Acid/Base-Steered Cascade Cyclization: An Efficient One-Pot Access to Diverse Isobenzofuranone and Isoindolobenzoxazinone Derivatives
Abstract
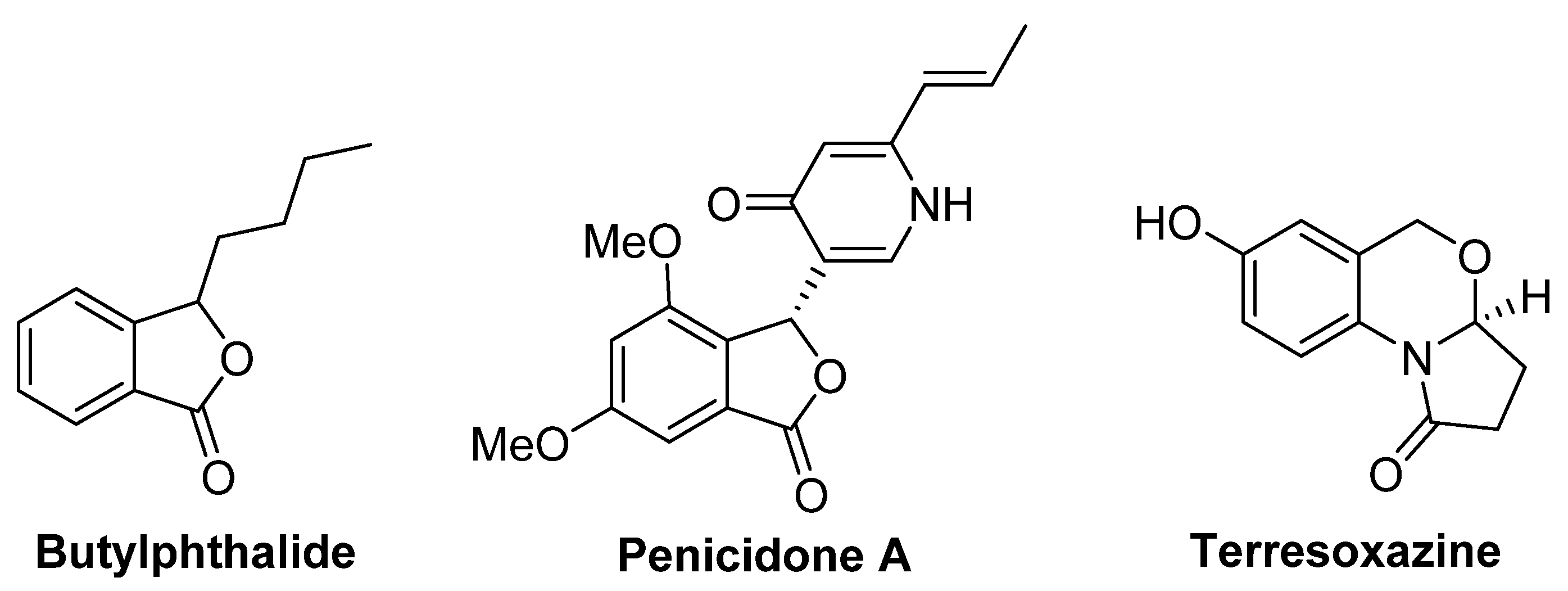
:1. Introduction
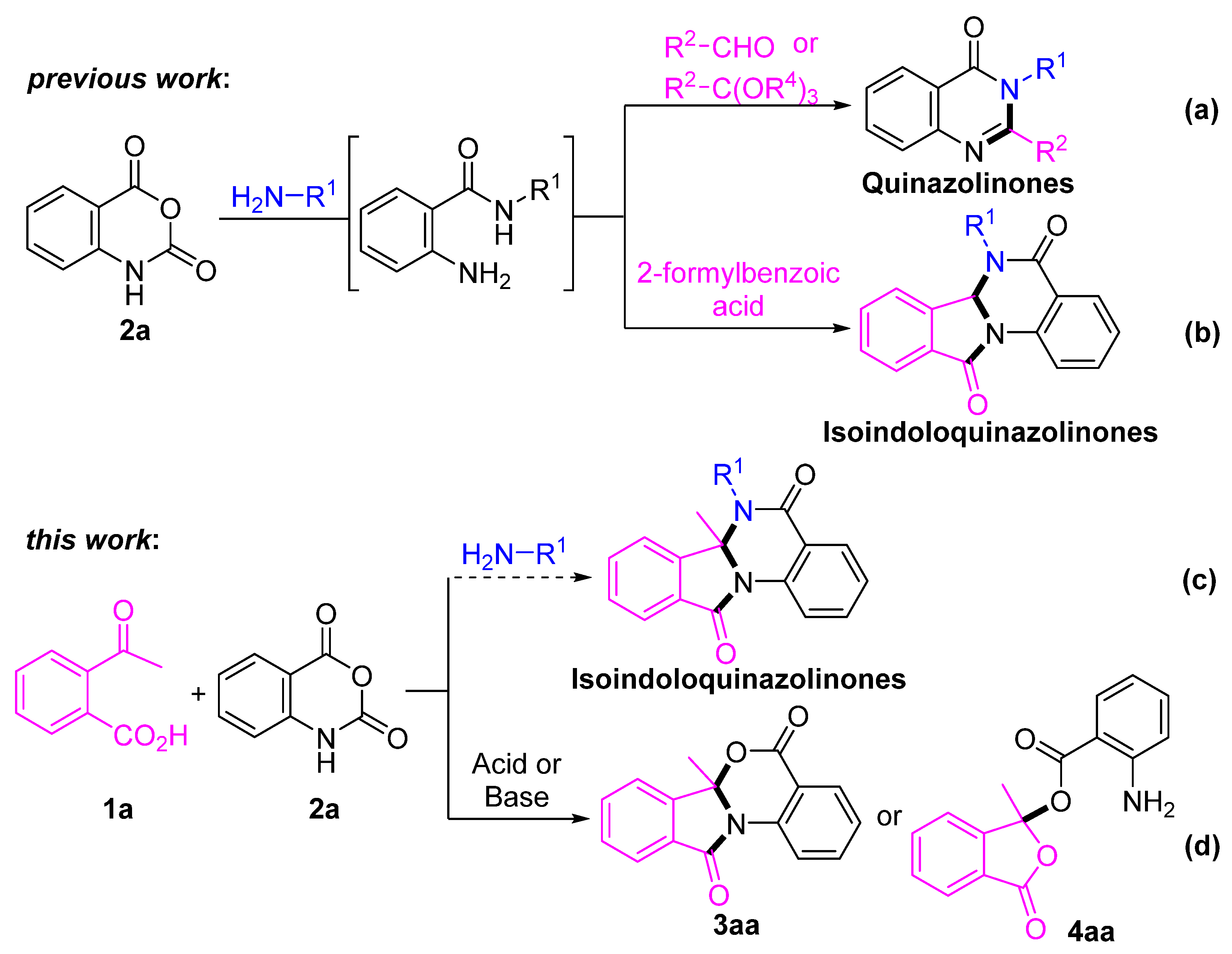
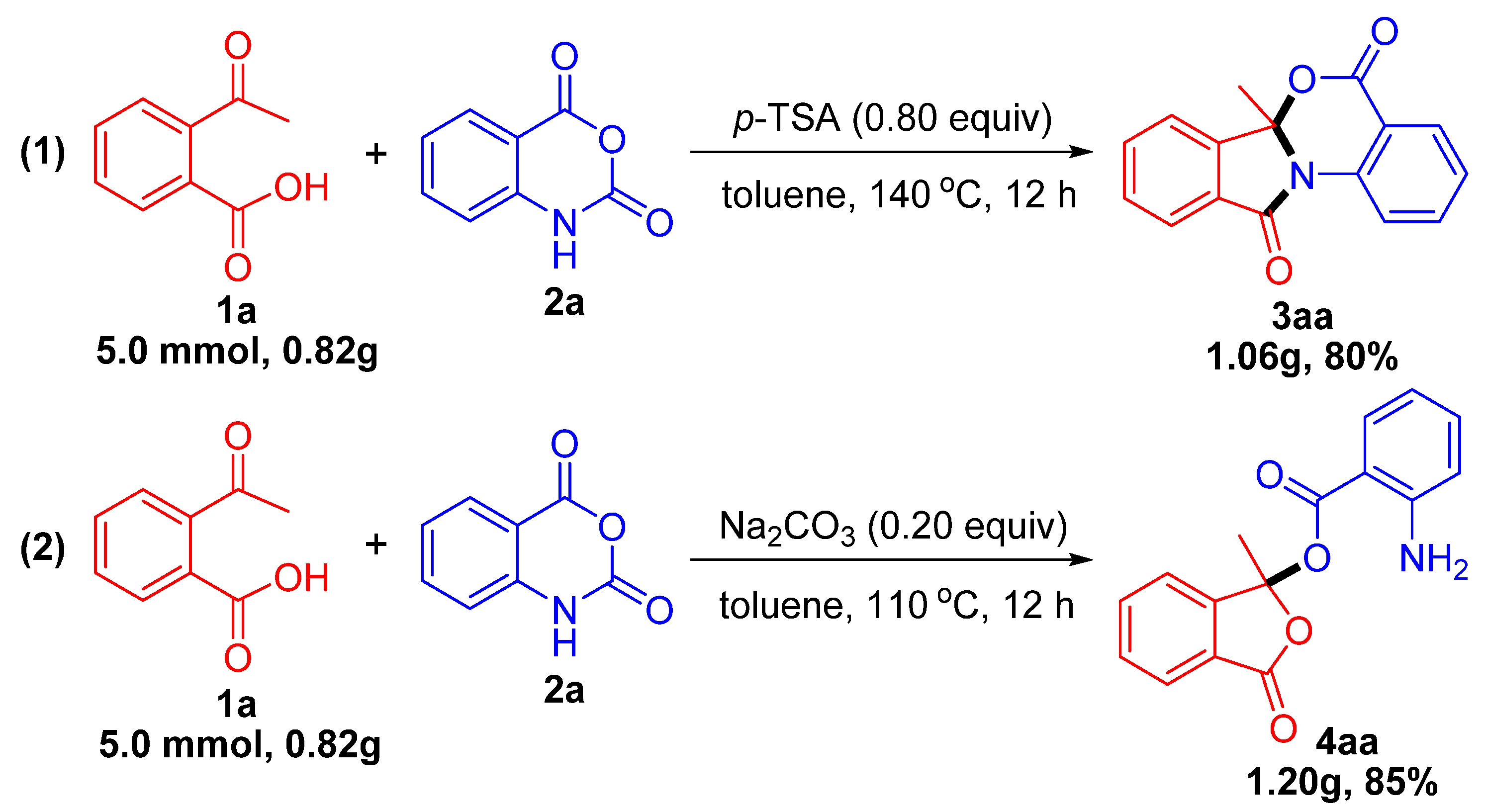
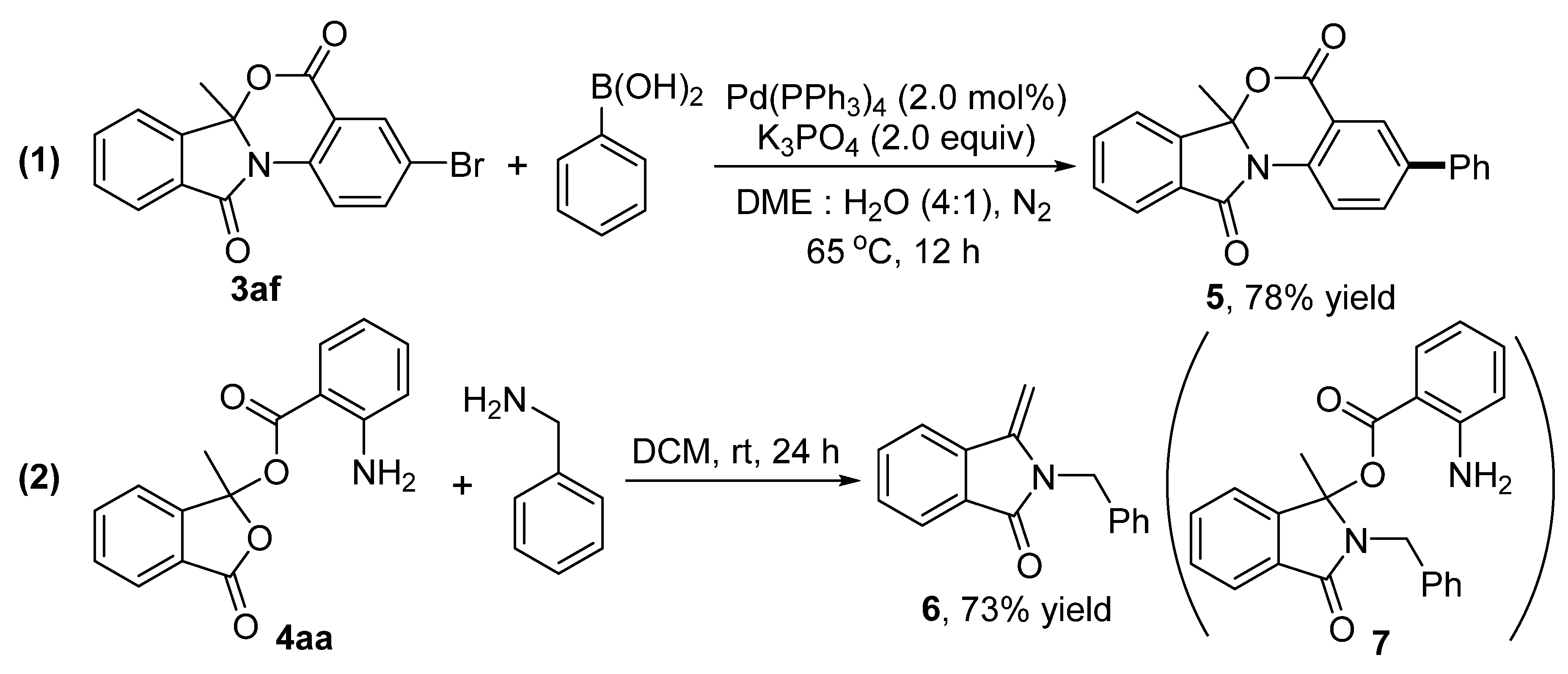
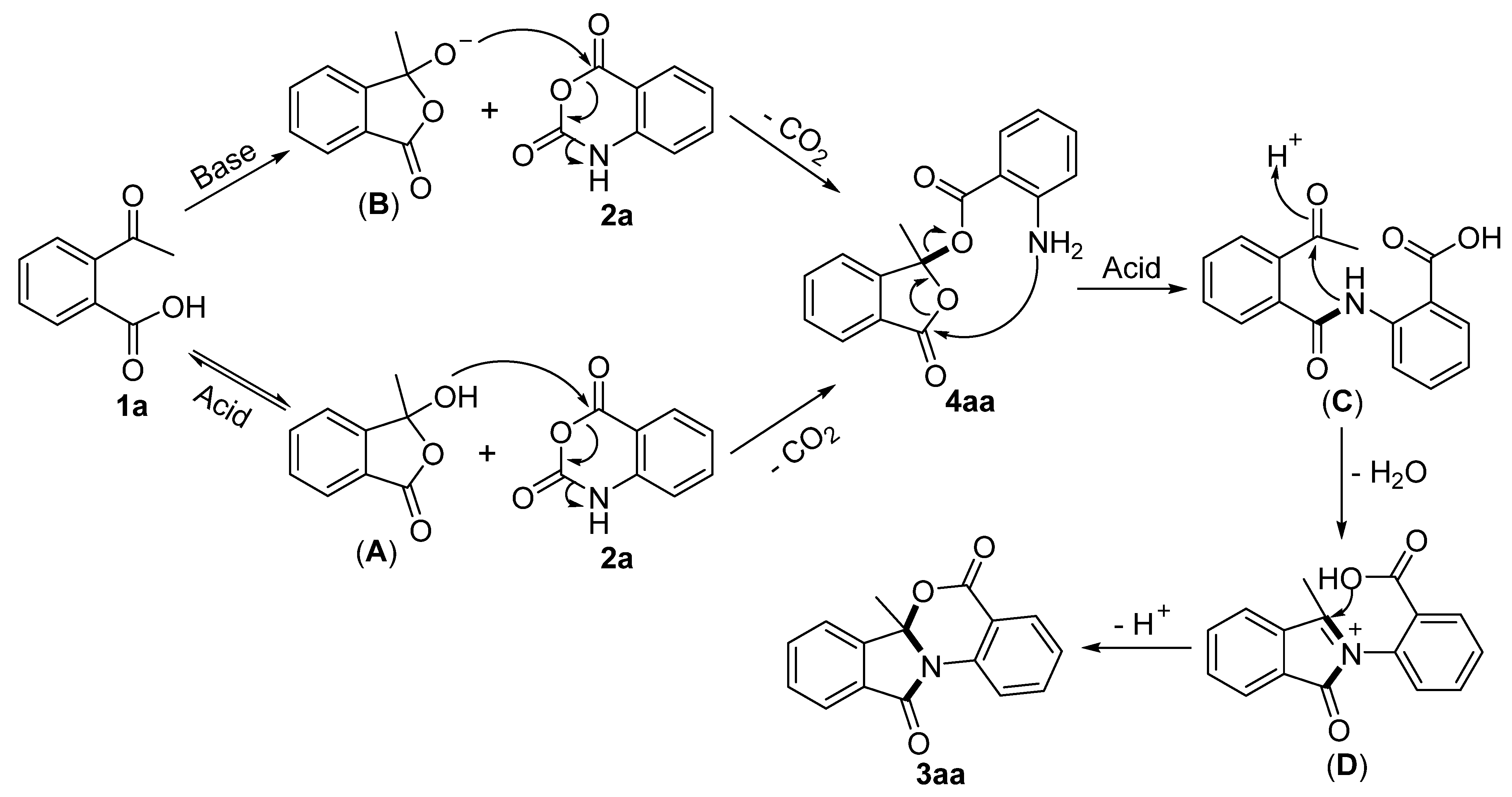
2. Results and Discussion
3. Materials and Methods
3.1. General Information
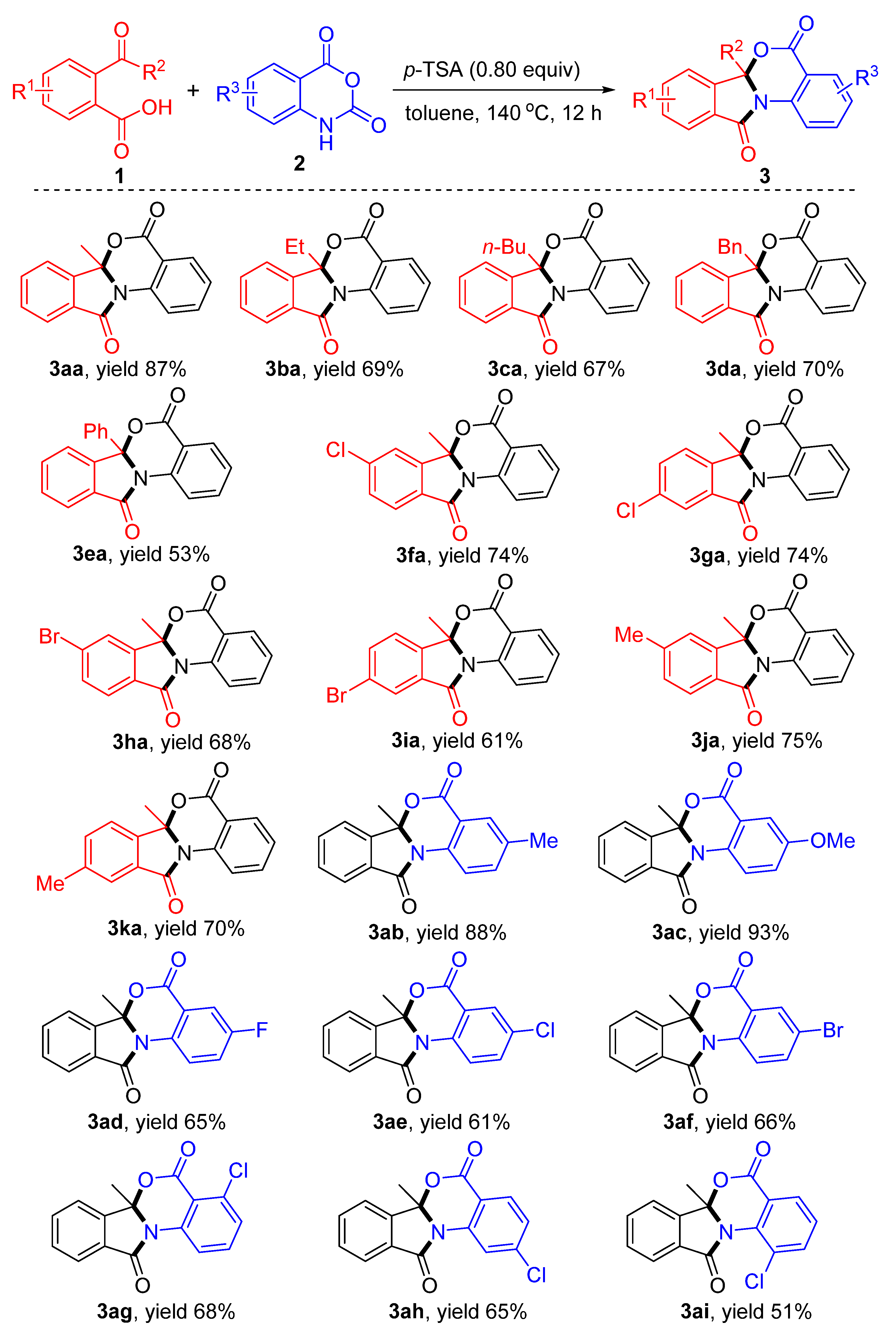
3.2. General Procedure for the Synthesis of Isoindolobenzoxazinones 3
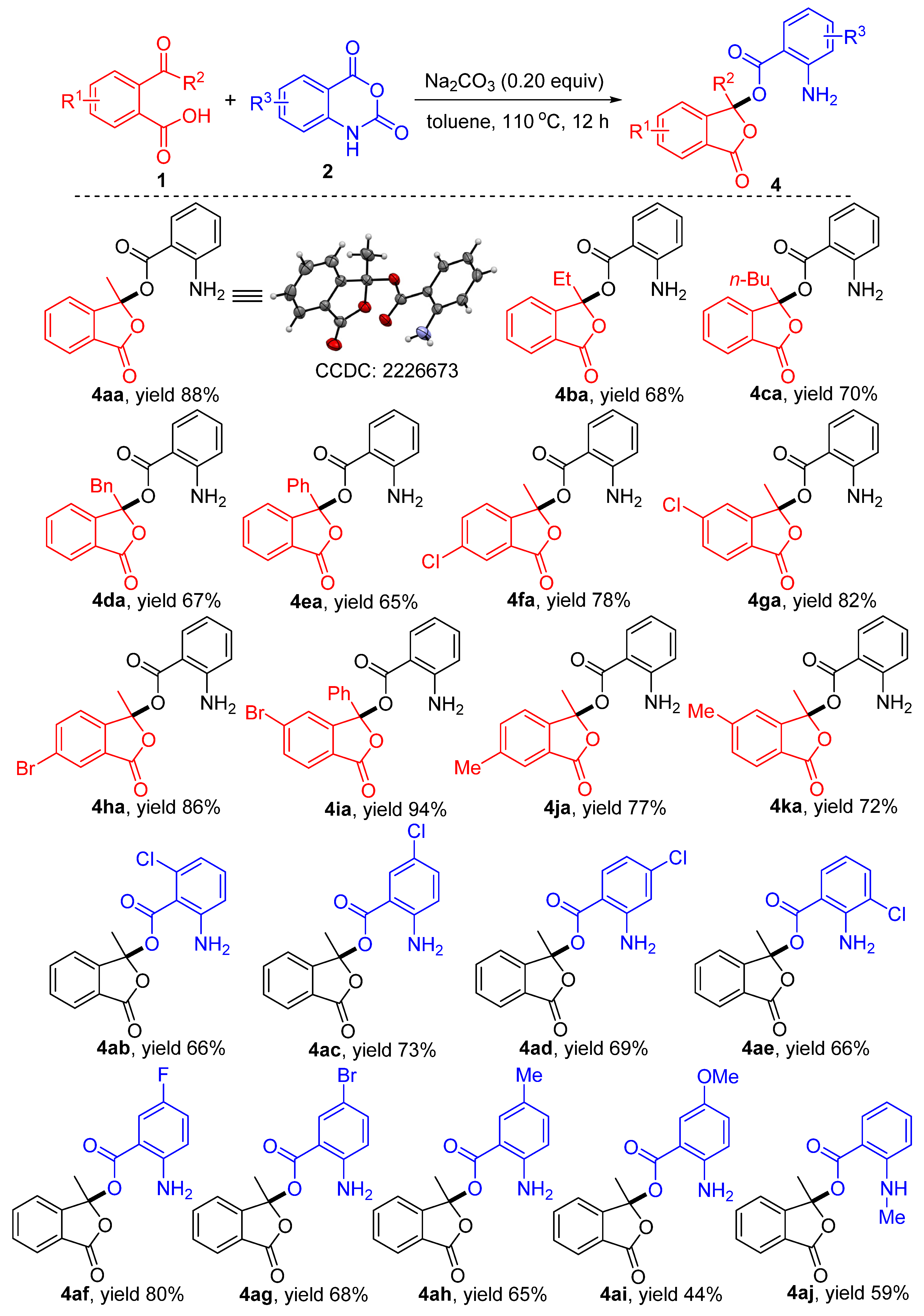
3.3. General Procedure for the Synthesis of Isobenzofuranones 4
3.4. The Procedure for the Suzuki-Miyaura Coupling Reaction
3.5. The Procedure for the Reaction of 4aa with Benzylamine
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Oh, S.; Park, S.B. A design strategy for drug-like polyheterocycles with privileged substructures for discovery of specific small-molecule modulators. Chem. Commun. 2011, 47, 12754–12761. [Google Scholar] [CrossRef]
- Tavassoli, A.; Hamilton, A.D.; Spring, D.R. Small molecules in biology. Chem. Soc. Rev. 2011, 40, 4269–4270. [Google Scholar] [CrossRef]
- Spring, D.R. Chemical genetics to chemical genomics: Small molecules offer big insights. Chem. Soc. Rev. 2005, 34, 472–482. [Google Scholar] [CrossRef]
- Dobson, C.M. Chemical space and biology. Nature 2004, 432, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.; Hopkins, A. Navigating chemical space for biology and medicine. Nature 2004, 432, 855–861. [Google Scholar] [CrossRef]
- Stockwell, B.R. Exploring biology with small organic molecules. Nature 2004, 432, 846–854. [Google Scholar] [CrossRef]
- Sari, A. Two new 3-benzylphthalides from Scorzonera veratrifolia Fenzl. Nat. Prod. Res. 2010, 24, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Knepper, K.; Ziegert, R.E.; Bräse, S. Solid-phase synthesis of isoindolinones and naturally-occurring benzobutyrolactones (phthalides) using a cyclative-cleavage approach. Tetrahedron 2004, 60, 8591–8603. [Google Scholar] [CrossRef]
- Sun, H.; Ho, C.L.; Ding, F.; Soehano, I.; Liu, X.-W.; Liang, Z.-X. Synthesis of (R)-mellein by a partially reducing iterative polyketide synthase. J. Am. Chem. Soc. 2012, 134, 11924–11927. [Google Scholar] [CrossRef] [PubMed]
