Role of CBFs as Integrators of Chloroplast Redox, Phytochrome and Plant Hormone Signaling during Cold Acclimation
Abstract
:1. Introduction
2. CBF–Hormone Interactions, Cold Acclimation and Dwarf Phenotype
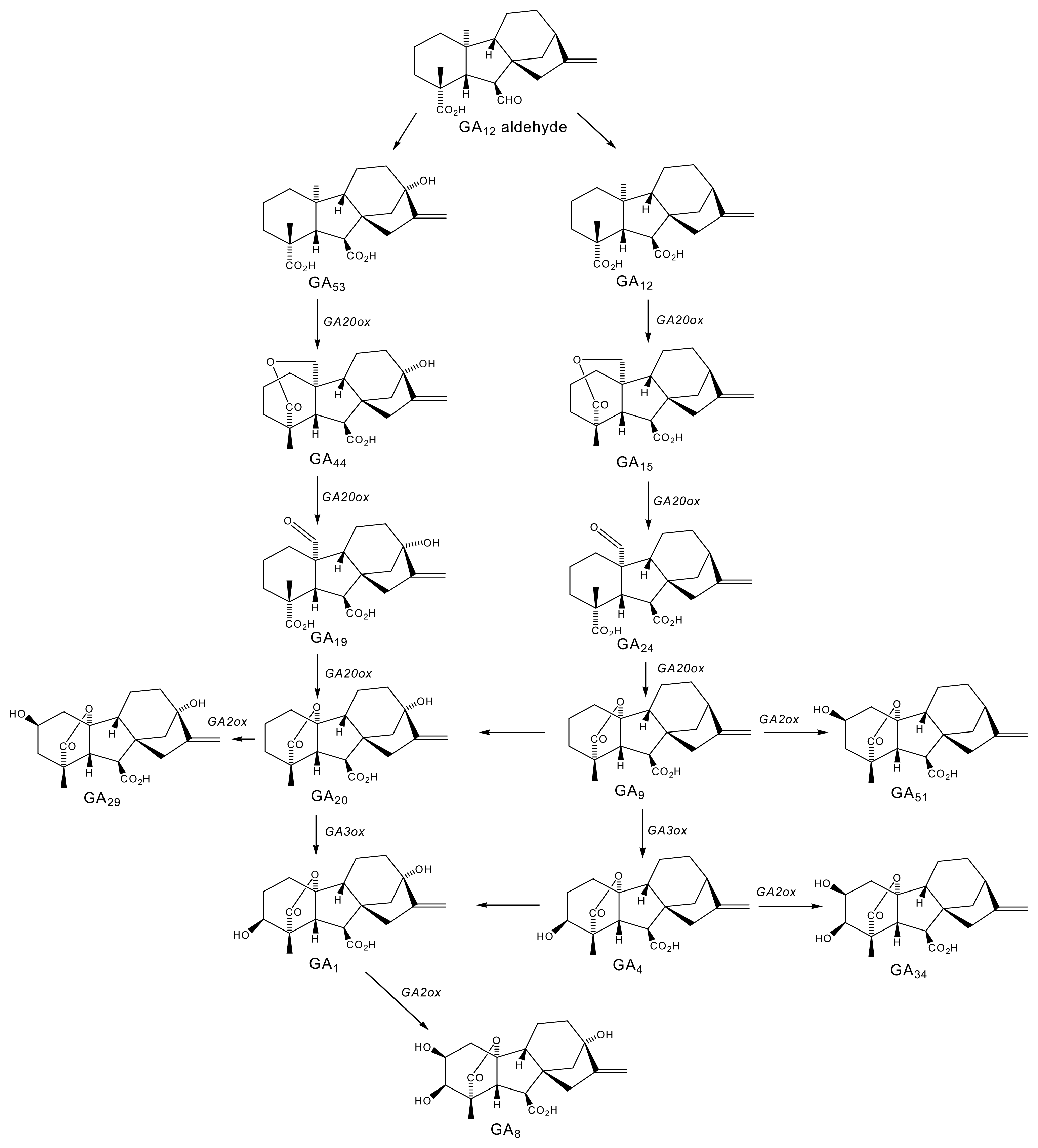
2.1. Gibberellins
2.2. Abscisic Acid
2.3. Cytokinins
2.4. Ethylene
2.5. Brassinosteroids
2.6. Salicylic Acid
3. Light, Cold Acclimation and Freezing Tolerance
3.1. Light Quality, Photoreceptors and Cold Acclimation
3.2. Light Intensity, Photosynthesis and Cold Acclimation
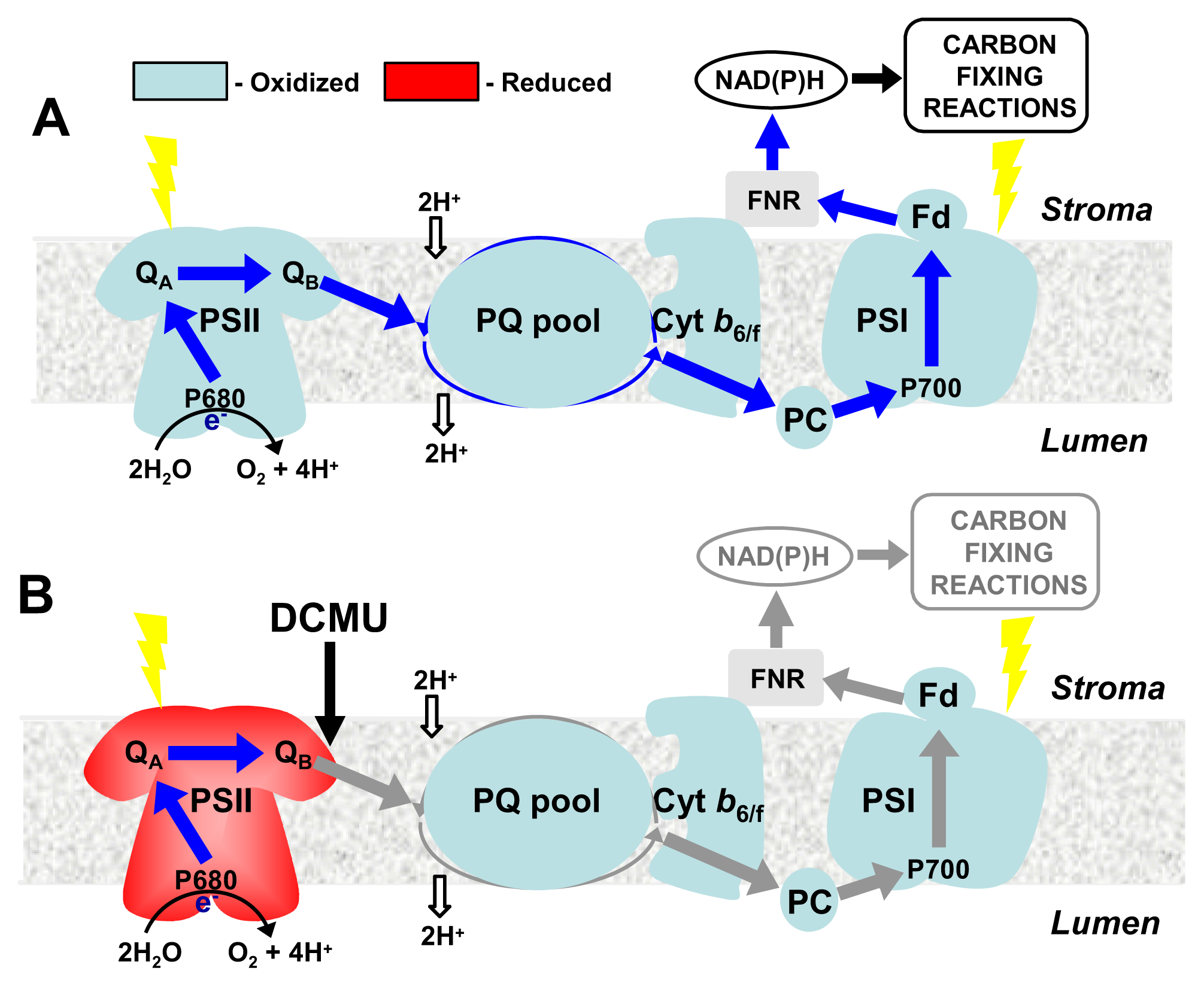
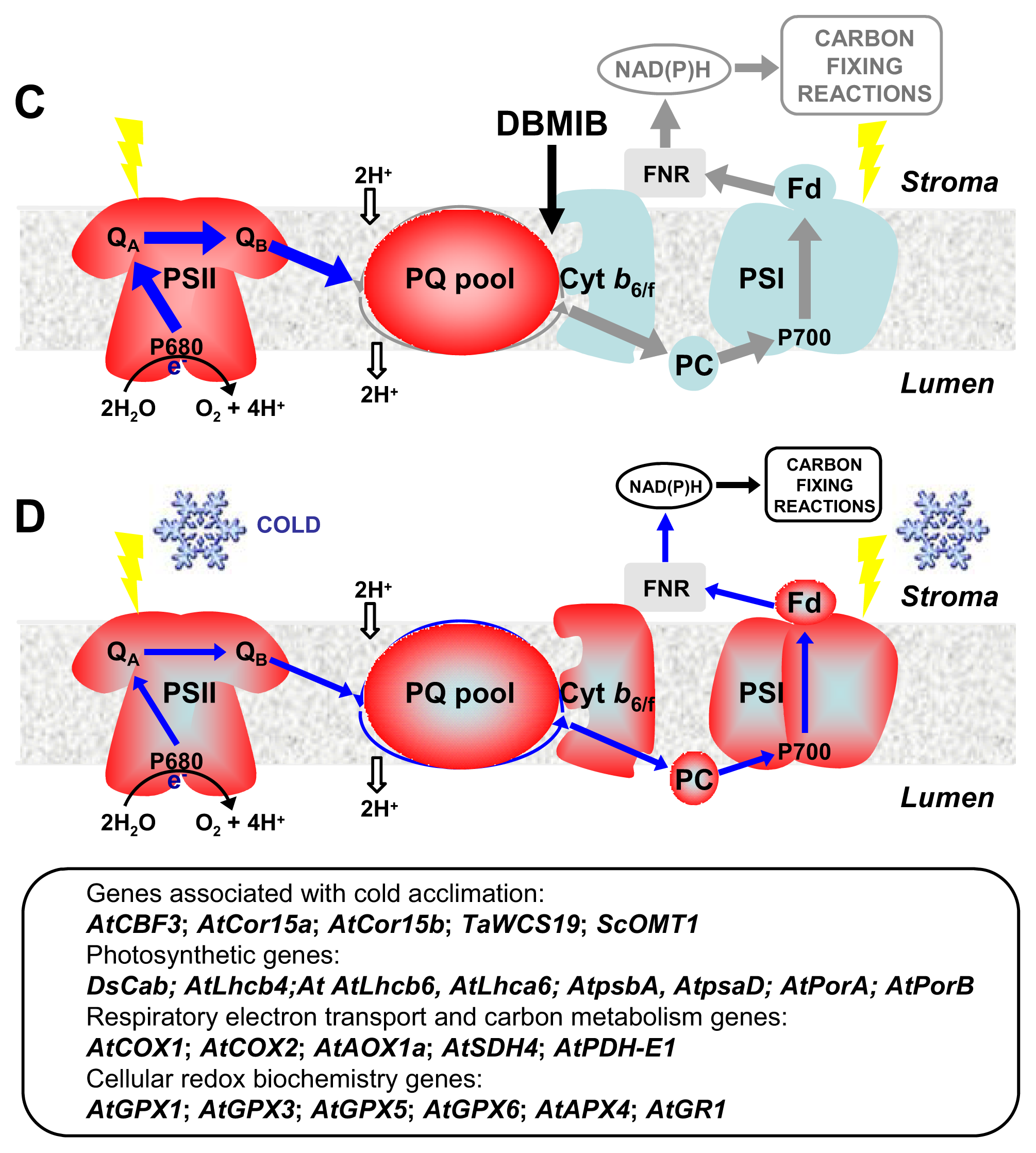
4. CBFs and Photosynthetic Performance
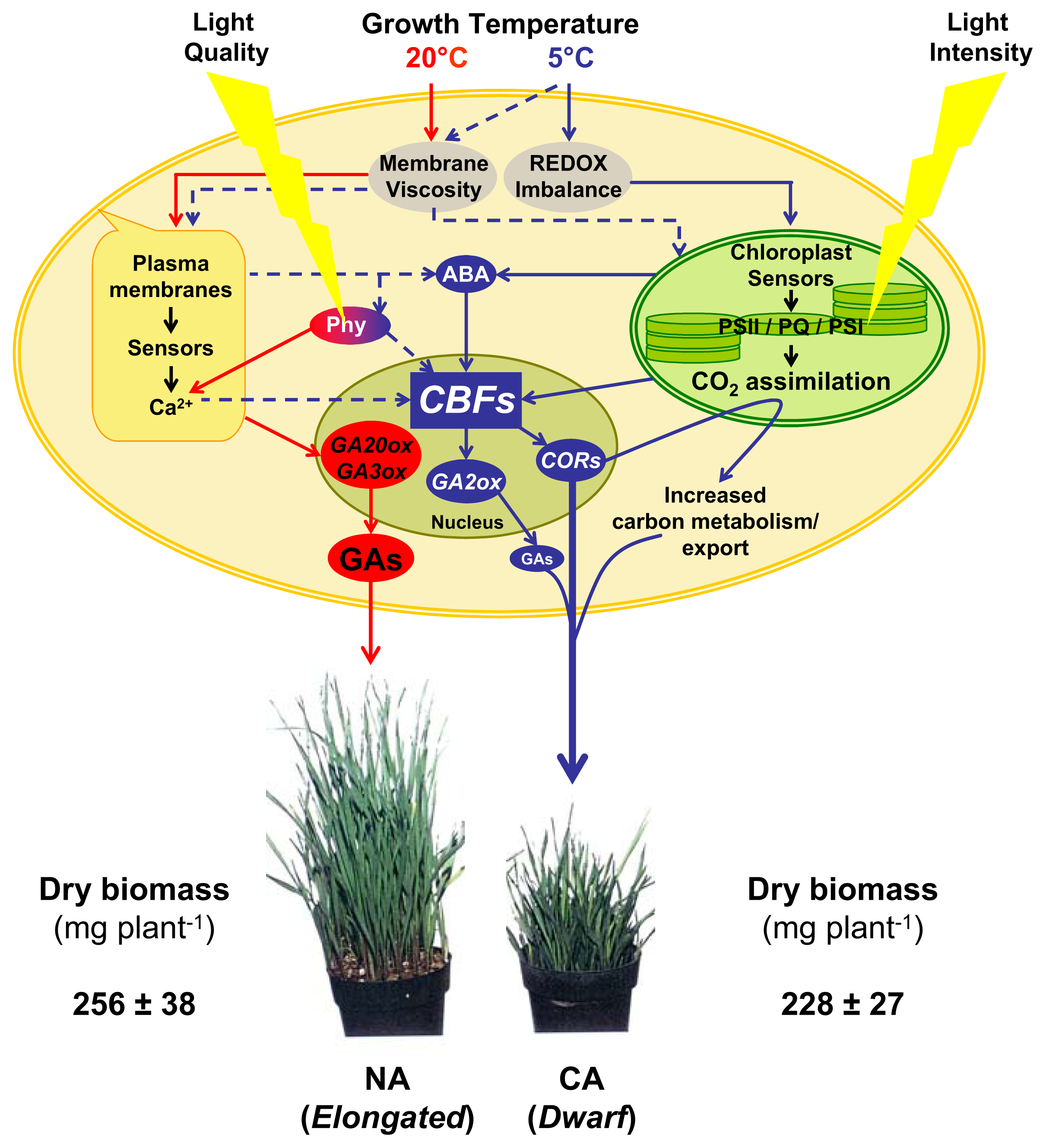
5. CBFs as Master Regulators
6. Conclusions
Acknowledgments
Conflict of Interest
References
- Thomashow, M.F. Plant cold acclimation: Freezing tolerance genes and regulatory mechanisms. Annu. Rev. Plant Physiol. Plant Mol. Biol 1999, 50, 571–599. [Google Scholar]
- Hüner, N.P.A.; Öquist, G.; Sarhan, F. Energy balance and acclimation to light and cold. Trends Plant Sci 1998, 3, 224–230. [Google Scholar]
- Wilson, K.E.; Ivanov, A.G.; Öquist, G.; Grodzinski, B.; Sarhan, F.; Hüner, N.P.A. Energy balance, organellar redox status, and acclimation to environmental stress. Can. J. Bot 2006, 84, 1355, –1370.. [Google Scholar]
- Hüner, N.P.A.; Bode, R.; Dahal, K.; Hollis, L.; Rosso, D.; Krol, M.; Ivanov, A.G. Chloroplast redox imbalance governs phenotypic plasticity: The “grand design of photosynthesis” revisited. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef]
- Hüner, N.P.A.; Bode, R.; Dahal, K.; Busch, F.A.; Possmayer, M.; Szyszka, B.; Rosso, D.; Ensminger, I.; Krol, M.; Ivanov, A.G.; et al. Shedding some light on cold acclimation, cold adaptation, and phenotypic plasticity. Can. J. Bot 2013, 91, 127–136. [Google Scholar]
- Gray, G.R.; Chauvin, L.-P.; Sarhan, F.; Hüner, N.P.A. Cold acclimation and freezing tolerance (A complex interaction of light and temperature). Plant Physiol 1997, 114, 467–474. [Google Scholar]
- Theocharis, A.; Clement, C.; Barka, E.A. Physiological and molecular changes in plants grown at low temperatures. Planta 2012, 235, 1091–1105. [Google Scholar]
- Chinnusamy, V.; Zhu, J; Zhu, J.K. Cold stress regulation of gene expression in plants. Trends Plant Sci 2007, 12, 444–451. [Google Scholar]
- Guy, C.; Kaplan, F.; Kopka, J.; Selbig, J.; Hincha, D.K. Metabolomics of temperature stress. Physiol. Plant 2008, 132, 220–235. [Google Scholar]
- Penfield, S. Temperature perception and signal transduction in plants. New Phytol 2008, 179, 615–628. [Google Scholar]
- Galiba, G.; Vagujfalvi, A.; Li, C.X.; Soltesz, A.; Dubcovsky, J. Regulatory genes involved in the determination of frost tolerance in temperate cereals. Plant Sci 2009, 176, 12–19. [Google Scholar]
- Hua, J. From freezing to scorching, transcriptional responses to temperature variations in plants. Curr. Opin. Plant Biol 2009, 12, 568–573. [Google Scholar]
- Ruelland, E.; Vaultier, M.N.; Zachowski, A.; Hurry, V. Cold signalling and cold acclimation in plants. Adv. Bot. Res. 2009, 49, 35–150. [Google Scholar]
- Thomashow, M.F. Molecular basis of plant cold acclimation: Insights gained from studying the CBF cold response pathway. Plant Physiol 2010, 154, 571–577. [Google Scholar]
- Medina, J.; Catala, R.; Salinas, J. The CBFs: Three Arabidopsis transcription factors to cold acclimate. Plant Sci 2011, 180, 3–11. [Google Scholar]
- Jaglo-Ottosen, K.R.; Gilmour, S.J.; Zarka, D.G.; Schabenberger, O.; Thomashow, M.F. Arabidopsis CBF1 overexpression induces COR genes and enhances freezing tolerance. Science 1998, 280, 104–106. [Google Scholar]
- Gilmour, S.J.; Fowler, S.G.; Thomashow, M.F. Arabidopsis transcriptional activators CBF1, CBF2, and CBF3 have matching functional activities. Plant Mol. Biol 2004, 54, 767–781. [Google Scholar]
- Lee, C.M.; Thomashow, M.F. Photoperiodic regulation of the C-repeat binding factor (CBF) cold acclimation pathway and freezing tolerance in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2012, 109, 15054–15059. [Google Scholar]
- Stockinger, E.J.; Gilmour, S.J.; Thomashow, M.F. Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat/DRE, a cis-acting DNA regulatory element that stimulates transcription in response to low temperature and water deficit. Proc Natl. Acad. Sci. USA 1997, 94, 1035–1040. [Google Scholar]
- Gilmour, S.J.; Zarka, D.G.; Stockinger, E.J.; Salazar, M.P.; Houghton, J.M.; Thomashow, M.F. Low temperature regulation of the Arabidopsis CBF family of AP2 transcriptional activators as an early step in cold-induced COR gene expression. Plant J 1998, 16, 433–442. [Google Scholar]
- Liu, Q.; Kasuga, M.; Sakuma, Y.; Abe, H.; Miura, S.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in drought- and low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 1998, 10, 1391–1406. [Google Scholar]
- Medina, J.; Bargues, M.; Terol, J.; Perez-Alonso, M.; Salinas, J. The Arabidopsis CBF gene family is composed of three genes encoding AP2 domain-containing proteins whose expression is regulated by low temperature but not by abscisic acid or dehydration. Plant Physiol 1999, 119, 463–470. [Google Scholar]
- Novillo, F.; Medina, J.; Salinas, J. Arabidopsis CBF1 and CBF3 have a different function than CBF2 in cold acclimation and define different gene classes in the CBF regulon. Proc. Natl. Acad. Sci. USA 2007, 104, 21002–21007. [Google Scholar]
- Novillo, F.; Alonso, J.M.; Ecker, J.R.; Salinas, J. CBF2/DREB1C is a negative regulator of CBF1/DREB1B and CBF3/DREB1A expression and plays a central role in stress tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2004, 101, 3985–3990. [Google Scholar]
- Novillo, F.; Medina, J.; Rodriguez-Franco, M.; Neuhaus, G.; Salinas, J. Genetic analysis reveals a complex regulatory network modulating CBF gene expression and Arabidopsis response to abiotic stress. J. Exp. Bot 2012, 63, 293–304. [Google Scholar]
- Chinnusamy, V.; Ohta, M.; Kanrar, S.; Lee, B.H.; Hong, X.; Agarwal, M.; Zhu, J.K. ICE1: A regulator of cold-induced transcriptome and freezing tolerance in Arabidopsis. Genes Dev 2003, 17, 1043–1054. [Google Scholar]
- Lee, H.; Xiong, L.; Gong, Z.; Ishitani, M.; Stevenson, B.; Zhu, J.K. The Arabidopsis HOS1 gene negatively regulates cold signal transduction and encodes a RING finger protein that displays cold-regulated nucleo–cytoplasmic partitioning. Genes Dev 2001, 15, 912–924. [Google Scholar]
- Dong, C.H.; Agarwal, M.; Zhang, Y.; Xie, Q.; Zhu, J.K. The negative regulator of plant cold responses, HOS1, is a RING E3 ligase that mediates the ubiquitination and degradation of ICE1. Proc. Natl. Acad. Sci. USA 2006, 103, 8281–8286. [Google Scholar]
