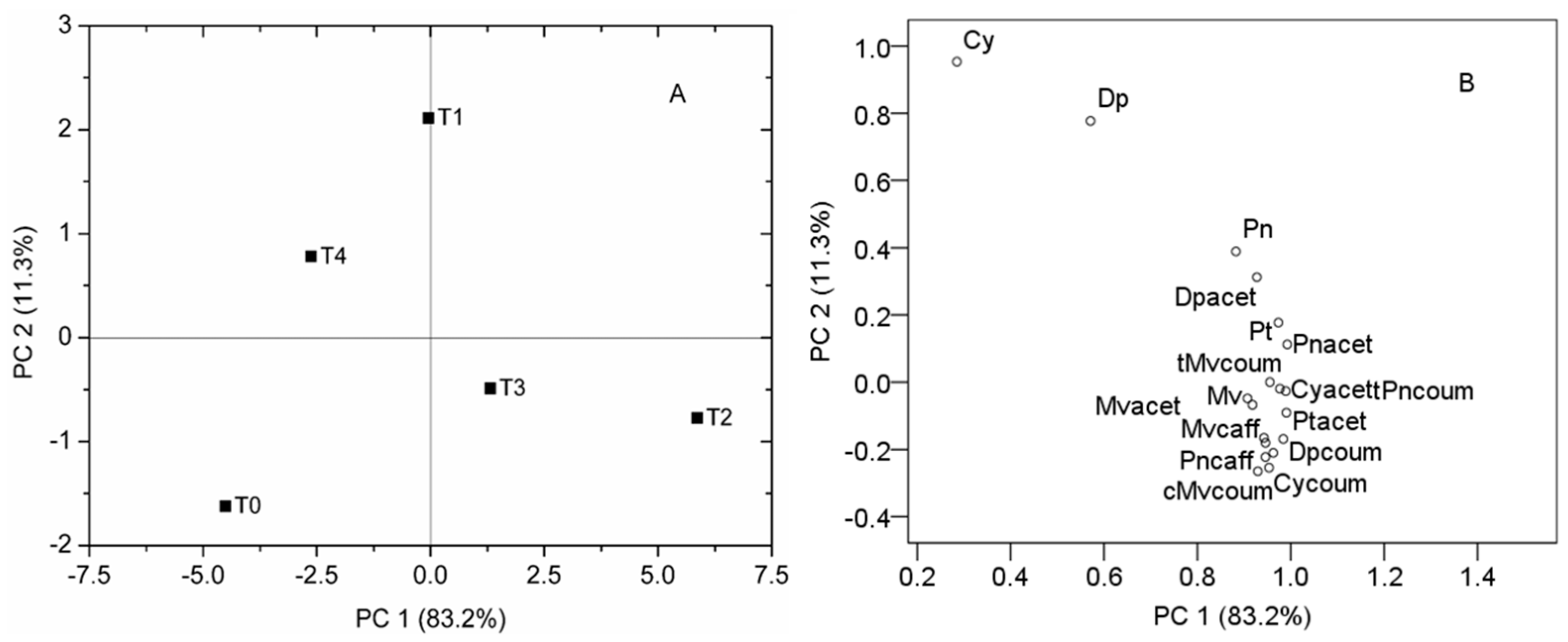
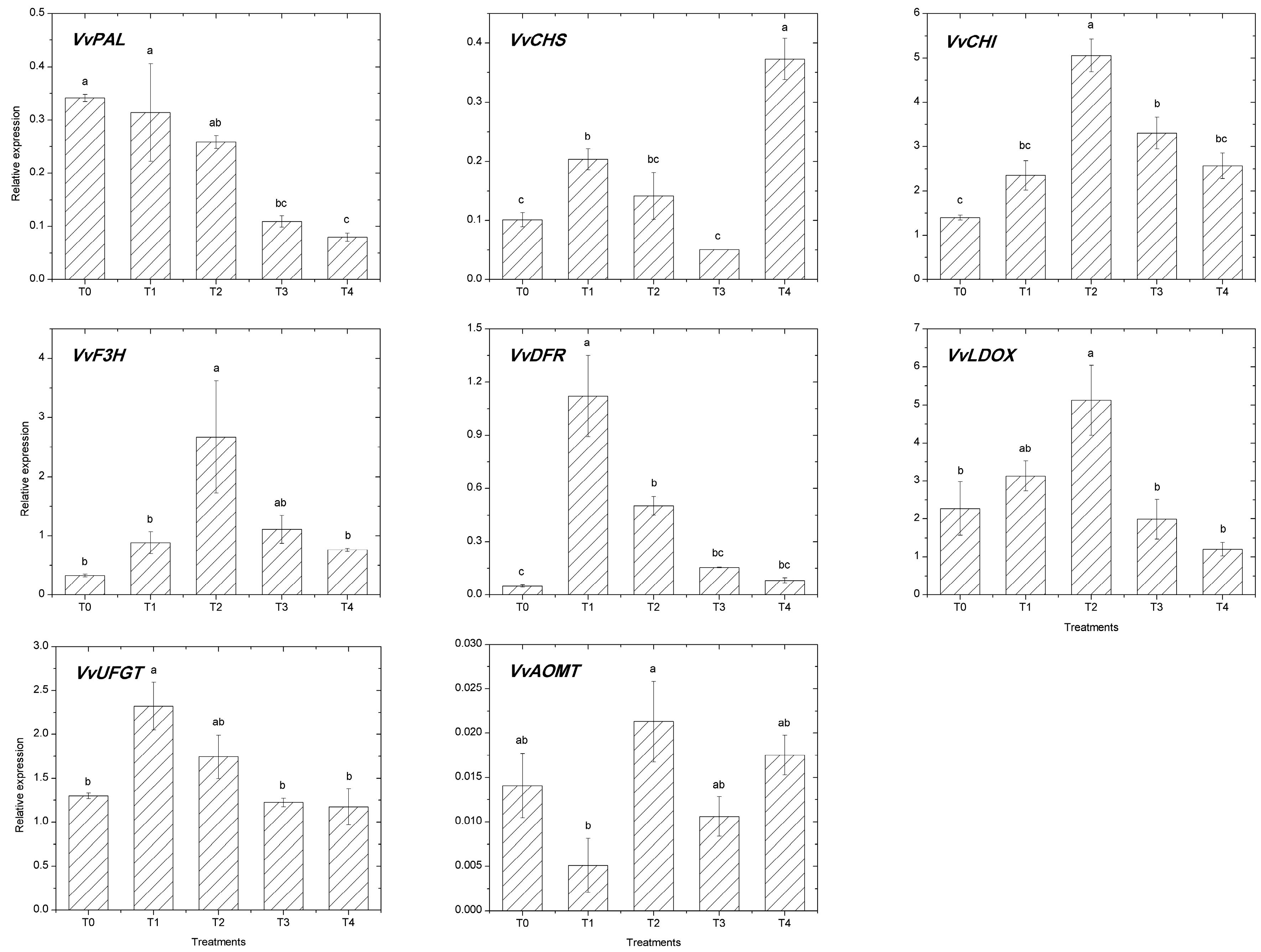
| Treatments | T0 | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|
| RS | 161.67 ± 0.85d | 173.56 ± 0.49b | 182.26 ± 0.54a | 168.86 ± 0.46c | 117.81 ± 0.22e |
| TAC | 7.87 ± 0.09b | 4.61 ± 0.05d | 3.31 ± 0.02e | 4.95 ± 0.06c | 9.58 ± 0.11a |
| RS/TAC | 20.55 ± 0.12d | 37.63 ± 0.32b | 55.03 ± 0.23a | 34.10 ± 0.29c | 12.30 ± 0.12e |
| pH | 3.69 ± 0.04c | 4.23 ± 0.02ab | 4.35 ± 0.03a | 4.11 ± 0.05b | 3.45 ± 0.04d |
| Berry weight (g) | 0.79 ± 0.04b | 0.69 ± 0.02b | 0.89 ± 0.02a | 0.71 ± 0.02b | 0.57 ± 0.03c |
| Skin/berry (%) | 9.41 ± 0.22a | 8.51 ± 0.59a | 8.31 ± 0.44a | 9.47 ± 0.29a | 9.10 ± 0.08a |
| Peak | Retention Time (min) | Molecular Ion M+ (m/z) | Fragment Ion M (m/z) | Tentative Identification | Abbreviations Used | Ref.(s) |
|---|---|---|---|---|---|---|
| 1 | 4.44 | 465 | 303 | delphinidin-3-O-glucoside | Dp | [28,31] |
| 2 | 6.27 | 449 | 287 | cyanidin-3-O-glucoside | Cy | [28,31] |
| 3 | 7.44 | 479 | 317 | petunidin-3-O-glucoside | Pt | [28,31] |
| 4 | 10.04 | 463 | 301 | peonidin-3-O-glucoside | Pn | [28,31] |
| 5 | 11.13 | 493 | 331 | malvidin-3-O-glucoside | Mv | [28,31] |
| 6 | 12.65 | 507 | 303, 465 | delphinidin-3-O-(6-O-acetyl)-glucoside | Dp-acet | [28,31] |
