Antioxidant, Antidiabetic, Anticholinergic, and Antiglaucoma Effects of Magnofluorine
Abstract
:1. Introduction
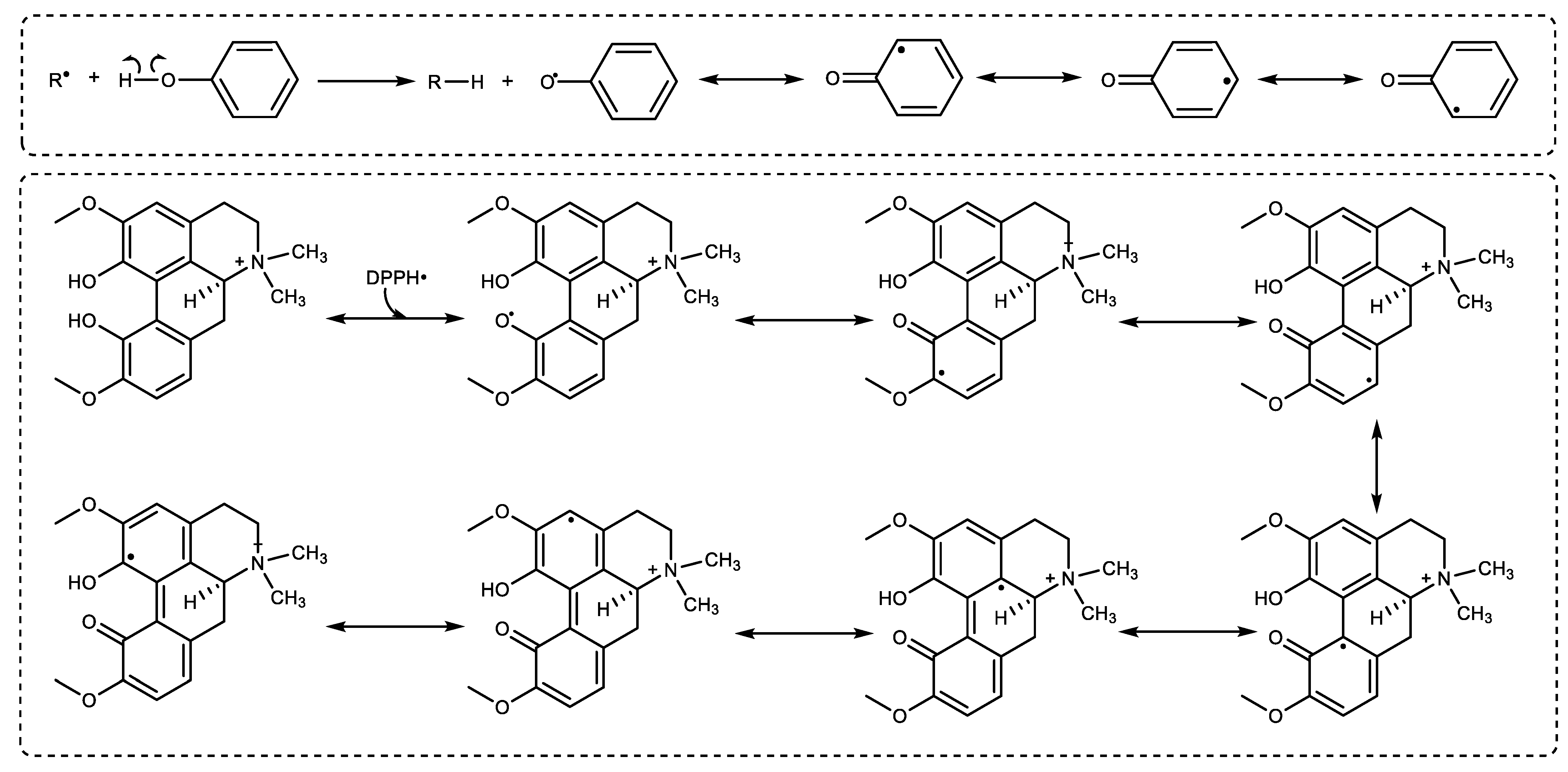
2. Results
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Antioxidant Assays
4.3. Anticholinergic Assay
4.4. Antidiabetic Assay
4.5. Antiglaucoma Assay
4.6. Inhibition Parameters
4.7. Molecular Docking Studies
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Gulcin, I. Antioxidants and antioxidant methods-An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.H.; Park, Y.J.; Namiesnik, J.; Gulcin, I.; Kim, T.C.; Kim, H.C.; Heo, B.G.; Gorinstein, S.; Ku, Y.G. Effects of artificial lighting on bioactivity of sweet red pepper (Capsicum annuum L.). Int. J. Food Sci. Technol. 2016, 51, 1378–1385. [Google Scholar] [CrossRef]
- Durmaz, L.; Ertürk, A.; Akyüz, M.; Polat Köse, L.; Uc, E.M.; Bingöl, Z.; Saglamtas, R.; Alwasel, S.; Gulcin, I. Screening of carbonic anhydrase, acetylcholinesterase, butyrylcholinesterase and α-glycosidase enzymes inhibition effects and antioxidant activity of coumestrol. Molecules 2022, 27, 3091. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Beydemir, S.; Sat, I.G.; Küfrevioğlu, O.I. Evaluation of antioxidant activity of cornelian cherry (Cornus mas L.). Acta Aliment. Hung. 2005, 34, 193–202. [Google Scholar] [CrossRef]
- Serbetci Tohma, H.; Gulcin, I. Antioxidant and radical scavenging activity of aerial parts and roots of Turkish liquorice (Glycyrrhiza glabra L.). Int. J. Food Prop. 2010, 13, 657–671. [Google Scholar] [CrossRef]
- Bingol, Z.; Kızıltas, H.; Goren, A.C.; Polat Kose, L.; Topal, M.; Durmaz, L.; Alwasel, S.H.; Gulcin, I. Antidiabetic, anticholinergic and antioxidant activities of aerial parts of shaggy bindweed (Convulvulus betonicifolia Miller subsp.)-profiling of phenolic compounds by LC-HRMS. Heliyon 2021, 7, e06986. [Google Scholar] [CrossRef]
- Gulcin, I.; Kufrevioglu, O.I.; Oktay, M.; Buyukokuroglu, M.E. Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.). J. Ethnopharmacol. 2004, 90, 205–215. [Google Scholar] [CrossRef]
- Gülçin, İ.; Buyukokuroglu, M.E.; Oktay, M.; Küfrevioğlu, O.I. Antioxidant and analgesic activities of turpentine of Pinus nigra Arn. Subsp. pallsiana (Lamb.) Holmboe. J. Ethnopharmacol. 2003, 86, 51–58. [Google Scholar] [CrossRef]
- Gulcin, I.; Beydemir, S.; Topal, F.; Gagua, N.; Bakuridze, A.; Bayram, R.; Gepdiremen, A. Apoptotic, antioxidant and antiradical effect of majdine and isomajdine from Vinca herbacea Waldst. and kit. J. Enzym. Inhib. Med. Chem. 2012, 27, 587–594. [Google Scholar] [CrossRef]
- Balaydın, H.T.; Gulcin, I.; Menzek, A.; Goksu, S.; Sahin, E. Synthesis and antioxidant properties of diphenylmethane derivative bromophenols including a natural product. J. Enzym. Inhib. Med. Chem. 2010, 25, 685–695. [Google Scholar] [CrossRef] [Green Version]
- Koksal, E.; Gulcin, I. Antioxidant activity of cauliflower (Brassica oleracea L.). Turk. J. Agric. For. 2008, 32, 65–78. [Google Scholar]
- Cetin Cakmak, K.; Gulcin, I. Anticholinergic and antioxidant activities of usnic acid-An activity-structure insight. Toxicol. Rep. 2019, 6, 1273–1280. [Google Scholar] [CrossRef]
- Kalin, P.; Gulcin, I.; Goren, A.C. Antioxidant activity and polyphenol content of cranberries (Vaccinium macrocarpon). Rec. Nat. Prod. 2015, 9, 496–502. [Google Scholar]
- Kızıltas, H.; Bingol, Z.; Goren, A.C.; Polat Kose, L.; Durmaz, L.; Topal, F.; Alwasel, S.H.; Gulcin, I. LC-HRMS profiling, antidiabetic, anticholinergic and anti-oxidant activities of aerial parts of kınkor (Ferulago stelleta). Molecules 2021, 26, 2469. [Google Scholar] [CrossRef]