- Miura, K.; Jin, J.B.; Lee, J.; Yoo, C.Y.; Stirm, V.; Miura, T.; Ashworth, E.N.; Bressan, R.A.; Yun, D.J.; Hasegawa, P.M. SIZ1-mediated sumoylation of ICE1 controls CBF3/DREB1A expression and freezing tolerance in Arabidopsis. Plant Cell 2007, 19, 1403–1414. [Google Scholar]
- Hsieh, T.H.; Lee, J.T.; Yang, P.T.; Chiu, L.H.; Charng, Y.Y.; Wang, Y.C.; Chan, M.T. Heterology expression of the Arabidopsis C-repeat/dehydration response element binding factor 1 gene confers elevated tolerance to chilling and oxidative stresses in transgenic tomato. Plant Physiol 2002, 129, 1086–1094. [Google Scholar]
- Savitch, L.V.; Allard, G.; Seki, M.; Robert, L.S.; Tinker, N.A.; Hüner, N.P.A.; Shinozaki, K.; Singh, J. The effect of overexpression of two Brassica CBF/DREB1-like transcription factors on photosynthetic capacity and freezing tolerance in Brassica napus. Plant Cell Physiol 2005, 46, 1525–1539. [Google Scholar]
- Benedict, C.; Skinner, J.S.; Meng, R.; Chang, Y.; Bhalerao, R.; Hüner, N.P.A.; Finn, C.E.; Chen, T.H.H.; Hurry, V. The CBF1-dependent low temperature signalling pathway, regulon and increase in freeze tolerance are conserved in Populus spp. Plant Cell Environ 2006, 29, 1259–1272. [Google Scholar]
- Achard, P.; Gong, F.; Cheminant, S.; Alioua, M.; Hedden, P.; Genschik, P. The cold-inducible CBF1 factor-dependent signaling pathway modulates the accumulation of the growth-repressing DELLA proteins via its effect on gibberellin metabolism. Plant Cell 2008, 20, 2117–2129. [Google Scholar]
- Huang, J.-G.; Yang, M.; Liu, P.; Yang, G.-D.; Wu, C.-A.; Zheng, C.-C. GhDREB1 enhances abiotic stress tolerance, delays GA-mediated development and represses cytokinin signalling in transgenic Arabidopsis. Plant Cell Environ 2009, 32, 1132–1145. [Google Scholar]
- Franklin, K.A. Light and temperature signal crosstalk in plant development. Curr. Opin. Plant Biol 2009, 12, 63–68. [Google Scholar]
- Clack, T.; Mathews, S.; Sharrock, R.A. The phytochrome apoprotein family in Arabidopsis is encoded by five genes: The sequences and expression of PHYD and PHYE. Plant Mol. Biol 1994, 25, 413–427. [Google Scholar]
- Devlin, P.F.; Halliday, K.J.; Harberd, N.P.; Whitelam, G.C. The rosette habit of Arabidopsis thaliana is dependent upon phytochrome action: Novel phytochromes control internode elongation and flowering time. Plant J 1996, 10, 1127–1134. [Google Scholar]
- Devlin, P.F.; Patel, S.R.; Whitelam, G.C. Phytochrome E influences internode elongation and flowering time in Arabidopsis. Plant Cell 1998, 10, 1479–1487. [Google Scholar]
- Devlin, P.F.; Robson, P.R.H.; Patel, S.R.; Goosey, L.; Sharrock, R.A.; Whitelam, G.C. Phytochrome D acts in the shade avoidance syndrome in Arabidopsis by controlling elongation growth and flowering time. Plant Physiol 1999, 119, 909–915. [Google Scholar]
- Franklin, K.A.; Davis, S.J.; Stoddart, W.M.; Vierstra, R.D.; Whitelam, G.C. Mutant analyses define multiple roles for phytochrome C in Arabidopsis photomorphogenesis. Plant Cell 2003, 15, 1981–1989. [Google Scholar]
- Shinomura, T.; Nagatani, A.; Chory, J.; Furuya, M. The induction of seed germination in Arabidopsis thaliana is regulated principally by phytochrome B and secondarily by phytochrome A. Plant Physiol 1994, 104, 363–371. [Google Scholar]
- Monte, E.; Alonso, J.M.; Ecker, J.R.; Zhang, Y.; Li, X.; Young, J.; Austin-Phillips, S.; Quail, P.H. Isolation and characterization of PHYC mutants in Arabidopsis reveals complex crosstalk between phytochrome signalling pathways. Plant Cell 2003, 15, 1962–1980. [Google Scholar]
- Aukerman, M.J.; Hirschfeld, M.; Wester, L.; Weaver, M.; Clack, T.; Amasino, R.M.; Sharrock, R.A. A deletion in the PHYD gene of the Arabidopsis Wassilewskija ecotype defines a role for phytochrome D in red/far-red light sensing. Plant Cell 1997, 9, 1317–1326. [Google Scholar]
- Yanovsky, M.J.; Casal, J.J.; Luppi, J.P. The VLF loci, polymorphic between ecotypes Landsberg erecta and Columbia, dissect two branches of phytochrome A signal transduction that correspond to very-low-fluence and high-irradiance responses. Plant J 1997, 12, 659–667. [Google Scholar]
- Kurepin, L.V.; Walton, L.J.; Hayward, A.; Emery, R.J.N.; Pharis, R.P.; Reid, D.M. Interactions between plant hormones and light quality signaling in regulating the shoot growth of Arabidopsis thaliana seedlings. Can. J. Bot 2012, 90, 237–246. [Google Scholar]
- Davies, P.J. The Plant Hormones: Their Nature, Occurrence, and Functions. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 1–15. [Google Scholar]
- Davies, P.J. Regulatory Factors in Hormone Action: Level, Location and Signal Transduction. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 16–35. [Google Scholar]
- Sponsel, V.M.; Hedden, P. Gibberellin Biosynthesis and Inactivation. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 63–94. [Google Scholar]
- Taiz, L.; Zeiger, E. Gibberellins. In Plant Physiology, 4th ed.; Sinauer Associates Inc: Sunderland, MA, USA, 2006; Volume Chapter 20, p. 511. [Google Scholar]
- Dahal, K. Plasticity in Photosynthetic Performance and Energy Utilization Efficiency in Triticum aestivum L., Secale cereale L. and Brassica napus in Response to Low Temperature and High CO2. Ph.D. Thesis, Western University, London, Canada, June 2012. [Google Scholar]
- Dahal, K.; Gadapati, W.; Savitch, L.V.; Singh, J.; Hüner, N.P.A. Cold acclimation and BnCBF17-over-expression enhance photosynthetic performance and energy conversion efficiency during long-term growth of Brassica napus under elevated CO2 conditions. Planta 2012, 236, 1639–1652. [Google Scholar]
- Krol, M.; Griffith, M.; Hüner, N.P.A. An appropriate physiological control for environmental temperature studies: Comparative growth kinetics for winter rye. Can. J. Bot 1984, 62, 1062–1068. [Google Scholar]
- Dahal, K.; Kane, K.; Gadapati, W.; Webb, E.; Savitch, L.V.; Singh, J.; Sharma, P.; Sarhan, F.; Longstaffe, F.J.; Grodzinski, B.; et al. The effects of phenotypic plasticity on photosynthetic performance in winter rye, winter wheat and Brassica napus. Physiol. Plant 2012, 144, 169–188. [Google Scholar]
- Sun, T.P. Gibberellin Signal Transduction in Stem Elongation & Leaf Growth. In Plant Hormones: Biosynthesis, Signal Transduction and Action! revised 3rd ed.; Davies, P.J., Ed.; Springer Dordrecht Heidelberg: London, New York, 2010; pp. 308–328. [Google Scholar]
- Kurepin, L.V.; Ozga, J.A.; Zaman, M.; Pharis, R.P. The physiology of plant hormones in cereal, oilseed and pulse crops. Prairie Soils Crops 2013, 6, 7–23. [Google Scholar]
- Reid, D.M.; Pharis, R.P.; Roberts, D.W.A. Effects of four temperature regimens on the gibberellin content of winter wheat cv. Kharkov. Physiol. Plant 1973, 30, 53–57. [Google Scholar]
