Na+, K+-ATPase Subunit Composition in a Human Chondrocyte Cell Line; Evidence for the Presence of α1, α3, β1, β2 and β3 Isoforms
Abstract
:1. Introduction
2. Results and Discussion
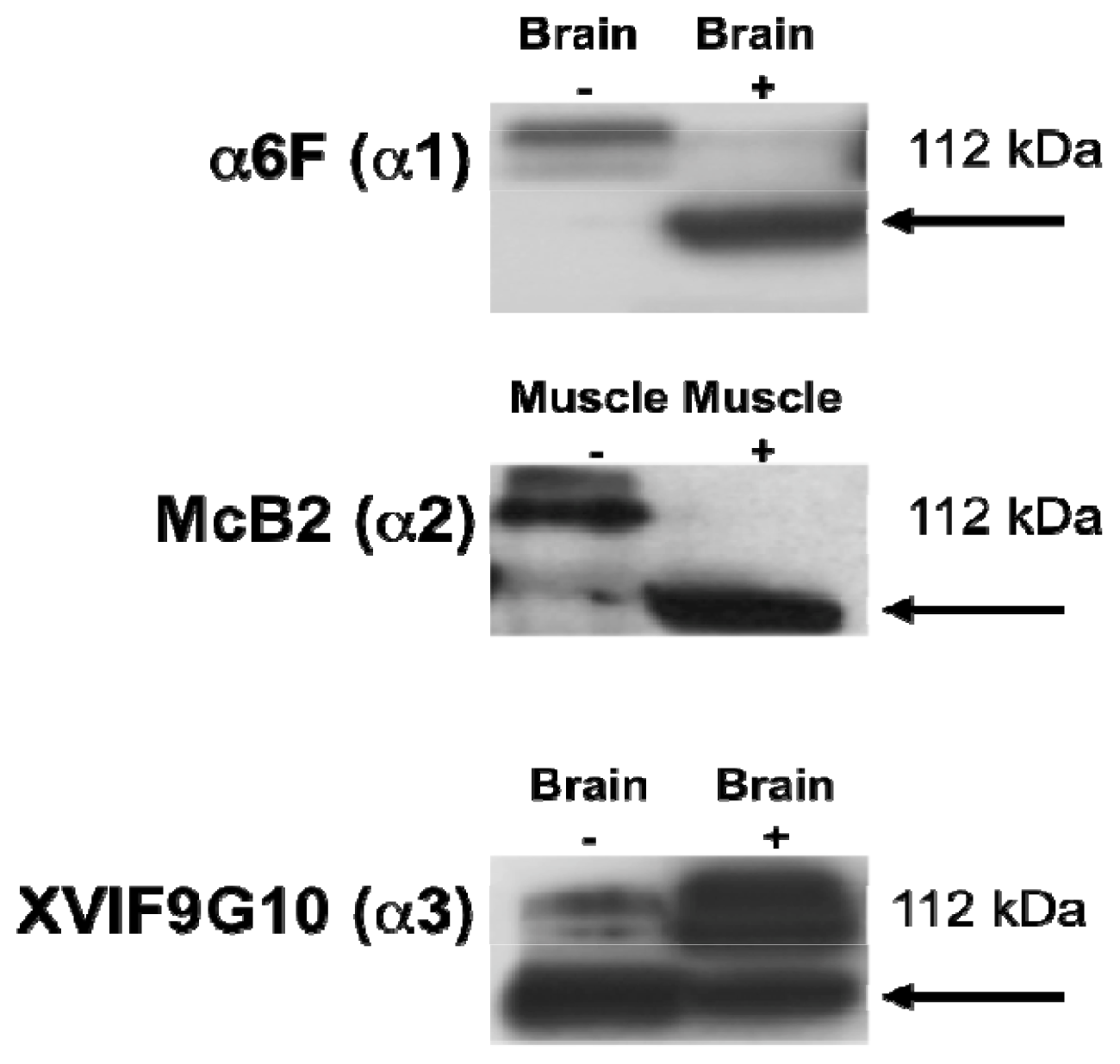
2.1. Characterization of the α Isoform Specific Antibodies
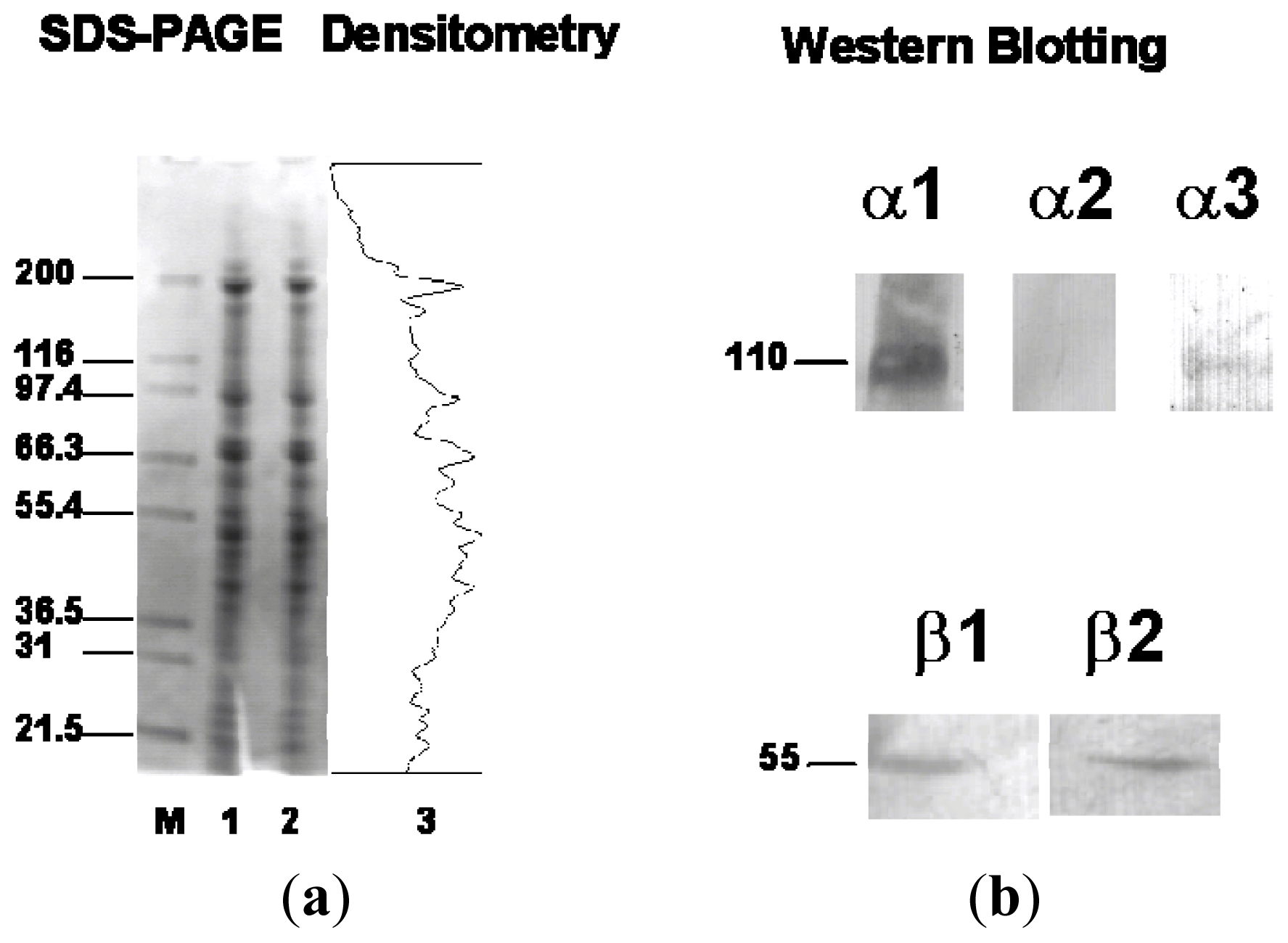
2.2. Western Blotting of α and β Isoforms in C-20/A4 Cells
2.3. FACS Analysis
2.4. Immunofluorescence and Immunocytochemistry
3. Experimental Section
3.1. Chemicals, Tissue Culture and Molecular Biology Kits
3.2. Human Tissues
3.3. Antibodies
3.4. Human Chondrocyte Cell Line
3.5. Preparation of Plasma Membrane Enriched Microsomes
3.6. Gel Electrophoresis and Western Blotting
3.7. Cell Fixation and Immunolabeling
3.8. Immunofluorescence Microscopy
3.9. FACS Analysis
4. Conclusions
Acknowledgments
References
- Lane Smith, R.; Trindade, M.C.; Ikenoue, T.; Mohtai, M.; Das, P.; Carter, D.R.; Goodman, S.B.; Schurman, D.J. Effects of shear stress on articular chondrocyte metabolism. Biorheology 2000, 37, 95–107. [Google Scholar]
- Archer, C.W.; Francis-West, P. The chondrocyte. Int. J. Biochem. Cell Biol 2003, 35, 401–404. [Google Scholar]
- Mobasheri, A.; Mobasheri, R.; Francis, M.J.; Trujillo, E.; Alvarez de la Rosa, D.; Martin-Vasallo, P. Ion transport in chondrocytes: Membrane transporters involved in intracellular ion homeostasis and the regulation of cell volume, free [Ca2+] and pH. Histol. Histopathol 1998, 13, 893–910. [Google Scholar]
- Barrett-Jolley, R.; Lewis, R.; Fallman, R.; Mobasheri, A. The emerging chondrocyte channelome. Front Physiol 2010, 1. [Google Scholar] [CrossRef]
- Mobasheri, A.; Vannucci, S.J.; Bondy, C.A.; Carter, S.D.; Innes, J.F.; Arteaga, M.F.; Trujillo, E.; Ferraz, I.; Shakibaei, M.; Martin-Vasallo, P. Glucose transport and metabolism in chondrocytes: A key to understanding chondrogenesis, skeletal development and cartilage degradation in osteoarthritis. Histol. Histopathol 2002, 17, 1239–1267. [Google Scholar]
