A Simplified Method for Simultaneous Determination of α-Lipoic Acid and Low-Molecular-Mass Thiols in Human Plasma
Abstract
:1. Introduction
2. Results and Discussion
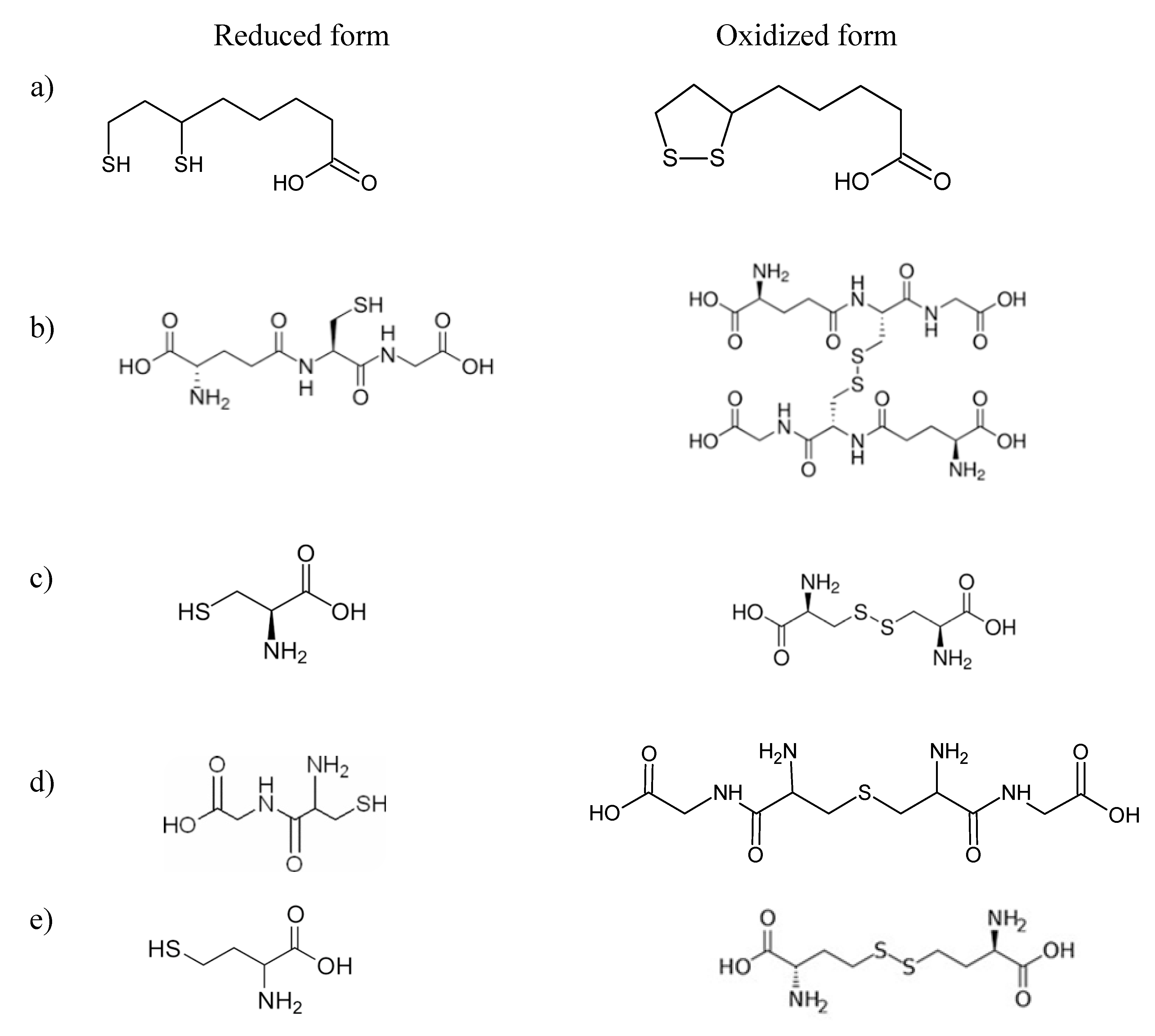
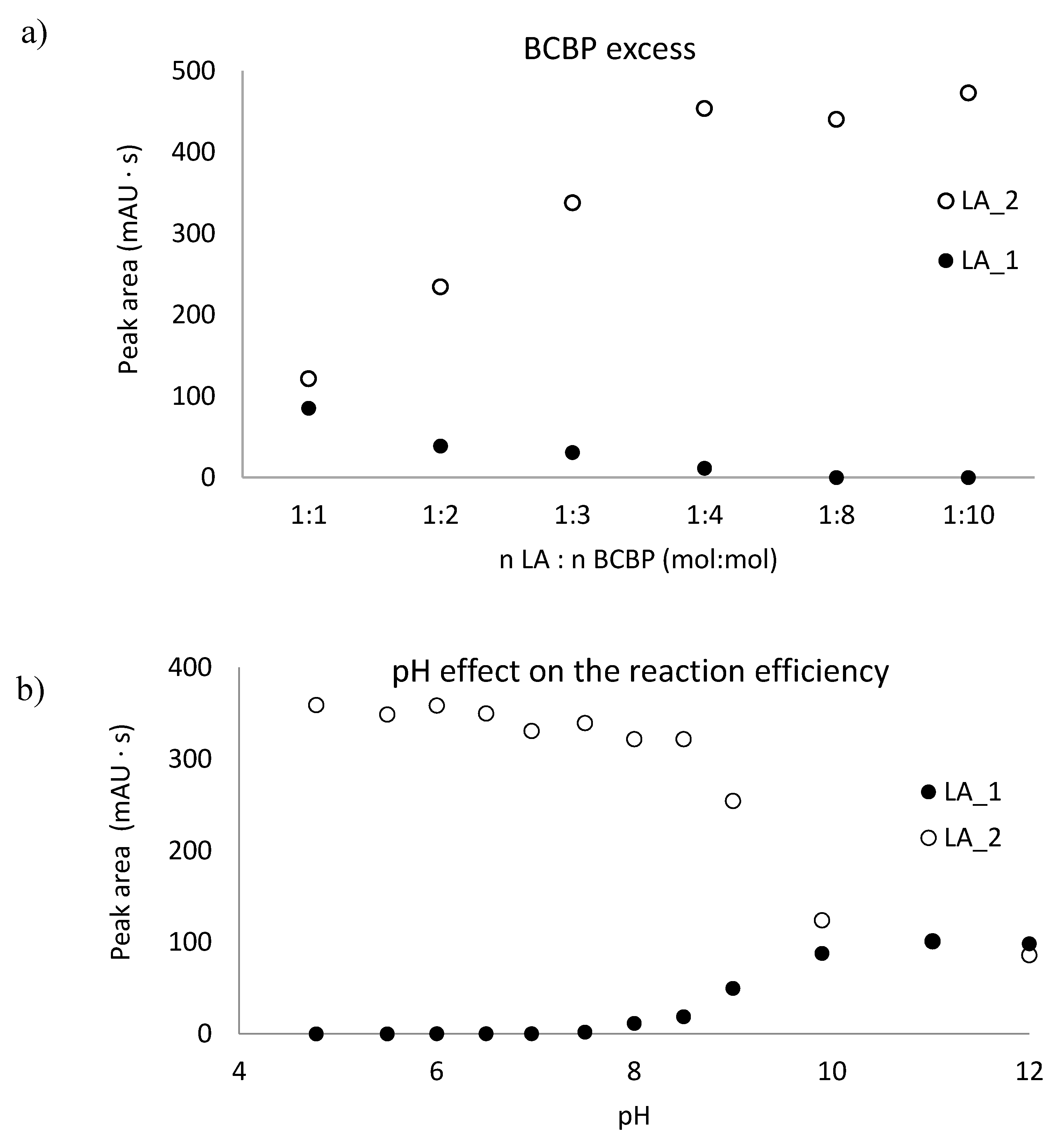
2.1. The Thiol Group Derivatization
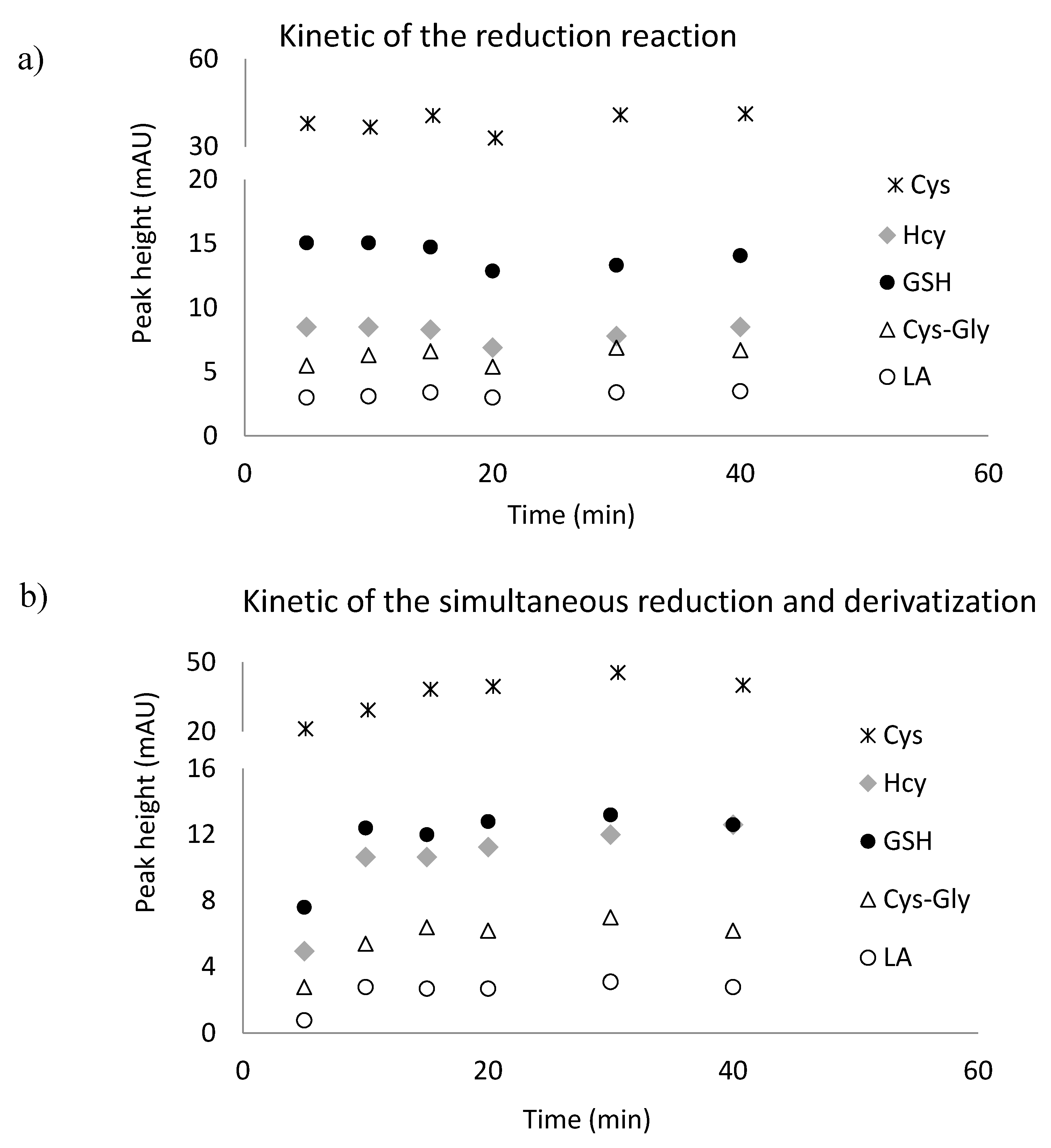
2.2. Reduction of Disulfide Bonds
2.3. LA and DHLA Separation from Proteins
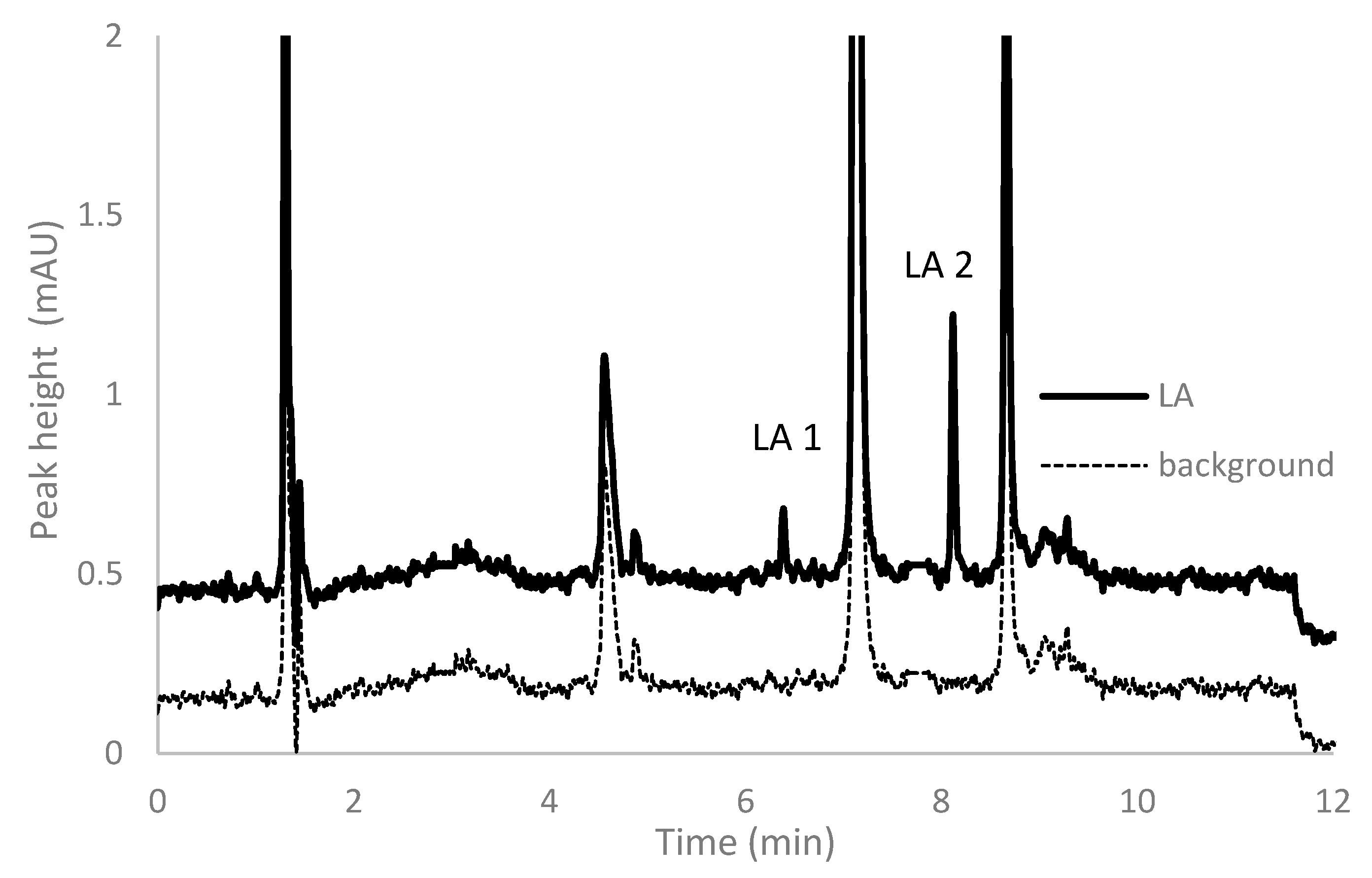
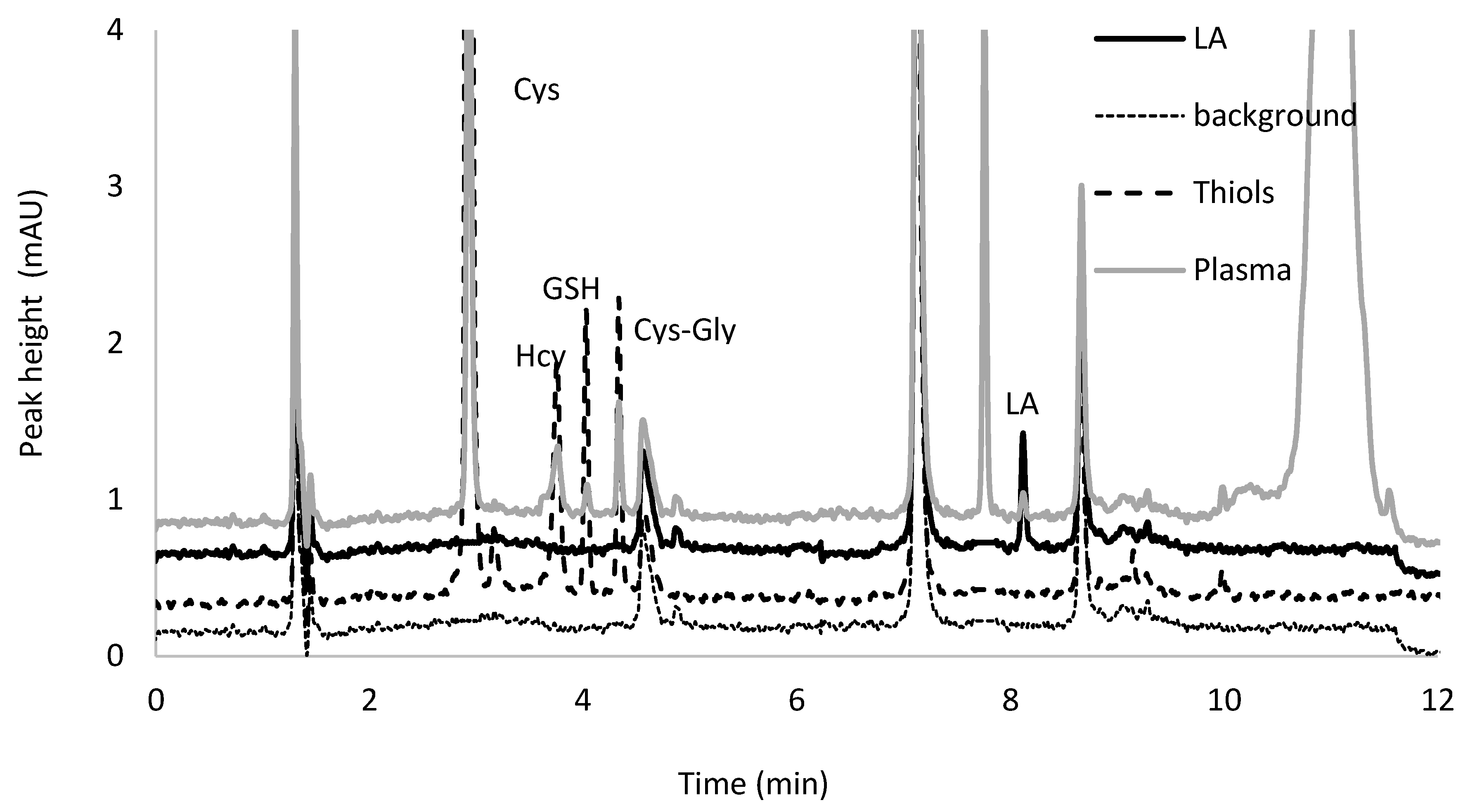
2.4. Chromatographic Conditions
2.5. Method Validation
2.5.1. LOD and LOQ
2.5.2. Linearity
2.5.3. Accuracy and Precision
2.6. Application to Authentic Plasma Samples
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Instrumentation
3.3. Human Plasma Samples
3.4. Stock Solutions
3.5. Human Plasma
3.6. HPLC conditions for Determination of LA and Low-Molecular-Mass Thiols in Human Plasma
3.7. Calibration and Validation Process
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BCPB | 1-benzyl-2-chloropyridinium bromide |
| Cys | cysteine |
| Cys–Gly | cysteinylglycine |
| DHLA | dihydrolipoic acid |
| GSH | glutathione |
| LDL-C | low-density lipoprotein cholesterol |
| Hcy | homocysteine |
| HSA | human serum albumin |
| LA | α-lipoic acid |
| MeCN | acetonitrile |
| TCA | trichloroacetic acid |
| TCEP | tris(2-carboxyethyl)phosphine |
References
- Rahimlou, M.; Asadi, M.; Jahromi, N.B.; Mansoori, A. Alpha-lipoic acid (ALA) supplementation effect on glycemic and inflammatory biomarkers: A Systematic Review and meta-analysis. Clin. Nutr. ESPEN. 2019, 32, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Farhata, D.; Linceta, H. Lipoic acid a multi-level molecular inhibitor of tumorigenesis. BBA Rev. Cancer. 2020, 1873, 188317. [Google Scholar] [CrossRef] [PubMed]
