Evaluation of the Cadmium Accumulation in Tamarillo Cells (Solanum betaceum) by Indirect Electrochemical Detection of Cysteine-Rich Peptides
Abstract
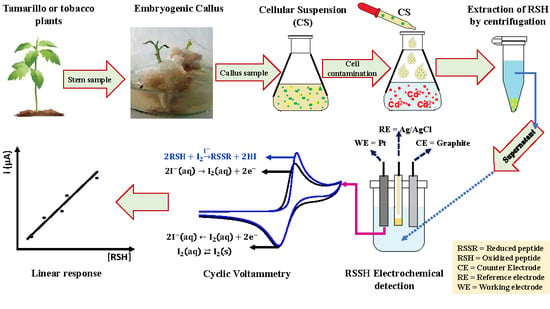
:1. Introduction
2. Results
2.1. Germination of Cell Extracts
2.2. Evaluation of the Analytical Method
2.3. Measurement of Peptides in Cell Suspensions
3. Materials and Methods
3.1. Materials
3.2. Preparation of Cell Extracts
3.3. Application of the Analytic Method
3.4. Measurement of Peptides in the Cell Suspensions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reyes, Y.C.; Vergara, I.; Torres, O.E.; Díaz, M.; González, E.E. Contaminación por metales pesados: Implicaciones en la salud, medio ambiente y seguridad alimentaria. Ing. Investig. y Desarro. 2016, 16, 66–77. [Google Scholar] [CrossRef]
- Salvo, A.; La Torre, G.L.; Mangano, V.; Casale, K.E.; Bartolomeo, G.; Santini, A.; Granata, T.; Dugo, G. Toxic inorganic pollutants in foods from agricultural producing areas of Southern Italy: Level and risk assessment. Ecotoxicol. Environ. Saf. 2018, 148, 114–124. [Google Scholar] [CrossRef] [PubMed]
- López-Climent, M.F.; Arbona, V.; Pérez-Clemente, R.M.; Zandalinas, S.I.; Gómez-Cadenas, A. Effect of cadmium and calcium treatments on phytochelatin and glutathione levels in citrus plants. Plant Biol. 2014, 16, 79–87. [Google Scholar] [CrossRef]
- Šelešovská-Fadrná, R.; Fojta, M.; Navrátil, T.; Chýlková, J. Brdička-type processes of cysteine and cysteine-containing peptides on silver amalgam electrodes. Anal. Chim. Acta 2007, 582, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Ghori, N.-H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Niu, L.-Y.; Guan, Y.-S.; Chen, Y.-Z.; Wu, L.-Z.; Tung, C.-H.; Yang, Q.-Z. BODIPY-Based Ratiometric Fluorescent Sensor for Highly Selective Detection of Glutathione over Cysteine and Homocysteine. J. Am. Chem. Soc. 2012, 134, 18928–18931. [Google Scholar] [CrossRef]
- Cereser, C.; Guichard, J.; Drai, J.; Bannier, E.; Garcia, I.; Boget, S.; Parvaz, P.; Revol, A. Quantitation of reduced and total glutathione at the femtomole level by high-performance liquid chromatography with fluorescence detection: Application to red blood cells and cultured fibroblasts. J. Chromatogr. B: Biomed. Sci. Appl. 2001, 752, 123–132. [Google Scholar] [CrossRef]
- Li, M.; Wu, X.; Wang, Y.; Li, Y.; Zhu, W.; James, T.D. A near-infrared colorimetric fluorescent chemodosimeter for the detection of glutathione in living cells. Chem. Commun. 2014, 50, 1751. [Google Scholar] [CrossRef]
- Dieckhaus, C.M.; Fernández-Metzler, C.L.; King, R.; Krolikowski, P.H.; Baillie, T.A. Negative Ion Tandem Mass Spectrometry for the Detection of Glutathione Conjugates. Chem. Toxicol. 2005, 18, 630–638. [Google Scholar] [CrossRef]
- Ju, J.; Zhang, R.; Chen, W. Photochemical deposition of surface-clean silver nanoparticles on nitrogen-doped graphene quantum dots for sensitive colorimetric detection of glutathione. Sens. Actuators B Chem. 2016, 228, 66–73. [Google Scholar] [CrossRef]