- Rood, S.B.; Major, D.J.; Pharis, R.P. Low temperature eliminates heterosis for growth and gibberellin content in maize. Crop Sci 1985, 25, 1063–1068. [Google Scholar]
- Kurepin, L.V.; Walton, L.J.; Pharis, R.P.; Emery, R.J.N.; Reid, D.M. Interactions of temperature and light quality on phytohormone-mediated elongation of Helianthus annuus hypocotyls. Plant Growth Regul 2011, 64, 147–154. [Google Scholar]
- Sarkar, S.; Perras, M.R.; Falk, D.E.; Zhang, R.; Pharis, R.P.; Fletcher, R.A. Relationship between gibberellins, height, and stress tolerance in barley (Hordeum vulgare L.) seedlings. Plant Growth Regul 2004, 42, 125–135. [Google Scholar]
- Hiller, L.K.; Kelly, W.C.; Powell, L.E. Temperature interactions with growth regulators and endogenous gibberellin-like activity during seedstalk elongation in carrots. Plant Physiol 1979, 63, 1055–1061. [Google Scholar]
- Pinthus, M.J.; Gale, M.D.; Appleford, N.E.J.; Lenton, J.R. Effect of temperature on gibberellin (GA) responsiveness and on endogenous GA1 content of tall and dwarf wheat genotypes. Plant Physiol 1989, 90, 854–859. [Google Scholar]
- Jensen, E.; Eilertsen, S.; Ernsten, A.; Juntilla, O.; Moe, R. Thermoperiodic control of stem elongation and endogenous gibberellins in Campanula isophylla. J. Plant Growth Regul 1996, 15, 167–171. [Google Scholar]
- Nishijima, T.; Nonaka, M.; Koshioka, M.; Ikeda, H.; Douzono, M.; Yamazaki, H.; Mander, L.N. Role of gibberellins in the thermoperiodic regulation of stem elongation in Dendranthema grandiflorum tzvelev. Biosci.Biotechnol. Biochem 1997, 61, 1362–1366. [Google Scholar]
- Fowler, S.; Thomashow, M.F. Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 2002, 14, 1675–1690. [Google Scholar]
- Kreps, J.A.; Wu, Y.; Chang, H.S.; Zhu, T.; Wang, X.; Harper, J.F. Transcriptome changes for Arabidopsis in response to salt, osmotic, and cold stress. Plant Physiol 2002, 130, 2129–2141. [Google Scholar]
- Seki, M.; Narusaka, M.; Ishida, J.; Nanjo, T.; Fujita, M.; Oono, Y.; Kamiya, A.; Nakajima, M.; Enju, A.; Sakurai, T.; Satou, M.; Akiyama, K.; Taji, T.; Yamaguchi-Shinozaki, K.; Carninci, P.; Kawai, J.; Hayashizaki, Y.; Shinozaki, K. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J 2002, 31, 279–292. [Google Scholar]
- Provart, N.J.; Gil, P.; Chen, W.; Han, B.; Chang, H.-S.; Wang, X.; Zhu, T. Gene expression phenotypes of Arabidopsis associated with sensitivity to low temperatures. Plant Physiol 2003, 132, 893–906. [Google Scholar]
- Yamauchi, Y.; Ogawa, M.; Kuwahara, A.; Hanada, A.; Kamiya, Y.; Yamaguchi, S. Activation of gibberellin biosynthesis and response pathways by low temperature during imbibition of Arabidopsis thaliana seeds. Plant Cell 2004, 16, 367–378. [Google Scholar]
- Kendall, S.A.; Hellwege, A.; Marriot, P.; Whalley, C.; Graham, I.A.; Penfield, S. Induction of dormancy in Arabidopsis summer annuals requires parallel regulation of DOG1 and hormone metabolism by low temperature and CBF transcription factors. Plant Cell 2011, 23, 2568–2580. [Google Scholar]
- Nadeau, C.D.; Ozga, J.A.; Kurepin, L.V.; Jin, A.; Pharis, R.P.; Reinecke, D.M. Tissue-specific regulation of gibberellin biosynthesis in developing pea seeds. Plant Physiol 2011, 156, 897–912. [Google Scholar]
- Magome, H.; Yamaguchi, S.; Hanada, A.; Kamiya, Y.; Oda, K. drarf and delayed-flowering 1, a novel Arabidopsis mutant deficient in gibberellin biosynthesis because of overexpression of a putative AP2 transcription factor. Plant J 2004, 377, 720–729. [Google Scholar]
- Kasuga, M.; Liu, Q.; Miura, S.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat. Biotechnol 1999, 17, 287–291. [Google Scholar]
- Gilmour, S.J.; Sebolt, A.M.; Salazar, M.P.; Everard, J.D.; Thomashow, M.F. Overexpression of the Arabidopsis CBF3 transcriptional activator mimics multiple biochemical changes associated with cold acclimation. Plant Physiol 2000, 124, 1854–1865. [Google Scholar]
- Magome, H.; Yamaguchi, S.; Hanada, A.; Kamiya, Y.; Oda, K. The DDF1 transcriptional activator upregulates expression of a gibberellin-deactivating gene, GA2ox7, under high-salinity stress in Arabidopsis. Plant J 2008, 56, 613–626. [Google Scholar]
- Siddiqua, M.; Nassuth, A. Vitis CBF1 and Vitis CBF4 differ in their effect on Arabidopsis abiotic stress tolerance, development and gene expression. Plant Cell Environ 2011, 34, 1345–1359. [Google Scholar]
- Shan, D.-P.; Huang, J.-G.; Yang, Y.-T.; Guo, Y.-H.; Wu, C.-A.; Yang, G.-D.; Gao, Z.; Zheng, C.C. Cotton GhDREB1 increases plant tolerance to low temperature and is negatively regulated by gibberellic acid. New Phytol 2007, 176, 70–81. [Google Scholar]
- Zhang, F.; Wan, X.Q.; Zhang, H.Q.; Liu, G.L.; Jiang, M.Y.; Pan, Y.Z.; Chen, Q.B. The effect of cold stress on endogenous hormones and CBF1 homolog in four contrasting bamboo species. J. For. Res 2012, 17, 72–78. [Google Scholar]
- Peng, J.; Carol, P.; Richards, D.E.; King, K.E.; Cowling, R.J.; Murphy, G.P.; Harberd, N.P. The Arabidopsis GAI gene defines a signaling pathway that negatively regulates gibberellin responses. Genes Dev. 1997, 11, 3194–3205. [Google Scholar]
- Irving, R.M.; Lanphear, F.O. Regulation of cold hardiness in Acer negundo. Plant Physiol 1968, 43, 9–13. [Google Scholar]
- Rikin, A.; Atsmon, D.; Gitler, C. Chilling injury in cotton (Gossypium hirsutum L.): Prevention by abscisic acid. Plant Cell Physiol 1979, 20, 1537–2546. [Google Scholar]
- Rikin, A.; Waldman, M.; Richmond, A.E.; Dovrat, A. Hormonal regulation of morphogenesis and cold resistance. I. Modifications by abscisic acid and by gibberellic acid in alfalfa (Medicago sativa L.) seedlings. J. Exp.Bot 1975, 16, 175–183. [Google Scholar]
- Chen, T.H.H.; Gusta, L.V. Abscisic acid-induced freezing resistance in cultured plant cells. Plant Physiol. 1983, 73, 71–75. [Google Scholar]
- Lalk, I.; Dorfling, K. Hardening, abscisic acid, praline and freezing resistance in two winter wheat varieties. Physiol. Plant 1985, 63, 287–292. [Google Scholar]
- Xing, W.; Rajashekar, C.B. Glycine betaine involvement in freezing tolerance and water stress in Arabidopsis thaliana. Environ. Exp. Bot 2001, 46, 21–28. [Google Scholar]
- Nayyar, H.; Bains, T.; Kumar, S. Low temperature induced floral abortion in chickpea: Relationship to abscisic acid and cryoprotectants in reproductive organs. Environ. Exp. Bot 2004, 52, 219–231. [Google Scholar]
- Dale, J.; Campbell, W.F. Response of tomato plants to stressful temperatures: Increase in abscisic acid concentrations. Plant Physiol 1981, 67, 26–29. [Google Scholar]
- Lang, V.; Mantyla, E.; Welin, B.; Sundberg, B.; Palva, E.T. Alterations in water status, endogenous abscisic acid content, and expression of rab18 gene during the development of freezing tolerance in Arabidopsis thaliana. Plant Physiol 1994, 104, 1341–1349. [Google Scholar]