- Gorąca, A.; Huk-Kolega, H.; Piechota, A.; Kleniewska, P.; Ciejka, E.; Skibska, B. Lipoic acid—biological activity and therapeutic potential. Pharmacol. Rep. 2011, 63, 849–858. [Google Scholar] [CrossRef]
- Golbidi, S.; Badranand, M.; Laher, I. Diabetes and alpha lipoic acid. Front. Pharmacol. 2011, 2, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wollin, S.D.; Jones, P.J. a-Lipoic acid and cardiovascular disease. J. Nutr. 2003, 133, 3327–3330. [Google Scholar] [CrossRef]
- Feuerecker, B.; Pirsig, S.; Seidl, C.; Aichler, M.; Feuchtinger, A.; Bruchelt, G.; Senekowitsch-Schmidtke, R. Lipoic acid inhibits cell proliferation of tumor cells in vitro and in vivo. Cancer Biol. Ther. 2012, 13, 1425–1435. [Google Scholar] [CrossRef] [Green Version]
- Waslo, C.; Bourdette, D.; Gray, N.; Wright, K.; Spain, R. Lipoic acid and other antioxidants as therapies for multiple sclerosis. Curr. Treat. Options Neurol. 2019, 21, 26. [Google Scholar] [CrossRef]
- Liu, W.; Shi, L.; Li, S. The immunomodulatory effect of alpha-lipoic acid in autoimmune diseases. BioMed Res. Int. 2019, 8086257. [Google Scholar] [CrossRef]
- Hernández-Rabaza, V.; López-Pedrajas, R.; Almansa, I. Progesterone, lipoic acid, and sulforaphane as promising antioxidants for retinal diseases: A review. Antioxidants 2019, 8, 53. [Google Scholar] [CrossRef] [Green Version]