- Mobasheri, A.; Bondy, C.A.; Moley, K.; Mendes, A.F.; Rosa, S.C.; Richardson, S.M.; Hoyland, J.A.; Barrett-Jolley, R.; Shakibaei, M. Facilitative glucose transporters in articular chondrocytes. Expression, distribution and functional regulation of GLUT isoforms by hypoxia, hypoxia mimetics, growth factors and pro-inflammatory cytokines. Adv. Anat. Embryol. Cell Biol 2008, 200, 1–84. [Google Scholar]
- Jay, D.G. Role of band 3 in homeostasis and cell shape. Cell 1996, 86, 853–854. [Google Scholar]
- Sarkadi, B.; Parker, J.C. Activation of ion transport pathways by changes in cell volume. Biochim. Biophys. Acta 1991, 1071, 407–427. [Google Scholar]
- Grinstein, S.; Swallow, C.J.; Rotstein, O.D. Regulation of cytoplasmic pH in phagocytic cell function and dysfunction. Clin. Biochem 1991, 24, 241–247. [Google Scholar]
- Brini, M.; Carafoli, E. Calcium signalling: A historical account, recent developments and future perspectives. Cell Mol. Life Sci 2000, 57, 354–370. [Google Scholar]
- Feraille, E.; Doucet, A. Sodium-potassium-adenosinetriphosphatase-dependent sodium transport in the kidney: Hormonal control. Physiol. Rev 2001, 81, 345–418. [Google Scholar]
- Guilak, F. The deformation behavior and viscoelastic properties of chondrocytes in articular cartilage. Biorheology 2000, 37, 27–44. [Google Scholar]
- Trickey, W.R.; Lee, G.M.; Guilak, F. Viscoelastic properties of chondrocytes from normal and osteoarthritic human cartilage. J. Orthop. Res 2000, 18, 891–898. [Google Scholar]
- Mobasheri, A.; Avila, J.; Cozar-Castellano, I.; Brownleader, M.D.; Trevan, M.; Francis, M.J.; Lamb, J.F.; Martin-Vasallo, P. Na+, K+-ATPase isozyme diversity; comparative biochemistry and physiological implications of novel functional interactions. Biosci. Rep 2000, 20, 51–91. [Google Scholar]
