Constructing of Bacillus subtilis-Based Lux-Biosensors with the Use of Stress-Inducible Promoters
Abstract
:1. Introduction
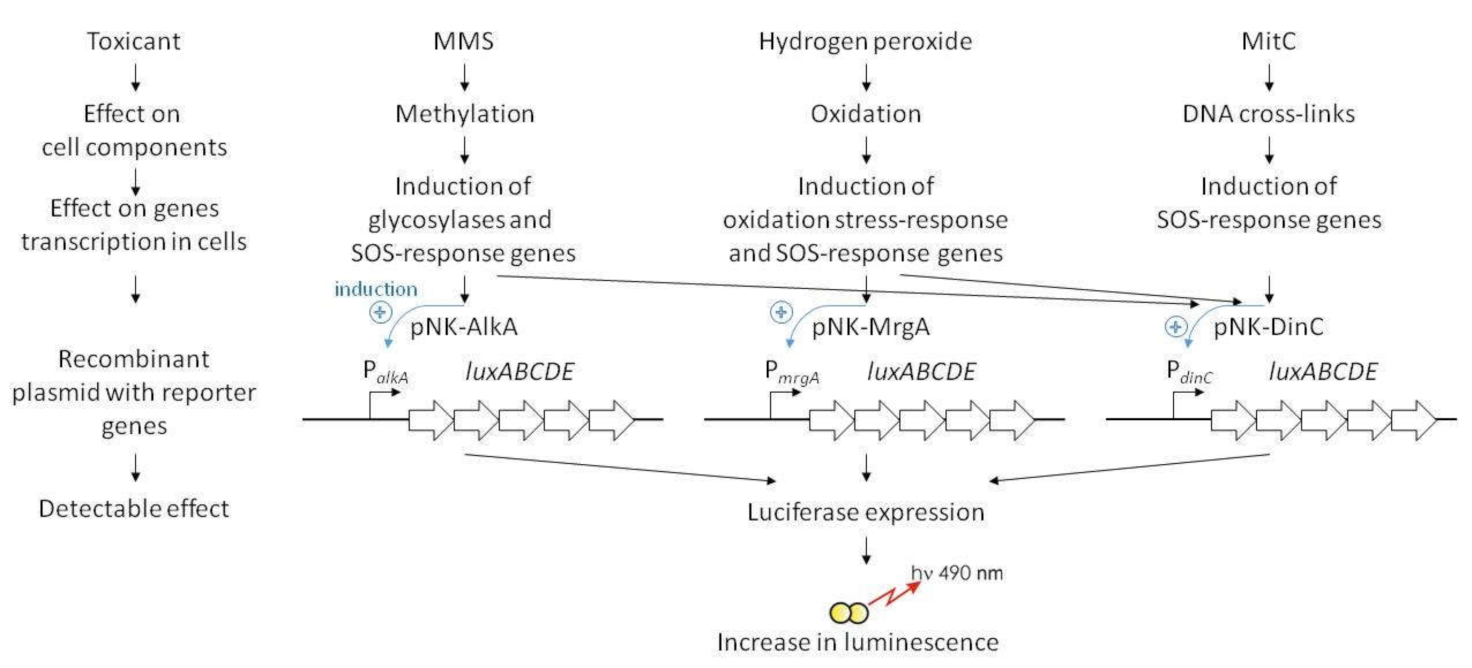
2. Results
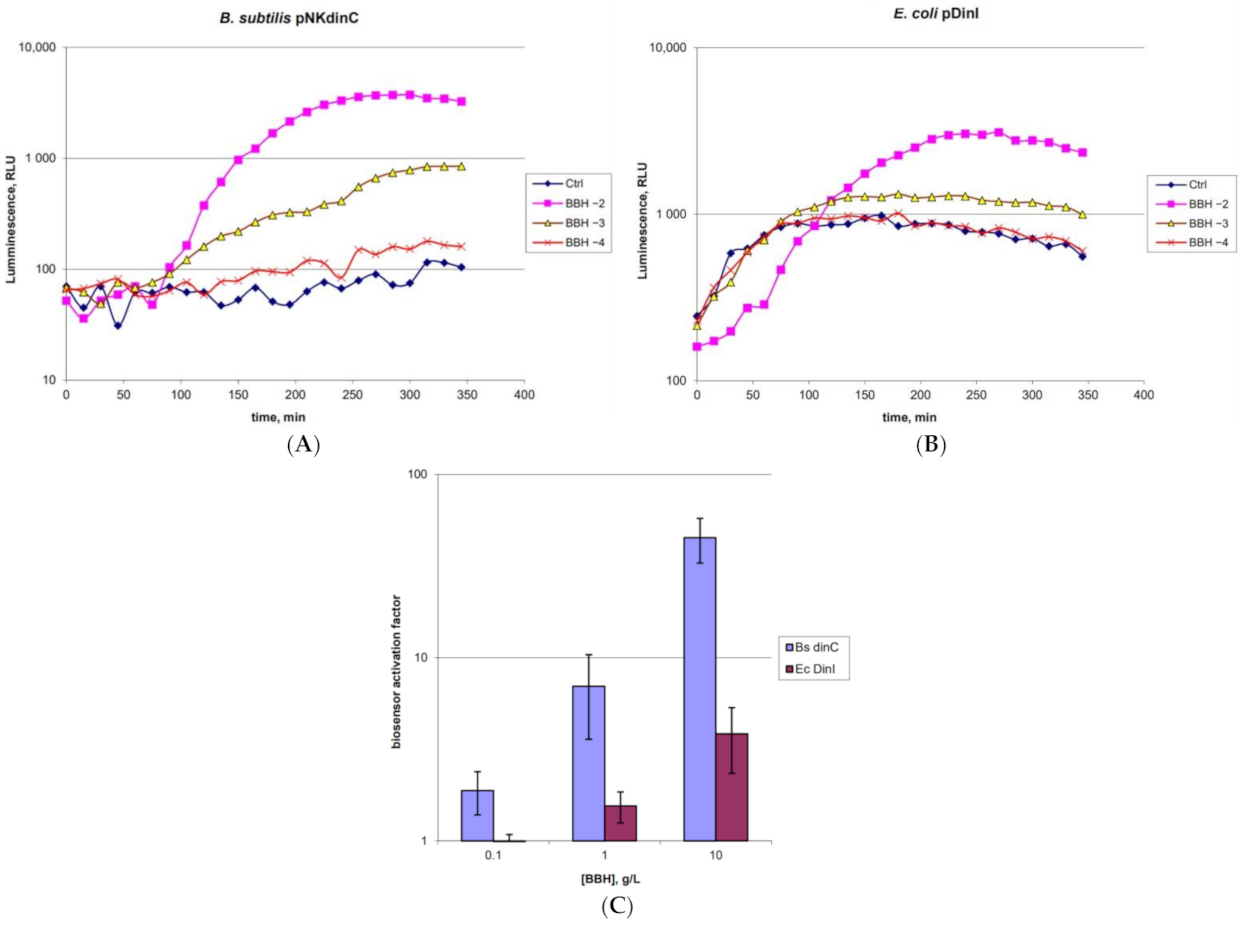
2.1. Characterization of the B. subtilis 168 pNK-DinC Lux-Biosensor for SOS-Response Detection
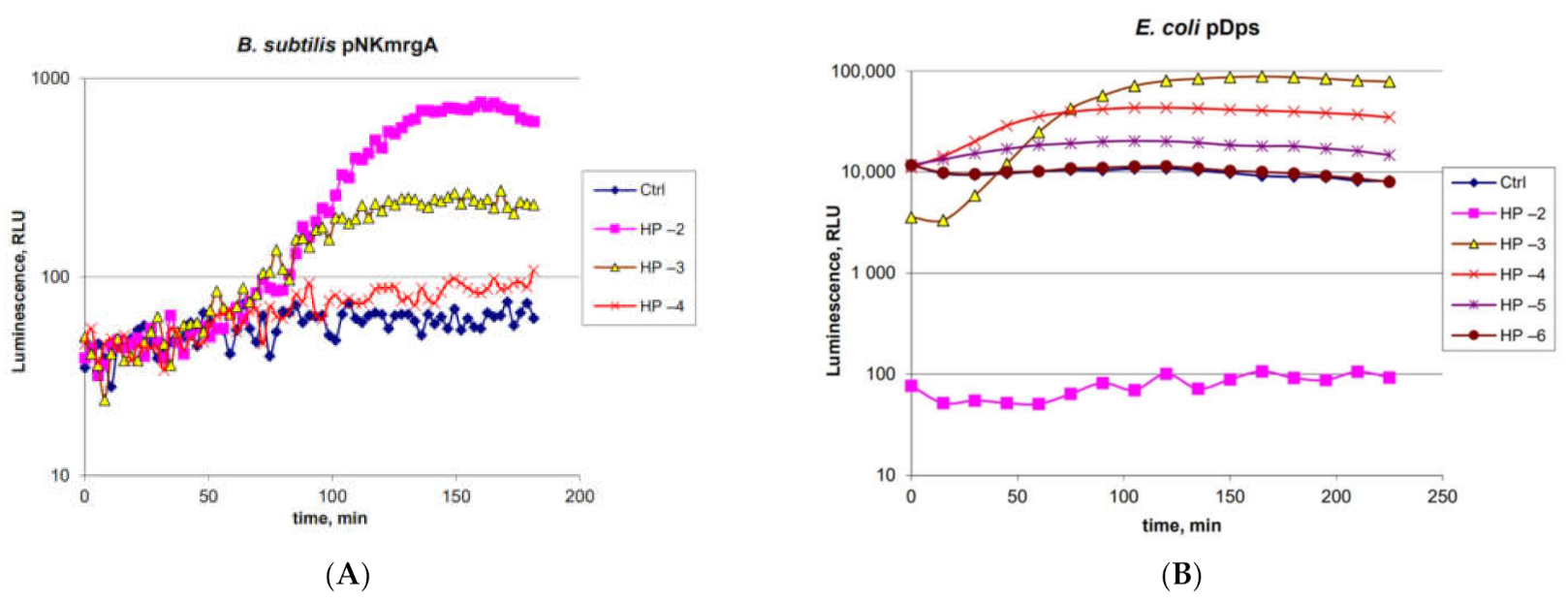
2.2. Characterization of the B. subtilis 168 pNK-MrgA Lux-Biosensor for Oxidative Stress Detection
2.3. Characterization of the B. subtilis 168 pNK-AlkA Lux-Biosensor for DNA Alkylation Detection
2.4. Main Characteristics of the Obtained Biosensors
2.5. Application of the Obtained Lux-Biosensors for Assessment of BBH Toxicity against Gram-Positive Bacteria
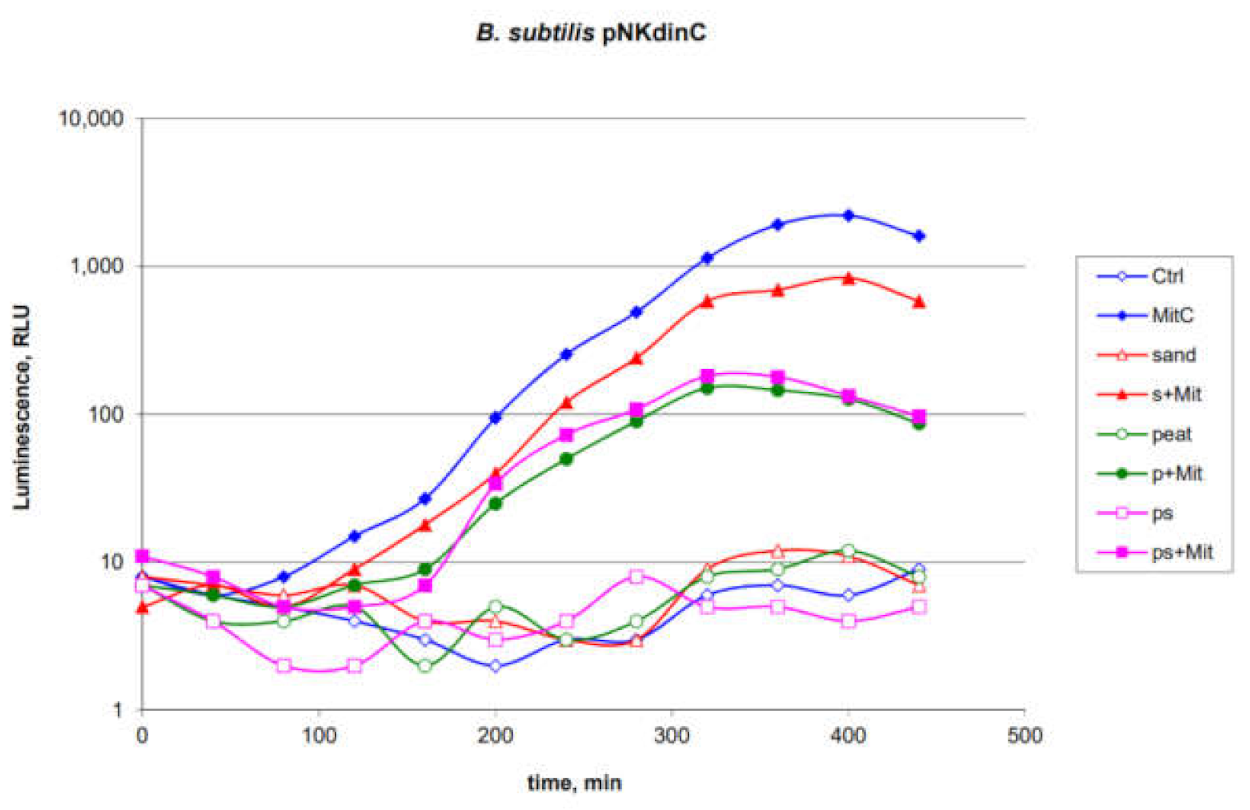
2.6. Application of the B. subtilis 168 pNKdinC Lux-Biosensor for Detection of Toxicants Introduced into the Soils
3. Discussion
4. Materials and Methods
4.1. Strains and Plasmids
4.2. Enzymes and DNA Manipulation
4.3. Chemicals
4.4. Constructing of Biosensor Plasmids
4.5. Culture Medium and Growth Conditions
4.6. Measurement of Bioluminescence
4.7. Data Processing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Danilov, V.S.; Ismailov, A.D. Bacterial luciferase as a biosensor of biologically active compounds. Biotechnology 1989, 11, 39–78. [Google Scholar] [PubMed]
- Deryabin, D.G.; Efremova, L.V.; Karimov, I.F.; Manukhov, I.V.; Gnuchikh, E.Y.; Miroshnikov, S.A. Comparative sensitivity of the luminescent Photobacterium phosphoreum, Escherichia coli, and Bacillus subtilis strains to toxic effects of carbon-based nanomaterials and metal nanoparticles. Microbiology 2016, 85, 198–206. [Google Scholar] [CrossRef]
- Gnuchikh, E.; Baranova, A.; Schukina, V.; Khaliullin, I.; Zavilgelsky, G.; Manukhov, I. Kinetics of the thermal inactivation and the refolding of bacterial luciferases in Bacillus subtilis and in Escherichia coli differ. PLoS ONE 2019, 14, e0226576. [Google Scholar] [CrossRef] [Green Version]
