Development of 3D in Vitro Technology for Medical Applications
Abstract
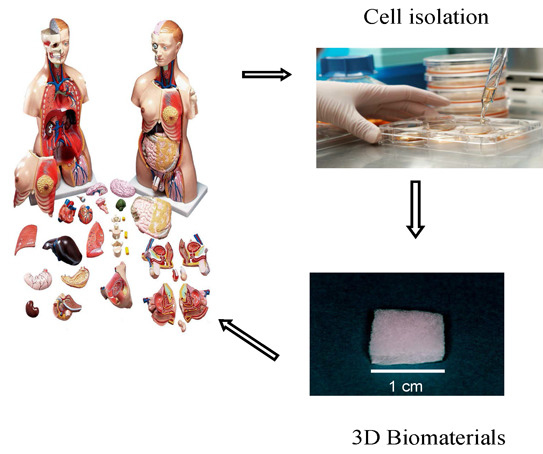
:1. Introduction

2. Three Dimensional (3D) Engineered Biomaterials
2.1. Microscale Biomaterials
2.2. Nanoscale Biomaterials
3. 3D Cellular Microenvironment
3.1. Extracellular Matrix
3.2. Cellular Microenvironment (Niche)

| Nanoscale | Microscale | Macroscale |
|---|---|---|
| support structures (<1 μm) to control individual cell behavior | support structures (1–100 μm) to control cell–cell interactions and cell-substrate interactions | support structures (>100 μm) for structural support |
| adhesion, migration proliferation | cell–ECM interaction | tissue–oran interaction |

4. 3D Technology for Development of Tissue Engineering
4.1. 3D in Vitro Technology
| 2D Culture (Tissue Culture Dish) | 3D Culture (Biomaterials) |
|---|---|
| The initial rate of cell growth is higher, but the proliferation stops once the cells reached confluence | Provides larger surface area available for cell attachment and spreading than that of 2D culture and can affect cell adhesion, spreading, and proliferation. Differentiation, cell viability and morphology of cultured cells in 3D biomaterials are significantly different with cells cultured in 2D tissue culture plate |
4.2. Bioreactor Technology


4.3. 3D Printing Technology

5. Development of Tissue Engineering in 3D Models
5.1. Potential of Biomaterials in Tissue Engineering


5.2. Advanced Technology behind 3D Biomaterials
| Advances in biomaterials for tissue engineering applications. | Nano-engineering of molecular templates and supra-molecular structures to engineer new structure of 3D biomaterials [69]. |
| Self-assembling amphiphilic peptide and protein systems that self-assemble to form various nanostructures like nanofibers, nanotubes, vesicles, helical ribbons and fibrous scaffolds [72]. | |
| Artificial proteins that self-assemble to form hydrogels in response to pH and environmental changes. Protein hydrogels can be used for advanced wound closure and tissue repair in regenerative medicine and tissue engineering [28]. | |
| Used as scaffolds to fabricate nanowires, templates for metallization (Ex: Histidine-rich peptide nanotubes were metallized with gold nanocrystals and the organic peptide scaffold was removed to make a conducting gold nanowires [66]. | |
| Targeted tissue engineering using ligand-receptors biomaterials that are chemically modified to mimic ECM to direct stem cells differentiation [30]. | |
| Combinational technology of micro- and nano-fabrication to mimic mechanical properties of ECM to accelerate tissue regeneration [65]. | |
| Controlled release technology of growth factors from implanted 3D biomaterials to regenerate new tissues [25]. | |
| Gene therapy technology in combination with 3D biomaterials for tissue engineering applications [65] | |
| Enhancement of angiogenesis by using 3D biomaterials to enhance survival of transplanted stem cells [4]. |
6. Future Prospects
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Taboas, J.; Maddox, R.; Krebsbach, P.; Hollister, S. Indirect solid free form fabrication of local and global porous, biomimetic and composite 3D polymer-ceramic scaffolds. Biomaterials 2003, 24, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.X.; Choi, J.-W. Biodegradable polymer scaffolds with well-defined interconnected spherical pore network. Tissue Eng. 2001, 7, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Hattori, S.; Matsuoka, R.; Kawaguchi, N. Micro and nano- scale in vitro 3D culture system for cardiac stem cells. J. Biomed. Mater. Res. Part A 2010, 94, 1–8. [Google Scholar] [CrossRef]
