FT-IR/ATR Solid Film Formation: Qualitative and Quantitative Analysis of a Piperacillin-Tazobactam Formulation
Abstract
1. Introduction
2. Results
2.1. Identification of PIP and TAZ
2.1.1. Powder Spectra
2.1.2. Film Spectra
2.2. Quantification of PIP and TAZ
3. Discussion
4. Materials and Methods
4.1. Preparation of Standard Mixtures
4.2. Preparation of Solutions of Standard Mixtures
4.3. Development of Film Forming Methodology
4.4. FT-IR/ATR Spectra Acquisition
4.5. Spectra Processing
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Larkin, P. Infrared and Raman Spectroscopy: Principles and Spectral Interpretation; Elsevier Science: Philadelphia, PA, USA, 2017. [Google Scholar]
- Skoog, D.A.; Holler, F.J.; Nieman, T.A. Instrumental Analysis Principles, 7th ed.; Cengage Learning: Boston, MA, USA, 2016; pp. 389–436. [Google Scholar]
- Hans, K.M.C.; Müller, S.; Sigrist, M.W. Infrared attenuated total reflection (IR-ATR) spectroscopy for detecting drugs in human saliva. Drug Test. Anal. 2012, 4, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Ghauch, A.; Deveau, P.A.; Jacob, V.; Baussand, P. Use of FTIR spectroscopy coupled with ATR for the determination of atmospheric compounds. Talanta 2006, 68, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Benramdane, L.; Bouatia, M.; Idrissi, M.O.B.; Draoui, M. Infrared Analysis of Urinary Stones, Using a Single Reflection Accessory and a KBr Pellet Transmission. Spectrosc. Lett. 2008, 41, 72–80. [Google Scholar] [CrossRef]
