SEPT12-Microtubule Complexes Are Required for Sperm Head and Tail Formation
Abstract
:1. Introduction
1.1. Septins
1.2. SEPT Interaction with Cytoskeletons
1.3. SEPT12 and Other SEPTs in Spermatogenesis
1.4. Roles of α- and β-Tubulins during Spermatogenesis
2. Results and Discussion
2.1. SEPT12 Interacts with α- and β-Tubulins in the Male Germ Cell Line
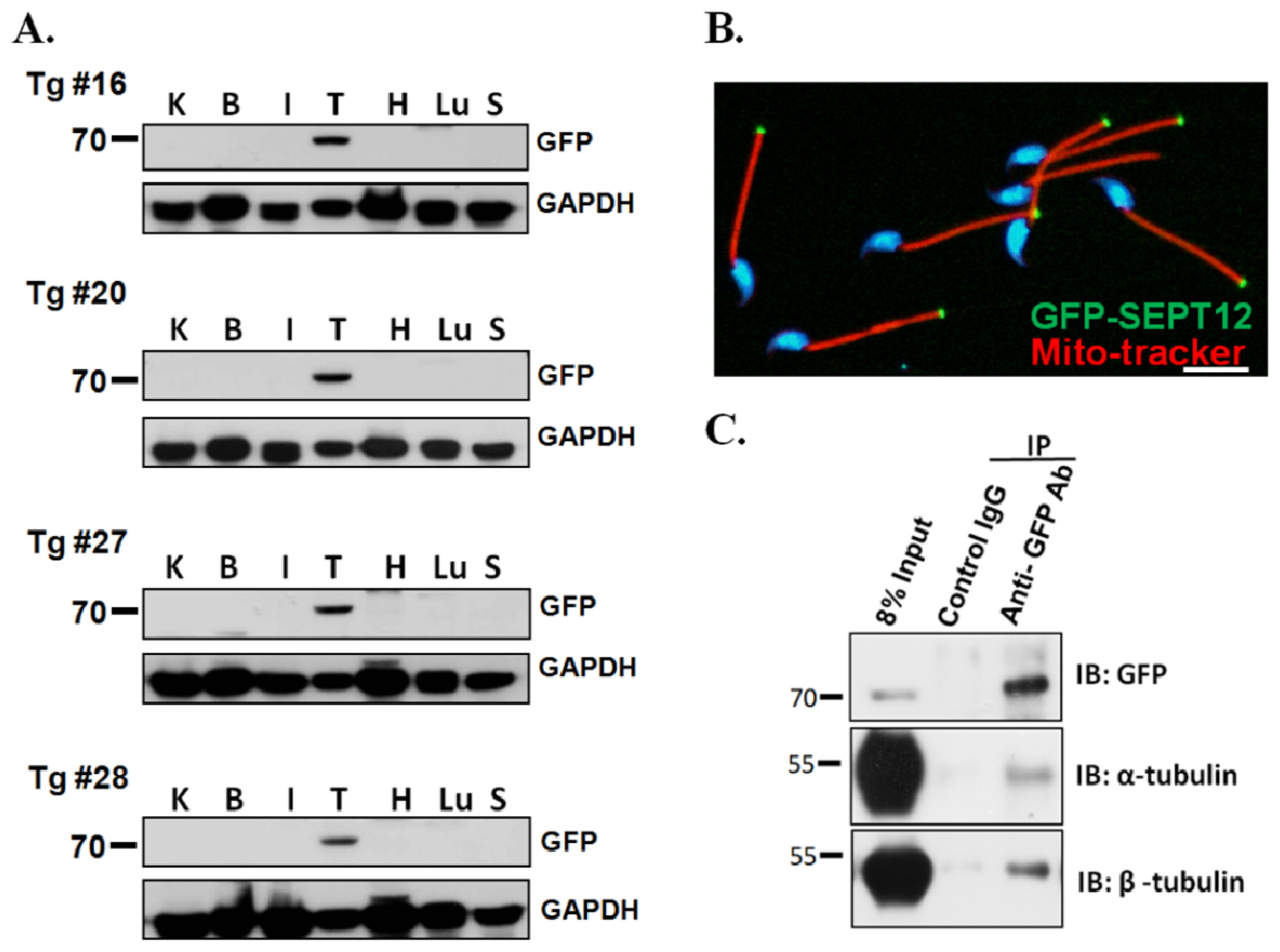
2.2. SEPT12, α- and β-Tubulins form the Complex In Vivo
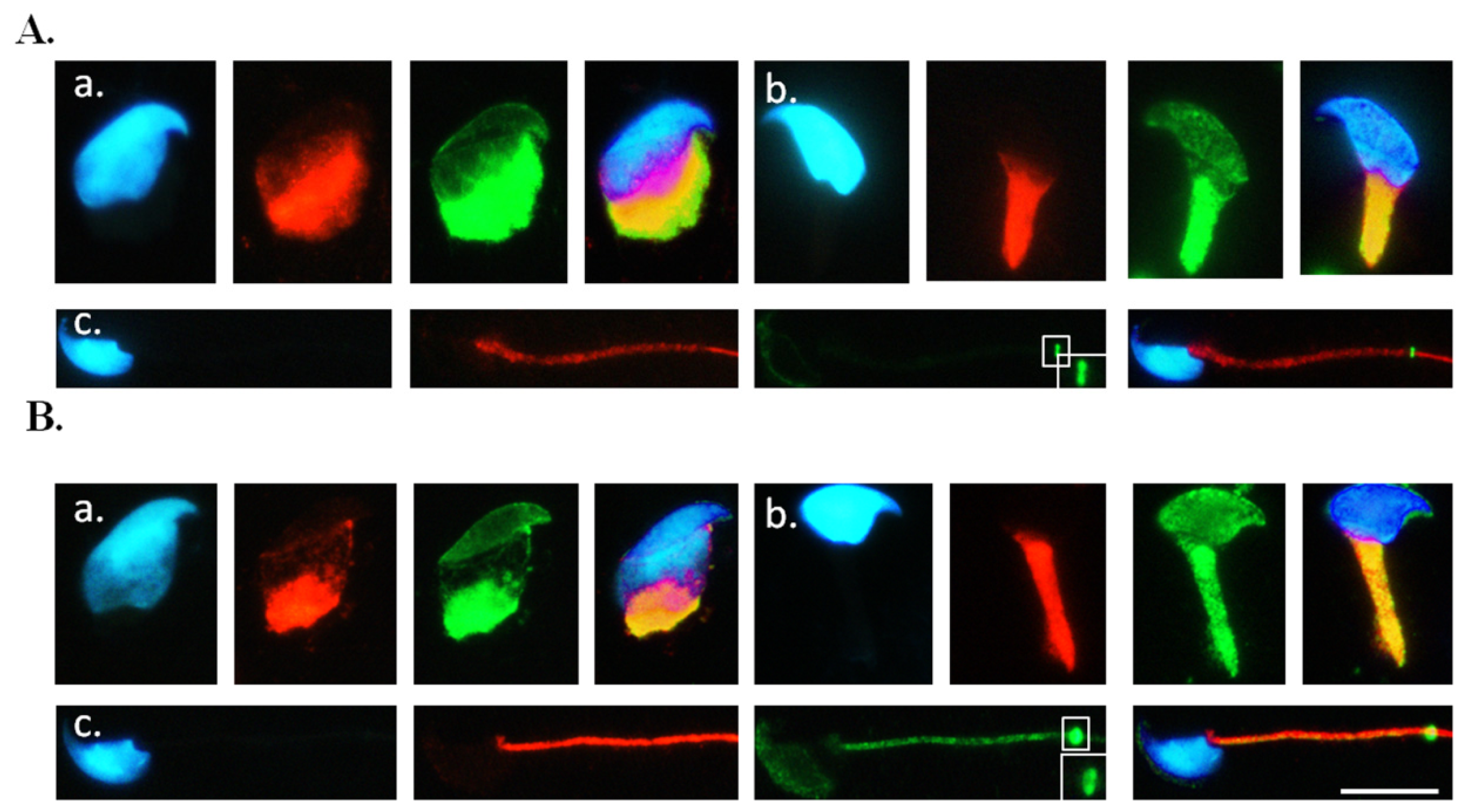
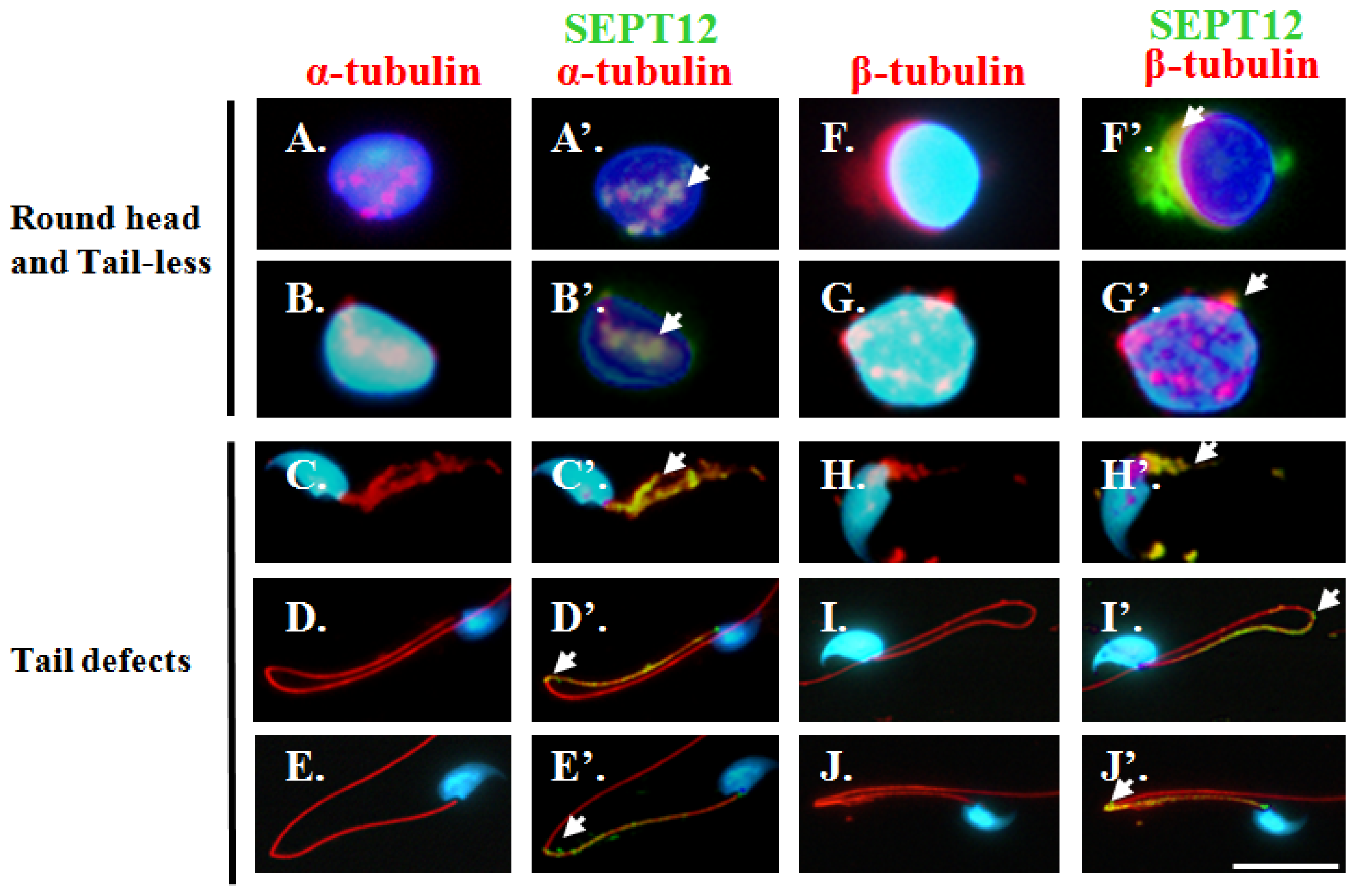
2.3. Co-Localisation of SEPT12 with α-/β-Tubulins in Vivo
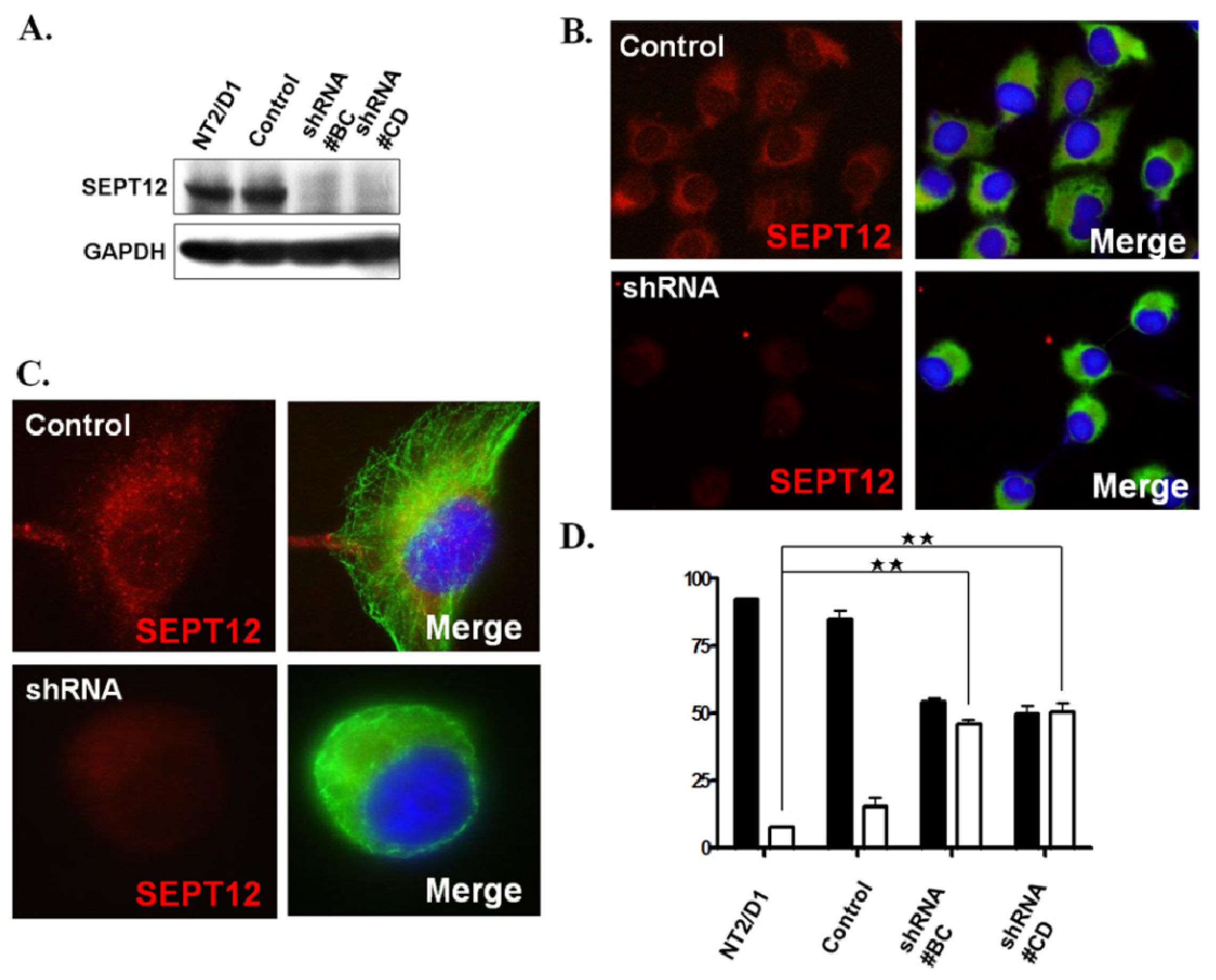
2.4 . SEPTIN12 Knockdown Disrupts the α- and β-Tubulin Patterns and Reshapes Cell Morphology
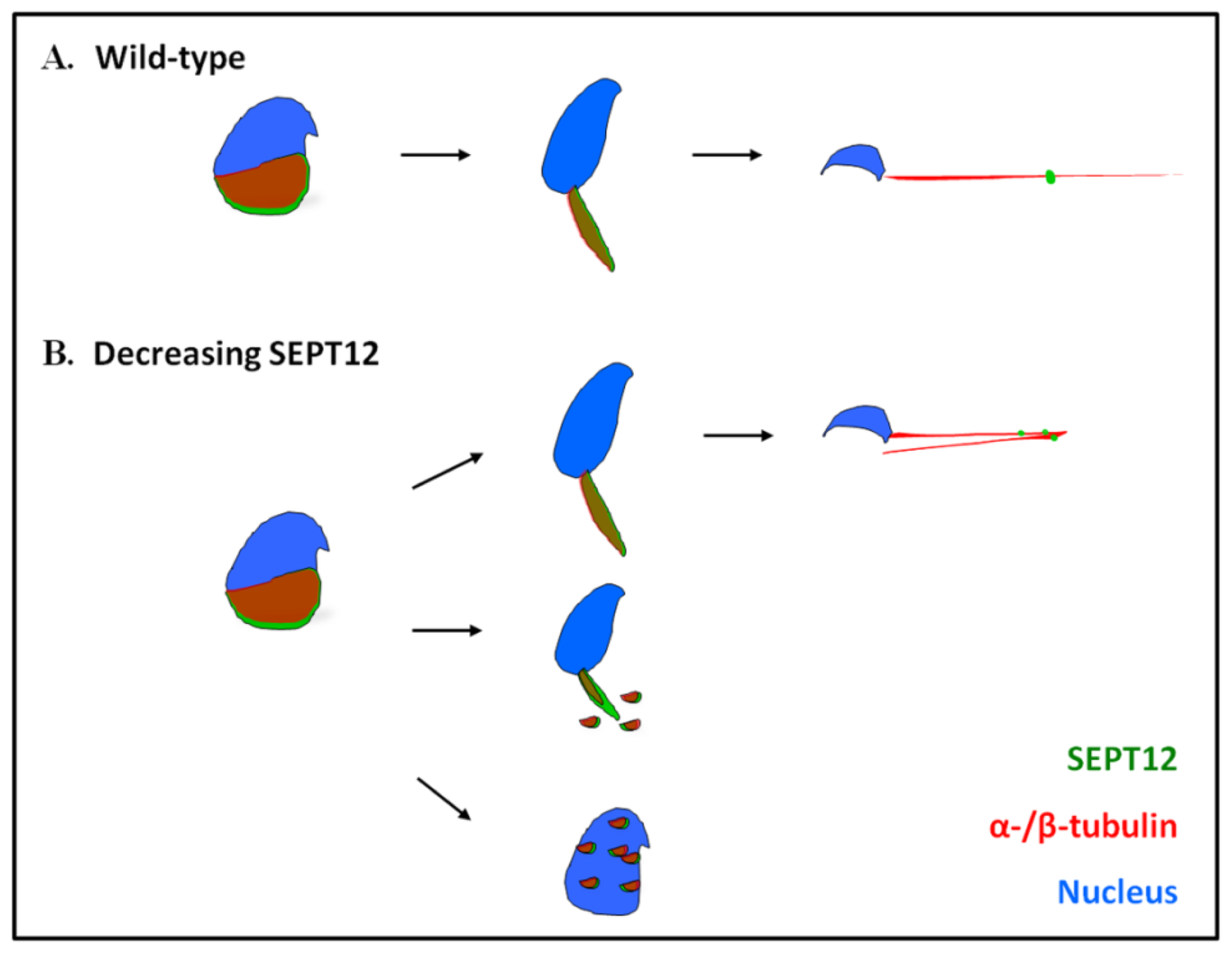
2.5. Reduced SEPT12 Disrupts Sperm Head and Tail Formation in SEPTIN12+/+/+/− Mice
3. Experimental Section
3.1. Cloning and Transfection
3.2. Co-Immunoprecipitation Assay
3.3. Creation of SEPTIN12-Transgenic Mice
3.4. Separation of Murine Testicular Germ Cell Populations and Sperm Preparation
3.5. Immunofluorescence Assay
3.6. Creation of a Stable Clone by Transfecting with SEPTIN12 shRNA
4. Conclusions
4.1. Coordination between SEPTs and Microtubules
4.2. SEPT12 Depletion May Affect Cytoskeleton Organization
4.3. Sperm Head and Tail Formation during Spermiogenesis
Supplementary Information
ijms-14-22102-s001.pdf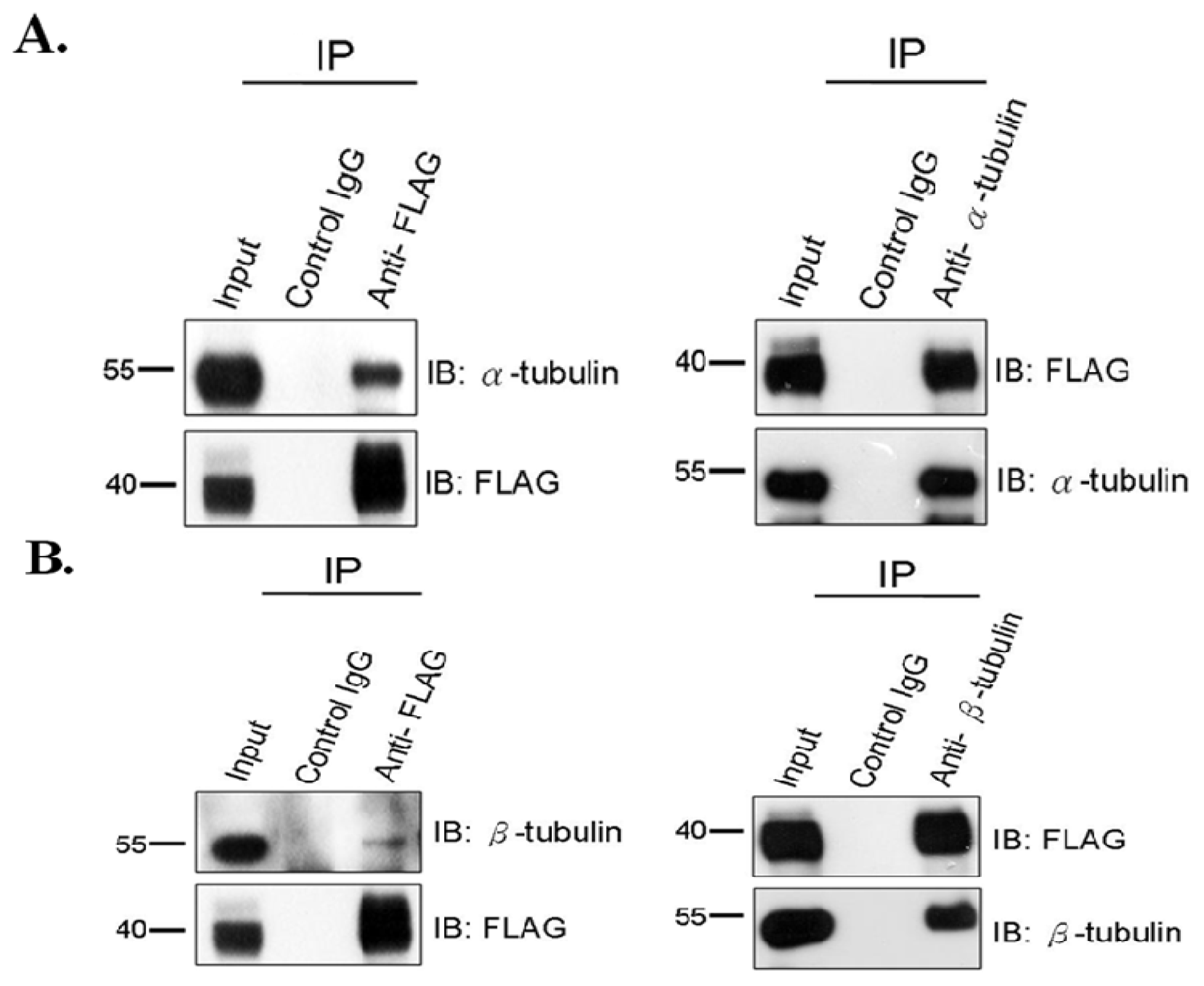
Acknowledgments
Conflicts of Interest
References