- Hosseinkhani, H.; Hosseinkhani, M.; Kobayashi, H. Design of tissue-engineered nanoscaffold through self-assembly of peptide amphiphile. J. Bioact. Compat. Polym. 2006, 21, 277–296. [Google Scholar] [CrossRef]
- Kalhor, H.R.; Shahin, V.F.; Fouani, M.H.; Hosseinkhani, H. Self-assembly of tissue transglutaminase into amyloid-like fibrils using physiological concentration of Ca2+. Langmuir 2011, 27, 10776–10784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohajeri, S.; Hosseinkhani, H.; Golshan Ebrahimi, N.; Nikfarjam, L.; Soleimani, M.; Kajbafzadeh, A.-M. Proliferation and differentiation of mesenchymal stem cell on collagen sponge reinforced with polypropylene/polyethylene terephthalate blend fibers. Tissue Eng. Part A 2010, 16, 3821–3830. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Aoyama, T.; Ogawa, O.; Tabata, Y. In vitro transfection of plasmid DNA by different-cationized gelatin with or without ultrasound irradiation. Proc. Jpn. Acad. Ser. B 2001, 77, 161–166. [Google Scholar] [CrossRef]
- Yang, F.; Murugan, R.; Wang, S.; Ramakrishna, S. Electrospinning of nano/micro scale poly (l-lactic acid) aligned fibers and their potential in neural tissue engineering. Biomaterials 2005, 26, 2603–2610. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Xu, C.; Kotaki, M.; Wang, S.; Ramakrishna, S. Characterization of neural stem cells on electrospun poly (l-lactic acid) nanofibrous scaffold. J. Biomater. Sci. Polym. Ed. 2004, 15, 1483–1497. [Google Scholar] [CrossRef] [PubMed]
- Katta, P.; Alessandro, M.; Ramsier, R.; Chase, G. Continuous electrospinning of aligned polymer nanofibers onto a wire drum collector. Nano Lett. 2004, 4, 2215–2218. [Google Scholar] [CrossRef]
- Rosenberg, M.D. Cell guidance by alterations in monomolecular films. Science 1963, 139, 411. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Kotaki, M.; Inai, R.; Ramakrishna, S. Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng. 2005, 11, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Price, R.L.; Waid, M.C.; Haberstroh, K.M.; Webster, T.J. Selective bone cell adhesion on formulations containing carbon nanofibers. Biomaterials 2003, 24, 1877–1887. [Google Scholar] [CrossRef]
- Elias, K.L.; Price, R.L.; Webster, T.J. Enhanced functions of osteoblasts on nanometer diameter carbon fibers. Biomaterials 2002, 23, 3279–3287. [Google Scholar] [CrossRef] [PubMed]
- Webster, T.J.; Schadler, L.S.; Siegel, R.W.; Bizios, R. Mechanisms of enhanced osteoblast adhesion on nanophase alumina involve vitronectin. Tissue Eng. 2001, 7, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Ma, P. Nano-fibrous scaffolds for tissue engineering. Colloids Surf. B 2004, 39, 125–131. [Google Scholar] [CrossRef]
- Kameoka, J.; Verbridge, S.S.; Liu, H.; Czaplewski, D.A.; Craighead, H. Fabrication of suspended silica glass nanofibers from polymeric materials using a scanned electrospinning source. Nano Lett. 2004, 4, 2105–2108. [Google Scholar] [CrossRef]
