Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors
Abstract
:1. Introduction
2. Results
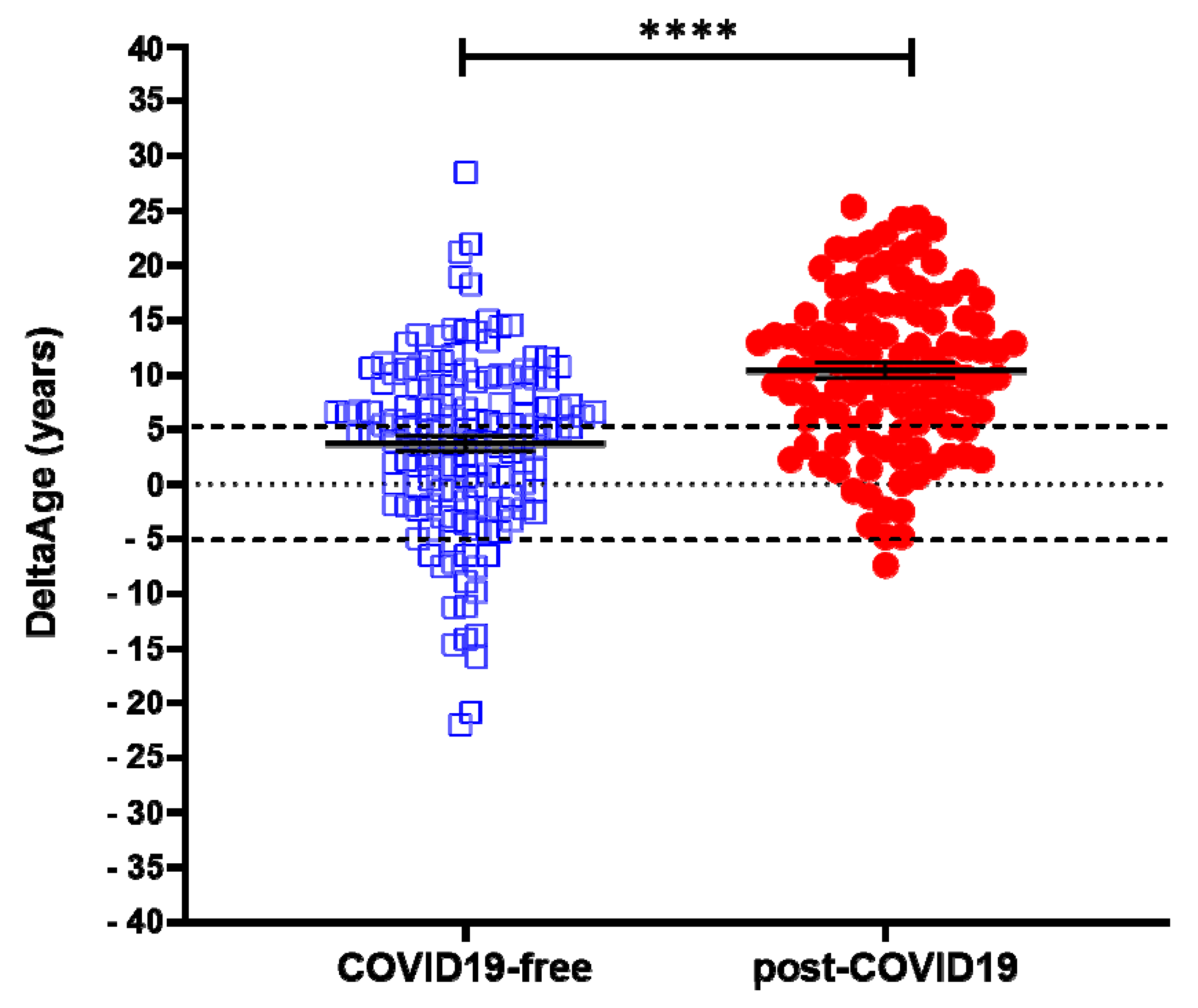
2.1. Evaluation of DNAmAge and DeltaAge in COVID-19 Survivors
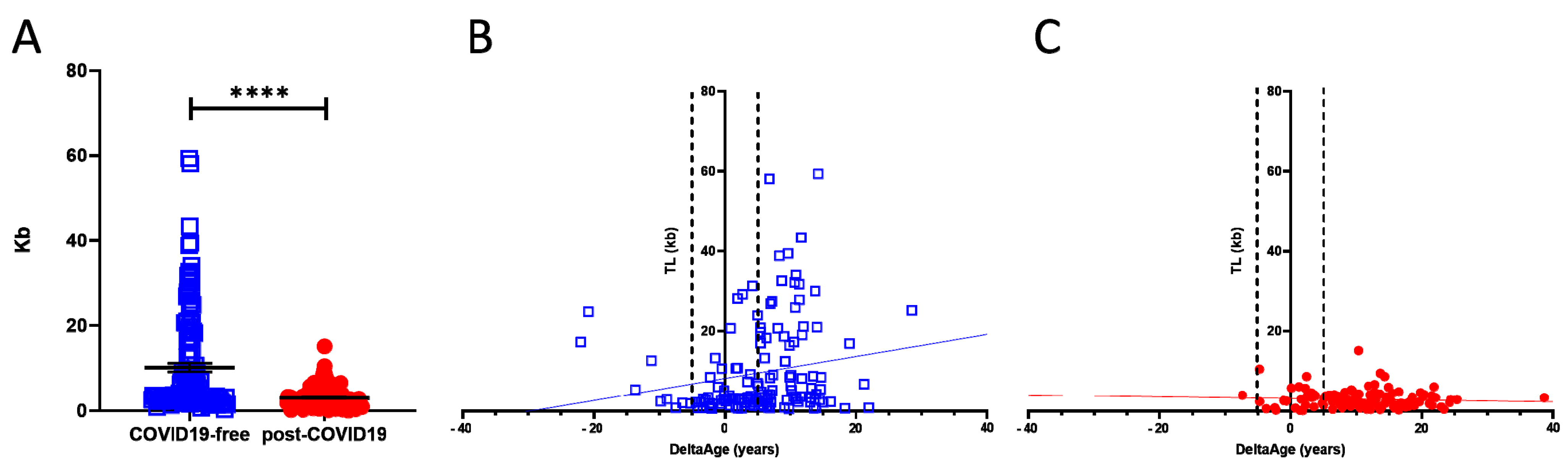
2.2. Telomere Length Quantification
2.3. Peripheral Blood Expression of ACE2 and DPP-4
3. Discussion
4. Materials and Methods
4.1. DNA Extraction from Whole Blood
4.2. Bisulfite Conversion
4.3. Polymerase Chain Reactions for Pyrosequencing
4.4. Pyrosequencing
4.5. DNAmAge Estimation
4.6. Telomere Length Quantification
4.7. RNA Extraction
4.8. cDNA Synthesis and qPCR Real-Time
- Initial denaturation: 95 °C, 5 min;
- Denaturation: 95 °C, 15 s;
- Annealing: 60 °C, 30 s;
- Elongation: 72 °C, 30 s;
- Final elongation: 72 °C, 1 min.
4.9. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ARDS | adult respiratory distress syndrome |
| ACE2 | angiotensin-converting enzyme 2 |
| DNAmAge | biological age based on DNA methylation |
| CVDs | cardiovascular diseases |
| COVID-19 | coronavirus disease 19 |
| CpGs | cytosine–guanine dinucleotides |
| DPP-4 | dipeptidyl-peptidase IV |
| MERS | Middle East respiratory syndrome |
| MERS-CoV | MERS coronavirus |
| PPCS | persistent post-COVID-19 syndrome |
| RAS | renin–angiotensin system |
| SARS | severe acute respiratory syndrome |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| TL | telomere length |
| T2DM | type 2 diabetes mellitus |
References
- Shi, H.; Han, X.; Jiang, N.; Cao, Y.; Alwalid, O.; Gu, J.; Fan, Y.; Zheng, C. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: A descriptive study. Lancet Infect. Dis. 2020, 20, 425–434. [Google Scholar] [CrossRef]
- Bansal, M. Cardiovascular disease and COVID-19. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Aboulhosn, J. The cardiovascular burden of coronavirus disease 2019 (COVID-19) with a focus on congenital heart disease. Int. J. Cardiol. 2020, 309, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, M.; Sitzia, C.; Baryshnikova, E.; Di Dedda, U.; Cardani, R.; Martelli, F.; Romanelli, M.C. Covid-19-Associated Coagulopathy: Biomarkers of Thrombin Generation and Fibrinolysis Leading the Outcome. J. Clin. Med. 2020, 9, 3487. [Google Scholar] [CrossRef]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Passen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–147. [Google Scholar] [CrossRef]
- Walkey, A.; Summer, R.; Ho, V.; Alkana, P. Acute respiratory distress syndrome: Epidemiology and management approaches. Clin. Epidemiol. 2012, 4, 159–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gue, Y.X.; Gorog, D. Reduction in ACE2 may mediate the prothrombotic phenotype in COVID-19. Eur. Heart J. 2020, 41, 3198–3199. [Google Scholar] [CrossRef] [PubMed]