- Wilson, R.H.; Tapp, H.S. Mid-infrared spectroscopy for food analysis: Recent new applications and relevant developments in sample presentation methods. TrAC Trends Anal. Chem. 1999, 18, 85–93. [Google Scholar] [CrossRef]
- Durak, T.; Depciuch, J. Effect of plant sample preparation and measuring methods on ATR-FTIR spectra results. Environ. Exp. Bot. 2020, 169, 103915. [Google Scholar] [CrossRef]
- Lawson, G. Counterfeit Tablet Investigations: Can ATR FT/IR Provide Rapid Targeted Quantitative Analyses? J. Anal. Bioanal. Tech. 2014, 5, 5. [Google Scholar] [CrossRef]
- Hartauer, K.J.; Guillory, J.K. Quantitative Fourier Transform-Infrared/Attenuated Total Reflectance (FT-IR/ATR) Analysis of Trimethoprim and Sulfamethoxazole in a Pharmaceutical Formulation Using Partial Least Squares. Pharm. Res. 1989, 6, 608–611. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.; Ayoko, G.; Collett, S.; Golding, G. Rapid quantification of methamphetamine: Using attenuated total reflectance fourier transform infrared spectroscopy (ATR-FTIR) and chemometrics. PLoS ONE 2013, 8, e69609. [Google Scholar] [CrossRef] [PubMed]