- Baker, S.C.; Atkin, N.; Gunning, P.A.; Granville, N.; Wilson, K.; Wilson, D.; Southgate, J. Characterisation of electrospun polystyrene scaffolds for three-dimensional in vitro biological studies. Biomaterials 2006, 27, 3136–3146. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Cui, D.; Schwarz, H.; Estrada, G.G.; Kobayashi, H. Cytotoxicity of single-wall carbon nanotubes on human fibroblasts. Toxicol. In Vitro 2006, 20, 1202–1212. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A.; Yokoyama, Y.; Estrada, G.G.; Kobayashi, H. Quantitative analysis of cell adhesion on aligned micro- and nanofibers. J. Biomed. Mater. Res. Part A 2008, 84, 291–299. [Google Scholar] [CrossRef]
- Hosseinkhani, H.; Hosseinkhani, M. Suppression effect of basic fibroblast growth factor on mesenchymal stem cell proliferation activity. Part I Release characteristics. Chimica Oggi 2008, 26, 10–13. [Google Scholar]
- Hosseinkhani, H.; Hosseinkhani, M. Suppression effect of basic fibroblast growth factor on mesenchymal stem cell proliferation activity. Part II Biological characteristics. Chimica Oggi 2008, 26, 64–66. [Google Scholar]
- Hong, Y.; Legge, R.L.; Zhang, S.; Chen, P. Effect of amino acid sequence and pH on nanofiber formation of self-assembling peptides EAK16-II and EAK16-IV. Biomacromolecules 2003, 4, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Hosseinkhani, H.; Hosseinkhani, M.; Boutry, S.; Simchi, A.; Journeay, W.S.; Subramani, K.; Laurent, S. Magnetic resonance imaging tracking of stem cells in vivo using iron oxide nanoparticles as a tool for the advancement of clinical regenerative medicine. Chem. Rev. 2010, 111, 253–280. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hong, P.-D.; Yu, D.-S. Self-assembled proteins and peptides for regenerative medicine. Chem. Rev. 2013, 113, 4837–4861. [Google Scholar] [CrossRef] [PubMed]
- Bandtlow, C.E.; Zimmermann, D.R. Proteoglycans in the developing brain: New conceptual for old proteins. Physiol. Rev. 2000, 80, 1267–1290. [Google Scholar]
- Yamaguchi, Y. Lecticans: Organizers of the brain extracellular matrix. Cell. Mol. Life Sci. 2000, 57, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Bissell, M.J.; LaBarge, M.A. Context, tissue plasticity, and cancer: Are tumor stem cells also regulated by the microenvironment? Cancer Cell 2005, 7, 17. [Google Scholar]
- Hosseinkhani, H.; Hong, P.-D.; Yu, D.-S.; Chen, Y.-R.; Ickowicz, D.; Farber, I.-Y.; Domb, A.J. Development of 3D in vitro platform technology to engineer mesenchymal stem cells. Int. J. Nanomed. 2012, 7, 3035. [Google Scholar] [CrossRef]
- Hosseinkhani, H.; Hosseinkhani, M. Tissue engineered Scaffolds for stem cells and regenerative medicine. In Trends in Stem Cell Biology and Technology; Springer: Berlin, Germany, 2009; pp. 367–387. [Google Scholar]
- Gingras, M.; Beaulieu, M.M.; Gagnon, V.; Durham, H.D.; Berthod, F. In vitro study of axonal migration and myelination of motor neurons in a three-dimensional tissue-engineered model. Glia 2008, 56, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Gingras, M.; Bergeron, J.; Déry, J.; Durham, H.D.; Berthod, F. In vitro development of a tissue-engineered model of peripheral nerve regeneration to study neurite growth. FASEB J. 2003, 17, 2124–2126. [Google Scholar] [PubMed]
- Horst, M.; Madduri, S.; Gobet, R.; Sulser, T.; Hall, H.; Eberli, D. Scaffold characteristics for functional hollow organ regeneration. Materials 2010, 3, 241–263. [Google Scholar] [CrossRef] [Green Version]