- Mantyla, E.; Lang, V.; Palva, E.T. Role of abscisic acid in drought-induced freezing tolerance, cold acclimation, and accumulation of LT178 and RAB18 proteins in Arabidopsis thaliana. Plant Physiol 1995, 107, 141–148. [Google Scholar]
- Gilmour, S.J.; Thomashow, M.F. Cold acclimation and cold-regulated gene expression in ABA mutants of Arabidopsis thaliana. Plant Mol. Biol 1991, 17, 1233–1240. [Google Scholar]
- Xiong, L.; Lee, B.H.; Ishitani, M.; Lee, H.; Zhu, J.K. Regulation of osmotic stress-responsive gene expression by the LOS6/ABA1 locus in Arabidopsis. J. Biol. Chem 2002, 56, 277–289. [Google Scholar]
- Fuchigami, L.H.; Evert, D.R.; Weiser, C.J. A translocatable cold hardiness promoter. Plant Physiol 1971, 47, 164–167. [Google Scholar]
- Fayyaz, M.M.; McCown, B.H.; Beck, G.H. Effect of temperature, photoperiod and several growth substances on the cold hardiness of Chrysanthemum morifolium rhizome. Physiol. Plant 1978, 44, 73–76. [Google Scholar]
- Gusta, L.V.; Fowler, D.B.; Tyler, N.J. The effect of abscisic acid and cytokinins on the cold hardiness of winter wheat. Can. J. Bot 1982, 60, 301–305. [Google Scholar]
- Holubowicz, T.; Cummins, J.N.; Forsline, P.L. Responses of Malus clones to programmed low-temperature stresses in late winter. J. Am. Soc. Hortic. Sci 1982, 107, 492–496. [Google Scholar]
- Holubowicz, T.; Pieniazek, J.; Khamis, M.A. Modification of Frost Resistance of Fruit Plants by Applied Growth Regulators. In Plant Hardiness and Freezing Stress; Academic Press: New York, NY, USA, 1982; pp. 541–559. [Google Scholar]
- Lang, V.; Palva, E.T. The expression of rab-related gene, rab18, is induced by abscisic acid during the cold acclimation process of Arabidopsis thaliana (L.) Heynh. Plant Mol. Biol 1992, 20, 951–962. [Google Scholar]
- Kurkela, S.; Franck, M. Structure and expression of kin2, one of two cold- and ABA-induced genes of Arabidopsis thaliana. Plant Mol. Biol 1992, 19, 689–692. [Google Scholar]
- Knight, H.; Zarka, D.G.; Okamoto, H.; Thomashow, M.F.; Knight, M.R. Abscisic acid induces CBF gene transcription and subsequent induction of cold-regulated genes via the CRT promoter element. Plant Physiol 2004, 135, 1710–1717. [Google Scholar]
- Xiao, H.; Siddiqua, M.; Braybrook, S.; Nassuth, A. Three grape CBF/DREB1 genes respond to low temperature, drought and abscisic acid. Plant Cell Environ 2006, 29, 1410–1421. [Google Scholar]
- Skinner, J.S.; von Zitzewitz, J.; Szucs, P.; Marquez-Cedillo, L.; Filichkin, T.; Amundsen, K.; Stockinger, E.J.; Thomasow, M.F.; Chen, T.N.N.; Hayes, P.M. Structural, functional, and phylogenetic characterization of a large CBF gene family in barley. Plant Mol. Biol 2005, 59, 533–551. [Google Scholar]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Molecular responses to dehydration and low temperature: Differences and cross-talk between two stress signaling pathways. Curr. Opin. Plant Biol 2000, 3, 217–223. [Google Scholar]
- Gusta, L.V.; Trischuk, R.; Weiser, C.J. Plant cold acclimation: The role of abscisic acid. J. Plant Growth Regul 2005, 24, 308–318. [Google Scholar]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu.Rev. Plant Biol 2006, 57, 781–803. [Google Scholar]
- Trewavas, A.J.; Jones, H.G. An Assessment of the Role of Aba in Plant Development. In Abscisic Acid: Physiology and Biochemistry; Davies, W.J., Jones, H.G., Eds.; Bios Scientific Publishers: Oxford, UK, 1991; pp. 169–188. [Google Scholar]
- Munns, R.; Cramer, G.R. Is coordination of leaf and root growth mediated by abscisic acid? Plant Soil 1996, 185, 33–49. [Google Scholar]
- Schwartz, S.H.; Zeevaart, J.A.D. Abscisic Acid Biosynthesis and Metabolism. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 137–155. [Google Scholar]
- Zentella, R.; Zhang, Z.L.; Park, M.; Thomas, S.G.; Endo, A.; Murase, K.; Fleet, C.M.; Jikumaru, Y.; Nambara, E.; Kamiya, Y.; et al. Global analysis of della direct targets in early gibberellin signaling in Arabidopsis. Plant Cell 2007, 19, 3037–3057. [Google Scholar]
- Toh, S.; Imamura, A.; Watanabe, A.; Nakabayashi, K.; Okamoto, M.; Jikumaru, Y.; Hanada, A.; Aso, Y.; Ishiyama, K.; Tamura, N.; et al. High temperature-induced abscisic acid biosynthesis and its role in the inhibition of gibberellin action in Arabidopsis seeds. Plant Physiol 2008, 146, 1368–1385. [Google Scholar]
- Roef, L.; van Onckelen, H. Cytokinin Regulation of the Cell Division Cycle. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 241–261. [Google Scholar]
- Ha, S.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Cytokinins: Metabolism and function in plant adaptation to environmental stresses. Trends Plant Sci 2012, 17, 172–179. [Google Scholar]
- Veselova, S.V.; Farhutdinov, R.G.; Veselov, S.Y.; Kudoyarova, G.R.; Veselov, D.S.; Hartung, W. The effect of root cooling on hormone content, leaf conductance and root hydraulic conductivity of durum wheat seedlings (Triticum durum L.). J. Plant Physiol 2005, 162, 21–26. [Google Scholar]
- Nishiyama, R.; Watanabe, Y.; Fujita, Y.; Le, D.T.; Kojima, M.; Werner, T.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Kakimoto, T.; et al. Analysis of cytokinin mutants and regulation of cytokinin metabolic genes reveals important regulatory roles of cytokinins in drought, salt and abscisic acid responses, and abscisic acid biosynthesis. Plant Cell 2011, 23, 2169–2183. [Google Scholar]
- Sakakibara, H. Cytokinin Biosynthesis Metabolism. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 95–114. [Google Scholar]
- Hwang, I.; Sheen, J.; Muller, B. Cytokinin signaling networks. Annu. Rev. Plant Biol 2012, 63, 353–380. [Google Scholar]
- Chaudhury, A.M.; Letham, S.; Craig, S.; Dennis, E.S. amp1—A mutant with high cytokinin levels and altered embryonic pattern, faster vegetative growth, constitutive photomorphogenesis and precocious flowering. Plant J 1993, 4, 907–916. [Google Scholar]
- Xia, J.; Zhao, H.; Liu, W.; Li, L.; He, Y. Role of cytokinin and salicylic acid in plant growth at low temperatures. Plant Growth Regul 2009, 57, 211–221. [Google Scholar]
- Jeon, J.; Kim, N.Y.; Kim, S.; Kang, N.Y.; Novak, O.; Ku, S.J.; Cho, C.; Lee, D.J.; Lee, E.J.; Strnad, M.; et al. A subset of cytokinin two-component signaling system plays a role in cold temperature stress response in Arabidopsis. J. Biol. Chem 2010, 285, 23371–23386. [Google Scholar]
- Shi, Y.; Tian, S.; Hou, L.; Huang, X.; Zhang, X.; Guo, H.; Yang, C. Ethylene signaling negatively regulates freezing tolerance by repressing expression of CBF and type-A ARR genes in Arabidopsis. Plant Cell 2012, 24, 2578–2595. [Google Scholar]
- Abeles, F.B.; Morgan, P.W.; Saltveit, M.E. Ethylene in Plant Biology, 2nd ed.; Academic Press: New York, NY, USA, 1992. [Google Scholar]