- Hall, P.A.; Jung, K.; Hillan, K.J.; Russell, S.E. Expression profiling the human septin gene family. J. Pathol 2005, 206, 269–278. [Google Scholar]
- Kinoshita, M.; Field, C.M.; Coughlin, M.L.; Straight, A.F.; Mitchison, T.J. Self- and actin-templated assembly of Mammalian septins. Dev. Cell 2002, 3, 791–802. [Google Scholar]
- Kremer, B.E.; Haystead, T.; Macara, I.G. Mammalian septins regulate microtubule stability through interaction with the microtubule-binding protein MAP4. Mol. Biol. Cell 2005, 16, 4648–4659. [Google Scholar]
- Hall, P.A.; Russell, S.E. The pathobiology of the septin gene family. J. Pathol 2004, 204, 489–505. [Google Scholar]
- Lin, Y.H.; Kuo, Y.C.; Chiang, H.S.; Kuo, P.L. The role of the septin family in spermiogenesis. Spermatogenesis 2011, 1, 298–302. [Google Scholar]
- Bi, E.; Maddox, P.; Lew, D.J.; Salmon, E.D.; McMillan, J.N.; Yeh, E.; Pringle, J.R. Involvement of an actomyosin contractile ring in Saccharomyces cerevisiae cytokinesis. J. Cell Biol 1998, 142, 1301–1312. [Google Scholar]
- Kinoshita, M.; Kumar, S.; Mizoguchi, A.; Ide, C.; Kinoshita, A.; Haraguchi, T.; Hiraoka, Y.; Noda, M. Nedd5, a mammalian septin, is a novel cytoskeletal component interacting with actin-based structures. Genes Dev 1997, 11, 1535–1547. [Google Scholar]
- Xie, H.; Surka, M.; Howard, J.; Trimble, W.S. Characterization of the mammalian septin H5: Distinct patterns of cytoskeletal and membrane association from other septin proteins. Cell Motil. Cytoskelet 1999, 43, 52–62. [Google Scholar]