| 7 | 16.11 | 491 | 287, 449 | cyanidin-3-O-(6-O-acetyl)-glucoside | Cy-acet | [28,31] |
| 8 | 17.60 | 521 | 317, 479 | petunidin-3-O-(6-O-acetyl)-glucoside | Pt-acet | [28,31] |
| 9 | 18.52 | 611 | 303, 465 | delphinidin-3-O-(6-O-coumaryl)-glucoside | Dp-coum | [28,31] |
| 10 | 21.35 | 505 | 301, 463 | peonidin-3-O-(6-O-acetyl)-glucoside | Pn-acet | [28,31] |
| 11 | 22.32 | 535 | 331, 493 | malvidin-3-O-(6-O-acetyl)-glucoside | Mv-acet | [28,31] |
| 12 | 24.52 | 625 | 301, 463 | peonidin-3-O-(6-O-caffeoyl)-glucoside | Pn-caff | [31] |
| 13 | 25.01 | 595 | 287, 449 | cyanidin-3-O-(6-O-coumaryl)-glucoside | Cy-coum | [28,31] |
| 14 | 25.52 | 655 | 331, 493 | malvidin-3-(6-O-caffeoyl)-glucoside | Mv-caff | [28,31] |
| 15 | 26.30 | 625 | 317, 479 | petunidin-3-O-(6-O-coumaryl)-glucoside | Pt-coum | [28,31] |
| 16 | 27.51 | 609 | 301, 463 | peonidin-3-O-(cis-6-O-coumaryl)-glucoside | cPn-coum | [28,32] |
| 17 | 28.09 | 639 | 331, 493 | Malvidin-3-O-(cis-6-O-coumaryl)-glucoside | cMv-coum | [28,32] |
| 18 | 29.45 | 609 | 301, 463 | peonidin-3-O-(trans-6-O-coumaryl)-glucoside | tPn-coum | [28,32] |
| 19 | 29.98 | 639 | 331, 493 | malvidin-3-O-(trans-6-O-coumaryl)-glucoside | tMv-coum | [28,32] |
| Anthocyanins | T0 | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|
| Dp | 7.39 ± 0.23e | 19.06 ± 0.30a | 17.20 ± 0.04b | 11.90 ± 0.02d | 15.35 ± 0.02c |