- Van Dyk, T.K.; Majarian, W.R.; Konstantinov, K.B.; Young, R.M.; Dhurjati, P.S.; LaRossa, R.A. Rapid and sensitive pollutant detection by induction of heat shock gene- bioluminescence gene fusions. Appl. Environ. Microbiol. 1994, 60, 1414–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Dyk, T.K.; Rosson, R.A. Photorhabdus luminescens luxCDABE promoter probe vectors. Methods Mol. Biol. 1998, 102, 85–95. [Google Scholar] [CrossRef]
- Vollmer, A.C.; Belkin, S.; Smulski, D.R.; Van Dyk, T.K.; Larossa, R.A. Detection of DNA damage by use of Escherichia coli carrying recA::lux, uvrA::lux, or alkA::lux reporter plasmids. Appl. Environ. Microbiol. 1997, 63, 2566–2571. [Google Scholar] [CrossRef] [Green Version]
- Zavilgelsky, G.B.; Kotova, V.Y.; Manukhov, I.V. Titanium dioxide (TiO2) nanoparticles induce bacterial stress response detectable by specific lux biosensors. Nanotechnol. Russ. 2011, 6, 401–406. [Google Scholar] [CrossRef]
- Kurittu, J.; Lonnberg, S.; Virta, M.; Karp, M. Qualitative detection of tetracycline residues in milk with a luminescence-based microbial method: The effect of milk composition and assay performance in relation to an immunoassay and a microbial inhibition assay. J. Food Prot. 2000, 63, 953–957. [Google Scholar] [CrossRef]
- Igonina, E.V.; Marsova, M.V.; Abilev, S.K. Lux biosensors: Screening biologically active compounds for genotoxicity. Russ. J. Genet. Appl. Res. 2018, 8, 87–95. [Google Scholar] [CrossRef]
- Zavilgelsky, G.B.; Kotova, V.Y.; Manukhov, I.V. Action of 1,1-dimethylhydrazine on bacterial cells is determined by hydrogen peroxide. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2007, 634, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Goryanin, I.; Kotova, V.; Krasnopeeva, E.; Manukhov, I.; Chubukov, P.; Balabanov, V.; Chalkin, S.; Shatrov, T.; Zavilgelsky, G. Genotoxic action of the 1,1-dimethylhydrazine determined by alkylating compounds appearing in the result of oxidation and hydrogen peroxide. Tr. MIPT 2013, 5, 103–111. [Google Scholar]