- Ghodsizadeh, A.; Hosseinkhani, H.; Piryaei, A.; Pournasr, B.; Najarasl, M.; Hiraoka, Y.; Baharvand, H. Galactosylated collagen matrix enhanced in vitro maturation of human embryonic stem cell-derived hepatocyte-like cells. Biotechnol. Lett. 2014, 36, 1095–1106. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.; Osborne, R.; Munger, R.; Xiong, X.; Doillon, C.J.; Laycock, N.L.; Hakim, M.; Song, Y.; Watsky, M.A. Functional human corneal equivalents constructed from cell lines. Science 1999, 286, 2169–2172. [Google Scholar] [CrossRef] [PubMed]
- Kloth, S.; Ebenbeck, C.; Kubitza, M.; Schmidbauer, A.; Röckl, W.; Minuth, W. Stimulation of renal microvascular development under organotypic culture conditions. FASEB J. 1995, 9, 963–967. [Google Scholar] [PubMed]
- Kloth, S.; Eckert, E.; Klein, S.J.; Monzer, J.; Wanke, C.; Minuth, W.W. Gastric epithelium under organotypic perfusion culture. In Vitro Cell. Dev. Biol.-Anim. 1998, 34, 515–517. [Google Scholar] [CrossRef]
- Minuth, W.W.; Dermietzel, R.; Kloth, S.; Hennerkes, B. A new method culturing renal cells under permanent superfusion and producing a luminal-basal medium gradient. Kidney Int. 1992, 41, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.M.; Palsson, B.O.; Emerson, S.G. Rapid medium perfusion rate significantly increases the productivity and longevity of human bone marrow cultures. Proc. Natl. Acad. Sci. USA 1991, 88, 6760–6764. [Google Scholar] [CrossRef]
- Glowacki, J.; Mizuno, S.; Greenberger, J.S. Perfusion enhances functions of bone marrow stromal cells in three-dimensional culture. Cell Transplant. 1998, 7, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.M.; Mizuno, S.; Gerstenfeld, L.C.; Glowacki, J. Medium perfusion enhances osteogenesis by murine osteosarcoma cells in three-dimensional collagen sponges. J. Bone Miner. Res. 1999, 14, 2118–2126. [Google Scholar] [CrossRef] [PubMed]
- Castilho, L.R.; Medronho, R.A. Cell retention devices for suspended-cell perfusion cultures. In Tools and Applications of Biochemical Engineering Science; Springer: Berlin, Germany, 2002; pp. 129–169. [Google Scholar]
- Ingber, D.E.; Prusty, D.; Sun, Z.; Betensky, H.; Wang, N. Cell shape, cytoskeletal mechanics, and cell cycle control in angiogenesis. J. Biomech. 1995, 28, 1471–1484. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Yang, S.; Kniss, D.A. Three-dimensional cell-scaffold constructs promote efficient gene transfection: Implications for cell-based gene therapy. Tissue Eng. 2001, 7, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Noshi, T.; Yoshikawa, T.; Ikeuchi, M.; Dohi, Y.; Ohgushi, H.; Horiuchi, K.; Sugimura, M.; Ichijima, K.; Yonemasu, K. Enhancement of the in vivo osteogenic potential of marrow/hydroxyapatite composites by bovine bone morphogenetic protein. J. Biomed. Mater. Res. 2000, 52, 621–630. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Ohgushi, H.; Akahane, M.; Tamai, S.; Ichijima, K. Analysis of gene expression in osteogenic cultured marrow/hydroxyapatite construct implanted at ectopic sites: A comparison with the osteogenic ability of cancellous bone. J. Biomed. Mater. Res. 1998, 41, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.R.; Bender, J.G.; Papoutsakis, E.T.; Miller, W.M. Beneficial effects of reduced oxygen tension and perfusion in long-term hematopoietic culturesa. Ann. N. Y. Acad. Sci. 1992, 665, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Chesler, N.C.; Thompson-Figueroa, J.; Millburne, K. Measurements of mouse pulmonary artery biomechanics. J. Biomech. Eng. 2004, 126, 309–313. [Google Scholar] [CrossRef]