- Koepfli, J.B.; Mead, J.F.; Brockman, J.A. An alkaloid with high antimalarial activity from dichroa ferbrifuga. J. Am. Chem. Soc. 1947, 69, 1837–1838. [Google Scholar] [CrossRef]
- Tsantrizos, Y.S.; Xu, X.-J. Novel quinazolinones and enniatins from fusarium lateritium Nees. Can. J. Chem. 1993, 71, 1362–1367. [Google Scholar] [CrossRef]
- Kikuchi, S.; Horoiwa, S.; Kasahara, R.; Hariguchi, N.; Matsumoto, M.; Oshima, Y. Synthesis of febrifugine derivatives and development of an effective and safe tetrahydroquinazoline-type antimalarial. Eur. J. Med. Chem. 2014, 76, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Hour, M.-J.; Huang, L.-J.; Kuo, S.-C.; Xia, Y.; Bastow, K.; Nakanishi, Y.; Hamel, E.; Lee, K.-H. 6-Alkylamino- and 2,3-dihydro-3′-methoxy-2-phenyl-4-quinazolinones and related compounds: their synthesis, cytotoxicity, and inhibition of tubulin polymerization. J. Med. Chem. 2000, 43, 4479–4487. [Google Scholar] [CrossRef] [PubMed]
- Chinigo, G.M.; Paige, M.; Grindrod, S.; Hamel, E.; Dakshanamurthy, S.; Chruszcz, M.; Minor, W.; Brown, M.L. Asymmetric synthesis of 2,3-dihydro-2-arylquinazolin-4-ones: Methodology and application to a potent fluorescent tubulin inhibitor with anticancer activity. J. Med. Chem. 2008, 51, 4620–4631. [Google Scholar] [CrossRef]
- Gromachevskaya, E.; Kvitkovskii, F.; Kosulina, T.; Kul’nevich, V. 4H-3,1-Benzoxazines, their salts and dihydro derivatives. Chem. Heterocycl. Compd. 2003, 39, 137–155. [Google Scholar] [CrossRef]
- Wang, W.; Cha, X.-X.; Reiner, J.; Gao, Y.; Qiao, H.-L.; Shen, J.-X.; Chang, J.-B. Synthesis and biological activity of n-butylphthalide derivatives. Eur. J. Med. Chem. 2010, 45, 1941–1946. [Google Scholar] [CrossRef]
- Shimojima, Y.; Hayashi, H.; Ooka, T.; Shibukawa, M.; Iitaka, Y. Studies on AI-77s, microbial products with gastroprotective activity. Structures and the chemical nature of AI-77s. Tetrahedron 1984, 40, 2519–2527. [Google Scholar] [CrossRef]
- Tu, F.X.; Pang, Q.Y.; Huang, T.T.; Zhao, Y.; Liu, M.X.; Chen, X. Apigenin ameliorates post-stroke cognitive deficits in rats through histone acetylation-mediated neurochemical alterations. Med. Sci. Monit. 2017, 23, 4004–4013. [Google Scholar] [CrossRef]