- Kurepin, L.V.; Walton, L.J.; Reid, D.M. Interaction of red to far red light ratio and ethylene in regulating stem elongation of Helianthus annuus. Plant Growth Regul 2007, 51, 53–61. [Google Scholar]
- Kurepin, L.V.; Walton, L.J.; Yeung, E.C.; Chinnappa, C.C.; Reid, D.M. The interaction of light irradiance with ethylene in regulating growth of Helianthus annuus shoot tissues. Plant Growth Regul 2010, 62, 43–50. [Google Scholar]
- Wang, C.Y.; Adams, D.O. Chilling-induced ethylene production in cucumbers (Cucumis sativus L.). Plant Physiol 1982, 69, 424–427. [Google Scholar]
- Ciardi, J.A.; Deikman, J.; Orzolek, M.D. Increased ethylene synthesis enhances chilling tolerance in tomato. Physiol. Plant 1997, 101, 333–340. [Google Scholar]
- Yu, X.M.; Griffith, M.; Wiseman, S.B. Ethylene induces antifreeze activity in winter rye leaves. Plant Physiol 2001, 126, 1232–1240. [Google Scholar]
- Zhang, Z.; Huang, R. Enhanced tolerance to freezing in tobacco and tomato overexpressing transcription factor TERF2/LeERF2 is modulated by ethylene biosynthesis. Plant Mol. Biol 2010, 73, 241–249. [Google Scholar]
- Field, R.J. The role of 1-aminocyclopropane-1-carboxylic acid in the control of low temperature induced ethylene production in leaf tissue of Phaseolus vulgaris L. Ann. Bot 1984, 54, 61–67. [Google Scholar]
- Machacckova, I.; Hanisova, A.; Krekule, J. Levels of ethylene, ACC, MACC, ABA and proline as indicators of cold hardening and frost resistance in winter wheat. Physiol. Plant 1989, 76, 603–607. [Google Scholar]
- Collins, G.G.; Nie, X.; Saltveit, M.E. Heat shock increases chilling tolerance of mung bean hypocotyls tissues. Physiol. Plant 1995, 89, 117–124. [Google Scholar]
- Chang, C.; Kwok, S.F.; Bleecker, A.B.; Meyerowitz, E.M. Arabidopsis ethylene-response gene ETR1: Similarity of product to two component regulators. Science 1993, 262, 539–544. [Google Scholar]
- Bleecker, A.B.; Esch, J.J.; Hall, A.E.; Rodriguez, F.I.; Binder, B.M. The ethylene-receptor family from Arabidopsis—Structure and function. Philos. Trans. R. Soc. B 1998, 353, 1405–1412. [Google Scholar]
- Solano, R.; Stepanova, A.; Chao, Q.; Ecker, J.R. Nuclear events in ethylene signaling: A transcriptional cascade mediated by ETHYLENE-INSENSITIVE3 and ETHYLENE-RESPONSE-FACTOR1. Genes Dev 1998, 12, 3703–3714. [Google Scholar]
- Sharabi-Schwager, M.; Samach, A.; Porat, R. Overexpression of the CBF2 transcriptional activator in Arabidopsis suppresses the responsiveness of leaf tissue to the stress hormone ethylene. Plant Biol 2010, 12, 630–638. [Google Scholar]
- Nagata, N.; Min, Y.K.; Nakano, T.; Asami, T.; Yoshida, S. Treatment of dark-grown Arabidopsis thaliana with a brassinosteroid biosynthesis inhibitor, brassinazole, induces some characteristics of light-grown plants. Planta 2000, 211, 781–790. [Google Scholar]
- Zullo, M.A.T.; Adam, G. Brassinosteroid phytohormones—Structure, bioactivity and applications. Braz. J. Plant Physiol 2002, 14, 143–181. [Google Scholar]
- Choe, S. Brassinosteroid Biosynthesis Metabolism. In Plant Hormones: Biosynthesis, Signal Transduction and Action! 3rd ed.; Davies, P.J., Ed.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2010; pp. 156–178. [Google Scholar]
- Kurepin, L.V.; Joo, S.H.; Kim, S.K.; Pharis, R.P.; Back, T.G. Interaction of brassinosteroids with light quality and plant hormones in regulating shoot growth of young sunflower and Arabidopsis seedlings. J. Plant Growth Regul 2012, 31, 156–164. [Google Scholar]
- Wilen, R.W.; Sacco, M.; Gusta, L.V.; Krishna, P. Effects of 24-epibrassinolide on freezing and thermotolerance of bromegrass (Bromus inermis) cell cultures. Physiol. Plant 1995, 95, 195–202. [Google Scholar]
- Khripach, V.A.; Zhabinskii, V.N.; de Groot, A.E. Brassinosteroids: A New Class of Plant Hormones; Academic Press: San Diego, CA, USA, 1999; pp. 263–277. [Google Scholar]
- Kurepin, L.V.; Qaderi, M.M.; Back, T.G.; Reid, D.M.; Pharis, R.P. A rapid effect of applied brassinolide on abscisic acid concentrations in Brassica napus leaf tissue subjected to short-term heat stress. Plant Growth Regul 2008, 55, 165–167. [Google Scholar]
- He, R.Y.; Wang, G.Y.; Wang, S.X. Effect of Brassinolide on Growth and Chilling Resistance of Maize Seedlings. In Brassinosteroids: Chemistry, Bioactivity and Applications; Cutler, H.G., Yokoda, T., Adam, G., Eds.; American Chemical Society: Washington, DC, USA, 1991; pp. 220–230. [Google Scholar]
- Kagale, S.; Divi, U.K.; Krochko, J.E.; Keller, W.A.; Krishna, P. Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta 2007, 225, 353–364. [Google Scholar]
- Kim, S.Y.; Kim, B.H.; Lim, C.J.; Lim, C.O.; Nam, K.H. Constitutive activation of stress-inducible genes in a brassinosteroid-insensitive 1 (bri1) mutant results in higher tolerance to cold. Physiol. Plant 2010, 138, 191–204. [Google Scholar]
- Wang, Z.Y.; Seto, H.; Fujioka, S.; Yoshida, S.; Chory, J. BRI1 is a critical component of a plasma-membrane receptor for plant steroids. Nature 2001, 410, 380–383. [Google Scholar]
- Li, J.; Wen, J.; Lease, K.A.; Doke, J.T.; Tax, F.E.; Walker, J.C. BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 2002, 110, 213–222. [Google Scholar]
- Divi, U.K.; Krishna, P. Overexpression of the brassinosteroid biosynthetic gene AtDWF4 in Arabidopsis seeds overcomes abscisic acid-induced inhibition of germination and increases cold tolerance in transgenic seedlings. J. Plant Growth Regul 2010, 29, 385–393. [Google Scholar]
- Kim, H.B.; Kwon, M.; Ryu, H.; Fujioka, S.; Takatsuto, S.; Yoshida, S.; An, C.S.; Lee, I.; Hwang, I.; Choe, S. The regulation of DWARF4 expression is likely a critical mechanism in maintaining the homeostasis of bioactive brassinosteroids in Arabidopsis. Plant Physiol 2006, 140, 548–557. [Google Scholar]
- Raskin, I. Role of salicylic acid in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol 1992, 43, 439–463. [Google Scholar]
- Raskin, I. Salicylic Acid. In Plant Hormones, Physiology, Biochemistry and Molecular Biology; Davies, P.J., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1995; pp. 188–205. [Google Scholar]
- Schettel, N.L.; Balke, N.E. Plant growth response to several allelopathic chemicals. Weed Sci. 1983, 31, 293–298. [Google Scholar]
- Hayat, Q.; Hayat, S.; Irfan, M.; Ahmad, A. Effect of exogenous salicylic acid under changing environment: A review. Environ. Exp. Bot 2010, 68, 14–25. [Google Scholar]
- Janda, T.; Szalai, G.; Antunovics, Z.S.; Horvath, E.; Paldi, E. Effect of benzoic acid and aspirin on chilling tolerance and photosynthesis in young maize plants. Maydica 2000, 45, 29–33. [Google Scholar]