- Vega, I.E.; Hsu, S.C. The septin protein Nedd5 associates with both the exocyst complex and microtubules and disruption of its GTPase activity promotes aberrant neurite sprouting in PC12 cells. Neuroreport 2003, 14, 31–37. [Google Scholar]
- Joo, E.; Surka, M.C.; Trimble, W.S. Mammalian SEPT2 is required for scaffolding nonmuscle myosin II and its kinases. Dev. Cell 2007, 13, 677–690. [Google Scholar]
- Spiliotis, E.T.; Kinoshita, M.; Nelson, W.J. A mitotic septin scaffold required for Mammalian chromosome congression and segregation. Science 2005, 307, 1781–1785. [Google Scholar]
- Zhu, M.; Wang, F.; Yan, F.; Yao, P.Y.; Du, J.; Gao, X.; Wang, X.; Wu, Q.; Ward, T.; Li, J.; et al. Septin 7 interacts with centromere-associated protein E and is required for its kinetochore localization. J. Biol. Chem 2008, 283, 18916–18925. [Google Scholar]
- Surka, M.C.; Tsang, C.W.; Trimble, W.S. The mammalian septin MSF localizes with microtubules and is required for completion of cytokinesis. Mol. Biol. Cell 2002, 13, 3532–3545. [Google Scholar]
- Nagata, K.; Kawajiri, A.; Matsui, S.; Takagishi, M.; Shiromizu, T.; Saitoh, N.; Izawa, I.; Kiyono, T.; Itoh, T.J.; Hotani, H.; et al. Filament formation of MSF-A, a mammalian septin, in human mammary epithelial cells depends on interactions with microtubules. J. Biol. Chem 2003, 278, 18538–18543. [Google Scholar]
- Greenbaum, M.P.; Ma, L.; Matzuk, M.M. Conversion of midbodies into germ cell intercellular bridges. Dev. Biol 2007, 305, 389–396. [Google Scholar]