- Ozbey, F.; Taslimi, P.; Gulcin, I.; Maras, A.; Goksu, S.; Supuran, C.T. Synthesis, acetylcholinesterase, butyrilcholinesterase, carbonic anhydrase inhibitory and metal chelating properties of some novel diaryl ether. J. Enzym. Inhib. Med. Chem. 2016, 31, 79–85. [Google Scholar] [CrossRef]
- Gulcin, I.; Taslimi, P.; Aygun, A.; Sadeghian, N.; Bastem, E.; Kufrevioglu, O.I.; Turkan, F.; Sen, F. Antidiabetic and antiparasitic potentials: Inhibition effects of some natural antioxidant compounds on α-glycosidase, α-amylase and human glutathione S-transferase enzymes. Int. J. Biol. Macromol. 2018, 119, 741–746. [Google Scholar] [CrossRef]
- Aktas Anil, D.; Polat, M.F.; Saglamtas, R.; Tarikogullari, A.H.; Alagoz, M.A.; Gulcin, I.; Algul, O.; Burmaoglu, S. Exploring enzyme inhibition profiles of novel halogenated chalcone derivatives on some metabolic enzymes: Synthesis, characterization and molecular modeling studies. Comput. Biol. Chem. 2022, 100, 107748. [Google Scholar] [CrossRef]
- Erdemir, F.; Barut Celepci, D.; Aktas, A.; Taslimi, P.; Gok, Y.; Karabıyık, H.; Gulcin, I. 2-Hydroxyethyl substituted NHC precursors: Synthesis, characterization, crystal structure and carbonic anhydrase, α-glycosidase, butyrylcholinesterase, and acetylcholinesterase inhibitory properties. J. Mol. Struc. 2018, 1155, 797–806. [Google Scholar] [CrossRef]
- Genc Bilgicli, H.; Ergon, D.; Taslimi, P.; Tuzun, B.; Akyazı Kuru, I.; Zengin, M.; Gulcin, I. Novel propanolamine derivatives attached to 2-metoxifenol moiety: Synthesis, characterization, biological properties, and molecular docking studies. Bioorg. Chem. 2020, 101, 103969. [Google Scholar] [CrossRef]
- Topal, M.; Gulcin, I. Evaluation of the in vitro antioxidant, antidiabetic and anticholinergic properties of rosmarinic acid from rosemary (Rosmarinus officinalis L.). Biocat. Agric. Biotechnol. 2022, 43, 102417. [Google Scholar] [CrossRef]
- Hamide, M.; Gök, Y.; Demir, Y.; Yakali, G.; Taşkın-Tok, T.; Aktaş, A.; Sevinçek, R.; Güzel, B.; Gulçin, İ. Pentafluorobenzyl-substituted benzimidazolium salts: Synthesis, characterization, crystal structures, computational studies and inhibitory properties of some metabolic enzymes. J. Mol. Struct. 2022, 1265, 133266. [Google Scholar] [CrossRef]
- Gulcin, I.; Petrova, O.V.; Taslimi, P.; Malysheva, S.F.; Schmidt, E.Y.; Sobenina, L.N.; Gusarova, N.K.; Trofimov, B.A.; Tuzun, B.; Farzaliyev, V.M.; et al. Synthesis, characterization, molecular docking, acetylcholinesterase and α-glycosidase ınhibition profiles of nitrogen-based novel heterocyclic compounds. ChemistrySelect 2022, 7, e20220037. [Google Scholar] [CrossRef]
- Taslimi, P.; Aslan, H.E.; Demir, Y.; Oztaskın, N.; Maras, A.; Gulcin, I.; Beydemir, S.; Goksu, S. Diarilmethanon, bromophenols and diarilmetan compounds: Discovery of potent aldose reductase, α-amylase and α-glycosidase inhibitors as new therapeutic approach in diabetes and functional hyperglycemia. Int. J. Biol. Macromol. 2018, 119, 857–863. [Google Scholar] [CrossRef]
- Turkan, F.; Cetin, A.; Taslimi, P.; Karaman, M.; Gulcin, I. Synthesis, biological evaluation and molecular docking of novel pyrazole derivatives as potent carbonic anhydrase and acetylcholinesterase inhibitors. Bioorg. Chem. 2019, 86, 420–427. [Google Scholar] [CrossRef]
- Yakan, H.; Kocyigit, Ü.M.; Muglu, H.; Ergul, M.; Erkan, S.; Guzel, E.; Taslimi, P.; Gulcin, I. Potential thiosemicarbazone-based enzyme inhibitors: Assessment of anti-proliferative activity, metabolic enzyme inhibition properties and molecular docking calculations. J. Biochem. Mol. Toxicol. 2022, 36, e23018. [Google Scholar] [CrossRef]
- Topal, F.; Gulcin, I.; Dastan, A.; Guney, M. Novel eugenol derivatives: Potent acetylcholinesterase and carbonic anhydrase inhibitors. Int. J. Biol. Macromol. 2017, 94, 845–851. [Google Scholar] [CrossRef]
