Benzothiazole Derivatives. 48. Synthesis of 3-Alkoxycarbonylmethyl-6-bromo-2-benzothiazolones and 3-Alkoxycarbonylmethyl-6-nitro-2-benzothiazolones as Potential Plant Growth Regulators
Abstract
:Introduction
Results and Discussion
Experimental
General
Results of 1H NMR analysis (80 MHz; CDCl3):
6-Bromobenzothiazole skeleton
6-Nitrobenzothiazole skeleton
-CH2-COOR substituents
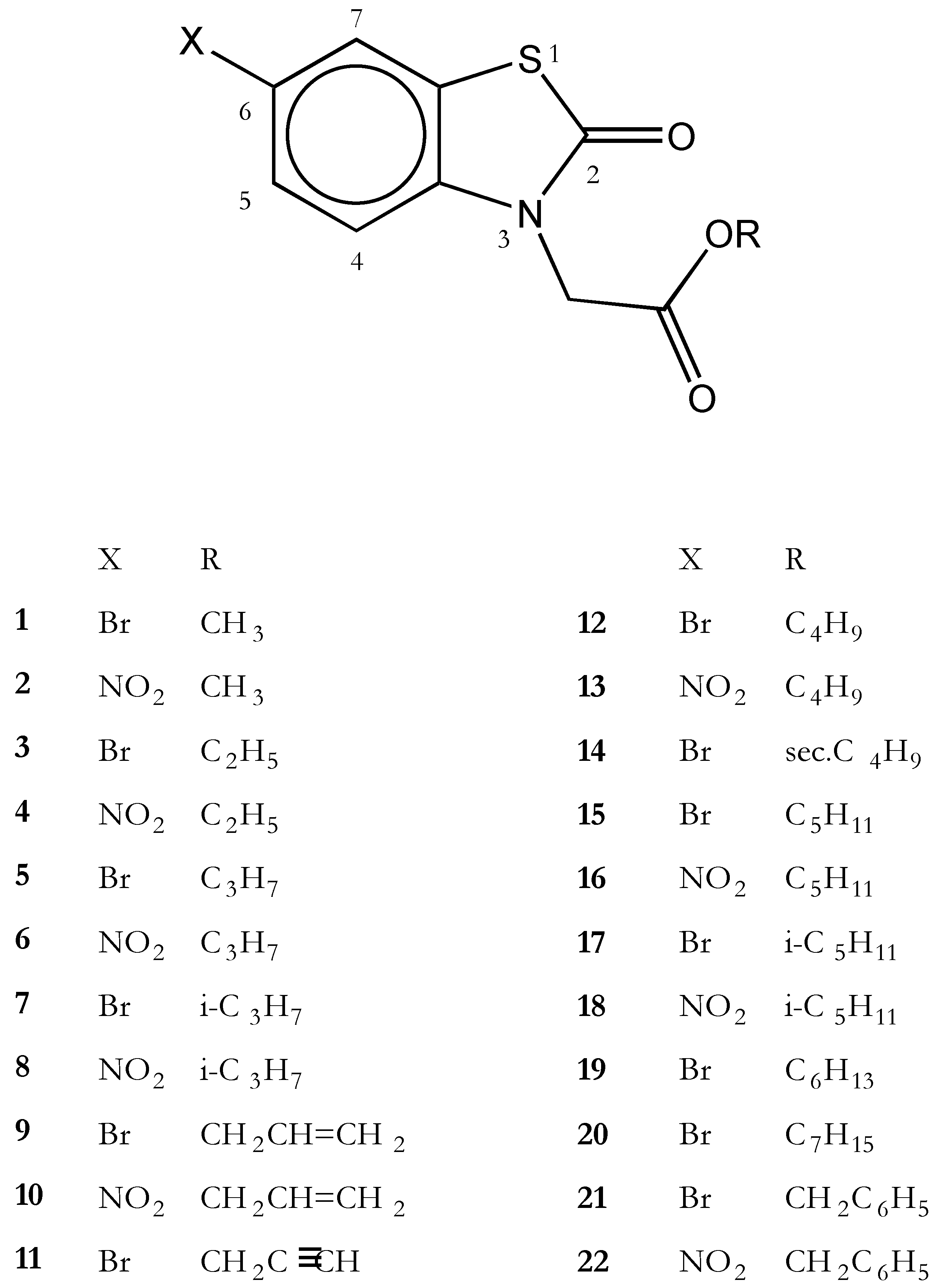
3-Alkoxycarbonylmethyl-6-bromo-2-bezothiazolones 1, 3, 5, 7, 9, 11, 12, 14, 15, 17, 19, 20, 21
3-Alkoxycarbonylmethyl-6-nitro-2-bezothiazolones 2, 4, 6, 8, 10, 13, 16, 18, 22
Acknowledgements
References and Notes
- Sutoris, V.; Sekerka, V.; Gaplovsky, A. Chem. Papers 1991, 45, 687.