- Ihara, M.; Kinoshita, A.; Yamada, S.; Tanaka, H.; Tanigaki, A.; Kitano, A.; Goto, M.; Okubo, K.; Nishiyama, H.; Ogawa, O.; et al. Cortical organization by the septin cytoskeleton is essential for structural and mechanical integrity of mammalian spermatozoa. Dev. Cell 2005, 8, 343–352. [Google Scholar]
- Kissel, H.; Georgescu, M.M.; Larisch, S.; Manova, K.; Hunnicutt, G.R.; Steller, H. The Sept4 septin locus is required for sperm terminal differentiation in mice. Dev. Cell 2005, 8, 353–364. [Google Scholar]
- Steels, J.D.; Estey, M.P.; Froese, C.D.; Reynaud, D.; Pace-Asciak, C.; Trimble, W.S. Sept12 is a component of the mammalian sperm tail annulus. Cell Motil. Cytoskelet 2007, 64, 794–807. [Google Scholar]
- Lhuillier, P.; Rode, B.; Escalier, D.; Lores, P.; Dirami, T.; Bienvenu, T.; Gacon, G.; Dulioust, E.; Toure, A. Absence of annulus in human asthenozoospermia: Case report. Hum. Reprod 2009, 24, 1296–1303. [Google Scholar]
- Sugino, Y.; Ichioka, K.; Soda, T.; Ihara, M.; Kinoshita, M.; Ogawa, O.; Nishiyama, H. Septins as diagnostic markers for a subset of human asthenozoospermia. J. Urol 2008, 180, 2706–2709. [Google Scholar]
- Lin, Y.H.; Lin, Y.M.; Teng, Y.N.; Hsieh, T.Y.; Lin, Y.S.; Kuo, P.L. Identification of ten novel genes involved in human spermatogenesis by microarray analysis of testicular tissue. Fertil Steril 2006, 86, 1650–1658. [Google Scholar]