- Polat Kose, L.; Gulcin, I.; Goren, A.C.; Namiesnik, J.; Martinez-Ayala, A.L.; Gorinstein, S. LC-MS/MS analysis, antioxidant and anticholinergic properties of galanga (Alpinia officinarum Hance) rhizomes. Ind. Crops Prod. 2015, 74, 712–721. [Google Scholar] [CrossRef]
- Oztaskin, N.; Cetinkaya, Y.; Taslimi, P.; Goksu, S.; Gulcin, I. Antioxidant and acetylcholinesterase inhibition properties of novel bromophenol derivatives. Bioorg. Chem. 2015, 60, 49–57. [Google Scholar] [CrossRef]
- Okon, E.; Kukula-Koch, W.; Jarzab, A.; Halasa, M.; Stepulak, A.; Wawruszak, A. Advances in chemistry and bioactivity of magnoflorine and magnoflorine-containing extracts. Int. J. Mol. Sci. 2020, 21, 1330. [Google Scholar] [CrossRef]
- Morris, J.S.; Facchini, P.J. Isolation and characterization of reticuline N-methyltransferase involved in biosynthesis of the aporphine alkaloid magnoflorine in opium poppy. J. Biol. Chem. 2016, 291, 23416–23427. [Google Scholar] [CrossRef]
- Hamad, H.O.; Alma, M.H.; Gulcin, I.; Yılmaz, M.A.; Karaogul, E. Evaluation of phenolic contents and bioactivity of root and nutgall extracts from Iraqian Quercus infectoria Olivier. Rec. Nat. Prod. 2017, 11, 205–210. [Google Scholar]
- Garibov, E.; Taslimi, P.; Sujayev, A.; Bingol, Z.; Cetinkaya, S.; Gulcin, I.; Beydemir, S.; Farzaliyev, V.; Alwasel, S.H.; Supuran, C.T. Synthesis of 4,5-disubstituted-2-thioxo-1,2,3,4-tetrahydropyrimidines and investigation of their acetylcholinesterase, butyrylcholinesterase, carbonic anhydrase I/II inhibitory and antioxidant activities. J. Enzym. Inhib. Med. Chem. 2016, 31, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Masters, C.L.; Bateman, R.; Blennow, K.; Rowe, C.C.; Sperling, R.A.; Cummings, J.L. Alzheimer’s disease. Nat. Rev. Dis. Primers 2015, 1, 15056. [Google Scholar] [CrossRef] [PubMed]
- Taslimi, P.; Osmanova, S.; Caglayan, C.; Turkan, F.; Sardarova, S.; Farzaliyev, F.; Sujayev, E.; Sadeghian, N.; Gulcin, I. Novel amides of 1,1-bis-(carboxymethylthio)-1-arylethanes: Synthesis, characterization, and acetylcholinesterase, butyrylcholinesterase, and carbonic anhydrase inhibitory properties. J. Biochem. Mol. Toxicol. 2018, 32, e22191. [Google Scholar] [CrossRef]
- Ozgeris, B.; Goksu, S.; Kose Polat, L.; Gulcin, I.; Salmas, R.E.; Durdagi, S.; Tumer, F.; Supuran, C.T. Acetylcholinesterase and carbonic anhydrase inhibitory properties of novel urea and sulfamide derivatives incorporating dopaminergic 2-aminotetralin scaffolds. Bioorg. Med. Chem. 2016, 24, 2318–2329. [Google Scholar] [CrossRef]
- Wang, W.; Fu, X.W.; Dai, X.L.; Hua, F.; Chu, G.X.; Chu, M.J.; Bao, G.H. Novel acetylcholinesterase inhibitors from Zijuan tea and biosynthetic pathway of caffeoylated catechin in tea plant. Food Chem. 2017, 237, 1172–1178. [Google Scholar] [CrossRef]
- Arabaci, B.; Gulcin, I.; Alwasel, S. Capsaicin: A potent inhibitor of carbonic anhydrase isoenzymes. Molecules 2014, 19, 10103–10114. [Google Scholar] [CrossRef]
- Gulcin, I.; Scozzafava, A.; Supuran, C.T.; Akıncıoğlu, H.; Koksal, Z.; Turkan, F.; Alwasel, S. The effect of caffeic acid phenethyl ester (CAPE) metabolic enzymes including acetylcholinesterase, butyrylcholinesterase, glutathione s-transferase, lactoperoxidase and carbonic anhydrase isoenzymes I, II, IX and XII. J. Enzym. Inhib. Med. Chem. 2016, 31, 1095–1101. [Google Scholar] [CrossRef]
- Gocer, H.; Topal, F.; Topal, M.; Kucuk, M.; Teke, D.; Gulcin, I.; Alwasel, S.H.; Supuran, C.T. Acetylcholinesterase and carbonic anhydrase isoenzymes I and II inhibition profiles of taxifolin. J. Enzym. Inhib. Med. Chem. 2016, 31, 441–447. [Google Scholar] [CrossRef]