- Blanco, G.; Mercer, R.W. Isozymes of the Na-K-ATPase: Heterogeneity in structure, diversity in function. Am. J. Physiol 1998, 275, F633–F650. [Google Scholar]
- Mercer, R.W. Structure of the Na,K-ATPase. Int. Rev. Cytol 1993, 137C, 139–168. [Google Scholar]
- Shull, G.E.; Schwartz, A.; Lingrel, J.B. Amino-acid sequence of the catalytic subunit of the (Na+ + K+)ATPase deduced from a complementary DNA. Nature 1985, 316, 691–695. [Google Scholar]
- Shamraj, O.I.; Lingrel, J.B. A putative fourth Na+,K+-ATPase alpha-subunit gene is expressed in testis. Proc. Natl. Acad. Sci. USA 1994, 91, 12952–12956. [Google Scholar]
- Shull, G.E.; Greeb, J.; Lingrel, J.B. Molecular cloning of three distinct forms of the Na+,K+-ATPase alpha-subunit from rat brain. Biochemistry 1986, 25, 8125–8132. [Google Scholar]
- Martin-Vasallo, P.; Dackowski, W.; Emanuel, J.R.; Levenson, R. Identification of a putative isoform of the Na,K-ATPase beta subunit. Primary structure and tissue-specific expression. J. Biol. Chem 1989, 264, 4613–4618. [Google Scholar]
- Shull, G.E.; Lane, L.K.; Lingrel, J.B. Amino-acid sequence of the β-subunit of the (Na+ + K+)ATPase deduced from a cDNA. Nature 1986, 321, 429–431. [Google Scholar]
- Malik, N.; Canfield, V.A.; Beckers, M.C.; Gros, P.; Levenson, R. Identification of the mammalian Na,K-ATPase 3 subunit. J. Biol. Chem 1996, 271, 22754–22758. [Google Scholar]
- Pestov, N.B.; Adams, G.; Shakhparonov, M.I.; Modyanov, N.N. Identification of a novel gene of the X,K-ATPase beta-subunit family that is predominantly expressed in skeletal and heart muscles. FEBS Lett 1999, 456, 243–248. [Google Scholar]