- Lin, Y.H.; Lin, Y.M.; Wang, Y.Y.; Yu, I.S.; Lin, Y.W.; Wang, Y.H.; Wu, C.M.; Pan, H.A.; Chao, S.C.; Yen, P.H.; et al. The expression level of septin12 is critical for spermiogenesis. Am. J. Pathol 2009, 174, 1857–1868. [Google Scholar]
- Lin, Y.H.; Chou, C.K.; Hung, Y.C.; Yu, I.S.; Pan, H.A.; Lin, S.W.; Kuo, P.L. SEPT12 deficiency causes sperm nucleus damage and developmental arrest of preimplantation embryos. Fertil Steril 2011, 95, 363–365. [Google Scholar]
- Kuo, Y.C.; Lin, Y.H.; Chen, H.I.; Wang, Y.Y.; Chiou, Y.W.; Lin, H.H.; Pan, H.A.; Wu, C.M.; Su, S.M.; Hsu, C.C.; et al. SEPT12 mutations cause male infertility with defective sperm annulus. Hum. Mutat 2012, 33, 710–719. [Google Scholar]
- Lin, Y.H.; Wang, Y.Y.; Chen, H.I.; Kuo, Y.C.; Chiou, Y.W.; Lin, H.H.; Wu, C.M.; Hsu, C.C.; Chiang, H.S.; Kuo, P.L. SEPTIN12 genetic variants confer susceptibility to teratozoospermia. PLoS One 2012, 7, e34011. [Google Scholar]
- Mochida, K.; Tres, L.L.; Kierszenbaum, A.L. Isolation of the rat spermatid manchette and its perinuclear ring. Dev. Biol 1998, 200, 46–56. [Google Scholar]
- Moreno, R.D.; Schatten, G. Microtubule configurations and post-translational alpha-tubulin modifications during mammalian spermatogenesis. Cell Motil. Cytoskelet 2000, 46, 235–246. [Google Scholar]
- Tachibana, M.; Terada, Y.; Murakawa, H.; Murakami, T.; Yaegashi, N.; Okamura, K. Dynamic changes in the cytoskeleton during human spermiogenesis. Fertil Steril 2005, 84, 1241–1248. [Google Scholar]
- Cole, A.; Meistrich, M.L.; Cherry, L.M.; Trostle-Weige, P.K. Nuclear and manchette development in spermatids of normal and azh/azh mutant mice. Biol. Reprod 1988, 38, 385–401. [Google Scholar]