| Cy | 1.74 ± 0.006c | 6.87 ± 0.002a | 3.95 ± 0.030b | 3.76 ± 0.853b | 4.45 ± 0.012b |
| Pt | 8.42 ± 0.027e | 15.44 ± 0.006b | 20.54 ± 0.002a | 14.07 ± 0.078c | 11.14 ± 0.012d |
| Pn | 15.70 ± 0.02e | 26.85 ± 0.13c | 28.88 ± 0.05a | 27.49 ± 0.18b | 21.87 ± 0.02d |
| Mv | 124.35 ± 0.23d | 203.60 ± 0.36c | 274.57 ± 0.40a | 269.56 ± 0.31b | 123.62 ± 0.10d |
| Dp-acet | 2.95 ± 0.001d | 6.23 ± 0.299b | 8.11 ± 0.001a | 5.34 ± 0.028c | 5.09 ± 0.003c |
| Cy-acet | 2.12 ± 0.001e | 4.68 ± 0.253c | 7.82 ± 0.019a | 5.95 ± 0.009b | 2.80 ± 0.010d |
| Pt-acet | 4.65 ± 0.04e | 7.86 ± 0.30c | 14.17 ± 0.04a | 8.92 ± 0.05b | 5.60 ± 0.01d |
| Dp-coum | 0.80 ± 0.001d | 1.84 ± 0.151c | 4.47 ± 0.048a | 2.95 ± 0.317b | 1.09 ± 0.024d |
| Pn-acet | 7.87 ± 0.02e | 13.68 ± 0.14c | 18.85 ± 0.06a | 14.69 ± 0.02b | 10.86 ± 0.02d |
| Mv-acet | 92.74 ± 0.37d | 151.50 ± 0.69c | 211.27 ± 0.11a | 202.62 ± 1.53b | 91.54 ± 0.15d |
| Pn-caff | 0.38 ± 0.012c | 0.82 ± 0.000b | 2.42 ± 0.003a | 1.00 ± 0.140b | 0.13 ± 0.007c |
| Cy-coum | 0.05 ± 0.005b | 0.28 ± 0.002b | 1.29 ± 0.023a | 0.68 ± 0.499a,b | 0.29 ± 0.016b |
| Mv-caff | 2.43 ± 0.01d | 4.02 ± 0.03c | 7.28 ± 0.02a | 4.39 ± 0.06b | 1.38 ± 0.03e |
| Pt-coum | 0.28 ± 0.005e | 0.95 ± 0.009c | 3.75 ± 0.010a | 1.36 ± 0.034b | 0.77 ± 0.019d |