- Pestov, N.B.; Korneenko, T.V.; Zhao, H.; Adams, G.; Shakhparonov, M.I.; Modyanov, N.N. Immunochemical demonstration of a novel beta-subunit isoform of X, K-ATPase in human skeletal muscle. Biochem. Biophys. Res. Commun 2000, 277, 430–435. [Google Scholar]
- Mobasheri, A.; Errington, R.J.; Golding, S.; Hall, A.C.; Urban, J.P. Characterization of the Na+, K+-ATPase in isolated bovine articular chondrocytes; molecular evidence for multiple alpha and beta isoforms. Cell Biol. Int 1997, 21, 201–212. [Google Scholar]
- Mobasheri, A.; Hall, A.C.; Urban, J.P.; France, S.J.; Smith, A.L. Immunologic and autoradiographic localisation of the Na+, K+-ATPase in articular cartilage: Upregulation in response to changes in extracellular Na+ concentration. Int. J. Biochem. Cell Biol 1997, 29, 649–657. [Google Scholar]
- Mobasheri, A. Correlation between [Na+], [glycosaminoglycan] and Na+/K+ pump density in the extracellular matrix of bovine articular cartilage. Physiol. Res 1998, 47, 47–52. [Google Scholar]
- Mobasheri, A. Regulation of Na+, K+-ATPase density by the extracellular ionic and osmotic environment in bovine articular chondrocytes. Physiol. Res 1999, 48, 509–512. [Google Scholar]
- Trujillo, E.; Alvarez de la Rosa, D.; Mobasheri, A.; Avila, J.; Gonzalez, T.; Martin-Vasallo, P. Sodium transport systems in human chondrocytes. I. Morphological and functional expression of the Na+,K+-ATPase alpha and beta subunit isoforms in healthy and arthritic chondrocytes. Histol. Histopathol 1999, 14, 1011–1022. [Google Scholar]
- Peng, L.; Martin-Vasallo, P.; Sweadner, K.J. Isoforms of Na,K-ATPase alpha and beta subunits in the rat cerebellum and in granule cell cultures. J. Neurosci 1997, 17, 3488–3502. [Google Scholar]
- Resources Page of the Scripps Research Institute’s FACS Core Facility. Available online: http://facs.scripps.edu/software.html accessed on 6 April 2012.