- Lv, Y.; Yang, L.; Mao, X.; Lu, M.; Zhao, J.; Yin, Y. Electrochemical detection of glutathione based on Hg2+-mediated strand displacement reaction strategy. Biosens. Bioelectron. 2016, 85, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Han, P.; Mao, X.; Yin, Y.; Cao, Y. Sensitive detection of glutathione by using DNA-templated copper nanoparticles as electrochemical reporters. Sens. Actuators B Chem. 2017, 238, 325–330. [Google Scholar] [CrossRef]
- De Rosso, V.V.; Mercadante, A.Z. HPLC–PDA–MS/MS of Anthocyanins and Carotenoids from Dovyalis and Tamarillo Fruits. J. Agric. Food Chem. 2007, 55, 9135–9141. [Google Scholar] [CrossRef] [PubMed]
- Arahana, V.S.; Cabrera, A.R.; de Lourdes Torres, M. Propagación de tomate de árbol (Solanum betaceum) vía embriogénesis somática. Avances 2010, 2, B16–B21. [Google Scholar]
- Correia, S.I.; Canhoto, J.M. Biotechnology of tamarillo (Cyphomandra betacea): From in vitro cloning to genetic transformation. Sci. Hortic. 2012, 148, 161–168. [Google Scholar] [CrossRef]
- Arias-Estévez, M.; López–Periago, E.; Martínez–Carballo, E.; Simal–Gándara, J.; Mejuto, J.C.; García–Río, L. The mobility and degradation of pesticides in soils and the pollution of groundwater resources Agric Ecosyst. Environ. 2008, 123, 247–260. [Google Scholar]
- Anjum, N.A.; Aref, I.M.; Duarte, A.C.; Pereira, E.; Ahmad, I.; Iqbal, M. Glutathione and proline can coordinately make plants withstand the joint attack of metal(loid) and salinity stresses. Front Plant Sci. 2014, 5, 1–4. [Google Scholar] [CrossRef]
- González-Sánchez, M.; Valero, E.; Compton, R. Iodine mediated electrochemical detection of thiols in plant extracts using platinum screen-printed electrodes. Sens. Actuators B Chem. 2016, 236, 1–7. [Google Scholar]
- Valero-Ruiz, E.; González-Sánchez, M.I.; Batchelor-McAuley, C.; Compton, R.G. Halogen mediated voltammetric oxidation of biological thiols and disulfides. Analyst 2016, 141, 144–149. [Google Scholar] [CrossRef]
- Lee, P.T.; Gonçalves, L.M.; Compton, R.G. Electrochemical determination of free and total glutathione in human saliva samples. Sens. Actuators B Chem. 2015, 221, 962–968. [Google Scholar] [CrossRef]
- Fojta, M.; Fojtová, M.; Havran, L.; Pivoňková, H.; Dorčák, V.; Šestáková, I. Electrochemical monitoring of phytochelatin accumulation in Nicotiana tabacum cells exposed to sub-cytotoxic and cytotoxic levels of cadmium. Anal. Chim. Acta 2006, 558, 171–178. [Google Scholar] [CrossRef]
- Sánchez-Calvo, L.; Alvarenga, S. Establecimiento in vitro y cultivo de células de la uña de gato (Uncaria tomentosa) (Willd.) D.C. Tecnol. en Marcha 2010, 23, 24–33. [Google Scholar]
- Curtis, W.R.; Emery, A.H. Plant cell suspension culture rheology. Biotechnol. Bioeng. 1993, 42, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Paredes, G.E.; Kato, M.J.; Rojas-Idrogo, C. Suspensiones celulares y producción de metabolitos secundarios en cultivos in vitro de Piper sp. Bol. Latinoam. y del Caribe Plantas Med. y Aromat. 2013, 12, 669–674. [Google Scholar]