- Makki, A.A.; Bonnier, F.; Respaud, R.; Chtara, F.; Tfayli, A.; Tauber, C.; Bertrand, D.; Byrne, H.J.; Mohammed, E.; Chourpa, I. Qualitative and quantitative analysis of therapeutic solutions using Raman and infrared spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 218, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Shorr, R.I.; Hoth, A.B.; Rawls, N. (Eds.) Drugs for the Geriatric Patient; W.B. Saunders: Philadelphia, PA, USA, 2007; pp. 930–1062. [Google Scholar]
- Drawz, S.M.; Bonomo, R.A. Three Decades of β-Lactamase Inhibitors. Clin. Microbiol. Rev. 2010, 23, 160. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, T.; Karasawa, T.; Williams, D.W. Chapter Thirteen—Antimicrobial Chemotherapy: Significance to Healthcare. In Biofilms in Infection Prevention and Control; Percival, S.L., Williams, D.W., Randle, J., Cooper, T., Eds.; Academic Press: Boston, MA, USA, 2014; pp. 209–244. [Google Scholar]
- Rama Krishna Veni, P.; Sharmila, N.; Narayana, K.J.P.; Hari Babu, B.; Satyanarayana, P.V.V. Simultaneous determination of piperacillin and tazobactam in pharmaceutical formulations by RP-HPLC method. J. Pharm. Res. 2013, 7, 127–131. [Google Scholar] [CrossRef]
- Atmakuri, L.R.; Krishna, K.; Kumar, C.H.; Raja, T.A. Simultaneous determination of piperacillin and tazobactum in bulk and pharmaceutical dosage forms by rphplc. Int. J. Pharm. Pharm. Sci. 2011, 3, 134–136. [Google Scholar]
- Pai, S.; Rao, G.K.; Murthy, M.S.; Prathibha, H. Simultaneous estimation of piperacillin and tazobactam in injection formulations. Indian J. Pharm. Sci. 2006, 68, 799–801. [Google Scholar] [CrossRef]