- Li, X.C.; Zhang, J.; Zhuo, J.L. The vasoprotective axes of the renin-angiotensin system: Physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney diseases. Pharmacol. Res. 2017, 125, 21–38. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Mohammadmoradi, S.; Chen, J.Z.; Sawada, H.; Daugherty, A.; Lu, H.S. Renin-Angiotensin System and Cardiovascular Functions. Arterioscler. Thromb. Vasc. Biol. 2018, 38, e108–e116. [Google Scholar] [CrossRef] [Green Version]
- Myall, K.J.; Mukherjee, B.; Castanheira, A.M.; Lam, J.L.; Benedetti, G.; Mak, S.M.; Preston, R.; Thillai, M.; Dewar, A.; Molyneaux, P.L.; et al. Persistent Post-COVID-19 Inflammatory Interstitial Lung Disease: An Observational Study of Corticosteroid Treatment. Ann. Am. Thorac. Soc. 2021, 18, 799–806. [Google Scholar] [CrossRef]
- Sargiacomo, C.; Sotgia, F.; Lisanti, M.P. COVID-19 and chronological aging: Senolytics and other anti-aging drugs for the treatment or prevention of corona virus infection? Aging 2020, 12, 6511–6517. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Shi, X.; Jiang, L.; Zhang, S.; Wang, D.; Tong, P.; Guo, D.; Fu, L.; Cui, Y.; Liu, X.; et al. Structure of MERS-CoV spike receptor-binding domain complexed with human receptor DPP4. Cell Res. 2013, 23, 986–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.M.; Noh, J.H.; Bodogai, M.; Martindale, J.L.; Yang, X.; Indig, F.E.; Basu, S.K.; Ohnuma, K.; Morimoto, C.; Johnson, P.F.; et al. Identification of senescent cell surface targetable protein DPP4. Genes Dev. 2017, 31, 1529–1534. [Google Scholar] [CrossRef] [Green Version]
- Enz, N.; Vliegen, G.; De Meester, I.; Jungraithmayr, W. CD26/DPP4—A potential biomarker and target for cancer therapy. Pharmacol. Ther. 2019, 198, 135–159. [Google Scholar] [CrossRef] [PubMed]
- Suda, M.; Shimizu, I.; Yoshida, Y.; Hayashi, Y.; Ikegami, R.; Katsuumi, G.; Wakasugi, T.; Yoshida, Y.; Okuda, S.; Soga, T.; et al. Inhibition of dipeptidyl peptidase-4 ameliorates cardiac ischemia and systolic dysfunction by up-regulating the FGF-2/EGR-1 pathway. PLoS ONE 2017, 12, e0182422. [Google Scholar] [CrossRef] [Green Version]
- Solerte, S.B.; Di Sabatino, A.; Galli, M.; Fiorina, P. Dipeptidyl peptidase-4 (DPP4) inhibition in COVID-19. Acta Diabetol. 2020, 57, 779–783. [Google Scholar] [CrossRef]