- Ge, H.M.; Shen, Y.; Zhu, C.H.; Tan, S.H.; Ding, H.; Song, Y.C.; Tan, R.X. Penicidones A–C, three cytotoxic alkaloidal metabolites of an endophytic Penicillium sp. Phytochemistry 2008, 69, 571–576. [Google Scholar] [CrossRef]
- Grande, F.; Occhiuzzi, M.A.; Ioele, G.; Ragno, G.; Garofalo, A. Benzopyrroloxazines containing a bridgehead nitrogen atom as promising scaffolds for the achievement of biologically active agents. Eur. J. Med. Chem. 2018, 151, 121–144. [Google Scholar] [CrossRef]
- Akagi, Y.; Yamada, S.I.; Etomi, N.; Kumamoto, T.; Nakanishi, W.; Ishikawa, T. Synthetic studies on jadomycins: Synthesis of dimethyljadomycin A. Tetrahedron Lett. 2010, 51, 1338–1340. [Google Scholar] [CrossRef]
- Karmakar, R.; Pahari, P.; Mal, D. A synthetic route to 1,3-dihydroisobenzofuran natural products: The synthesis of methyl ethers of pestacin. Tetrahedron Lett. 2009, 50, 4042–4045. [Google Scholar] [CrossRef]
- Kumar, P.; Gupta, M.; Bahadur, V.; Parmar, V.S.; Singh, B.K. Radical-induced, palladium-catalyzed C–H activation: An approach to functionalize 4H-benzo[d][1,3]oxazin-4-one derivatives by using toluenes, aldehydes, and benzyl alcohols. Eur. J. Org. Chem. 2018, 13, 1552–1558. [Google Scholar] [CrossRef]
- Kumar Lalji, R.S.; Kumar, P.; Gupta, M.; Bahadur, V.; Parmar, V.S.; Singh, B.K. Palladium-catalyzed decarboxylative synthesis of 5H-benzo [4,5][1,3]oxazino[2,3-a]isoindole-5,11(6aH)-diones using 2-phenyl-4H-benzo[d][1,3]oxazin-4-ones and α-oxo carboxylic acids. Adv. Synth. Catal. 2020, 362, 552–560. [Google Scholar] [CrossRef]
- Karmakar, R.; Pahari, P.; Mal, D. Phthalides and phthalans: Synthetic methodologies and their applications in the total synthesis. Chem. Rev. 2014, 114, 6213–6284. [Google Scholar] [CrossRef]
- Lu, L.; Yang, K.; Zhang, M.-M.; Wang, X.-S. Synthesis of isoindolo[2,1-a]quinazoline derivatives in ionic liquid catalyzed by iodine. J. Heterocycl. Chem. 2014, 51, 630–634. [Google Scholar] [CrossRef]
- Kolotaev, A.V.; Matevosyan, K.R.; Osipov, V.N.; Khachatryan, D.S. Synthesis of new quinazoline-containing hydroxamic acids as potential HDAC/VEGFR inhibitors. Unusual rearrangements with pyrrolidone ring opening and dehydration of 3-N-hydroxyquinazoline fragment containing tetracycles. Tetrahedron Lett. 2019, 60, 151315. [Google Scholar] [CrossRef]