- Council of Europe, European Directorate for the Quality of Medicines & Healthcare. European Pharmacopoeia 10.4; Council of Europe: Strasburg, France, 2020; p. 1168. [Google Scholar]
- Naicker, S.; Guerra Valero, Y.C.; Ordenez Meija, J.L.; Lipman, J.; Roberts, J.A.; Wallis, S.C.; Parker, S.L. A UHPLC-MS/MS method for the simultaneous determination of piperacillin and tazobactam in plasma (total and unbound), urine and renal replacement therapy effluent. J. Pharm. Biomed. Anal. 2018, 148, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Zander, J.; Maier, B.; Suhr, A.; Zoller, M.; Frey, L.; Teupser, D.; Vogeser, M. Quantification of piperacillin, tazobactam, cefepime, meropenem, ciprofloxacin and linezolid in serum using an isotope dilution UHPLC-MS/MS method with semi-automated sample preparation. Clin. Chem. Lab. Med. 2015, 53, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Karpova, S.P.; Blazheyevskiy, M.; Mozgova, O. Development and validation of UV spectrophotometric area under curve method quantitative estimation of piperacillin. Int. J. Pharm. Sci. Res. 2018, 9, 3556–3560. [Google Scholar]
- Toral, M.I.; Nova-Ramírez, F.; Nacaratte, F. Simultaneous determination of piperacillin and tazobactam in the pharmaceutical formulation Tazonam® by derivative spectrophotometry. J. Chil. Chem. Soc. 2012, 57, 1189–1193. [Google Scholar] [CrossRef][Green Version]