- Hayflick, L.; Moorhead, P. The serial cultivation of human diploid cell strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Zhan, Y.; Hägg, S. Telomere length and cardiovascular disease risk. Curr. Opin. Cardiol. 2019, 34, 270–274. [Google Scholar] [CrossRef]
- Bekaert, S.; De Meyer, T.; Van Oostveldt, P. Telomere attrition as ageing biomarker. Anticancer. Res. 2005, 25, 3011–3021. [Google Scholar]
- Bekaert, B.; Kamalandua, A.; Zapico, S.C.; Van De Voorde, W.; Decorte, R. Improved age determination of blood and teeth samples using a selected set of DNA methylation markers. Epigenetics 2015, 10, 922–930. [Google Scholar] [CrossRef] [Green Version]
- Froidure, A.; Mahieu, M.; Hoton, D.; Laterre, P.-F.; Yombi, J.C.; Koenig, S.; Ghaye, B.; Defour, J.-P.; Decottignies, A. Short telomeres increase the risk of severe COVID-19. Aging 2020, 12, 19911–19922. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S.; Raj, K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat. Rev. Genet. 2018, 19, 371–384. [Google Scholar] [CrossRef]
- Zubakov, D.; Liu, F.; Kokmeijer, I.; Choi, Y.; van Meurs, J.B.; van Ijcken, W.; Uitterlinden, A.G.; Hofman, A.; Broer, L.; van Duijn, C.M.; et al. Human age estimation from blood using mRNA, DNA methylation, DNA rearrangement, and telomere length. Forensic Sci. Int. Genet. 2016, 24, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Zapico, C.S.; Ubelaker, D.H. Applications of physiological bases of ageing to forensic sciences. Estimation of age-at-death. Ageing Res. Rev. 2013, 12, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Jylhävä, J.; Pedersen, N.L.; Hägg, S. Biological Age Predictors. EBioMedicine 2017, 21, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 2013, 14, R115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, S.-E.; Shin, K.-J.; Lee, A.H.Y. DNA methylation-based age prediction from various tissues and body fluids. BMB Rep. 2017, 50, 546–553. [Google Scholar] [CrossRef] [Green Version]
- Vidaki, A.; Kayser, M. Recent progress, methods and perspectives in forensic epigenetics. Forensic Sci. Int. Genet. 2018, 37, 180–195. [Google Scholar] [CrossRef]
- Weidner, C.I.; Lin, Q.; Koch, C.M.; Eisele, L.; Beier, F.; Ziegler, P.; Bauerschlag, D.O.; Jöckel, K.-H.; Erbel, R.; Mühleisen, T.W.; et al. Aging of blood can be tracked by DNA methylation changes at just three CpG sites. Genome Biol. 2014, 15, R24. [Google Scholar] [CrossRef] [Green Version]
- Thong, Z.; Chan, X.L.S.; Tan, J.Y.Y.; Loo, E.S.; Syn, C.K.C. Evaluation of DNA methylation-based age prediction on blood. For. Sci. Int. Gen. Suppl. Ser. 2017, 6, e249–e251. [Google Scholar] [CrossRef] [Green Version]