- Trujillo, E.; Alvarez de la Rosa, D.; Mobasheri, A.; Gonzalez, T.; Canessa, C.M.; Martin-Vasallo, P. Brefeldin A influences the cell surface abundance and intracellular pools of low and high ouabain affinity Na+, K+-ATPase alpha subunit isoforms in articular chondrocytes. Histol. Histopathol 1999, 14, 427–438. [Google Scholar]
- Trujillo, E.; Alvarez de la Rosa, D.; Mobasheri, A.; Gonzalez, T.; Canessa, C.M.; Martin-Vasallo, P. Sodium transport systems in human chondrocytes. II. Expression of ENaC, Na+/K+/2Cl− cotransporter and Na+/H+ exchangers in healthy and arthritic chondrocytes. Histol. Histopathol 1999, 14, 1023–1031. [Google Scholar]
- Arystarkhova, E.; Sweadner, K.J. Isoform-specific monoclonal antibodies to Na,K-ATPase alpha subunits. Evidence for a tissue-specific post-translational modification of the alpha subunit. J. Biol. Chem 1996, 271, 23407–23417. [Google Scholar]
- Russo, J.J.; Sweadner, K.J. Na+-K+-ATPase subunit isoform pattern modification by mitogenic insulin concentration in 3T3-L1 preadipocytes. Am. J. Physiol 1993, 264, C311–C316. [Google Scholar]
- Lytton, J. Insulin affects the sodium affinity of the rat adipocyte (Na+,K+)-ATPase. J. Biol. Chem 1985, 260, 10075–10080. [Google Scholar]
- Antonicek, H.; Persohn, E.; Schachner, M. Biochemical and functional characterization of a novel neuron-glia adhesion molecule that is involved in neuronal migration. J. Cell. Biol 1987, 104, 1587–1595. [Google Scholar]
- Lecuona, E.; Luquin, S.; Avila, J.; Garcia-Segura, L.M.; Martin-Vasallo, P. Expression of the beta 1 and beta 2(AMOG) subunits of the Na,K-ATPase in neural tissues: Cellular and developmental distribution patterns. Brain Res. Bull 1996, 40, 167–174. [Google Scholar]
- Schmalzing, G.; Kroner, S.; Schachner, M.; Gloor, S. The adhesion molecule on glia (AMOG/beta 2) and alpha 1 subunits assemble to functional sodium pumps in Xenopus oocytes. J. Biol. Chem 1992, 267, 20212–20216. [Google Scholar]
- Gloor, S.; Antonicek, H.; Sweadner, K.J.; Pagliusi, S.; Frank, R.; Moos, M.; Schachner, M. The adhesion molecule on glia (AMOG) is a homologue of the beta subunit of the Na,K-ATPase. J. Cell. Biol 1990, 110, 165–174. [Google Scholar]
- Magyar, J.P.; Bartsch, U.; Wang, Z.Q.; Howells, N.; Aguzzi, A.; Wagner, E.F.; Schachner, M. Degeneration of neural cells in the central nervous system of mice deficient in the gene for the adhesion molecule on Glia, the beta 2 subunit of murine Na,K-ATPase. J. Cell. Biol 1994, 127, 835–845. [Google Scholar]