- Janda, T.; Szalai, G.; Tari, I.; Paldi, E. Hydroponic treatment with salicylic acid decreases the effect of chilling injury in maize (Zea mays L.) plants. Planta 1999, 208, 175–180. [Google Scholar]
- Ding, C.K.; Wang, C.Y.; Gross, K.C.; Smith, D.L. Jasmonate and salicylate induce the expression of pathogenesis-related-protein genes and increase resistance to chilling injury in tomato fruit. Planta 2002, 214, 895–901. [Google Scholar]
- Kang, G.Z.; Wang, C.H.; Sun, G.C.; Wang, Z.X. Salicylic acid changes activities of H2O2-metabolizing enzymes and increases the chilling tolerance of banana seedlings. Environ. Exp. Bot 2003, 50, 9–15. [Google Scholar]
- Kang, H.M.; Saltveit, M.E. Chilling tolerance of maize, cucumber and rice seedling leaves and roots are differentially affected by salicylic acid. Physiol. Plant 2002, 115, 571–576. [Google Scholar]
- Kurepin, L.V.; Dahal, K.P.; Zaman, M.; Pharis, R.P. Interplay between Environmental Signals and Endogenous Salicylic Acid Concentration. In Salicylic Acid: Plant Growth and Development; Hayat, S., Ahmad, A., Alyemini, M.N., Eds.; Springer Science+Business Media B.V.: Dordrecht, The Netherlands, 2013; pp. 61–82. [Google Scholar]
- Farooq, M.; Aziz, T.; Basra, S.M.A.; Cheema, M.A.; Rehman, H. Chilling tolerance in hybrid maize induced by seed priming with salicylic acid. J. Agron. Crop Sci 2008, 194, 161–168. [Google Scholar]
- Lei, T.; Feng, H.; Sun, X.; Dai, Q.-L.; Zhang, F.; Liang, H.-G.; Lin, H.-H. The alternative pathway in cucumber seedlings under low temperature stress was enhanced by salicylic acid. Plant Growth Regul 2010, 60, 35–42. [Google Scholar]
- Mora-Herrera, M.E.; Lopez-Delgado, H.; Castillo-Morales, A.; Foyer, C.H. Salicylic acid and H2O2 function by independent pathways in the induction of freezing tolerance in potato. Physiol. Plant 2005, 125, 430–440. [Google Scholar]
- Wang, L.; Chen, S.; Kong, W.; Li, S.; Archbold, D.D. Salicylic acid pretreatment alleviates chilling injury and affects the antioxidant system and heat shock proteins of peaches during cold storage. Postharvest Biol. Technol 2006, 41, 244–251. [Google Scholar]
- Bowling, S.A.; Clarke, J.D.; Liu, Y.; Klessig, D.F.; Dong, X. The cpr5 mutant of Arabidopsis expresses both NPR1-dependent and NPR1-independent resistance. Plant Cell 1997, 9, 1573–1584. [Google Scholar]
- Falk, A.; Feys, B.J.; Frost, L.N.; Jones, J.D.; Daniels, M.J.; Parker, J.E. EDS1, an essential component of R gene-mediated disease resistance in Arabidopsis has homology to eukaryotic lipases. Proc. Natl. Acad. Sci. USA 1999, 96, 3292–3297. [Google Scholar]
- Scott, I.M.; Clarke, S.M.; Wood, J.E.; Mur, L.A.J. Salicylate accumulation inhibits growth at chilling temperature in Arabidopsis. Plant Physiol 2004, 135, 1040–1049. [Google Scholar]
- Miura, K.; Ohta, M. SIZ1, a small ubiquitin-related modifier ligase, controls cold signaling through regulation of salicylic acid accumulation. J. Plant Physiol 2010, 167, 555–560. [Google Scholar]
- Levitt, J. Responses of Plants to Environmental Stresses. In Chilling, Freezing, and High Temperature Stresses; Academic Press: New York, NY, USA, 1980; Volume 1, p. 497. [Google Scholar]
- Demmig-Adams, B; Adams, W.W., III. Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol 1992, 43, 599–626. [Google Scholar]
- Anderson, J.M.; Chow, W.S.; Park, Y.-I. The grand design of photosynthesis: Acclimation of the photosynthetic apparatus to environmental cues. Photosynth. Res 1995, 46, 129–139. [Google Scholar]
- Fey, V.; Wagner, R.; Brautigam, K.; Pfannschmidt, T. Photosynthetic redox control of nuclear gene expression. J. Exp. Bot 2005, 56, 1491–1498. [Google Scholar]
- Ensminger, I.; Busch, F.; Hüner, N.P.A. Photostasis and cold acclimation: Sensing low temperature through photosynthesis. Physiol. Plant 2006, 126, 28–44. [Google Scholar]
- Murchie, E.H.; Pinto, M.; Horton, P. Agriculture and the new challenges for photosynthesis research. New Phytol 2009, 181, 532–552. [Google Scholar]
- Kurepin, L.V.; Emery, R.J.N.; Pharis, R.P.; Reid, D.M. The interaction of light quality and irradiance with gibberellins, cytokinins and auxin in regulating growth of Helianthus annuus hypocotyls. Plant Cell Environ 2007, 30, 147–155. [Google Scholar]
- Kurepin, L.V.; Emery, R.J.N.; Pharis, R.P.; Reid, D.M. Uncoupling light quality from light irradiance effects in Helianthus annuus shoots: Putative roles for plant hormones in leaf and internode growth. J. Exp. Bot 2007, 58, 2145–2157. [Google Scholar]
- Walters, R.G.; Rogers, J.J.M.; Shephard, F.; Horton, P. Acclimation of Arabidopsis thaliana to the light environment: The role of photoreceptors. Planta 1999, 209, 517–527. [Google Scholar]
- Pogson, B.J.; Woo, N.S.; Förster, B.; Small, I.D. Plastid signalling to the nucleus and beyond. Trends Plant Sci 2008, 13, 602–609. [Google Scholar]
- Estavillo, G.M.; Chan, K.X.; Phua, S.Y.; Pogson, B.J. Reconsidering the nature and mode of action of metabolite retrograde signals from the chloroplast. Front. Plant Physiol. 2013, 3. doi:310.3389/fpls.2012.00300. [Google Scholar]
- Wilson, K.E.; Krol, M.; Hüner, N.P.A. Temperature-induced greening of Chlorella vulgaris. The role of the cellular energy balance and zeaxanthin-dependent nonphotochemical quenching. Planta 2003, 217, 616–627. [Google Scholar]
- Kim, H.-J.; Kim, Y.-K.; Park, J.-Y.; Kim, J. Light signalling mediated by phytochrome plays an important role in cold-induced gene expression through the C-repeat/dehydration responsive element (C/DRE) in Arabidopsis thaliana. Plant J 2002, 2, 693–704. [Google Scholar]
- Nott, A.; Jung, H.-S.; Koussevitzky, S.; Chory, J. Plastid-to-nucleus retrograde signaling. Annu. Rev. Plant Biol 2006, 57, 739–759. [Google Scholar]
- Fernandez, A.P.; Strand, A. Retrograde signaling and plant stress: Plastid signals initiate cellular stress responses. Curr. Opin. Plant Biol 2008, 11, 509–513. [Google Scholar]
- Jung, H.-S.; Chory, J. Signaling between chloroplasts and the nucleus: Can a systems biology approach bring clarity to a complex and highly regulated pathway? Plant Physiol 2010, 152, 453–459. [Google Scholar]
- Foyer, C.H.; Neukermans, J.; Queval, G.; Noctor, G.; Harbinson, J. Photosynthetic control of electron transport and the regulation of gene expression. J. Exp. Bot 2012, 63, 1637–1661. [Google Scholar]
- Munné-Bosch, S.; Queval, G.; Foyer, C.H. The impact of global change factors on redox signaling underpinning stress tolerance. Plant physiol 2013, 161, 5–19. [Google Scholar]
- Dietz, K.-J. Redox signal integration: From stimulus to networks and genes. Physiol. Plant 2008, 133, 459–468. [Google Scholar]
- Dietz, K.-J.; Pfannschmidt, T. Novel regulators in photosynthetic redox control of plant metabolism and gene expression. Plant physiol 2011, 155, 1477–1485. [Google Scholar]