- Pocernich, C.B.; Butterfield, D.A. Elevation of glutathione as a therapeutic strategy in Alzheimer disease. Bioch. Biophys. Acta. 2012, 1822, 625–630. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.; Choi, J. Age-associated decline in gamma-glutamylcysteine synthetase gene expression in rats. Free Radic. Biol. Med. 2000, 28, 566–574. [Google Scholar] [CrossRef]
- Tsugawa, S.; Noda, Y.; Tarumi, R.; Mimura, Y.; Yoshida, K.; Iwata, Y.; Elsalhy, M.; Kuromiya, M.; Kurose, S.; Masuda, F.; et al. Glutathione levels and activities of glutathione metabolism enzymes in patients with schizophrenia: A systematic review and meta-analysis. J. Psychopharm. 2019, 33, 1199–1214. [Google Scholar] [CrossRef] [PubMed]
- Zmorzyński, S.; Świderska-Kołacz, G.; Koczkodaj, D.; Filip, A.A. Significance of polymorphisms and expression of enzyme-encoding genes related to glutathione in hematopoietic cancers and solid tumors. Biomed. Res. Int. 2015, 2015, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, S.C. Glutathione synthesis. Biochim. Biophys. Acta. 2014, 1830, 3143–3153. [Google Scholar] [CrossRef] [Green Version]
- Corso, C.R.; Acco, A. Glutathione system in animal model of solid tumors: From regulation to therapeutic target. Crit. Rev. Oncol. Hematol. 2018, 128, 43–57. [Google Scholar] [CrossRef]
- Jakubowski, H. Homocysteine is a protein amino acid in humans. J. Biol. Chem. 2002, 277, 30425–30428. [Google Scholar] [CrossRef] [Green Version]
- Jakubowski, H. Homocysteine modification in protein structure/function and human disease. Physiol. Rev. 2019, 99, 555–604. [Google Scholar] [CrossRef]