- Nelson, W.J.; Veshnock, P.J. Ankyrin binding to (Na+ + K+)ATPase and implications for the organization of membrane domains in polarized cells. Nature 1987, 328, 533–536. [Google Scholar]
- Morrow, J.S.; Cianci, C.D.; Ardito, T.; Mann, A.S.; Kashgarian, M. Ankyrin links fodrin to the alpha subunit of Na,K-ATPase in Madin-Darby canine kidney cells and in intact renal tubule cells. J. Cell. Biol 1989, 108, 455–465. [Google Scholar]
- Nelson, W.J.; Hammerton, R.W. A membrane-cytoskeletal complex containing Na+,K+-ATPase, ankyrin, and fodrin in Madin-Darby canine kidney (MDCK) cells: Implications for the biogenesis of epithelial cell polarity. J. Cell. Biol 1989, 108, 893–902. [Google Scholar]
- Cantiello, H.F. Actin filaments stimulate the Na+-K+-ATPase. Am. J. Physiol 1995, 269, F637–F643. [Google Scholar]
- Arystarkhova, E.; Sweadner, K.J. Tissue-specific expression of the Na,K-ATPase beta3 subunit. The presence of beta3 in lung and liver addresses the problem of the missing subunit. J. Biol. Chem 1997, 272, 22405–22408. [Google Scholar]
- Choi, Y.; Dubel, S.J.; Pacioaiou, M.L.; Omori, A.; Ito, T.; Copeland, T.D.; Takahashi, M.; McEnery, M.W. Parallel detection of Na,K-ATPase alpha subunit isoforms by pan-specific monoclonal mAb 9A7. Arch. Biochem. Biophys 1997, 344, 165–175. [Google Scholar]
- Lebovitz, R.M.; Takeyasu, K.; Fambrough, D.M. Molecular characterization and expression of the (Na+ + K+)-ATPase alpha-subunit in Drosophila melanogaster. EMBO J 1989, 8, 193–202. [Google Scholar]
- Takeyasu, K.; Tamkun, M.M.; Renaud, K.J.; Fambrough, D.M. Ouabain-sensitive (Na+ + K+)-ATPase activity expressed in mouse L cells by transfection with DNA encoding the alpha-subunit of an avian sodium pump. J. Biol. Chem 1988, 263, 4347–4354. [Google Scholar]
- Gottardi, C.J.; Caplan, M.J. Molecular requirements for the cell-surface expression of multisubunit ion-transporting ATPases. Identification of protein domains that participate in Na,K-ATPase and H,K-ATPase subunit assembly. J. Biol. Chem 1993, 268, 14342–14347. [Google Scholar]
- Gottardi, C.J.; Caplan, M.J. Delivery of Na+,K+-ATPase in polarized epithelial cells. Science 1993, 260, 552–554, author reply 554–556. [Google Scholar]
- Urayama, O.; Shutt, H.; Sweadner, K.J. Identification of three isozyme proteins of the catalytic subunit of the Na,K-ATPase in rat brain. J. Biol. Chem 1989, 264, 8271–8280. [Google Scholar]
- Gonzalez-Martinez, L.M.; Avila, J.; Marti, E.; Lecuona, E.; Martin-Vasallo, P. Expression of the beta-subunit isoforms of the Na,K-ATPase in rat embryo tissues, inner ear and choroid plexus. Biol. Cell 1994, 81, 215–222. [Google Scholar]