- Fowler, S.G.; Cook, D.; Thomashow, M.F. Low temperature induction of Arabidopsis CBF1, 2, and 3 is gated by the circadian clock. Plant Physiol 2005, 137, 961–968. [Google Scholar]
- Franklin, K.A.; Whitelam, G.C. Light-quality regulation of freezing tolerance in Arabidopsis thaliana. Nat. Genet 2007, 39, 1410–1413. [Google Scholar]
- Kidokoro, S.; Maruyama, K.; Nakashima, K.; Imura, Y.; Narusaka, Y.; Shinwari, Z.K.; Osakabe, Y.; Fujita, Y.; Mizoi, J.; Shinozaki, K.; et al. The phytochrome-interacting factor PIF7 negatively regulates DREB1 expression under circadian control in Arabidopsis. Plant Physiol 2009, 151, 2046–2057. [Google Scholar]
- De Lucas, M.; Daviere, J.M.; Rodrıguez-Falcon, M.; Pontin, M.; Iglesias-Pedraz, J.M.; Lorrain, S.; Fankhauser, C.; Blasquez, M.A.; Titarenko, E.; Prat, S. A molecular framework for light and gibberellin control of cell elongation. Nature 2008, 451, 480–486. [Google Scholar]
- Feng, S.; Martinez, C.; Gusmaroli, G.; Wang, Y.; Zhou, J.; Wang, F.; Chen, L.; Yu, L.; Iglesias-Pedraz, J.M.; Kircher, S. Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 2008, 451, 475–479. [Google Scholar]
- Hurry, V.M.; Hüner, N.P.A. Low growth temperature effects a differential inhibition of photosynthesis in spring and winter wheat. Plant Physiol 1991, 96, 491–497. [Google Scholar]
- Hurry, V.M.; Strand, A.; Tobiaeson, M.; Gardeström, P.; Öquist, G. Cold hardening of spring and winter wheat and rape results in differential effects on growth, carbon metabolism, and carbohydrate content. Plant Physiol 1995, 109, 697–706. [Google Scholar]
- Hurry, V.; Strand, Å.; Furbank, R.; Stitt, M. The role of inorganic phosphate in the developpment of freezing tolerance and the acclimatization of photosynthesis to low temperature is revealed by the pho mutantsof Arabidopsis thaliana. Plant J 2000, 24, 383–396. [Google Scholar]
- Stitt, M.; Hurry, V. A plant for all seasons: Alterations in photosynthetic carbon metabolism during cold acclimation in Arabidopsis. Curr. Opin. Plant Biol 2002, 5, 199–206. [Google Scholar]
- Öquist, G.; Hüner, N.P.A. Photosynthesis of overwintering evergreen plants. Annu. Rev. Plant Biol 2003, 54, 329–355. [Google Scholar]
- Hurry, V.M.; Malmberg, G.; Gardeström, P.; Öquist, G. Effects of a short-term shift to low temperature and of long-term cold hardening on photosynthesis and ribulose 1,5-bisphosphate carboxylase/oxygenase and sucrose phosphate synthase activity in leaves of winter rye (Secale cereale L.). Plant Physiol 1994, 106, 983–990. [Google Scholar]
- Strand, A.; Hurry, V.; Henkes, S.; Hüner, N.P.A.; Gustafsson, P.; Gardestrom, P.; Stitt, M. Acclimation of Arabidopsis leaves developing at low temperatures. Increasing cytoplasmic volume accompanies increased activities of enzymes in the Calvin cycle and in the sucrose-biosynthesis pathway. Plant Physiol 1999, 119, 1387–1397. [Google Scholar]
- Savitch, L.V.; Leonardos, E.D.; Krol, M.; Jansson, S.; Grodzinski, B.; Hüner, N.P.A.; Öquist, G. Two different strategies for light utilization in photosynthesis in relation to growth and cold acclimation. Plant Cell Environ 2002, 25, 761–771. [Google Scholar]
- Rapacz, M.; Wolanin, B.; Hura, K.; Tyrka, M. The effects of cold acclimation on photosynthetic apparatus and the expression of COR14b in four genotypes of barley (Hordeum vulgare) contrasting in their tolerance to freezing and high-light treatment in cold conditions. Ann. Bot 2008, 101, 689–699. [Google Scholar]
- Leonardos, E.D.; Savitch, L.V.; Hüner, N.P.A.; Öquist, G.; Grodzinski, B. Daily photosynthetic and C-export patterns in winter wheat leaves during cold stress and acclimation. Physiol. Plant 2003, 117, 521–531. [Google Scholar]
- Lundmark, M.; Cavaco, A.M.; Trevanion, S.; Hurry, V. Carbon partitioning and export in transgenic Arabidopsis thaliana with altered capacity for sucrose synthesis grown at low temperature: A role for metabolite transporters. Plant Cell Environ 2006, 29, 1703–1714. [Google Scholar]
- Pollock, C.J.; Cairns, A.J. Fructan metabolism in grasses and cereals. Annu. Rev. Plant Biol 1991, 42, 77–101. [Google Scholar]
- Savitch, L.V.; Harney, T.; Hüner, N.P.A. Sucrose metabolism in spring and winter wheat in response to high irradiance, cold stress and cold acclimation. Physiol. Plant 2000, 108, 270–278. [Google Scholar]
- Hurry, V.M.; Gardeström, P.; Öquist, G. Reduced sensitivity to photoinhibition following frost-hardening of winter rye is due to increased phosphate availability. Planta 1993, 190, 484–490. [Google Scholar]
- Strand, A.; Foyer, C.H.; Gustafsson, P.; Gardestrom, P.; Hurry, V. Altering flux through the sucrose biosynthesis pathway in transgenic Arabidopsis thaliana modifies photosynthetic acclimation at low temperatures and the development of freezing tolerance. Plant Cell Environ 2003, 26, 523–535. [Google Scholar]
- Hurry, V.; Keerberg, O.; Pärnik, T.; Öquist, G.; Gardeström, P. Effect of cold hardening on the components of respiratory decarboxylation in the light and in the dark in leaves of winter rye. Plant Physiol 1996, 111, 713–719. [Google Scholar]
- Gray, G.R.; Heath, D. A global reorganization of the metabolome in Arabidopsis during cold acclimation is revealed by metabolic finger printing. Physiol. Plant 2005, 124, 236–248. [Google Scholar]
- Öquist, G.; Hurry, V.M.; Hüner, N.P.A. The temperature dependence of the redox state of QA and the susceptibility of photosynthesis to photoinhibition. Plant Physiol. Biochem 1993, 31, 683–691. [Google Scholar]
- Krause, G.H. Photoinhibition of photosynthesis: An evaluation of damaging and protective mechanisms. Physiol. Plant 1988, 74, 566–574. [Google Scholar]
- Boese, S.R.; Hüner, N.P.A. Effect of growth temperature and temperature shifts on spinach leaf morphology and photosynthesis. Plant Physiol 1990, 94, 1830–1836. [Google Scholar]
- Gray, G.R.; Savitch, L.V.; Ivanov, A.G.; Hüner, N.P.A. Photosystem II excitation pressure and development of resistance to photoinhibition: II. Adjustment of photosynthetic capacity in winter wheat and winter rye. Plant Physiol 1996, 110, 61–71. [Google Scholar]
- Pocock, T.H.; Hurry, V.; Savitch, L.V.; Hüner, N. Susceptibility to low-temperature photoinhibition and the acquisition of freezing tolerance in winter and spring wheat: The role of growth temperature and irradiance. Physiol. Plant 2001, 113, 499–506. [Google Scholar]
- Savitch, L.V.; Barker-Åström, J.; Ivanov, A.G.; Hurry, V.; Öquist, G.; Hüner, N.P.A.; Gardeström, P. Cold acclimation of Arabidopsis thaliana results in incomplete recovery of photosynthetic capacity, associated with an increased reduction of the chloroplast stroma. Planta 2001, 214, 295–303. [Google Scholar]