- Barton, N.; Wiegand, T.J. Potassium Iodide. In Encyclopedia of Toxicology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Peters, F.T.; Drummer, O.H.; Musshoff, F. Validation of new methods. Forensic Sci. Int. 2007, 165, 216–224. [Google Scholar]
- Boqué, R. La selectividad en Análisis Químico Univ. Rovira i Virgili 2001, 299, 878–881. [Google Scholar]
- Rodríquez Serrano, M.; Martínez-de la Casa, N.; Romero Puertas, M.C.; del Rio, L.A.; Sandalio, L.M. Toxicidad del Cadmio en Plantas. Ecosistemas 2008, 17, 139–146. [Google Scholar]
- Navarro-Aviño, J.P.; Alonso, I.A.; Moya-López, J.R.L. Aspectos bioquímicos y genéticos de la tolerancia y acumulación de metales pesados en plantas. Ecosistemas 2007, 16, 10–25. [Google Scholar]
- Coté, R.J. Aseptic Technique for Cell Culture Curr. Protoc. Cell Biol. 2004. [Google Scholar] [CrossRef]
- Borrero, E. Protocolo para la Regeneración de Plántulas a partir de Explantes de Hojas de Cinco Variedades Ecuatorianas de Tomate de Árbol (Solanum betaceum). 2007. Available online: http://repositorio.usfq.edu.ec/handle/23000/795 (accessed on 1 March 2019).
- Fujii, S.; Kasuya, M.; Kurihara, K. Characterization of Platinum Electrode Surfaces by Electrochemical Surface Forces Measurement. J. Phys. Chem. C 2017, 121, 26406–26413. [Google Scholar] [CrossRef]
- Solla-Gullón, J.; Rodríguez, P.; Herrero, E.; Aldaz, A.; Feliu, J.M. Surface characterization of platinum electrodes. Phys. Chem. Chem. Phys. 2008, 10, 1359–1373. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |






| Medium | 2,4-D (mg L−1) | ANA (mg L−1) | Stems | Leaves | ||
|---|---|---|---|---|---|---|
| % 1 | % 2 | % 1 | % 2 | |||
| 1 | 0.5 | 0 | 70 | 65 | 10 | 10 |
| 2 | 1 | 0 | 90 | 95 | 0 | 10 |
| 3 | 0 | 5 | 30 | 40 | 50 | 50 |
| 4 | 0 | 7 | 10 | 10 | 25 | 30 |
| p value | 0.002 | 0.0011 | ||||
| Parameter | Accepted * | Result |
|---|---|---|
| Repeatability | CV < 2% | 1.57% |
| Reproducibility | CV < 3% | 2.25% |
| Linearity | r2 > 0.98 | 0.9972 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montero-Jiménez, M.; Fernández, L.; Alvarado, J.; Criollo, M.; Jadán, M.; Chuquer, D.; Espinoza-Montero, P. Evaluation of the Cadmium Accumulation in Tamarillo Cells (Solanum betaceum) by Indirect Electrochemical Detection of Cysteine-Rich Peptides. Molecules 2019, 24, 2196. https://doi.org/10.3390/molecules24122196
Montero-Jiménez M, Fernández L, Alvarado J, Criollo M, Jadán M, Chuquer D, Espinoza-Montero P. Evaluation of the Cadmium Accumulation in Tamarillo Cells (Solanum betaceum) by Indirect Electrochemical Detection of Cysteine-Rich Peptides. Molecules. 2019; 24(12):2196. https://doi.org/10.3390/molecules24122196
Chicago/Turabian StyleMontero-Jiménez, Marjorie, Lenys Fernández, José Alvarado, Mauricio Criollo, Mónica Jadán, David Chuquer, and Patricio Espinoza-Montero. 2019. "Evaluation of the Cadmium Accumulation in Tamarillo Cells (Solanum betaceum) by Indirect Electrochemical Detection of Cysteine-Rich Peptides" Molecules 24, no. 12: 2196. https://doi.org/10.3390/molecules24122196