- Russell, L.D.; Russell, J.A.; MacGregor, G.R.; Meistrich, M.L. Linkage of manchette microtubules to the nuclear envelope and observations of the role of the manchette in nuclear shaping during spermiogenesis in rodents. Am. J. Anat 1991, 192, 97–120. [Google Scholar]
- Kierszenbaum, A.L. Spermatid manchette: Plugging proteins to zero into the sperm tail. Mol. Reprod. Dev 2001, 59, 347–349. [Google Scholar]
- Kierszenbaum, A.L. Intramanchette transport (IMT): Managing the making of the spermatid head, centrosome, and tail. Mol. Reprod. Dev 2002, 63, 1–4. [Google Scholar]
- Kierszenbaum, A.L.; Rivkin, E.; Tres, L.L. Molecular biology of sperm head shaping. Soc. Reprod. Fertil. Suppl 2007, 65, 33–43. [Google Scholar]
- Qi, Y.; Jiang, M.; Yuan, Y.; Bi, Y.; Zheng, B.; Guo, X.; Huang, X.; Zhou, Z.; Sha, J. ADP-ribosylation factor-like 3, a manchette-associated protein, is essential for mouse spermiogenesis. Mol. Hum. Reprod 2013, 19, 327–335. [Google Scholar]
- Bao, J.; Zhang, J.; Zheng, H.; Xu, C.; Yan, W. UBQLN1 interacts with SPEM1 and participates in spermiogenesis. Mol. Cell. Endocrinol 2010, 327, 89–97. [Google Scholar]
- Mochida, K.; Tres, L.L.; Kierszenbaum, A.L. Structural and biochemical features of fractionated spermatid manchettes and sperm axonemes of the azh/azh mutant mouse. Mol. Reprod. Dev 1999, 52, 434–444. [Google Scholar]