- Gulcin, I.; Scozzafava, A.; Supuran, C.T.; Koksal, Z.; Turkan, F.; Cetinkaya, S.; Bingol, Z.; Huyut, Z.; Alwasel, S.H. Rosmarinic acid inhibits some metabolic enzymes including glutathione S-transferase, lactoperoxidase, acetylcholinesterase, butyrylcholinesterase, and carbonic anhydrase isoenzymes. J. Enzym. Inhib. Med. Chem. 2016, 31, 1698–1702. [Google Scholar] [CrossRef]
- Kanner, E.; Tsai, J.C. Glaucoma medications. Drugs Aging 2006, 23, 321–332. [Google Scholar] [CrossRef]
- Hou, Z.; Li, C.; Liu, Y.; Zhang, M.; Wang, Y.; Fan, Z.; Guo, C.; Lin, B.; Liu, Y. Design, synthesis and biological evaluation of carbohydrate-based sulphonamide derivatives as topical antiglaucoma agents through selective inhibition of carbonic anhydrase II. J. Enzym. Inhib. Med. Chem. 2020, 35, 383–390. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid). Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef]
- Gulcin, I.; Oktay, M.; Koksal, E.; Serbetci, H.; Beydemir, S.; Kufrevioglu, Ö.I. Antioxidant and radical scavenging activities of uric acid. Asian J. Chem. 2008, 20, 2079–2090. [Google Scholar]
- Gocer, H.; Gulcin, I. Caffeic acid phenethyl ester (CAPE): Correlation of structure and antioxidant properties. Int. J. Food Sci. Nutr. 2011, 62, 821–825. [Google Scholar] [CrossRef]
- Gulcin, I.; Huyut, Z.; Elmastas, M.; Aboul-Enein, H.Y. Radical scavenging and antioxidant activity of tannic acid. Arab. J. Chem. 2010, 3, 43–53. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant properties of resveratrol: A structure-activity insight. Innov. Food Sci. Emerg. 2010, 11, 210–218. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of eugenol-a structure and activity relationship study. J. Med. Food 2011, 14, 975–985. [Google Scholar] [CrossRef]
- Taslimi, P.; Gulcin, I. Antioxidant and anticholinergic properties of olivetol. J. Food Biochem. 2018, 42, e12516. [Google Scholar] [CrossRef]
- Topal, F.; Topal, M.; Gocer, H.; Kalın, P.; Kocyigit, U.M.; Gulcin, I.; Alwasel, S.H. Antioxidant activity of taxifolin: An activity-structure relationship. J. Enzym. Inhib. Med. Chem. 2016, 31, 674–683. [Google Scholar] [CrossRef]
- Koksal, E.; Gulcin, I.; Ozturk Sarikaya, S.B.; Bursal, E. On the in vitro antioxidant activity of silymarin. J. Enzym. Inhib. Med. Chem. 2009, 24, 395–405. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of L-Adrenaline: An activity-structure insight. Chem. Biol. Interact. 2009, 179, 71–80. [Google Scholar] [CrossRef]
- Ak, T.; Gulcin, I. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 2008, 174, 27–37. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, Y.; Cheng, Y.; Wang, Y. Rapid screening and identification of α-glucosidase inhibitors from mulberry leaves using enzyme-immobilized magnetic beads coupled with HPLC/MS and NMR. Biomed. Chromatogr. 2013, 27, 148–155. [Google Scholar] [CrossRef]
- Elmastas, M.; Turkekul, I.; Ozturk, L.; Gulcin, I.; Isıldak, O.; Aboul-Enein, H.Y. The antioxidant activity of two wild edible mushrooms (Morchella vulgaris and Morchella esculanta). Comb. Chem. High. Throughput Screen 2006, 9, 443–448. [Google Scholar] [CrossRef]
- Gulcin, I.; Topal, F.; Ozturk Sarikaya, S.B.; Bursal, E.; Gören, A.C.; Bilsel, M. Polyphenol contents and antioxidant properties of medlar (Mespilus germanica L.). Rec. Nat. Prod. 2011, 5, 158–175. [Google Scholar]
- Gulcin, I.; Sat, I.G.; Beydemir, S.; Kufrevioglu, Ö.İ. Evaluation of the in vitro antioxidant properties of extracts of broccoli (Brassica oleracea L.). Ital. J. Food Sci. 2004, 16, 17–30. [Google Scholar]
- Eruygur, N.; Atas, M.; Tekin, M.; Taslimi, P.; Kocyigit, U.M.; Gulcin, I. In vitro antioxidant, antimicrobial, anticholinesterase and antidiabetic activities of Turkish endemic Achillea cucullata (Asteraceae) from ethanol extract. S. Afr. J. Bot. 2019, 120, 141–145. [Google Scholar] [CrossRef]