- Boomhoff, M.; Ukis, R.; Schneider, C. A Highly Stereocontrolled, one-pot approach toward pyrrolobenzoxazinones and pyrroloquinazolinones through a lewis acid-catalyzed [3 + 2]-cycloannulation process. J. Org. Chem. 2015, 80, 8236–8244. [Google Scholar] [CrossRef]
- Aeberli, P.; Houlihan, J.W. Reaction of some oxo acids with anthranilic acid, anthranilamides, orthanilamides, and salicylamide. J. Org. Chem. 1968, 33, 2402–2407. [Google Scholar] [CrossRef]
- Gromachevskaya, E.V.; Finko, A.V.; Butin, A.V.; Pushkareva, K.S.; Strelkov, V.D.; Isakova, L.I.; Krapivin, G.D. Substituted 2-formylbenzoic acids in the synthesis of 11H-isoindolo[2,1-a]benzimidazol-11-ones, 5H-isoindolo[2,1-a][3,1]benzoxazine-5,11(6aH)-diones, and 6,6a-dihydroisoindolo-[2,1-a]quinazoline-5,11-diones. Chem. Heterocycl. Comp. 2013, 49, 1331–1344. [Google Scholar] [CrossRef]
- Feng, E.; Zhou, Y.; Zhang, D.; Zhang, L.; Sun, H.; Jiang, H.; Liu, H. Gold(I)-catalyzed tandem transformation: A simple approach for the synthesis of pyrrolo/Pyrido[2,1-a][1,3]benzoxazinones and pyrrolo/Pyrido[2,1-a]quinazolinones. J. Org. Chem. 2010, 75, 3274–3282. [Google Scholar] [CrossRef]
- Qiao, J.; Jia, X.; Li, P.; Liu, X.; Zhao, J.; Zhou, Y.; Wang, J.; Liu, H.; Zhao, F. Gold-catalyzed rapid construction of nitrogen-containing heterocyclic compound library with scaffold diversity and molecular complexity. Adv. Synth. Catal. 2019, 361, 1419–1440. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhai, Y.; Ji, X.; Liu, G.; Feng, E.; Ye, D.; Zhao, L.; Jiang, H.; Liu, H. Gold(I)-catalyzed one-pot tandem coupling/cyclization: An efficient synthesis of pyrrolo-/Pyrido[2,1-b]benzo[d][1,3]oxazin- 1-ones. Adv. Synth. Catal. 2010, 352, 373–378. [Google Scholar] [CrossRef]
- Conde, N.; Herrero, M.T.; Urgoitia, G.; SanMartin, R. Palladium-Catalyzed Domino Cycloisomerization/Double Condensation of Acetylenic Acids with Dinucleophiles. Catalysts 2022, 12, 127. [Google Scholar] [CrossRef]
- Sarralde, J.D.; Astobieta, E.; Sevilla, A.; Rincón, Y.; Herrero, M.T.; Urgoitia, G.; SanMartin, R. Iron-catalyzed cascade synthesis of nitrogen polycycles from alkynoic acids and functionalized amines. Environ. Chem. Lett. 2022, 20, 3421–3427. [Google Scholar] [CrossRef]
- Coppola, G.M. The chemistry of isatoic anhydride. Synthesis 1980, 7, 505–536. [Google Scholar] [CrossRef]