- ICH Harmonized Tripartite Guideline Q2 (R1) Validation of Analytical Procedures. Available online: https://www.ema.europa.eu/en/ich-q2-r1-validation-analytical-procedures-text-methodology (accessed on 20 March 2020).


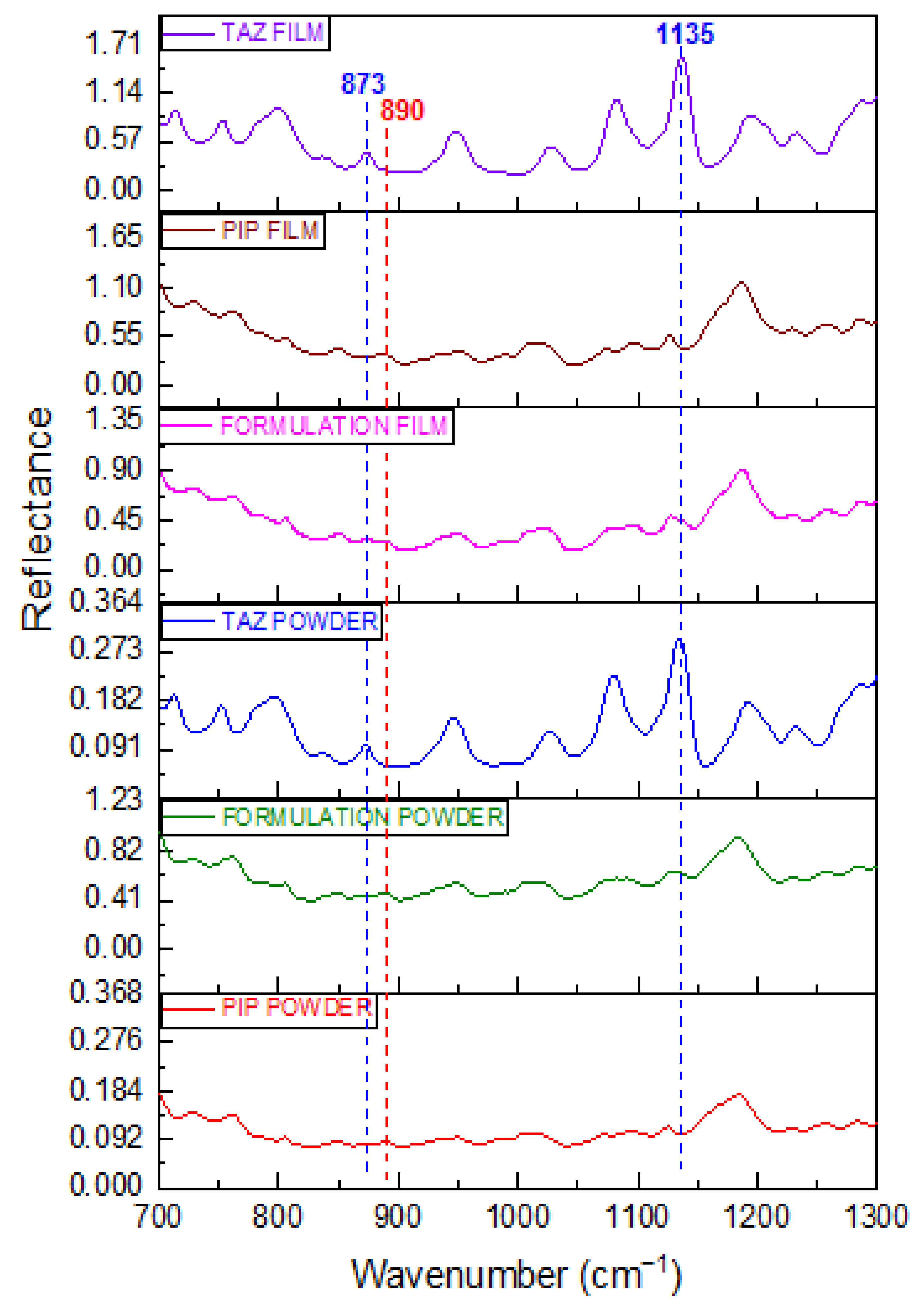
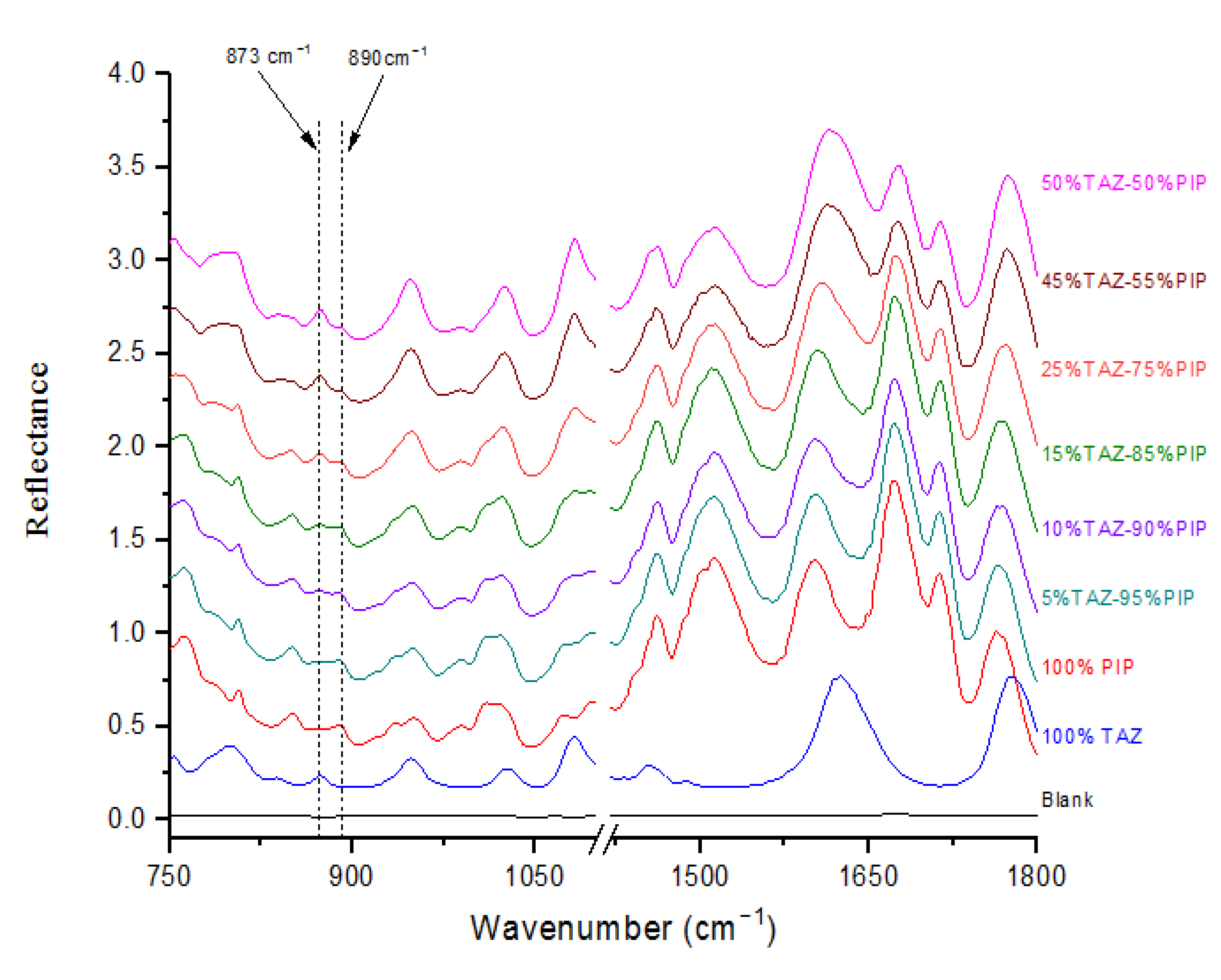
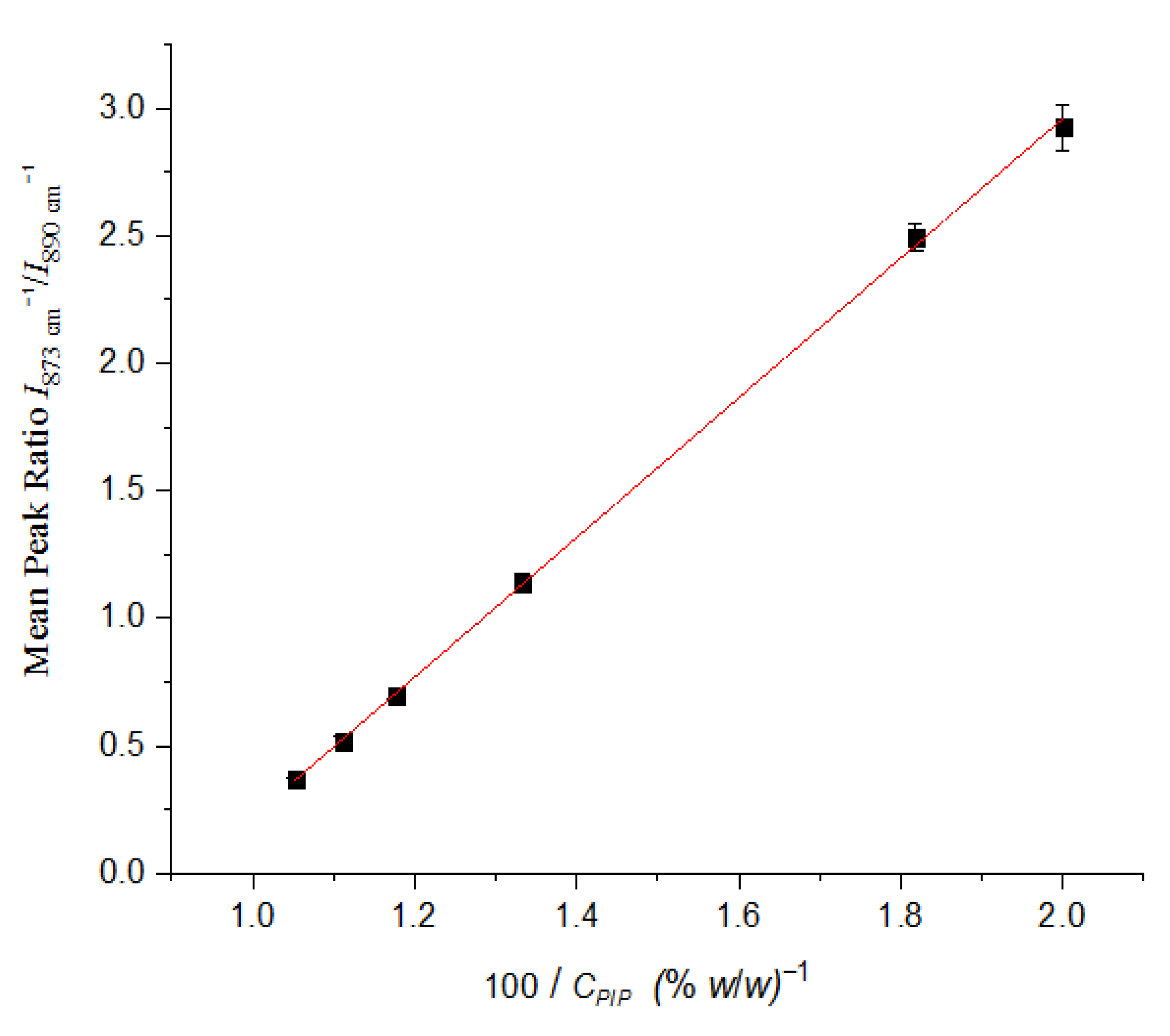
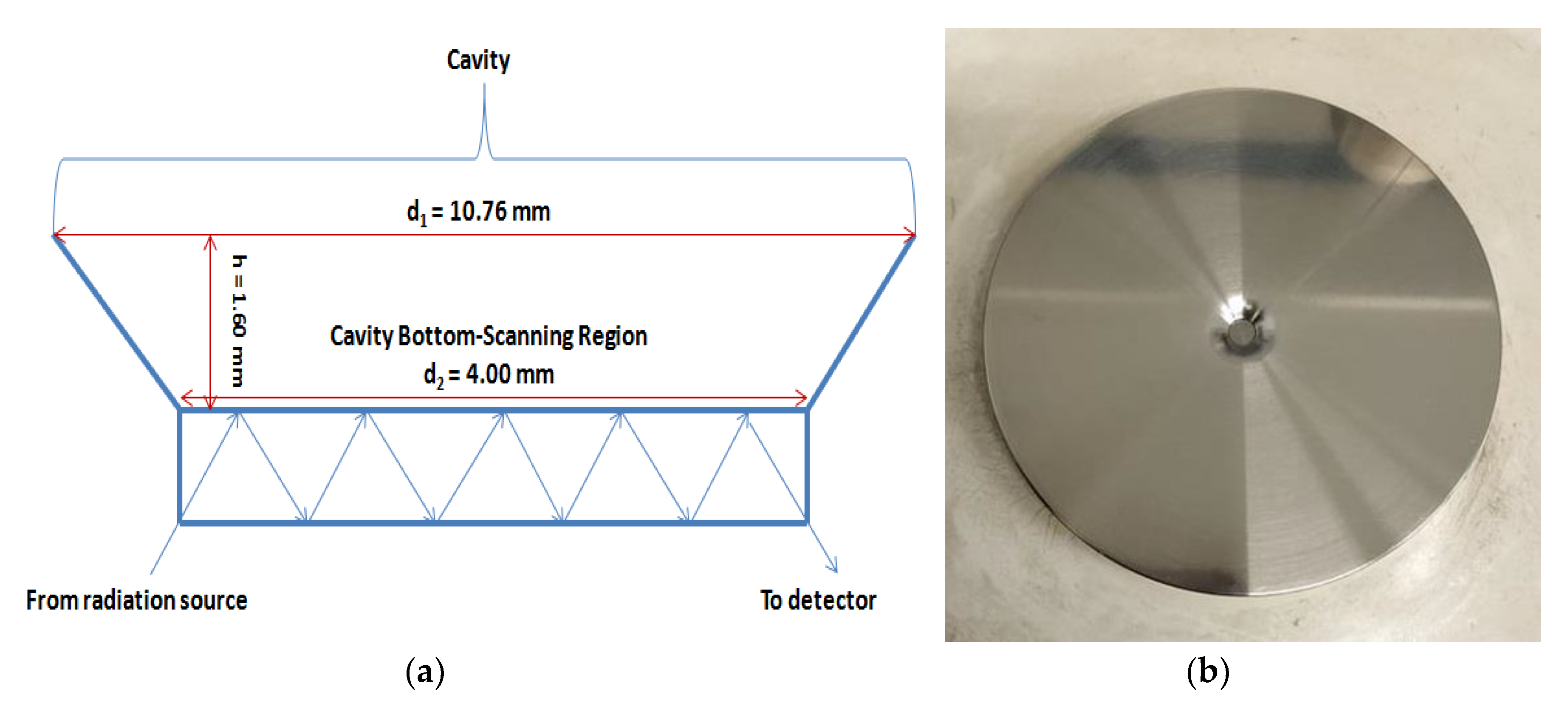
| API | Wavenumber (cm−1) | Vibration Mode |
|---|---|---|
| PIP | 728, 762, 850, 890 | Aromatic CH wagging |
| 1072, 1094 | In-plane movement of aromatic carbons | |
| 1461, 1602 | Semi-circle stretching and Quadrum stretching of aromatic ring | |
| 1713, 1764 | Carbonyl group (CO) stretching | |
| TAZ | 1135, 1191, 1312 | In-phase and Out-of-phase stretching of C-SO2-C |
| 1232, 1452 | N=N stretching | |
| 1385, 1616 | Carbonyl group (CO) stretching |
| Wavenumber (cm−1) | Observation |
|---|---|
| 873 | Intensity enhancement and change in intensity ratio against the characteristic PIP peak at 890 cm−1 |
| 945 | Intensity enhancement and change in intensity ratio between neighboring peaks at 933 and 949 cm−1 (area A) |
| 1023 | Intensity enhancement and change in intensity ratio against the neighboring peak at 1007 cm−1 |
| 1081 | Intensity enhancement and change in intensity ratio between neighboring peaks at 1072 and 1094 cm−1 (area B) |