- Daunay, A.; Baudrin, L.G.; Deleuze, J.-F.; How-Kit, A. Evaluation of six blood-based age prediction models using DNA methylation analysis by pyrosequencing. Sci. Rep. 2019, 9, 8862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Declerck, K.; Berghe, W.V. Back to the future: Epigenetic clock plasticity towards healthy aging. Mech. Aging Dev. 2018, 174, 18–29. [Google Scholar] [CrossRef]
- Horvath, S.; Levine, A.J. HIV-1 Infection Accelerates Age According to the Epigenetic Clock. J. Infect. Dis. 2015, 212, 1563–1573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esteban-Cantos, A.; Rodríguez-Centeno, J.; Barruz, P.; Alejos, B.; Saiz-Medrano, G.; Nevado, J.; Martin, A.; Gayá, F.; De Miguel, R.I.; Bernardino, J.; et al. Epigenetic age acceleration changes 2 years after antiretroviral therapy initiation in adults with HIV: A substudy of the NEAT001/ANRS143 randomised trial. Lancet HIV 2021, 8, e197–e205. [Google Scholar] [CrossRef]
- Levine, A.J.; Quach, A.; Moore, D.J.; Achim, C.L.; Soontornniyomkij, V.; Masliah, E.; Singer, E.J.; Gelman, B.; Nemanim, N.; Horvath, S. Accelerated epigenetic aging in brain is associated with pre-mortem HIV-associated neurocognitive disorders. J. Neuro Virol. 2016, 22, 366–375. [Google Scholar] [CrossRef] [Green Version]
- Kananen, L.; Nevalainen, T.; Jylhävä, J.; Marttila, S.; Hervonen, A.; Jylhä, M.; Hurme, M. Cytomegalovirus infection accelerates epigenetic aging. Exp. Gerontol. 2015, 72, 227–229. [Google Scholar] [CrossRef]
- Oronsky, B.; Larson, C.; Hammond, T.C.; Oronsky, A.; Kesari, S.; Lybeck, M.; Reid, T.R. A Review of Persistent Post-COVID Syndrome (PPCS). Clin. Rev. Allergy Immunol. 2021, 1–9. [Google Scholar] [CrossRef]
- Walitt, B.; Bartrum, E. A clinical primer for the expected and potential post-COVID-19 syndromes. Pain Rep. 2021, 6, 1–7. [Google Scholar]
- Sabry, D.; Ahmed, R.; Abdalla, S.; Fathy, W.; Eldemery, A.; Elamir, A. Braf, Kras and Helicobacter pylori epigenetic changes-associated chronic gastritis in Egyptian patients with and without gastric cancer. World J. Microbiol. Biotechnol. 2016, 32, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hannum, G.; Guinney, J.; Zhao, L.; Zhang, L.; Hughes, G.; Sadda, S.; Klotzle, B.; Bibikova, M.; Fan, J.-B.; Gao, Y.; et al. Genome-wide Methylation Profiles Reveal Quantitative Views of Human Aging Rates. Mol. Cell 2013, 49, 359–367. [Google Scholar] [CrossRef] [Green Version]
- Bonkowski, M.S.; Sinclair, D.A. Slowing ageing by design: The rise of NAD (+) and sirtuin-activating compounds. Nat. Rev. Mol. Cell Biol. 2016, 17, 679–690. [Google Scholar] [CrossRef]