- Saeed, S.; Faramarz, F.; Mojtaba, S.; Davoodi, G.; Mahmoodian, M.; Fallah, N.; Darvish, S.; Karimi, A. Tehran Heart Center. Homocysteine, vitamin B12 and folate levels in premature coronary artery disease. BMC Cardiovasc. Disord. 2006, 6, 38. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Jiang, X.; Yang, F.; Gaubatz, J.W.; Ma, L.; Magera, M.J.; Yang, X.F.; Berger, P.B.; Durante, W.; Pownall, H.J.; et al. Hyperhomocysteinemia accelerates atherosclerosis in cystathionine beta-synthase and apolipoprotein E double knock-out mice with and without dietary perturbation. Blood 2003, 101, 3901–3907. [Google Scholar] [CrossRef]
- Dong, X.; Yao, Z.; Hu, Y.; Yang, N.; Gao, X.; Xu, Y.; Wang, G. Potential harmful correlation between homocysteine and low-density lipoprotein cholesterol in patients with hypothyroidism. Medicine 2016, 95, e4291. [Google Scholar] [CrossRef]
- Chwatko, G.; Krawczyk, M.; Iciek, M.; Kamińska, A.; Bilska-Wilkosz, A.; Marcykiewicz, B.; Głowacki, R. Determination of lipoic acid in human plasma by high-performance liquid chromatography with ultraviolet detection. Arab. J. Chem. 2019, 12, 4878–4886. [Google Scholar] [CrossRef]
- Witt, W.; Rustow, B. Determination of lipoic acid by precolumn derivatization with monobromobimane and reversed-phase high-performance liquid chromatography. J. Chromatogr. B 1998, 705, 127–131. [Google Scholar] [CrossRef]
- Satoh, S.; Toyo’oka, T.; Fukushima, T.; Inagaki, S. Simultaneous determination of α-lipoic acid and its reduced form by high-performance liquid chromatography with fluorescence detection. J. Chromatogr. B. 2007, 854, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Borowczyk, K.; Olejarz, P.; Chwatko, G. Application of simultaneous separation and derivatization for the determination of α-lipoic acid in urine samples by high-performance liquid chromatography with spectrofluorimetric detection. Biomed. Chromatogr. 2019, 33, e4576. [Google Scholar] [CrossRef]