- Hüner, N.P.A.; Hopkins, W.G. Growth and development of winter rye at cold-hardening temperatures results in thylakoid membranes with increased sensitivity to low concentrations of osmoticum. Physiol. Plant 1985, 64, 468–476. [Google Scholar]
- Sarhan, F.; Ouellet, F.; Vazquez-Tello, A. The wheat wcs120 gene family. A useful model to understand the molecular genetics of freezing tolerance in cereals. Physiol. Plant 1997, 101, 439–445. [Google Scholar]
- Thomashow, M.F. So what’s new in the field of plant cold acclimation? Lots! Plant Physiol 2001, 125, 89–93. [Google Scholar]
- Hüner, N.P.A.; Palta, J.P.; Li, P.H.; Carter, J.V. Anatomical changes in leaves of puma rye in response to growth at cold-hardening temperatures. Bot. Gaz 1981, 142, 55–62. [Google Scholar]
- Gorsuch, P.A.; Pandey, S.; Atkin, O.K. Temporal heterogeneity of cold acclimation phenotypes in Arabidopsis leaves. Plant Cell Environ 2010, 33, 244–258. [Google Scholar]
- Gorsuch, P.A.; Pandey, S.; Atkin, O.K. Thermal de-acclimation: How permanent are leaf phenotypes when cold-acclimated plants experience warming? Plant Cell Environ 2010, 33, 1124–1137. [Google Scholar]
- Hüner, N.P.A.; Elfman, B.; Krol, M.; MacIntosh, A. Growth and development at cold hardening temperatures. Chloroplast ultrastructure, pigment content and composition. Can. J. Bot 1984, 62, 53–60. [Google Scholar]
- Guy, C.L.; Huber, J.L.A.; Huber, S.C. Sucrose phosphate synthase and sucrose accumilation at low temperature. Plant Physiol 1992, 100, 502–508. [Google Scholar]
- Miśkiewicz, E.; Ivanov, A.G.; Williams, J.P.; Khan, M.U.; Falk, S.; Hüner, N.P.A. Photosynthetic acclimation of the filamentous cyanobacterium, Plectonema boryanum UTEX 485, to temperature and light. Plant Cell Physiol 2000, 41, 767–775. [Google Scholar]
- Rosso, D.; Bode, R.; Li, W.; Krol, M.; Saccon, D.; Wang, S.; Schillaci, L.; Rodermel, S.R.; Maxwell, D.P.; Hüner, N.P.A. Photosynthetic redox imbalance governs leaf sectoring in the Arabidopsis thaliana variegation mutants immutans, spotty, var1, and var2. Plant Cell 2009, 21, 3473–3492. [Google Scholar]
- Bode, R. Effects of excitation pressure on variegation and global gene expression in Arabidopsis thaliana. Ph.D. Thesis, Western University, London, Canada, January 2013. [Google Scholar]
- Yang, J.S.; Wang, R.; Meng, J.J.; Bi, Y.P.; Xu, P.L.; Guo, F.; Wan, S.B.; He, Q.W.; Li, X.G. Overexpression of Arabidopsis CBF1 gene in transgenic tobacco alleviates photoinhibition of PSII and PSI during chilling stress under low irradiance. J. Plant Physiol 2010, 167, 534–539. [Google Scholar]
- Xu, Y.Z.; Santamaria, R.R.; Virdi, K.S.; Arrieta-Montiel, M.P.; Razvi, F.; Li, S.; Ren, G.; Yu, B.; Alexander, D.; Guo, L.; et al. The chloroplast triggers developmental reprogramming when MUTS HOMOLOG1 is suppressed in plants. Plant Physiol 2012, 159, 710–720. [Google Scholar]
- Murata, N.; Los, D.A. Membrane fluidity and temperature perception. Plant Physiol 1997, 115, 875–879. [Google Scholar]
- Los, D.A.; Murata, N. Sensing and Response to Low Temperature in Cyanobacteria. In Sensing, signaling and cell adaptation: Cell and Molecular Responses to Stress; Storey, K.B., Storey, J.M., Eds.; Elsevier Press: Amsterdam, Holland, 2002; Volume 3, pp. 139–153. [Google Scholar]
- Xin, Z. Acquired Freezing Tolerance in Higher Plants: The Sensing and Molecular Responses to Low Nonfreezing Temperatures. In Sensing, Signaling and Cell Adaptation: Cell and Molecular Responses to Stress; Storey, K.B., Storey, J.M., Eds.; Elsevier Press: Amsterdam, Holland, 2002; Volume 3, pp. 121–137. [Google Scholar]
- Monroy, A.F.; Sangwan, V.; Dhindsa, R.S. Low temperature signal transduction during cold acclimation: Protein phosphatase 2A as an early target for cold-inactivation. Plant J 1998, 13, 653–660. [Google Scholar]
- Plieth, C.; Hansen, U.F.; Knight, H.; Knight, M.R. Temperature sensing by plants: The primary characteristics of signal perception and calcium response. Plant J 1999, 18, 491–497. [Google Scholar]
- Örvar, B.L.; Sangwan, V.; Omann, F.; Dhindsa, R.S. Early steps in cold sensing by plant cells: The role of actin cytoskeleton and membrane fluidity. Plant J 2000, 23, 785–794. [Google Scholar]
- Zarka, D.G.; Vogel, J.T.; Cook, D.; Thomashow, M.F. Cold induction of Arabidopsis CBF genes involves multiple ICE (Inducer of CBF Expression) promoter elements and a cold-regulatory circuit that is desensitized by low temperature. Plant Physiol 2003, 133, 910–918. [Google Scholar]
- Benedict, C.; Geisler, M.; Trygg, J.; Hüner, N.P.A.; Hurry, V. Consensus by democracy. Using meta-analyses of microarray and genomic data to model the cold acclimation signaling pathway in Arabidopsis. Plant Physiol 2006, 141, 1219–1232. [Google Scholar]
- Waters, M.T.; Langdale, J.A. The making of a chloroplast. EMBO J 2009, 28, 2861–2873. [Google Scholar]
- Foyer, C.H.; Noctor, G. Photosynthetic Nitrogen Assimilation: Inter-Pathway Control and Signalling. In Photosynthetic Nitrogen Assimilation and Associated Carbon and Respiratory Metabolism; Foyer, C.H., Noctor, G., Eds.; Kluwer: Amsterdam, Holland, 2002; Volume 12, pp. 1–22. [Google Scholar]
- Hüner, N.P.A.; Grodzinski, B. Photosynthesis and Photoautotrophy. In Comprehensive Biotechnology; Moo-Young, M., Ed.; Elsevier: London, UK, 2011; Volume 1, pp. 315–322. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kurepin, L.V.; Dahal, K.P.; Savitch, L.V.; Singh, J.; Bode, R.; Ivanov, A.G.; Hurry, V.; Hüner, N.P.A. Role of CBFs as Integrators of Chloroplast Redox, Phytochrome and Plant Hormone Signaling during Cold Acclimation. Int. J. Mol. Sci. 2013, 14, 12729-12763. https://doi.org/10.3390/ijms140612729
Kurepin LV, Dahal KP, Savitch LV, Singh J, Bode R, Ivanov AG, Hurry V, Hüner NPA. Role of CBFs as Integrators of Chloroplast Redox, Phytochrome and Plant Hormone Signaling during Cold Acclimation. International Journal of Molecular Sciences. 2013; 14(6):12729-12763. https://doi.org/10.3390/ijms140612729
Chicago/Turabian StyleKurepin, Leonid V., Keshav P. Dahal, Leonid V. Savitch, Jas Singh, Rainer Bode, Alexander G. Ivanov, Vaughan Hurry, and Norman P. A. Hüner. 2013. "Role of CBFs as Integrators of Chloroplast Redox, Phytochrome and Plant Hormone Signaling during Cold Acclimation" International Journal of Molecular Sciences 14, no. 6: 12729-12763. https://doi.org/10.3390/ijms140612729
APA StyleKurepin, L. V., Dahal, K. P., Savitch, L. V., Singh, J., Bode, R., Ivanov, A. G., Hurry, V., & Hüner, N. P. A. (2013). Role of CBFs as Integrators of Chloroplast Redox, Phytochrome and Plant Hormone Signaling during Cold Acclimation. International Journal of Molecular Sciences, 14(6), 12729-12763. https://doi.org/10.3390/ijms140612729