- Lin, Y.H.; Lin, Y.M.; Kuo, Y.C.; Wang, Y.Y.; Kuo, P.L. Identification and characterization of a novel Rab GTPase-activating protein in spermatids. Int. J. Androl 2011, 34, e358–e367. [Google Scholar]
- Hall, P.A.; Russell, S.E. Mammalian septins: Dynamic heteromers with roles in cellular morphogenesis and compartmentalization. J. Pathol 2012, 226, 287–299. [Google Scholar]
- Silverman-Gavrila, R.V.; Silverman-Gavrila, L.B. Septins: New microtubule interacting partners. Sci. World J 2008, 8, 611–620. [Google Scholar]
- Hanai, N.; Nagata, K.; Kawajiri, A.; Shiromizu, T.; Saitoh, N.; Hasegawa, Y.; Murakami, S.; Inagaki, M. Biochemical and cell biological characterization of a mammalian septin, Sept11. FEBS Lett 2004, 568, 83–88. [Google Scholar]
- Spiliotis, E.T.; Hunt, S.J.; Hu, Q.; Kinoshita, M.; Nelson, W.J. Epithelial polarity requires septin coupling of vesicle transport to polyglutamylated microtubules. J. Cell Biol 2008, 180, 295–303. [Google Scholar]
- Hu, J.; Bai, X.; Bowen, J.R.; Dolat, L.; Korobova, F.; Yu, W.; Baas, P.W.; Svitkina, T.; Gallo, G.; Spiliotis, E.T. Septin-driven coordination of actin and microtubule remodeling regulates the collateral branching of axons. Curr. Biol. CB 2012, 22, 1109–1115. [Google Scholar]
- Bowen, J.R.; Hwang, D.; Bai, X.; Roy, D.; Spiliotis, E.T. Septin GTPases spatially guide microtubule organization and plus end dynamics in polarizing epithelia. J. Cell Biol 2011, 194, 187–197. [Google Scholar]
- Walenta, J.H.; Didier, A.J.; Liu, X.; Kramer, H. The Golgi-associated hook3 protein is a member of a novel family of microtubule-binding proteins. J. Cell Biol 2001, 152, 923–934. [Google Scholar]