- Khan, A.; Iqbal, Z.; Watson, D.G.; Khan, A.; Khan, I.; Muhammad, N.; Muhammad, S.; Nasib, H.A.; Iqbal, N.; Faiz-ur-rehman Kashif, M. Simultaneous determination of lipoic acid (LA) and dihydrolipoic acid (DHLA) in human plasma using high performance liquid chromatography coupled with electrochemical detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 1725–1731. [Google Scholar] [CrossRef]
- Khan, M.I.; Iqbal, Z.; Ahmad, L. Simultaneous determination of the endogenous free a-lipoic acid and dihydrolipoic acid in human plasma and erythrocytes by RP-HPLC with electrochemical detection. Chromatographia 2011, 73, 929–939. [Google Scholar] [CrossRef]
- Montero, O.; Ramırez, M.; Sanchez-Guijo, A.; Gonzalez, C. Determination of lipoic acid, Trolox methyl ether and tocopherols in human plasma by liquid-chromatography and ion-trap tandem mass spectrometry. Biomed. Chromatogr. 2012, 26, 1228–1233. [Google Scholar] [CrossRef]
- Kuśmierek, K.; Chwatko, G.; Głowacki, R.; Kubalczyk, P.; Bald, E. Ultraviolet derivatization of low-molecular-mass thiols for high performance liquid chromatography and capillary electrophoresis analysis. J. Chromatogr. B. 2011, 879, 1290–1307. [Google Scholar] [CrossRef]
- Borowczyk, K.; Krawczyk, M.; Kubalczyk, P.; Chwatko, G. Determination of lipoic acid in biological samples. Bioanalysis 2015, 7, 1785–1798. [Google Scholar] [CrossRef]