- The Developmental Studies Hybridoma Bank at the University of Iowa. Available online: http://dshb.biology.uiowa.edu accessed on 6 April 2012.
- Goldring, M.B.; Birkhead, J.R.; Suen, L.F.; Yamin, R.; Mizuno, S.; Glowacki, J.; Arbiser, J.L.; Apperley, J.F. Interleukin-1 beta-modulated gene expression in immortalized human chondrocytes. J. Clin. Invest 1994, 94, 2307–2316. [Google Scholar]
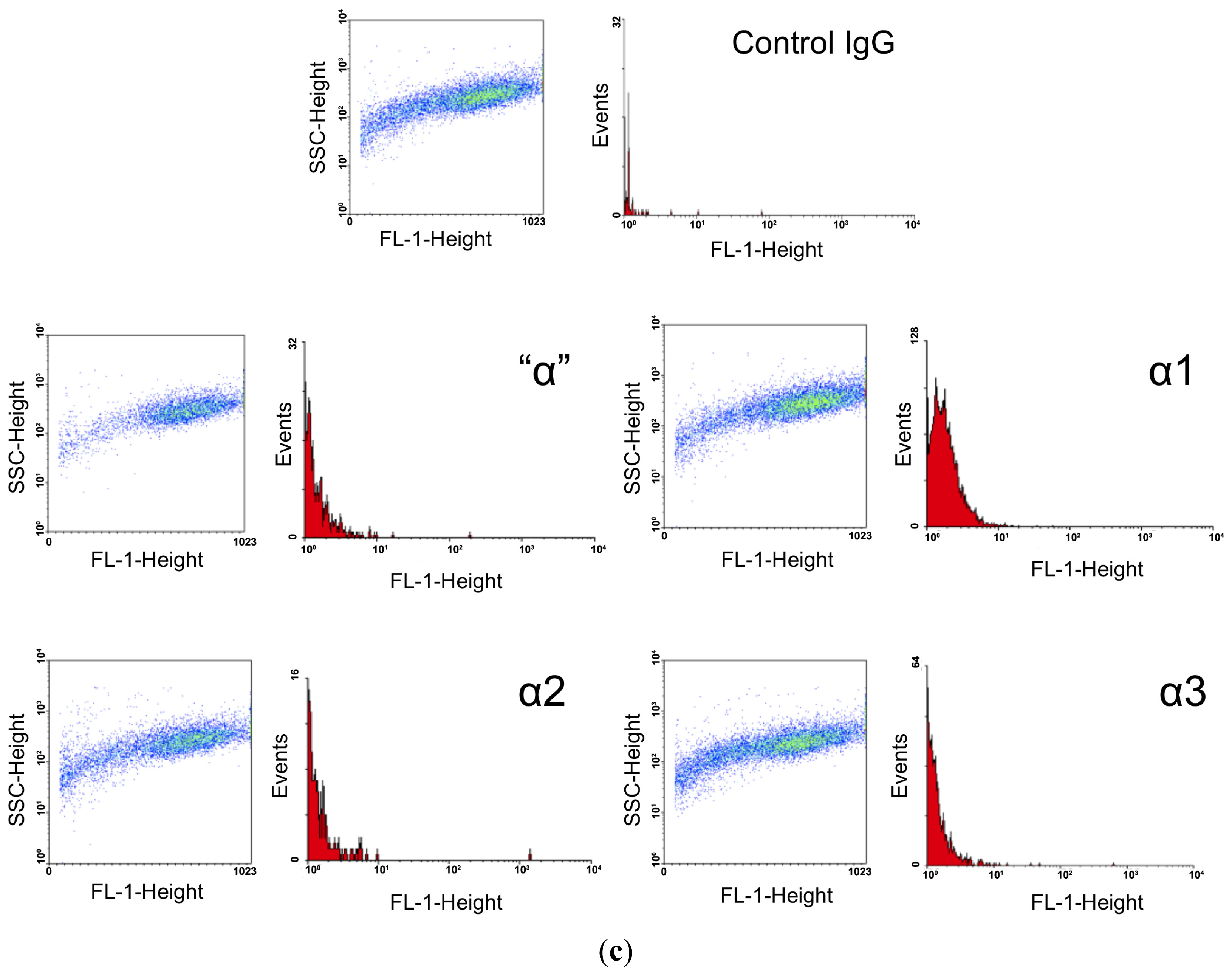


| Antigen | Number of Cells Counted | Number of FITC Positive Cells | % FITC Labeling | Mean Fluorescence Intensity |
|---|---|---|---|---|
| α1 | 6800 | 4568 | 67.18 | 19.2 |
| α2 | 3963 | 45 | 1.14 | 16.5 |
| α3 | 5904 | 312 | 5.28 | 16.1 |
| β1 | 195 | 155 | 79.49 | 36.18 |
| β2 | 3287 | 2866 | 87.19 | 45.83 |
| Antibody | Isoform | Working Dilution | Source | Reference |
|---|---|---|---|---|
| mAb 9A7 (pan α monoclonal) | α isoforms | 1:100 | M. Takahashi | [47] |
| α5 § (monoclonal) | α isoforms | Neat Supernatant | D.M. Fambrough and DSHB * | [48] |
| α6F (monoclonal) | α1 | Neat Supernatant | D.M. Fambrough and DSHB * | [49] |
| α620 (polyclonal) | α1 | 1:100–1:200 | M. Caplan | [50,51] |
| McB2 (monoclonal) | α2 | Neat Supernatant | K.J. Sweadner | [52] |
| McBX3 (monoclonal) | α3 | Neat Supernatant | K.J. Sweadner | [30,46] |
| XVIF9G10 (monoclonal) | α3 | Neat Supernatant | K.J. Sweadner | [30,46] |
| SpETb1 (polyclonal) | β1 | 1:200–1:400 | P. Martín-Vasallo | [53] |
| SpETb2 (polyclonal) | β2 | 1:200–1:400 | P. Martín-Vasallo | [53] |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mobasheri, A.; Trujillo, E.; Arteaga, M.-F.; Martín-Vasallo, P. Na+, K+-ATPase Subunit Composition in a Human Chondrocyte Cell Line; Evidence for the Presence of α1, α3, β1, β2 and β3 Isoforms. Int. J. Mol. Sci. 2012, 13, 5019-5034. https://doi.org/10.3390/ijms13045019
Mobasheri A, Trujillo E, Arteaga M-F, Martín-Vasallo P. Na+, K+-ATPase Subunit Composition in a Human Chondrocyte Cell Line; Evidence for the Presence of α1, α3, β1, β2 and β3 Isoforms. International Journal of Molecular Sciences. 2012; 13(4):5019-5034. https://doi.org/10.3390/ijms13045019
Chicago/Turabian StyleMobasheri, Ali, Elisa Trujillo, Mari-Francis Arteaga, and Pablo Martín-Vasallo. 2012. "Na+, K+-ATPase Subunit Composition in a Human Chondrocyte Cell Line; Evidence for the Presence of α1, α3, β1, β2 and β3 Isoforms" International Journal of Molecular Sciences 13, no. 4: 5019-5034. https://doi.org/10.3390/ijms13045019