- Clark, R.H.; Wagner, E.C. Isatoic anhydride. I. reactions with primary and secondary amines and with some amides. J. Org. Chem. 1944, 9, 55–67. [Google Scholar] [CrossRef]
- Ji, F.; Lv, M.-F.; Yi, W.-B.; Cai, C. One-pot synthesis of 2-amino-4(3H)-quinazolinones via ring-opening of isatoic anhydride and palladium-catalyzed oxidative isocyanide-insertion. Org. Biomol. Chem. 2014, 12, 5766–5772. [Google Scholar] [CrossRef]
- Patel, S.M.; Chada, H.; Biswal, S.; Sharma, S.; Sharada, D.S. Copper-catalyzed intramolecular α-C–H amination via ring-opening cyclization strategy to quinazolin-4-ones: Development and qpplication in rutaecarpine synthesis. Synthesis 2019, 51, 3160–3170. [Google Scholar] [CrossRef]
- Zhang, J.; Ren, D.; Ma, Y.; Wang, W.; Wu, H. CuO nanoparticles catalyzed simple and efficient synthesis of 2,3-dihydroquinazolin-4(1H)-ones and quinazolin-4(3H)-ones under ultrasound irradiation in aqueous ethanol under ultrasound irradiation in aqueous ethanol. Tetrahedron 2014, 70, 5274–5282. [Google Scholar] [CrossRef]
- Chen, Y.; Shan, W.; Lei, M.; Hu, L. Thiamine hydrochloride (VB1) as an efficient promoter for the one-pot synthesis of 2,3-dihydroquinazolin-4(1H)-ones. Tetrahedron Lett. 2012, 53, 5923–5925. [Google Scholar] [CrossRef]
- Bui, H.T.; Do, K.M.; Nguyen, H.D.; Mai, H.V.; Danh, T.D.; Tran, D.Q.; Morita, H. Efficient one-pot tandem synthesis and cytotoxicity evaluation of 2,3-disubstituted quinazolin-4(3H)-one derivatives. Tetrahedron 2021, 98, 132426. [Google Scholar] [CrossRef]
- Chakraborti, A.K.; Kumar, D.; Jadhavar, P.S.; Nautiyal, M.; Sharma, H.; Meena, P.K.; Adane, L.; Pancholia, S. Convenient synthesis of 2,3-disubstituted quinazolin-4(3H)-ones and 2-styryl-3-substituted quinazolin-4(3H)-ones: Applications towards the synthesis of drugs. RSC Adv. 2015, 5, 30819–30825. [Google Scholar]
- Mohammadi, A.A.; Sadat Hossini, S.S. KAl(SO4)2·12H2O (Alum) catalyzed one-pot three-component synthesis of 2-alkyl and 2-aryl-4(3H)-quinazolinone under microwave irradiation and solvent free conditions. Chin. J. Chem. 2011, 29, 1982–1984. [Google Scholar] [CrossRef]
- Salehi, P.; Dabiri, M.; Zolfigol, M.A.; Baghbanzaden, M. A new approach to the facile synthesis of mono- and disubstituted quinazolin-4(3H)-ones under solvent-free conditions. Tetrahedron Lett. 2005, 46, 7051–7053. [Google Scholar] [CrossRef]