- Borowczyk, K.; Wyszczelska-Rokiel, M.; Kubalczyk, P.; Głowacki, R. Simultaneous determination of albumin and low-molecular-mass thiols in plasma by HPLC with UV detection. J. Chromatogr. B. 2015, 981, 57–64. [Google Scholar] [CrossRef]
- Kuśmierek, K.; Bald, E. Reversed-phase liquid chromatography method for the determination of total plasma thiols after derivatization with 1-benzyl-2-chloropyridinium bromide. Biomed. Chromatogr. 2009, 23, 770–775. [Google Scholar] [CrossRef]
- Głowacki, R.; Bald, E.; Jakubowski, H. Identification and origin of Nε-homocysteinyl-lysine isopeptide in humans and mice. Amino Acids. 2010, 39, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Kuśmierek, K.; Chwatko, G.; Głowacki, R.; Bald, E. Determination of endogenous thiols and thiol drugs in urine by HPLC with ultraviolet detection. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2009, 877, 3300–3308. [Google Scholar] [CrossRef] [PubMed]
- Kamińska, A.; Głowacka, I.E.; Pasternak, B.; Głowacki, R.; Chwatko, G. The first method for determination of lipoyllysine in human urine after oral lipoic acid supplementation. Bioanalysis 2019, 11, 1359–1373. [Google Scholar] [CrossRef]
- Chwatko, G.; Kubalczyk, P.; Bald, E. Determination of lipoic acid in the form of 2-S-pyridinium derivative by high-performance liquid chromatography with ultraviolet detection. Curr. Org. Chem. 2014, 10, 320–325. [Google Scholar] [CrossRef]