- Khrulnova, S.A.; Baranova, A.; Bazhenov, S.V.; Goryanin, I.I.; Konopleva, M.N.; Maryshev, I.V.; Salykhova, A.I.; Vasilyeva, A.V.; Manukhov, I.V.; Zavilgelsky, G.B. Lux-operon of the marine psychrophilic bacterium Aliivibrio logei: A comparative analysis of the LuxR1/LuxR2 regulatory activity in Escherichia coli cells. Microbiology 2016, 162, 717–724. [Google Scholar] [CrossRef]
- Bazhenov, S.; Novoyatlova, U.; Scheglova, E.; Fomin, V.; Khrulnova, S.; Melkina, O.; Chistyakov, V.; Manukhov, I. Influence of the luxR regulatory gene dosage and expression level on the sensitivity of the whole-cell biosensor to acyl-homoserine lactone. Biosensors 2021, 11, 166. [Google Scholar] [CrossRef] [PubMed]
- Manukhov, I.; Balabanov, V.; Kotova, V.; Khrulnova, S.; Melkina, O.; Kraynov, A.; Pustovoit, K.; Krechetov, P.; Koroleva, T.; Shatrov, T.; et al. Use of Lux biosensors for detection of UDMH in soil. Dual Technol. Russ. 2008, 44, 50–56. [Google Scholar]
- Deryabin, D.G.; Karimov, I.F.; Manukhov, I.V.; Tolmacheva, N.A.; Balabanov, V.P. Differential analysis of bactericidal systems of blood serum with recombinant luminescent Escherichia coli and Bacillus subtilis strains. Bull. Exp. Biol. Med. 2012, 154, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Francis, K.P.; Yu, J.; Bellinger-Kawahara, C.; Joh, D.; Hawkinson, M.J.; Xiao, G.; Purchio, T.F.; Caparon, M.G.; Lipsitch, M.; Contag, P.R. Visualizing Pneumococcal Infections in the Lungs of Live Mice Using Bioluminescent Streptococcus pneumoniae Transformed with a Novel Gram-Positive lux Transposon. Infect. Immun. 2001, 69, 3350. [Google Scholar] [CrossRef] [Green Version]