- Gulcin, I.; Tel, A.Z.; Goren, A.C.; Taslimi, P.; Alwasel, S. Sage (Salvia pilifera): Determination its polyphenol contents, anticholinergic, antidiabetic and antioxidant activities. J. Food Meas. Charact. 2019, 13, 2062–2074. [Google Scholar] [CrossRef]
- Oktay, M.; Yildirim, A.; Bilaloglu, V.; Gulcin, I. Antioxidant activity of different parts of isgin (Rheum ribes L.). Asian J. Chem. 2007, 19, 3047–3055. [Google Scholar]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I.; Tel, A.Z.; Kirecci, E. Antioxidant, antimicrobial, antifungal and antiradical activities of Cyclotrichium niveum (Boiss.) Manden and Scheng. Int. J. Food Propert. 2008, 11, 450–471. [Google Scholar] [CrossRef]
- Bursal, E.; Aras, A.; Kılıc, O.; Taslimi, P.; Goren, A.C.; Gulcin, I. Phytochemical content, antioxidant activity and enzyme inhibition effect of Salvia eriophora Boiss. & Kotschy against acetylcholinesterase, α-amylase, butyrylcholinesterase and α-glycosidase enzymes. J. Food Biochem. 2019, 43, e12776. [Google Scholar] [PubMed]
- Gulcin, I.; Topal, F.; Cakmakcı, R.; Goren, A.C.; Bilsel, M.; Erdogan, U. Pomological features, nutritional quality, polyphenol content analysis and antioxidant properties of domesticated and three wild ecotype forms of raspberries (Rubus idaeus L.). J. Food Sci. 2011, 76, C585–C593. [Google Scholar] [CrossRef] [PubMed]
- Oktay, M.; Gulcin, I.; Kufrevioglu, Ö.İ. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. Lebensm. Wissen. Technol. 2003, 36, 263–271. [Google Scholar] [CrossRef]
- Moodie, L.W.K.; Sepcic, K.; Turk, T.; Frangez, R.; Svenson, J. Natural cholinesterase inhibitors from marine organisms. Nat. Prod. Rep. 2019, 36, 1053–1092. [Google Scholar] [CrossRef] [PubMed]
- Polat Kose, L.; Gulcin, I. Inhibition effects of some lignans on carbonic anhydrase, acetylcholinesterase and butyrylcholinesterase enzymes. Rec. Nat. Prod. 2017, 11, 558–561. [Google Scholar] [CrossRef]
- Gulcin, İ. Measurement of antioxidant ability of melatonin and serotonin by the DMPD and CUPRAC methods as trolox equivalent. J. Enzyme Inhib.Med. Chem. 2003, 23, 871–876. [Google Scholar] [CrossRef]
- Talaz, O.; Gulcin, I.; Goksu, S.; Saracoglu, N. Antioxidant activity of 5,10-dihydroindeno [1,2-b]indoles containing substituents on dihydroindeno part. Bioorg. Med. Chem. 2009, 17, 6583–6589. [Google Scholar] [CrossRef]
- Artunc, T.; Menzek, A.; Taslimi, P.; Gulcin, I.; Kazaz, C.; Sahin, E. Synthesis and antioxidant activities of phenol derivatives from 1,6-bis(dimethoxyphenyl)hexane-1,6-dione. Bioorg. Chem. 2020, 100, 103884. [Google Scholar] [CrossRef]
- Rezai, M.; Bayrak, C.; Taslimi, P.; Gulcin, I.; Menzek, A. The first synthesis, antioxidant and anticholinergic activities of 1-(4,5-dihydroxybenzyl)pyrrolidin-2-one derivative bromophenols including natural products. Turk. J. Chem. 2018, 42, 808–825. [Google Scholar]
- Kiziltas, H.; Bingol, Z.; Goren, A.C.; Pınar, S.M.; Alwasel, S.H.; Gulcin, I. LC-HRMS profiling of phytochemicals, antidiabetic, anticholinergic and antioxidant activities of evaporated ethanol extract of Astragalus brachycalyx FISCHER. J. Chem. Metrol. 2021, 15, 135–151. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 26, 1199–1200. [Google Scholar] [CrossRef]
- Gulcin, I.; Dastan, A. Synthesis of dimeric phenol derivatives and determination of in vitro antioxidant and radical scavenging activities. J. Enzym. Inhib. Med. Chem. 2007, 22, 685–695. [Google Scholar] [CrossRef]
- Gulcin, I.; Alwasel, S.H. Metal ions, metal chelators and metal chelating assay as antioxidant method. Processes 2022, 10, 132. [Google Scholar] [CrossRef]