- Sittinger, M.; Schultz, O.; Keyszer, G.; Minuth, W.; Burmester, G. Artificial tissues in perfusion culture. Int. J. Artif. Organs 1997, 20, 57–62. [Google Scholar] [PubMed]
- Bancroft, G.N.; Sikavitsas, V.I.; van den Dolder, J.; Sheffield, T.L.; Ambrose, C.G.; Jansen, J.A.; Mikos, A.G. Fluid flow increases mineralized matrix deposition in 3D perfusion culture of marrow stromal osteoblasts in a dose-dependent manner. Proc. Natl Acad. Sci. USA 2002, 99, 12600–12605. [Google Scholar] [CrossRef] [PubMed]
- Cartmell, S.H.; Porter, B.D.; García, A.J.; Guldberg, R.E. Effects of medium perfusion rate on cell-seeded three-dimensional bone constructs in vitro. Tissue Eng. 2003, 9, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Inatsugu, Y.; Hiraoka, Y.; Inoue, S.; Shimokawa, H.; Tabata, Y. Impregnation of plasmid DNA into three-dimensional scaffolds and medium perfusion enhance in vitro DNA expression of mesenchymal stem cells. Tissue Eng. 2005, 11, 1459–1475. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Inatsugu, Y.; Hiraoka, Y.; Inoue, S.; Tabata, Y. Perfusion culture enhances osteogenic differentiation of rat mesenchymal stem cells in collagen sponge reinforced with poly (glycolic acid) fiber. Tissue Eng. 2005, 11, 1476–1488. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Li, Y.; Yang, S.T.; Kniss, D.A. Effects of pore size in 3-D fibrous matrix on human trophoblast tissue development. Biotechnol. Bioeng. 2000, 70, 606–618. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, A.S.; Juarez, T.M.; Helmke, C.D.; Gustin, M.C.; Mikos, A.G. Effect of convection on osteoblastic cell growth and function in biodegradable polymer foam scaffolds. Biomaterials 2001, 22, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Mueller-Klieser, W. Three-dimensional cell cultures: From molecular mechanisms to clinical applications. Am. J. Physiol-Cell Physiol. 1997, 273, C1109–C1123. [Google Scholar]
- Hosseinkhani, H.; Hosseinkhani, M.; Tian, F.; Kobayashi, H.; Tabata, Y. Ectopic bone formation in collagen sponge self-assembled peptide–amphiphile nanofibers hybrid scaffold in a perfusion culture bioreactor. Biomaterials 2006, 27, 5089–5098. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.S.; Won, Y.J.; Cooke, P.H.; Coffin, D.R.; Fishman, M.L.; Hicks, K.B.; Ma, P.X. Pectin/poly(lactide-co-glycolide) composite matrices for biomedical applications. Biomaterials 2004, 25, 3201–3210. [Google Scholar] [CrossRef] [PubMed]
- Wintermantel, E.; Mayer, J.; Blum, J.; Eckert, K.-L.; Lüscher, P.; Mathey, M. Tissue engineering scaffolds using superstructures. Biomaterials 1996, 17, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.X.; Langer, R. Morphology and mechanical function of long-term in vitro engineered cartilage. J. Biomed. Mater. Res. 1999, 44, 217–221. [Google Scholar] [CrossRef]