- Au, N.; Kuester-Schoeck, E.; Mandava, V.; Bothwell, L.E.; Canny, S.P.; Chachu, K.; Colavito, S.A.; Fuller, S.N.; Groban, E.S.; Hensley, L.A.; et al. Genetic composition of the Bacillus subtilis SOS system. J. Bacteriol. 2005, 187, 7655. [Google Scholar] [CrossRef] [Green Version]
- Cheo, D.L.; Bayles, K.W.; Yasbin, R.E. Cloning and characterization of DNA damage-inducible promoter regions from Bacillus subtilis. J. Bacteriol. 1991, 173, 1696–1703. [Google Scholar] [CrossRef] [Green Version]
- Morohoshi, F.; Hayashi, K.; Munakata, N. Bacillus subtilis alkA gene encoding inducible 3-methyladenine DNA glycosylase is adjacent to the ada operon. J. Bacteriol. 1993, 175, 6010–6017. [Google Scholar] [CrossRef] [Green Version]
- Pomposiello, P.J.; Demple, B. Redox-operated genetic switches: The SoxR and OxyR transcription factors. Trends Biotechnol. 2001, 19, 109–114. [Google Scholar] [CrossRef]
- Zheng, M.; Storz, G. Redox sensing by prokaryotic transcription factors. Biochem. Pharmacol. 2000, 59, 1–6. [Google Scholar] [CrossRef]
- Kotova, V.Y.; Manukhov, I.V.; Zavilgelskii, G.B. Lux-biosensors for detection of SOS-response, heat shock, and oxidative stress. Appl. Biochem. Microbiol. 2010, 46, 781–788. [Google Scholar] [CrossRef]
- Mostertz, J.; Scharf, C.; Hecker, M.; Homuth, G. Transcriptome and proteome analysis of Bacillus subtilis gene expression in response to superoxide and peroxide stress. Microbiology 2004, 150, 497–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helmann, J.D.; Wu, M.F.W.; Gaballa, A.; Kobel, P.A.; Morshedi, M.M.; Fawcett, P.; Paddon, C. The global transcriptional response of Bacillus subtilis to peroxide stress is coordinated by three transcription factors. J. Bacteriol. 2003, 185, 243. [Google Scholar] [CrossRef] [Green Version]