- Yigit, M.; Barut Celepci, D.; Taslimi, P.; Yigit, B.; Cetinkaya, B.; Ozdemir, I.; Aygün, M.; Gulcin, I. Selenourea and thiourea derivatives of chiral and achiral enetetramines: Synthesis, characterization and enzyme inhibitory properties. Bioorg. Chem. 2022, 120, 105566. [Google Scholar] [CrossRef]
- Aktas, A.; Barut Celepci, D.; Gok, Y.; Taslimi, P.; Akıncıoğlu, H.; Gulcin, I. A novel Ag-N-heterocyclic carbene complex bearing the hydroxyethyl ligand: Synthesis, characterization, crystal and spectral structures and bioactivity properties. Crystals 2020, 10, 171. [Google Scholar] [CrossRef]
- Topal, M.; Gulcin, I. Rosmarinic acid: A potent carbonic anhydrase isoenzymes inhibitor. Turk. J. Chem. 2014, 38, 894–902. [Google Scholar] [CrossRef]
- Mahmudov, I.; Demir, Y.; Sert, Y.; Abdullayev, Y.; Sujayev, E.; Alwasel, S.H.; Gulcin, I. Synthesis and inhibition profiles of N-benzyl- and N-allyl aniline derivatives against carbonic anhydrase and acetylcholinesterase-A molecular docking study. Arab. J. Chem. 2022, 15, 103645. [Google Scholar] [CrossRef]
- Burmaoglu, S.; Akın Kazancioglu, E.; Kazancioglu, M.Z.; Saglamtas, R.; Yalcın, G.; Gulcin, I.; Algul, O. Synthesis, molecular docking and some metabolic enzyme inhibition properties of biphenyl-substituted chalcone derivatives. J. Mol. Struct. 2022, 1254, 132358. [Google Scholar] [CrossRef]
- Topal, M.; Gocer, H.; Topal, F.; Kalin, P.; Polat Kose, P.; Gulcin, I.; Cetin Cakmak, K.; Kucuk, M.; Durmaz, L.; Goren, A.C.; et al. Antioxidant, antiradical and anticholinergic properties of cynarin purified from the illyrian thistle (Onopordum illyricum L.). J. Enzym. Inhib. Med. Chem. 2016, 31, 266–275. [Google Scholar] [CrossRef]
- Bursal, E.; Gulcin, I. Polyphenol contents and in vitro antioxidant activities of lyophilized aqueous extract of kiwifruit (Actinidia deliciosa). Food Res. Int. 2011, 44, 1482–1489. [Google Scholar] [CrossRef]
- Bulduran, K.; Turan, N.; Bursal, E.; Aras, A.; Mantarcı, A.; Çolak, N.; Türkan, F.; Durgun, M.; Gulcin, I. Synthesis, characterization, powder X-ray diffraction analysis, thermal stability, antioxidant properties and enzyme inhibitions of M(II)-Schiff base ligand complexes. J. Biomol. Struct. Dyn. 2021, 39, 6480–6487. [Google Scholar] [CrossRef]
- Gulcin, I.; Goren, A.C.; Taslimi, P.; Alwasel, S.H.; Kilic, O.; Bursal, E. Anticholinergic, antidiabetic and antioxidant activities of Anatolian pennyroyal (Mentha pulegium)-Analysis of its polyphenol contents by LC-MS/MS. Biocat. Agric. Biotechnol. 2020, 23, 101441. [Google Scholar] [CrossRef]
- Takım, K.; Yigin, A.; Koyuncu, I.; Kaya, R.; Gulcin, I. Anticancer, anticholinesterase and antidiabetic activities of Tunceli garlic (Allium tuncelianum)-Determining its phytochemical content by LC-MS/MS analysis. J. Food Meas. Charact. 2021, 15, 3323–3335. [Google Scholar] [CrossRef]
- Hashmi, S.; Khan, S.; Shafiq, Z.; Taslimi, P.; Ishaq, M.; Sadeghian, N.; Karaman, S.H.; Akhtar, N.; Islam, M.; Asari, A.; et al. Probing 4-(diethylamino)-salicylaldehyde-based thiosemicarbazones as multi-target directed ligands against cholinesterases, carbonic anhydrases and α-glycosidase enzymes. Bioorg. Chem. 2021, 107, 104554. [Google Scholar] [CrossRef]
- Kazancı, A.; Gok, Y.; Kaya, R.; Aktas, A.; Taslimi, P.; Gulcin, I. Synthesis, characterization and bioactivities of dative donor ligand N-heterocyclic carbene (NHC) precursors and their Ag(I)NHC coordination compounds. Polyhedron 2021, 193, 114866. [Google Scholar] [CrossRef]
- Karimov, A.; Orujova, A.; Taslimi, P.; Sadeghian, N.; Mammadov, B.; Karaman, H.S.; Farzaliyev, V.; Sujayev, A.; Tas, R.; Alwasel, S.; et al. Novel functionally substituted esters based on sodium diethyldithiocarbamate derivatives: Synthesis, characterization, biological activity and molecular docking studies. Bioorg. Chem. 2020, 99, 103762. [Google Scholar] [CrossRef] [PubMed]