| cPn-coum | 0.14 ± 0.003d | 0.39 ± 0.012c | 1.94 ± 0.114a | 0.90 ± 0.017b | 0.35 ± 0.004c |
| tPn-coum | 1.40 ± 0.03d | 4.39 ± 0.14c | 9.71 ± 0.07a | 6.27 ± 0.02b | 4.20 ± 0.02c |
| cMv-coum | 0.72 ± 0.04d | 1.20 ± 0.16c | 4.99 ± 0.19a | 1.90 ± 0.07b | 0.94 ± 0.03c,d |
| tMv-coum | 11.81 ± 0.08e | 31.24 ± 0.26c | 54.03 ± 0.22a | 47.66 ± 0.09b | 23.79 ± 0.24d |
| Total anthocyanins | 285.95 ± 0.30e | 500.90 ± 0.68c | 695.24 ± 0.79a | 631.43 ± 1.66b | 325.25 ± 0.30d |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, P.; Li, B.; Chen, H.; Song, C.; Meng, J.; Xi, Z.; Zhang, Z. Iron Supply Affects Anthocyanin Content and Related Gene Expression in Berries of Vitis vinifera cv. Cabernet Sauvignon. Molecules 2017, 22, 283. https://doi.org/10.3390/molecules22020283
Shi P, Li B, Chen H, Song C, Meng J, Xi Z, Zhang Z. Iron Supply Affects Anthocyanin Content and Related Gene Expression in Berries of Vitis vinifera cv. Cabernet Sauvignon. Molecules. 2017; 22(2):283. https://doi.org/10.3390/molecules22020283
Chicago/Turabian StyleShi, Pengbao, Bing Li, Haiju Chen, Changzheng Song, Jiangfei Meng, Zhumei Xi, and Zhenwen Zhang. 2017. "Iron Supply Affects Anthocyanin Content and Related Gene Expression in Berries of Vitis vinifera cv. Cabernet Sauvignon" Molecules 22, no. 2: 283. https://doi.org/10.3390/molecules22020283