- Hanzarkhani, H.; Karimi, B. A facile synthesis of new 3-(2-benzimidazolyl)-2-alkyl-4-(3H)-quinazolinones under microwave irradiation. Tetrahedron 2003, 59, 4757–4760. [Google Scholar] [CrossRef]
- Reddy, G.R.; Reddy, T.R.; Chary, R.G.; Joseph, S.C.; Mukherjee, S.; Pal, M. β-Cyclodextrin mediated MCR in water: Synthesis of dihydroisoindolo[2,1-a]quinazoline-5,11-dione derivatives under microwave irradiation. Tetrahedron Lett. 2013, 54, 6744–6746. [Google Scholar] [CrossRef]
- Sashidhara, K.V.; Palnai, G.R.; Dodda, R.P.; Avula, S.R.; Swami, P. Studies on novel synthetic methodologies, part XII: An efficient one-pot access to 6,6a-dihydroisoindolo[2,1-a]quinazoline-5,11-diones and 5-phenylisoindolo[2,1-a]quinazolin-11(6aH)-ones. Synlett 2013, 24, 105–113. [Google Scholar] [CrossRef]
- Kumar, K.S.; Kumar, P.M.; Kumar, K.A.; Jafar, A.A.; Krishna, G.R.; Reddy, C.M.; Shivakumar, K.; Priya, K.K.; Parsa, K.L.; Pal, M. A new three-component reaction: Green synthesis of novel isoindolo[2,1-a]quinazoline derivatives as potent inhibitors of TNF-α. Chem. Commun. 2011, 47, 5010–5012. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, W.; Xu, X.-M.; Wang, Z.-L. Tandem reaction of tertiary enamides as a synthetic strategy to construct the fused N-pentacyclic skeleton of erythrina alkaloid derivatives. Org. Lett. 2020, 22, 8814–8818. [Google Scholar] [CrossRef]
- Xu, X.-M.; Chen, D.; Wang, Z.-L. Recent advances in sulfenylation of C(sp3)-H bond under transition metal-free conditions. Chin. Chem. Lett. 2020, 31, 49–57. [Google Scholar] [CrossRef]
- Li, W.; Wang, Y.; Qi, H.; Shi, R.; Li, J.; Chen, S.; Xu, X.-M.; Wang, W.-L. Diverse privileged N-polycyclic skeletons accessed from a metal-free cascade cyclization reaction. Org. Biomol. Chem. 2021, 19, 8086–8095. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.-Q.; Sun, Y.-Y.; Yang, S.-H.; Song, J.-C.; Wang, Z.-L. Persulfate promoted tandem radical cyclization of ortho-cyanoarylacrylamides with oxamic acids for construction of carbamoyl quinoline-2,4-diones under metal-free conditions. Chin. Chem. Lett. 2021, 32, 3632–3635. [Google Scholar] [CrossRef]