- Aras, A.; Bursal, E.; Turkan, F.; Tohma, H.; Kılıç, O.; Gulcin, I.; Koksal, E. Phytochemical content antidiabetic, anticholinergic, and antioxidant activities of endemic Lecokia cretica extracts. Chem. Biodivers. 2019, 16, e1900341. [Google Scholar] [CrossRef]
- Bicer, A.; Taslimi, P.; Yakali, G.; Gulcin, I.; Gultekin, M.S.; Turgut Cin, G. Synthesis, characterization, crystal structure of novel bis-thiomethylcyclohexanone derivatives and their inhibitory properties against some metabolic enzymes. Bioorg. Chem. 2019, 82, 393–404. [Google Scholar] [CrossRef]
- Taslimi, P.; Akıncıoğlu, H.; Gulcin, I. Synephrine and phenylephrine act as α-amylase, α-glycosidase, acetylcholinesterase, butyrylcholinesterase and carbonic anhydrase enzymes inhibitors. J. Biochem. Mol. Toxicol. 2017, 31, e21973. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Scozzafava, A.; Kalın, P.; Supuran, C.T.; Gulcin, I.; Alwasel, S. The impact of hydroquinone on acetylcholine esterase and certain human carbonic anhydrase isoenzymes (hCA I, II, IX, and XII). J. Enzym. Inhib. Med. Chem. 2015, 30, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Ozmen Ozgun, D.; Gul, H.I.; Yamali, C.; Sakagami, H.; Gulcin, I.; Sukuroglu, M.; Supuran, C.T. Synthesis and bioactivities of pyrazoline benzensulfonamides as carbonic anhydrase and acetylcholinesterase inhibitors with low cytotoxicity. Bioorg. Chem. 2019, 84, 511–517. [Google Scholar] [CrossRef]
- Kocyigit, U.M.; Budak, Y.; Gurdere, M.B.; Tekin, S.; Kul Koprulu, T.; Erturk, F.; Ozcan, K.; Gulcin, I.; Ceylan, M. Synthesis, characterization, anticancer, antimicrobial and carbonic anhydrase inhibition profiles of novel (3aR,4S,7R,7aS)-2-(4-((E)-3-(3-aryl)acryloyl) phenyl)-3a,4,7,7a-tetrahydro-1H-4,7-methanoisoindole-1,3(2H)-dione derivatives. Bioorg. Chem. 2017, 70, 118–125. [Google Scholar] [CrossRef]
- Pedrood, K.; Sherefati, M.; Taslimi, P.; Mohammadi-Khanaposhtani, M.; Asgari, M.S.; Hosseini, S.; Rastegar, H.; Larijani, B.; Mahdavi, M.; Taslimi, P.; et al. Design, synthesis, characterization, enzymatic inhibition evaluations, and docking study of novel quinazolinone derivatives. Int. J. Biol. Macromol. 2021, 170, 1–12. [Google Scholar] [CrossRef]
- Lineweaver, H.; Burk, D. The determination of enzyme dissociation constants. J. Am. Chem. Soc. 1934, 56, 658–666. [Google Scholar] [CrossRef]
- Dasgin, S.; Gok, Y.; Barut Celepci, D.; Taslimi, P.; İzmirli, M.; Aktas, A.; Gulcin, I. Synthesis, characterization, crystal structure and bioactivity properties of the benzimidazole-functionalized PEPPSI type of Pd(II)NHC complexes. J. Mol. Struct. 2021, 1228, 129442. [Google Scholar] [CrossRef]
- Cheung, J.; Rudolph, M.J.; Burshteyn, F.; Cassidy, M.S.; Gary, E.N.; Love, J.; Franklin, M.C.; Height, J.J. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef]
- Roig-Zamboni, V.; Cobucci-Ponzano, B.; Iacono, R.; Ferrara, M.C.; Germany, S.; Bourne, Y.; Parenti, G.; Moracci, M.; Sulzenbacher, G. Structure of human lysosomal acid α-glucosidase-A guide for the treatment of Pompe disease. Nat. Commun. 2017, 8, 1111. [Google Scholar] [CrossRef]
- Marakovic, N.; Knezevic, A.; Roncevic, I.; Brazzolotto, X.; Kovarik, Z.; Sinko, G. Enantioseparation, in vitro testing, and structural characterization of triple-binding reactivators of organophosphate-inhibited cholinesterases. Biochem. J. 2020, 477, 2771–2790. [Google Scholar] [CrossRef]
- Sippel, K.H.; Robbins, A.H.; Domsic, J.; Genis, C.; Agbandje-Mckenna, M.; McKenna, R. High-resolution structure of human carbonic anhydrase II complexed with acetazolamide reveals insights into inhibitor drug design. Acta Cryst. F Struct. Biol. Cryst. Commun. 2009, 65, 992–995. [Google Scholar] [CrossRef]