- Antelmann, H.; Engelmann, S.; Schmid, R.; Hecker, M. General and oxidative stress responses in Bacillus subtilis: Cloning, expression, and mutation of the alkyl hydroperoxide reductase operon. J. Bacteriol. 1996, 178, 6571–6578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, K.; Ohsawa, T.; Kobayashi, K.; Ogasawara, N.; Ogura, M. The H2O2 stress-responsive regulator PerR positively regulates srfA expression in Bacillus subtilis. J. Bacteriol. 2005, 187, 6659–6667. [Google Scholar] [CrossRef] [Green Version]
- Albert Vericat, J.; Barbé, J.; Guerrero, R. Expression of the SOS response following simultaneous treatment with methyl-nitrosoguanidine and mitomycin C in Escherichia coli. Mutat. Res. Repair Rep. 1984, 132, 15–20. [Google Scholar] [CrossRef]
- Abilev, S.K.; Kotova, V.Y.; Smirnova, S.V.; Shapiro, T.N.; Zavilgelsky, G.B. Specific Lux Biosensors of Escherichia coli Containing pRecA::lux, pColD::lux, and pDinI::lux Plasmids for Detection of Genotoxic Agents. Russ. J. Genet. 2020, 56, 666–673. [Google Scholar] [CrossRef]
- Chen, L.; Helmann, J.D. Bacillus subtilis MrgA is a Dps(PexB) homologue: Evidence for metalloregulation of an oxidative-stress gene. Mol. Microbiol. 1995, 18, 295–300. [Google Scholar] [CrossRef]
- Nakabeppu, Y.; Miyata, T.; Kondo, H.; Iwanaga, S.; Sekiguchi, M. Structure and expression of the alkA gene of Escherichia coli involved in adaptive response to alkylating agents. J. Biol. Chem. 1984, 259, 13730–13736. [Google Scholar] [CrossRef]
- Goerlich, O.; Quillardet, P.; Hofnung, M. Induction of the SOS response by hydrogen peroxide in various Escherichia coli mutants with altered protection against oxidative DNA damage. J. Bacteriol. 1989, 171, 6141. [Google Scholar] [CrossRef] [Green Version]
- Kessenikh, A.; Gnuchikh, E.; Bazhenov, S.; Bermeshev, M.; Pevgov, V.; Samoilov, V.; Shorunov, S.; Maksimov, A.; Yaguzhinsky, L.; Manukhov, I. Genotoxic effect of 2,2′-bis(bicyclo[2.2.1] heptane) on bacterial cells. PLoS ONE 2020, 15, e0228525. [Google Scholar] [CrossRef]
- Morozova, R.M.; Fedorets, N.G.; Bahmet, O.N. Soils and soil cover of Karelian Zaonezhje. Tr. Karel. Nauchnogo Cent. Ross. Akad. Nauk 2004, 6, 69–89. [Google Scholar]