- Culak, I.; Gaplovsky, A.; Sutoris, V.; Sekerka, V. Chem. Papers 1990, 44, 381.
- Tran Thi Tuu; Sutoris, V.; Sekerka, V.; Gaplovsky, A.; Loos, D. Chem. Papers 1989, 431, 567.
- Sutoris, V.; Bajci, P.; Sekerka, V.; Halgas, J. Chem. Papers 1988, 42, 249.
- Mikulasek, S.; Sutoris, V.; Perjessy, A.; Foltinova, P. Chem. Papers 1986, 40, 779.
- Sutoris, V.; Mikulasek, S.; Sekerka, V.; Konecny, V. CS Pat. 252502 1989.
- Beska, E.; Konecny, V.; Sutoris, V.; Varkonda, S.; Mikulasek, S.; Sekerka, V. CS Pat. 245866 1989.
- Sekerka, V.; Sutoris, V.; Mikulasek, S.; Konecny, V. CS Pat. 245313 1989.
- Varkonda, S.; Hyblova, O.; Sutoris, V.; Konecny, V.; Mikulasek, S. CS Pat. 252503 1989.
- Sutoris, V.; Sekerka, V.; Halgas, J.; Bajci, P.; Konecny, V.; Varkonda, S. CS Pat. 237706 1987.
- Sutoris, V.; Sekerka, V. CS Pat. 239422 1988.
- Sutoris, V.; Sekerka, V.; Konecny, V. Chem. Papers 1992, 58, 46.
- Sutoris, V.; Gaplovsky, A.; Sekerka, V.; Ertl, P. Chem. Papers 1993, 47, 251.
- Sutoris, V.; Sekerka, V.; Gaplovsky, A. Chem. Papers 1993, 47, 256.
- Sutoris, V.; Gaplovsky, A.; Sekerka, V. Chem. Papers 1993, 47, 260.
- Sutoris, V.; Gaplovsky, A.; Sekerka, V. Chem. Papers 1994, 48, 195.
- Sidoova, E. CS Pat. 265777 1989.
- Sidoova, E. CS Pat. 265776 1989.
- Gunther, H. NMR Spectroscopy; John Wiley: Chichester, 1995; p. 102. [Google Scholar]
- PCMODEL; Serena Software: Bloomington, 1989.
- Dewar, M. J. S.; Zoebisch, E.; Healy, E.; Stewart, J. P. P. J. Am. Chem. Soc. 1985, 107, 3902. [CrossRef]
- Ertl, P. Chem. Listy 1992, 86, 465.