- Shahmirzadi, A.A.; Edgar, D.; Liao, C.-Y.; Hsu, Y.-M.; Lucanic, M.; Wiley, C.D.; Gan, G.; Kim, D.E.; Kasler, H.G.; Kuehnemann, C.; et al. Alpha-Ketoglutarate, an Endogenous Metabolite, Extends Lifespan and Compresses Morbidity in Aging Mice. Cell Metab. 2020, 32, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; D’Alessio, A.C.; Taranova, O.V.; Hong, K.; Sowers, L.C.; Zhang, Y. Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nat. Cell Biol. 2010, 466, 1129–1133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atlante, S.; Mongelli, A.; Barbi, V.; Martelli, F.; Farsetti, A.; Gaetano, C. The epigenetic implication in coronavirus infection and therapy. Clin. Epigenet. 2020, 12, 1–12. [Google Scholar] [CrossRef]
- Sang, E.; Tian, Y.; Miller, L.; Sang, Y. Epigenetic Evolution of ACE2 and IL-6 Genes: Non-Canonical Interferon-Stimulated Genes Correlate to COVID-19 Susceptibility in Vertebrates. Genes 2021, 12, 154. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ma, F.; Yokota, T.; Garcia, G.; Palermo, A.; Wang, Y.; Farrell, C.; Wang, Y.C.; Wu, R.; Zhou, Z.; et al. Metabolic reprogramming and epigenetic changes of vital organs in SARS-CoV-2–induced systemic toxicity. JCI Insight. 2021, 6, 1–18. [Google Scholar] [CrossRef]
- Castro de Moura, M.; Davalos, V.; Planas-Serra, L.; Alvarez-Errico, D.; Arribas, C.; Ruiz, M.; Aguilera-Albesa, S.; Troya, J.; Valencia-Ramos, J.; Vélez-Santamaria, V.; et al. Epigenome-wide association study of COVID-19 severity with respiratory failure. EBioMedicine 2021. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Carroll, L.; Joglekar, M.V.; Januszewski, A.S.; Wong, K.K.; Hardikar, A.; Jenkins, A.J.; Ma, R.C.W. Diabetes, metabolic disease, and telomere length. Lancet Diabetes Endocrinol. 2021, 9, 117–126. [Google Scholar] [CrossRef]
- Sanchez-Vazquez, R.; Guío-Carrión, A.; Zapatero-Gaviria, A.; Martínez, P.; Blasco, M.A. Shorter telomere lengths in patients with severe COVID-19 disease. Aging 2021, 13, 1–15. [Google Scholar] [CrossRef]
- Amendola, A.; Garoffolo, G.; Songia, P.; Nardacci, R.; Ferrari, S.; Bernava, G.; Canzano, P.; Myasoedova, V.; Colavita, F.; Castilletti, C.; et al. Human cardiosphere-derived stromal cells exposed to SARS-CoV-2 evolve into hyper-inflammatory/pro-fibrotic phenotype and produce infective viral particles depending on the levels of ACE2 receptor expression. Cardiovasc Res. 2021, 117, 1557–1566. [Google Scholar] [CrossRef]



| Clinical Data | COVID-19-Free | Post-COVID-19 |
|---|---|---|
| Samples (n) | 144 (Male 66.0%; Female 34.0%) | 117 (Male 60.7%; Female 39.3%) |