- Atukeren, P.; Aydin, S.; Uslu, E.; Gumustas, M.K.; Cakatay, U. Redox homeostasis of albumin in relation to alpha-lipoic acid and dihydrolipoic acid. Oxidat. Med. Cell. Longev. 2010, 3, 206–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawakami, A.; Kubota, K.; Yamada, N.; Tagami, U.; Takehana, K.; Sonaka, I.; Suzuki, E.; Hirayama, K. Identification and characterization of oxidized human serum albumin. A slight structural change impairs its ligand-binding and antioxidant functions. FEBS J. 2006, 273, 3346–3357. [Google Scholar] [CrossRef] [PubMed]
- Chng, H.T.; New, L.S.; Neo, A.H.; Goh, C.W.; Browne, E.R.; Chan, E.C.Y. A sensitive LC/MS/MS bioanalysis assay of orally administered lipoic acid in rat blood and brain tissue. J. Pharm. Biomed. Anal. 2010, 51, 754–757. [Google Scholar] [CrossRef]
- Khan, A.; Khan, M.I.; Iqbal, Z.; Ahmad, L.; Shah, Y.; Watson, D.G. Determination of lipoic acid in human plasma by HPLC ECD using liquid–liquid and solid-phase extraction: Method development, validation and optimization of experimental parameters. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2010, 878, 2782–2788. [Google Scholar] [CrossRef]
- Bald, E.; Chwatko, G.; Głowacki, R.; Kuśmierek, K. Analysis of plasma thiols by high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. A. 2004, 1032, 109–115. [Google Scholar] [CrossRef]
- Głowacki, R.; Bald, E. Fully automated method for simultaneous determination of total cysteine, cysteinylglycine, glutathione and homocysteine in plasma by HPLC with UV absorbance detection. J. Chromatogr. B 2009, 877, 3400–3404. [Google Scholar] [CrossRef] [PubMed]
- FDA. Guidance for Industry Bioanalytical Method Validation. Available online: http://www.fda.gov/downloads/Drugs/Guidance/ucm070107.pdf (accessed on 6 March 2019).