- Chistyakov, V.A.; Prazdnova, E.V.; Mazanko, M.S.; Bren, A.B. The use of biosensors to explore the potential of probiotic strains to reduce the SOS response and mutagenesis in bacteria. Biosensors 2018, 8, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jing, W.; Liu, Q.; Wang, M.; Zhang, X.; Chen, J.; Sui, G.; Wang, L. A method for particulate matter 2.5 (PM2.5) biotoxicity assay using luminescent bacterium. Ecotoxicol. Environ. Saf. 2019, 170, 796–803. [Google Scholar] [CrossRef]
- Manukhov, I.; Gorbunov, M.; Degtev, D.; Zavilgelsky, G.; Kessenikh, A.; Konopleva, M.; Kotova, V.; Krasnopeeva, E.; Motovilov, K.; Osetrova, M.; et al. A Set of Lux Biosensors for Determining the Genotoxic Products of Incomplete Oxidation of Asymmetric Dimethylhydrazine in a Medium. Patent of Russia No 2,569,156, 20 October 2015. [Google Scholar]
- Kessenikh, A.G.; Manukhov, I.V.; Yaguzhinsky, L.S.; Bermeshev, M.V.; Zisman, M.A.; Pevgov, V.G.; Samoilov, V.O.; Shorunov, S.V.; Maksimov, A.L. Toxic effect of 2-ethy l(bicyclo[2.2.1] heptane) on bacterial cells. Biotekhnologiya 2019, 35, 67–72. [Google Scholar] [CrossRef]
- Gnuchikh, E.Y.; Manukhov, I.V.; Zavilgelsky, G. Biosensors for the determination of promoters and chaperones activity in Bacillus subtilis cells. Biotekhnologiya 2020, 36, 68–77. [Google Scholar] [CrossRef]
- Melkina, O.E.; Goryanin, I.I.; Zavilgelsky, G.B. The DNA–mimic antirestriction proteins ArdA ColIB-P9, Arn T4, and Ocr T7 as activators of H-NS-dependent gene transcription. Microbiol. Res. 2016, 192, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Green, M.R.; Sambrook, J. Molecular Cloning: A Laboratory Manual, 4th ed.; Cold Spring Harbor Laboratory Press: Berlin/Heidelberg, Germany, 2012; ISBN 978-1-936113-41-5. [Google Scholar]