- Alibolandi, M.; Abnous, K.; Ramezani, M.; Hosseinkhani, H.; Hadizadeh, F. Synthesis of AS1411-Aptamer-Conjugated CdTe quantum dots with high fluorescence strength for probe labeling tumor cells. J. Fluoresc. 2014, 24, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.E.; Ribeiro, A.; Malafaya, P.; Reis, R.; Cunha, A. A new approach based on injection moulding to produce biodegradable starch-based polymeric scaffolds: Morphology, mechanical and degradation behaviour. Biomaterials 2001, 22, 883–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosseinkhani, H.; Azzam, T.; Kobayashi, H.; Hiraoka, Y.; Shimokawa, H.; Domb, A.J.; Tabata, Y. Combination of 3D tissue engineered scaffold and non-viral gene carrier enhance in vitro DNA expression of mesenchymal stem cells. Biomaterials 2006, 27, 4269–4278. [Google Scholar] [CrossRef] [PubMed]
- Pachenari, M.; Seyedpour, S.; Janmaleki, M.; Shayan, S.B.; Taranejoo, S.; Hosseinkhani, H. Mechanical properties of cancer cytoskeleton depend on actin filaments to microtubules content: Investigating different grades of colon cancer cell lines. J. Biomech. 2014, 47, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Rahbarghazi, R.; Nassiri, S.M.; Ahmadi, S.H.; Mohammadi, E.; Rabbani, S.; Araghi, A.; Hosseinkhani, H. Dynamic induction of pro-angiogenic milieu after transplantation of marrow-derived mesenchymal stem cells in experimental myocardial infarction. Int. J. Cardiol. 2014, 173, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Tatu, R.; Liu, Q.; Hosseinkhani, H. Stem cell-based tissue engineering for regenerative medicine. Nano LIFE 2014, 4, 1430001. [Google Scholar] [CrossRef]
- Langer, R. Biomaterials in drug delivery and tissue engineering: One laboratory’s experience. Acc. Chem. Res. 2000, 33, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.W.Y.; Hosseinkhani, H.; Rahman, S.A.; Rosli, R.; Domb, A.J.; Abdullah, S. Safety profile of dextran-spermine gene delivery vector in mouse lungs. J. Nanosci. Nanotechnol. 2014, 14, 3328–3336. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.-J.; Oh, J.-W.; Kwak, K.; Lee, B.Y.; Meyer, J.; Wang, E.; Hexemer, A.; Lee, S.-W. Biomimetic self-templating supramolecular structures. Nature 2011, 478, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Marini, D.M.; Hwang, W.; Santoso, S. Design of nanostructured biological materials through self-assembly of peptides and proteins. Curr. Opin. Chem. Biol. 2002, 6, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A. Tissue regeneration through self-assembled peptide amphiphile nanofibers. Yakhte Med. J. 2006, 6, 204–209. [Google Scholar]
- Hosseinkhani, H.; Yamamoto, M.; Inatsugu, Y.; Hiraoka, Y.; Inoue, S.; Shimokawa, H.; Tabata, Y. Enhanced ectopic bone formation using combination of impregnation of plasmid DNA into 3-D scaffold and bioreactor perfusion culture. Biomaterials 2006, 27, 1387–1398. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Khademhosseini, A.; Kobayashi, H. Bone regeneration through controlled release of bone morphogenetic protein-2 from 3-D tissue engineered nano-scaffold. J. Control. Release 2007, 117, 380–386. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ou, K.-L.; Hosseinkhani, H. Development of 3D in Vitro Technology for Medical Applications. Int. J. Mol. Sci. 2014, 15, 17938-17962. https://doi.org/10.3390/ijms151017938
Ou K-L, Hosseinkhani H. Development of 3D in Vitro Technology for Medical Applications. International Journal of Molecular Sciences. 2014; 15(10):17938-17962. https://doi.org/10.3390/ijms151017938
Chicago/Turabian StyleOu, Keng-Liang, and Hossein Hosseinkhani. 2014. "Development of 3D in Vitro Technology for Medical Applications" International Journal of Molecular Sciences 15, no. 10: 17938-17962. https://doi.org/10.3390/ijms151017938