| BMI ≥ 30 | 9.0% | 15.3% |
| Smokers | 37.5% | 16.9% |
| Diabetics | 11.1% | 12.1% |
| Hypertension | 40.3% | 36.3% |
| Clinical history of CVDs | 33.3% | 27.4% |
| Antecedent lung involvement | 1.6% | 20.2% |
| COVID-19-related complications | ||
| Pneumonia | / | 57.3% |
| Oxygen therapy | / | 52.4% |
| Artificial ventilation | / | 35.5% |
| Length of viral positivity (average) in weeks | / | 4.84 |
| COVID-19-Free | Post-COVID-19 | p-Value | |
|---|---|---|---|
| Samples (n) | 144 (Male 66.0%; Female 34.0%) | 117 (Male 60.7%; Female 39.3%) | |
| Chronological age (years) | 62.48 ± 9.04 | 58.44 ± 14.66 | Ns |
| Biological age (years) | 63.81 ± 13.66 | 67.18 ± 10.86 | Ns |
| Chronological vs. biological (p-value) | Ns | <0.0001 | |
| DeltaAge (years) Ratio | 3.68 ± 8.17 1 | 10.45 ± 7.29 2.84 | <0.0001 |
| DeltaAge distribution | |||
| Decelerated (%) | 12.8 | 0.9 | |
| Normal (%) | 39.0 | 22.5 | |
| Accelerated (%) | 48.2 | 76.6 | |
| Telomere length (kb) | 10.67 ± 11.69 | 3.03 ± 2.39 | <0.0001 |
| ACE2 expression (2^(-dct)) | 0.001390 ± 0.002298 | 0.0003801 ± 0.0004463 | <0.0001 |
| DPP-4 expression (2^(-dct)) | 0.1038 ± 0.089 | 0.1152 ± 0.069 | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mongelli, A.; Barbi, V.; Gottardi Zamperla, M.; Atlante, S.; Forleo, L.; Nesta, M.; Massetti, M.; Pontecorvi, A.; Nanni, S.; Farsetti, A.; et al. Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors. Int. J. Mol. Sci. 2021, 22, 6151. https://doi.org/10.3390/ijms22116151
Mongelli A, Barbi V, Gottardi Zamperla M, Atlante S, Forleo L, Nesta M, Massetti M, Pontecorvi A, Nanni S, Farsetti A, et al. Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors. International Journal of Molecular Sciences. 2021; 22(11):6151. https://doi.org/10.3390/ijms22116151
Chicago/Turabian StyleMongelli, Alessia, Veronica Barbi, Michela Gottardi Zamperla, Sandra Atlante, Luana Forleo, Marialisa Nesta, Massimo Massetti, Alfredo Pontecorvi, Simona Nanni, Antonella Farsetti, and et al. 2021. "Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors" International Journal of Molecular Sciences 22, no. 11: 6151. https://doi.org/10.3390/ijms22116151
APA StyleMongelli, A., Barbi, V., Gottardi Zamperla, M., Atlante, S., Forleo, L., Nesta, M., Massetti, M., Pontecorvi, A., Nanni, S., Farsetti, A., Catalano, O., Bussotti, M., Dalla Vecchia, L. A., Bachetti, T., Martelli, F., La Rovere, M. T., & Gaetano, C. (2021). Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors. International Journal of Molecular Sciences, 22(11), 6151. https://doi.org/10.3390/ijms22116151