- Gibson, D.G.; Young, L.; Chuang, R.Y.; Venter, J.C.; Hutchison, C.A.; Smith, H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 2009, 6, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Zhang, X.; Zhong, L.; Lu, Z. A modified electro-transformation method for Bacillus subtilis and its application in the production of antimicrobial lipopeptides. Biotechnol. Lett. 2011, 33, 1047–1051. [Google Scholar] [CrossRef]
- Spizizen, J. Transformation of biochemically deficient strains of Bacillus subtilis by deoxyribonucleate. Proc. Natl. Acad. Sci. USA 1958, 44, 1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Toxicant | Biosensor | E. coli pAlkA-Lux | B. subtilis pNK-AlkA | E. coli pDps | B. subtilis pNK-MrgA | E. coli pDinI | B. subtilis pNK-DinC | |
|---|---|---|---|---|---|---|---|---|
| Characteristic | ||||||||
| MitC | Threshold concentration (LOD), M | n/a | n/a | n/a | n/a | (5 ± 3) × 10−9 | (1 ± 0.4) × 10−8 | |
| Induction amplitude | n/a | n/a | n/a | n/a | 21 ± 9 | 45 ± 16 | ||
| Dynamic range, log10 (Cmax/Cmin) | n/a | n/a | n/a | n/a | 4 ± 0.3 | 4 ± 0.3 | ||
| Induction start time, min | n/a | n/a | n/a | n/a | 29 ± 8 | 60 ± 20 | ||
| H2O2 | Threshold concentration (LOD), M | n/a | n/a | (1 ± 0.5) × 10−5 | (1.3 ± 0.7) × 10−4 | (3.0 ± 1.6) × 10−4 | (3.4 ± 2.7) × 10−4 | |
| Induction amplitude | n/a | n/a | 15 ± 4 | 14 ± 3 | 8 ± 5 | 4.2 ± 2.5 | ||
| Dynamic range, log10 (Cmax/Cmin) | n/a | n/a | 2.3 ± 0.3 | 2.3 ± 0.6 | 1.2 ± 0.3 | 1.2 ± 0.3 | ||
| Induction start time, min | n/a | n/a | 15 ± 5 | 60 ± 12 | 18 ± 7 | 64 ± 19 | ||
| MMS | Threshold concentration (LOD), M | (5 ± 3) × 10−4 | (1.1 ± 0.5) × 10−4 | n/a | n/a | (2.0 ± 1.2) × 10−4 | (1.5 ± 0.6) × 10−4 | |
| Induction amplitude | 12 ± 7 | 8 ± 4 | n/a | n/a | 3.2 ± 0.8 | 2.5 ± 1.0 | ||
| Dynamic range, log10 (Cmax/Cmin) | 2.0 ± 0.3 | 1.2 ± 0.4 | n/a | n/a | 1.5 ± 0.3 | 2.2 ± 0.4 | ||
| Induction start time, min | 60 ± 15 | 108 ± 24 | n/a | n/a | 82 ± 20 | 95 ± 24 | ||