| 1135 | Detection of TAZ shoulder |
| % Mass Ratio TAZ-PIP | I(873)/I(890) | RSD% |
|---|---|---|
| 50:50 | 2.925 | 3.14 |
| 45:55 | 2.493 | 2.19 |
| 25:75 | 1.138 | 3.48 |
| 15:85 | 0.699 | 2.22 |
| 10:90 | 0.517 | 3.89 |
| 5:95 | 0.367 | 1.90 |
Sample Availability: Samples of the compounds PIP and TAZ are available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrisikou, I.; Orkoula, M.; Kontoyannis, C. FT-IR/ATR Solid Film Formation: Qualitative and Quantitative Analysis of a Piperacillin-Tazobactam Formulation. Molecules 2020, 25, 6051. https://doi.org/10.3390/molecules25246051
Chrisikou I, Orkoula M, Kontoyannis C. FT-IR/ATR Solid Film Formation: Qualitative and Quantitative Analysis of a Piperacillin-Tazobactam Formulation. Molecules. 2020; 25(24):6051. https://doi.org/10.3390/molecules25246051
Chicago/Turabian StyleChrisikou, Ioanna, Malvina Orkoula, and Christos Kontoyannis. 2020. "FT-IR/ATR Solid Film Formation: Qualitative and Quantitative Analysis of a Piperacillin-Tazobactam Formulation" Molecules 25, no. 24: 6051. https://doi.org/10.3390/molecules25246051
APA StyleChrisikou, I., Orkoula, M., & Kontoyannis, C. (2020). FT-IR/ATR Solid Film Formation: Qualitative and Quantitative Analysis of a Piperacillin-Tazobactam Formulation. Molecules, 25(24), 6051. https://doi.org/10.3390/molecules25246051