- Berman, H.M.; Battistuz, T.; Bhat, T.N.; Bluhm, W.F.; Bourne, P.E.; Burkhardt, K.; Feng, Z.; Gilliland, G.L.; Iype, L.; Jain, S.; et al. The protein data bank. Acta Crystallogr. D 2002, 58, 899–907. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. Software news and updates gabedit-A graphical user interface for computational chemistry softwares. J. Comp. Chem. 2009, 31, 456–461. [Google Scholar]
- Salentin, S.; Schreiber, S.; Haupt, V.J.; Adasme, M.F.; Schroeder, M. PLIP: Fully automated protein-ligand interaction profiler. Nucleic Acids Res. 2015, 43, W443–W447. [Google Scholar] [CrossRef]
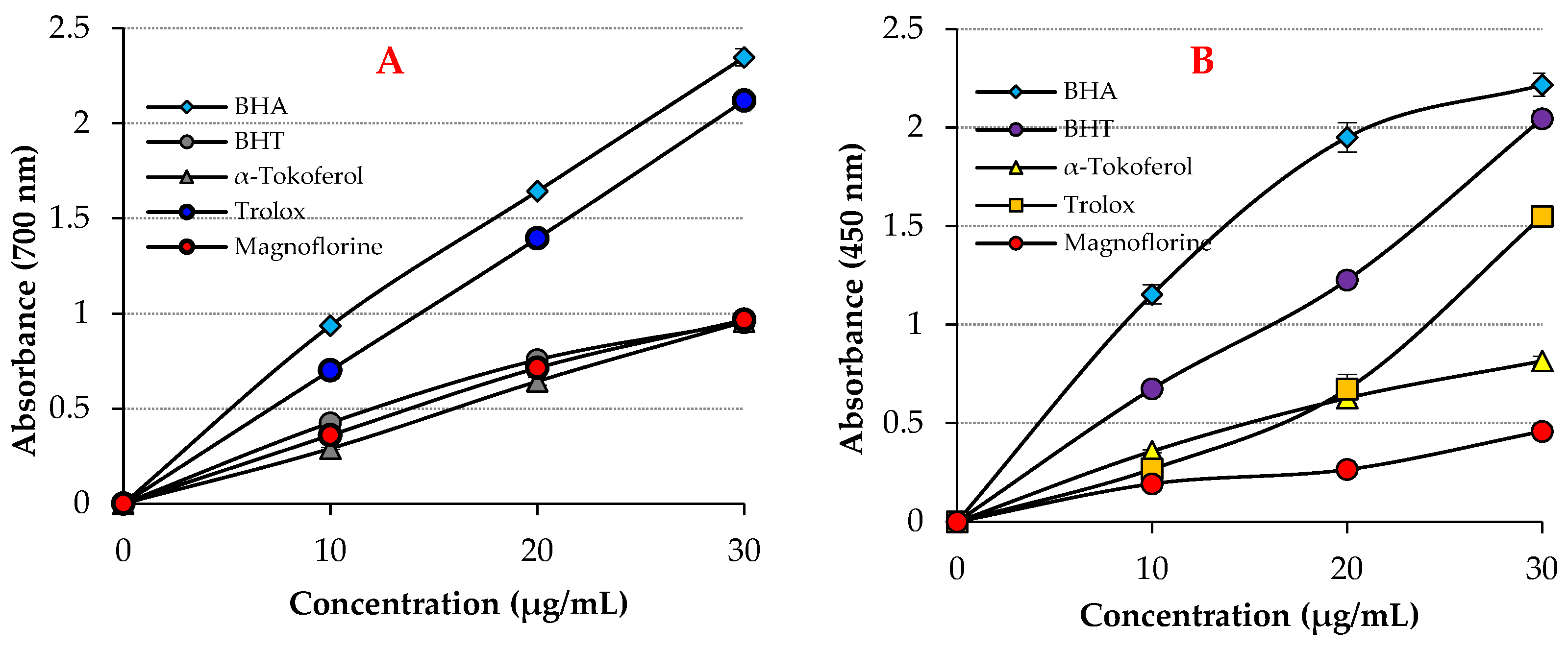

| Antioxidants | Fe3+ Reducing | Cu2+ Reducing | ||
|---|---|---|---|---|
| λ (700 nm) | r2 | λ (450 nm) | r2 | |
| BHA | 2.347 | 0.9086 | 2.216 | 0.9928 |
| BHT | 0.952 | 0.9154 | 2.044 | 0.9937 |
| Trolox | 2.119 | 0.9586 | 1.548 | 0.9305 |
| α-Tocopherol | 0.957 | 0.9863 | 0.816 | 0.9897 |
| Magnofluorine | 0.967 | 0.9938 | 0.458 | 0.9729 |
| Antioxidants | DPPH• Scavenging | ABTS•+ Scavenging | DMPD•+ Scavenging | |||
|---|---|---|---|---|---|---|
| IC50 | r2 | IC50 | r2 | IC50 | r2 | |
| BHA | 10.10 | 0.9015 | 5.07 | 0.9356 | 11.99 | 0.9580 |
| BHT | 25.95 | 0.9221 | 6.99 | 0.9350 | 8.72 | 0.9375 |
| Trolox | 7.05 | 0.9614 | 6.16 | 0.9692 | 4.33 | 0.9447 |
| α-Tocopherol | 11.31 | 0.9642 | 8.37 | 0.9015 | 7.11 | 0.9509 |
| Magnofluorine | 10.58 | 0.9908 | 27.61 | 0.9006 | 15.16 | 0.9966 |
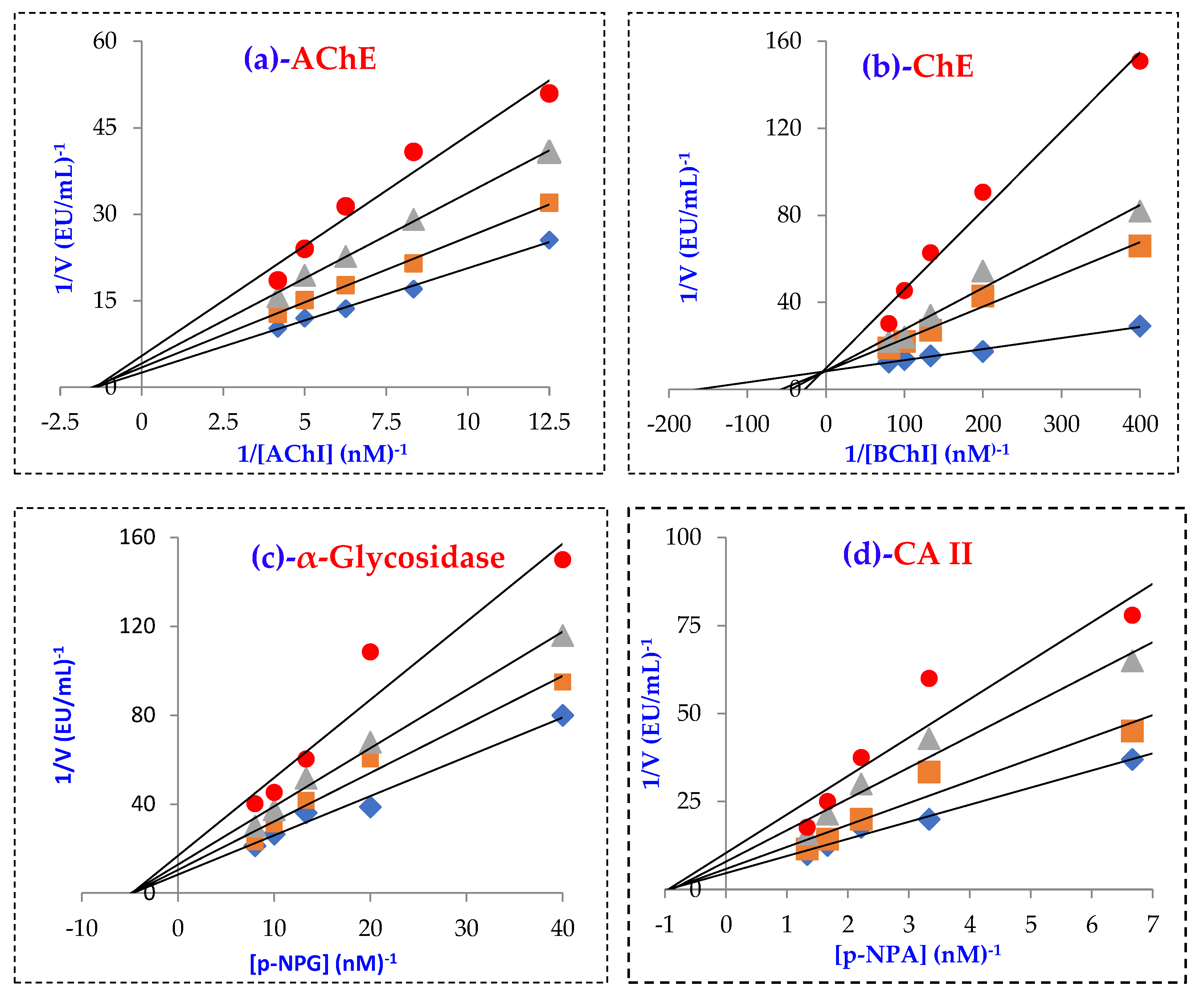
| Compounds | IC50 (nM) | Ki (nM) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CA II | r2 | AChE | r2 | BChE | r2 | α-Gly | r2 | CA II | AChE | BChE | α-Gly | |
| Magnofluorine | 26.03 | 0.9313 | 10.01 | 0.9429 | 8.71 | 0.9825 | 31.02 | 0.9364 | 25.41 ± 1.10 | 10.25 ± 1.94 | 2.47 ± 0.70 | 30.56 ± 3.36 |
| Acetazolamide * | 8.37 | 0.9825 | - | - | - | - | - | - | 4.41 ± 0.35 | - | - | - |
| Tacrine ** | - | - | 5.97 | 0.9706 | 8.37 | 0.9846 | - | - | - | 2.43 ± 0.92 | 5.99 ± 1.79 | - |
| Acarbose *** | - | - | - | - | - | - | 22,800 | - | - | - | - | - |
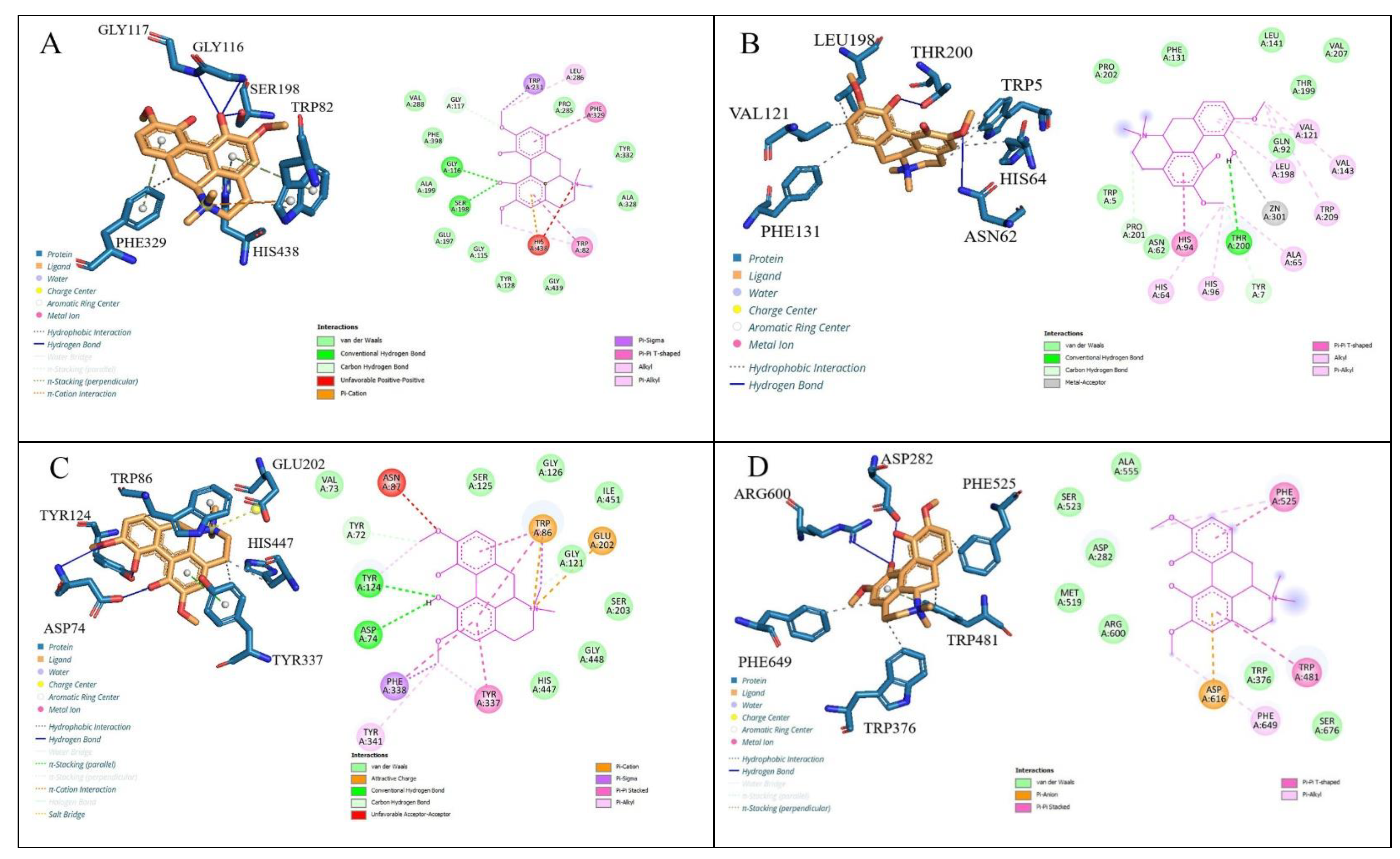
| Complex | Docking Scores (kcal/mol) | Types of Interactions | Interacting Residues |
|---|---|---|---|
| hCA II (3HS4)-Magnofluorine | −8.2 | Hydrogen bonding Hydrophobic interactions π-π T-shaped | Asn62, Thr200, Trp5; His64, His96, Val121, Val143, Leu198, Phe131, His94 |
| AChE (4EY7)-Magnofluorine | −9.5 | Hydrogen bonding Hydrophobic interactions π-π stacked, π cation | Tyr124, Asp74 Trp86, Tyr337, His447 Tyr337, Phe338, Trp86 |
| BChE (6T9P)-Magnofluorine | −9.8 | Hydrogen bonding Hydrophobic interactions π-π stacked, π cation, π sigma | Gly116, Gly117, Ser198, Trp82 Phe329 Trp82, Phe329, His438, Trp231 |
| α-Gly (5NN8)-Magnofluorine | −7.2 | Hydrogen bonding Hydrophobic interactions π-π stacked, π anion | Arg600, Asp282 Trp481, Trp376, Phe525, Phe649 Trp481, Asp616 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durmaz, L.; Kiziltas, H.; Guven, L.; Karagecili, H.; Alwasel, S.; Gulcin, İ. Antioxidant, Antidiabetic, Anticholinergic, and Antiglaucoma Effects of Magnofluorine. Molecules 2022, 27, 5902. https://doi.org/10.3390/molecules27185902
Durmaz L, Kiziltas H, Guven L, Karagecili H, Alwasel S, Gulcin İ. Antioxidant, Antidiabetic, Anticholinergic, and Antiglaucoma Effects of Magnofluorine. Molecules. 2022; 27(18):5902. https://doi.org/10.3390/molecules27185902
Chicago/Turabian StyleDurmaz, Lokman, Hatice Kiziltas, Leyla Guven, Hasan Karagecili, Saleh Alwasel, and İlhami Gulcin. 2022. "Antioxidant, Antidiabetic, Anticholinergic, and Antiglaucoma Effects of Magnofluorine" Molecules 27, no. 18: 5902. https://doi.org/10.3390/molecules27185902