- Mendoza-Lujambio, I.; Burfeind, P.; Dixkens, C.; Meinhardt, A.; Hoyer-Fender, S.; Engel, W.; Neesen, J. The Hook1 gene is non-functional in the abnormal spermatozoon head shape (azh) mutant mouse. Hum. Mol. Genet 2002, 11, 1647–1658. [Google Scholar]
- Kierszenbaum, A.L.; Tres, L.L. Bypassing natural sperm selection during fertilization: The azh mutant offspring experience and the alternative of spermiogenesis in vitro. Mol. Cell. Endocrinol. 2002, 187, 133–138. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kuo, P.-L.; Chiang, H.-S.; Wang, Y.-Y.; Kuo, Y.-C.; Chen, M.-F.; Yu, I.-S.; Teng, Y.-N.; Lin, S.-W.; Lin, Y.-H. SEPT12-Microtubule Complexes Are Required for Sperm Head and Tail Formation. Int. J. Mol. Sci. 2013, 14, 22102-22116. https://doi.org/10.3390/ijms141122102
Kuo P-L, Chiang H-S, Wang Y-Y, Kuo Y-C, Chen M-F, Yu I-S, Teng Y-N, Lin S-W, Lin Y-H. SEPT12-Microtubule Complexes Are Required for Sperm Head and Tail Formation. International Journal of Molecular Sciences. 2013; 14(11):22102-22116. https://doi.org/10.3390/ijms141122102
Chicago/Turabian StyleKuo, Pao-Lin, Han-Sun Chiang, Ya-Yun Wang, Yung-Che Kuo, Mei-Feng Chen, I-Shing Yu, Yen-Ni Teng, Shu-Wha Lin, and Ying-Hung Lin. 2013. "SEPT12-Microtubule Complexes Are Required for Sperm Head and Tail Formation" International Journal of Molecular Sciences 14, no. 11: 22102-22116. https://doi.org/10.3390/ijms141122102