- Li, W.; Shi, R.; Chen, S.; Zhang, X.; Peng, W.; Chen, S.; Li, J.; Xu, X.-M.; Zhu, Y.-P.; Wang, X. Synthesis of diverse pentasubstituted pyrroles by a gold(I)-catalyzed cascade rearrangement-cyclization of tertiary enamide. J. Org. Chem. 2022, 87, 3014–3024. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wan, B.; Shi, R.; Chen, S.; Li, J.; Wang, F.; Niu, H.; Xu, X.-M.; Wang, W.-L. Catalyst-free one-pot cascade cyclization: An efficient synthesis of isoindolobenzoxazinones and isoindoloquinazolinones derivatives. Tetrahedron 2022, 104, 132571. [Google Scholar] [CrossRef]
- Li, G.-H.; Han, Q.-Q.; Sun, Y.-Y.; Chen, D.-M.; Wang, Z.-L.; Xu, X.-M.; Yu, X.-Y. Visible-light induced cascade radical cyclization of sulfinic acids and o-(allyloxy)arylaldehydes towards functionalized chroman-4-ones. Chin. Chem. Lett. 2020, 31, 3255–3258. [Google Scholar] [CrossRef]
- Kolsaker, P.; Berner, E. Selfcondensation of some γ-keto acids in sulphuric acid. Tetrahedron 1973, 29, 1095–1100. [Google Scholar] [CrossRef]
- Nishio, T.; Yamamoto, H. Preparation of 3-hydroxyisoindolin-1-ones and o-acylbenzamides. A study of ring-chain tautomerism. J. Heterocycl. Chem. 1995, 32, 883–891. [Google Scholar] [CrossRef]
- Flippin, L.A.; Carter, D.S.; Berger, J.; Clark, R.D.; Bonhaus, D.W.; Leung, E.; Eglen, R.N. (R)-3-(6-chloro-1-isopropylbenzimidazole-4-carboxamido)quinuclidine: A high affinity ligand for the (R)-zacopride binding site. Bioorg. Med. Chem. Lett. 1996, 6, 477–480. [Google Scholar] [CrossRef]
- Jia, X.; Li, P.; Liu, X.; Lin, J.; Chu, Y.; Yu, J.; Wang, J.; Liu, H.; Zhao, F. Green and facile assembly of diverse fused N-heterocycles using gold-catalyzed cascade reactions in water. Molecules 2019, 24, 988. [Google Scholar] [CrossRef]
- Li, W.; Shi, R.; Zhang, X.; Chen, S.; Wang, Y.; Wang, M.; Yang, B.; Li, J.; Xu, X.-M. Different lewis acid promotor-steered highly regioselective phosphorylation of tertiary enamides. J. Org. Chem. 2022, 87, 9769–9781. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-M.; Li, W.; Li, Q.; Chen, S.; Zhang, X.; Yang, B.; Wang, W.-L. Manganese(III)-promoted highly stereoselective phosphorylation of acyclic tertiary enamides to synthesize E-selective β-phosphoryl enamides. Org. Biomol. Chem. 2022, 20, 5566–5574. [Google Scholar] [CrossRef] [PubMed]