- European Medicines Agency (EMA); Committee for Medicinal Products for Human Use (CHMP). Guideline on Bioanalytical Method Validation. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-bioanalytical-method-validation_en.pdf (accessed on 13 May 2019).
- Bald, E.; Sypniewski, S.; Drzewoski, J.; Stępień, M. Application of 2-halopyridinium salts as ultraviolet derivatization reagents and solid-phase extraction for determination of captopril in human plasma by high-performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 1996, 681, 283–289. [Google Scholar] [CrossRef]
- Borowczyk, K.; Olejarz, P.; Kamińska, A.; Głowacki, R.; Chwatko, G. Application of butylamine as a conjugative reagent to on-column derivatization for the determination of antioxidant amino acids in brain tissue, plasma, and urine samples. Int. J. Mol. Sci. 2019, 20, 3340. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Analyte | Linear Range (nmol mL−1) | Regression Equation | R2 | Imprecision (%) | Recovery (%) | ||
|---|---|---|---|---|---|---|---|
| Min. | Max. | Min. | Max. | ||||
| Cys | 40.0–400.0 | y = 3.78x + 16.80 | 0.999 | 0.2 | 6.5 | 99.1 | 100.8 |
| Hcy | 2.0–20.0 | y = 0.24x + 1.76 | 0.999 | 1.5 | 9.7 | 94 | 105.5 |
| GSH | 2.0–20.0 | y = 0.24x + 0.87 | 0.998 | 1.2 | 8.4 | 98.4 | 106.2 |
| Cys-Gly | 2.0–20.0 | y = 0.17x + 2.15 | 0.999 | 0.6 | 13.4 | 100.3 | 105.2 |
| LA | 0.12–5.0 | y = 0.38x + 0.04 | 0.999 | 2.3 | 14.7 | 97.2 | 101.4 |
| Analyte | Concentration (nmol mL −1) | Precision (%) | Accuracy (%) | ||
|---|---|---|---|---|---|
| Intra-day | Inter-day | Intra-day | Inter-day | ||
| Cys | 40 | 11.3 | 3.3 | 100.9 | 113.4 |
| 200 | 2.3 | 4.6 | 99.1 | 98.9 | |
| 400 | 0.5 | 0.5 | 95.3 | 93.8 | |
| Hcy | 2 | 5.8 | 4.2 | 94 | 103 |
| 10 | 5.8 | 0.5 | 105.5 | 100.5 | |
| 20 | 1.7 | 5.3 | 103 | 99.7 | |
| GSH | 2 | 5.6 | 5.6 | 98.4 | 106.2 |
| 10 | 4.2 | 2.9 | 104.1 | 102.2 | |
| 20 | 1.8 | 3.8 | 106.2 | 99 | |
| Cys–Gly | 2 | 6.2 | 4 | 105.2 | 98.6 |
| 10 | 4.2 | 1.1 | 103.6 | 100.3 | |
| 20 | 1.2 | 4 | 99.2 | 100.4 | |
| LA | 0.12 | 4.1 | 9.5 | 101.4 | 101.1 |
| 2.5 | 4 | 9.2 | 97.2 | 99.5 | |
| 5 | 3.6 | 3.7 | 101.1 | 100.2 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borowczyk, K.; Olejarz, P.; Chwatko, G.; Szylberg, M.; Głowacki, R. A Simplified Method for Simultaneous Determination of α-Lipoic Acid and Low-Molecular-Mass Thiols in Human Plasma. Int. J. Mol. Sci. 2020, 21, 1049. https://doi.org/10.3390/ijms21031049
Borowczyk K, Olejarz P, Chwatko G, Szylberg M, Głowacki R. A Simplified Method for Simultaneous Determination of α-Lipoic Acid and Low-Molecular-Mass Thiols in Human Plasma. International Journal of Molecular Sciences. 2020; 21(3):1049. https://doi.org/10.3390/ijms21031049
Chicago/Turabian StyleBorowczyk, Kamila, Patrycja Olejarz, Grażyna Chwatko, Marcin Szylberg, and Rafał Głowacki. 2020. "A Simplified Method for Simultaneous Determination of α-Lipoic Acid and Low-Molecular-Mass Thiols in Human Plasma" International Journal of Molecular Sciences 21, no. 3: 1049. https://doi.org/10.3390/ijms21031049