- Samples Availability: Available from the authors.
| Compound | Stimulation | Compound | Stimulation | ||
| Δl (mm) | Δ (%) | Δl (mm) | Δ (%) | ||
| 1 | 1.59 | 26.04a | 13 | 0.26 | 5.71 |
| 2 | 0.26 | 5.78 | 14 | 1.01 | 22.19a |
| 3 | 1.64 | 27.37a | 15 | 1.61 | 26.87a |
| 4 | 0.12 | 2.63 | 16 | 0.42 | 9.23 |
| 5 | 1.50 | 25.04a | 17 | 1.12 | 22.73a |
| 6 | -0.04 | -0.67 | 18 | 0.02 | 0.43 |
| 7 | 1.50 | 25.04 | 19 | 1.80 | 29.10a |
| 8 | 0.56 | 12.30 | 20 | 1.72 | 27.31a |
| 9 | 1.79 | 29.88a | 21 | 0.92 | 20.21a |
| 10 | 0.50 | 9.34 | 22 | 0.29 | 4.84 |
| 11 | 1.60 | 26.11a | IAAb | 5.93 | 100.33 |
| 12 | 1.37 | 22.87a | 2,4-Dc | 2.56 | 51.09 |
| Compound | Q1(S).103 | Q2(C).103 | -Q6(C).103 | μ.1030 /Cm | log P |
| 1 | 299 | 184 | 181 | 11.6 | 1.41 |
| 2 | 325 | 179 | 226 | 11.6 | 0.58 |
| 3 | 298 | 184 | 182 | 11.0 | 1.76 |
| 4 | 324 | 178 | 227 | 11.1 | 0.92 |
| 5 | 298 | 183 | 182 | 11.2 | 2.22 |
| 6 | 325 | 178 | 227 | 10.9 | 1.39 |
| 7 | 296 | 182 | 182 | 11.8 | 2.17 |
| 8 | 323 | 177 | 227 | 12.0 | 1.33 |
| 9 | 299 | 185 | 182 | 10.8 | 2.15 |
| 10 | 325 | 180 | 227 | 10.2 | 1.32 |
| 11 | 299 | 182 | 181 | 11.4 | 1.69 |
| 12 | 298 | 185 | 182 | 9.9 | 2.62 |
| 13 | 324 | 180 | 227 | 10.7 | 1.78 |
| 14 | 296 | 181 | 181 | 11.6 | 2.64 |
| 15 | 297 | 184 | 181 | 10.6 | 3.02 |
| 16 | 324 | 180 | 227 | 10.6 | 2.15 |
| 17 | 297 | 185 | 181 | 10.0 | 3.02 |
| 18 | 324 | 180 | 227 | 10.5 | 2.11 |
| 19 | 298 | 184 | 182 | 10.6 | 3.41 |
| 20 | 297 | 184 | 181 | 10.5 | 3.81 |
| 21 | 293 | 184 | 182 | 9.5 | 3.19 |
| 22 | 319 | 178 | 227 | 11.0 | 2.35 |
 |
  |
© 1999 MDPI. All rights reserved.
Share and Cite
Loos, D.; Sidoova, E.; Sutoris, V. Benzothiazole Derivatives. 48. Synthesis of 3-Alkoxycarbonylmethyl-6-bromo-2-benzothiazolones and 3-Alkoxycarbonylmethyl-6-nitro-2-benzothiazolones as Potential Plant Growth Regulators. Molecules 1999, 4, 81-93. https://doi.org/10.3390/40400081
Loos D, Sidoova E, Sutoris V. Benzothiazole Derivatives. 48. Synthesis of 3-Alkoxycarbonylmethyl-6-bromo-2-benzothiazolones and 3-Alkoxycarbonylmethyl-6-nitro-2-benzothiazolones as Potential Plant Growth Regulators. Molecules. 1999; 4(4):81-93. https://doi.org/10.3390/40400081
Chicago/Turabian StyleLoos, Dusan, Eva Sidoova, and Viktor Sutoris. 1999. "Benzothiazole Derivatives. 48. Synthesis of 3-Alkoxycarbonylmethyl-6-bromo-2-benzothiazolones and 3-Alkoxycarbonylmethyl-6-nitro-2-benzothiazolones as Potential Plant Growth Regulators" Molecules 4, no. 4: 81-93. https://doi.org/10.3390/40400081