| Entry | Cat. (equiv) | Temp. (°C) | Solvent (0.2 M) | 3aa 2 (%) | 4aa 2 (%) |
|---|---|---|---|---|---|
| 1 3 | Na2CO3 (0.2) | 110 | toluene | -- | 90 |
| 2 | Na2CO3 (0.2) | 110 | toluene | -- | 88 |
| 3 | Na2CO3 (1.0) | 110 | toluene | -- | 87 |
| 4 | K2CO3 (1.0) | 110 | toluene | -- | 85 |
| 5 | Cs2CO3 (1.0) | 110 | toluene | -- | 82 |
| 6 | NaHCO3 (1.0) | 110 | toluene | trace | 79 |
| 7 | p-TSA (1.0) | 110 | toluene | 45 | n.d. 4 |
| 8 | AcOH (1.0) | 110 | toluene | trace | n.d. 4 |
| 9 | HCl (1.0) | 110 | toluene | trace | n.d. 4 |
| 10 | CF3SO3H (1.0) | 110 | toluene | trace | n.d. 4 |
| 11 | p-TSA (2.0) | 110 | toluene | 36 | n.d. 4 |
| 12 | p-TSA (0.8) | 110 | toluene | 56 | n.d. 4 |
| 13 | p-TSA (0.6) | 110 | toluene | 28 | n.d. 4 |
| 14 | p-TSA (0.4) | 110 | toluene | 23 | n.d. 4 |
| 15 | p-TSA (0.8) | 120 | toluene | 70 | n.d. 4 |
| 16 | p-TSA (0.8) | 130 | toluene | 83 | n.d. 4 |
| 17 | p-TSA (0.8) | 140 | toluene | 87 | n.d. 4 |
| 18 | p-TSA (0.8) | 150 | toluene | 84 | n.d. 4 |
| 19 5 | p-TSA (0.8) | 140 | toluene | 81 | n.d. 4 |
| 20 6 | p-TSA (0.8) | 140 | toluene | 86 | n.d. 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, R.; Wang, X.; Zhang, X.; Chen, S.; Wang, Z.-L.; Qi, H.; Xu, X.-M. Acid/Base-Steered Cascade Cyclization: An Efficient One-Pot Access to Diverse Isobenzofuranone and Isoindolobenzoxazinone Derivatives. Molecules 2023, 28, 1443. https://doi.org/10.3390/molecules28031443
Shi R, Wang X, Zhang X, Chen S, Wang Z-L, Qi H, Xu X-M. Acid/Base-Steered Cascade Cyclization: An Efficient One-Pot Access to Diverse Isobenzofuranone and Isoindolobenzoxazinone Derivatives. Molecules. 2023; 28(3):1443. https://doi.org/10.3390/molecules28031443
Chicago/Turabian StyleShi, Ran, Xiangmin Wang, Xuesi Zhang, Sen Chen, Zu-Li Wang, Huijing Qi, and Xin-Ming Xu. 2023. "Acid/Base-Steered Cascade Cyclization: An Efficient One-Pot Access to Diverse Isobenzofuranone and Isoindolobenzoxazinone Derivatives" Molecules 28, no. 3: 1443. https://doi.org/10.3390/molecules28031443
APA StyleShi, R., Wang, X., Zhang, X., Chen, S., Wang, Z.-L., Qi, H., & Xu, X.-M. (2023). Acid/Base-Steered Cascade Cyclization: An Efficient One-Pot Access to Diverse Isobenzofuranone and Isoindolobenzoxazinone Derivatives. Molecules, 28(3), 1443. https://doi.org/10.3390/molecules28031443