| Name | Description | Source |
|---|---|---|
| Bacterial strains | ||
| E. coli K12 MC1061 | F–D(araA-leu)7697 [araD139]B/r ∆(codB-lacI)3 galK16 galE15(GalS) λ–e14- mcrA0 relA1 rpsL150 spoT1 mcrB1 hsdR2 | VKPM (Moscow, Russia) |
| E. coli K12 MG1655 | F- ilvG rfb-50 rph-1 | VKPM (Moscow, Russia) |
| B. subtilis 168 | trpC2 | VKPM (Moscow, Russia) |
| Plasmids | ||
| pPL_ABCDExen | Promoterless shuttle vector with the luxABCDE genes from Photorhabdus luminescens. The order of genes in the lux-operon and RBS upstream of each gene are optimized for B. subtilis expression. Two replication origins (from pMW118 and pBS72). Resistance to trimethoprim (Tpr), chloramphenicol (Cmr), and ampicillin (Apr). | [38] |
| pNK-AlkA | pPL_ABCDExen vector with insertion of the B. subtilis PalkA promoter; PalkA is transcriptionally fused to luxCDABE P. luminescens | This study |
| pNK-DinC | pPL_ABCDExen vector with insertion of the B. subtilis PalkA promoter; PdinC is transcriptionally fused to luxCDABE P. luminescens | This study |
| pNK-MrgA | pPL_ABCDExen vector with insertion of the B. subtilis PalkA promoter; PmrgA is transcriptionally fused to luxCDABE P. luminescens | This study |
| pDps | The E. coli Pdps promoter was cloned into pDEW201 [5] vector and transcriptionally fused to the reporter genes luxCDABE P. luminescens. Apr | [39] |
| pDinI | The E. coli PdinI promoter was cloned into pDEW201 [5] vector and transcriptionally fused to the reporter genes luxCDABE P. luminescens. Apr | [28] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kessenikh, A.G.; Novoyatlova, U.S.; Bazhenov, S.V.; Stepanova, E.A.; Khrulnova, S.A.; Gnuchikh, E.Y.; Kotova, V.Y.; Kudryavtseva, A.A.; Bermeshev, M.V.; Manukhov, I.V. Constructing of Bacillus subtilis-Based Lux-Biosensors with the Use of Stress-Inducible Promoters. Int. J. Mol. Sci. 2021, 22, 9571. https://doi.org/10.3390/ijms22179571
Kessenikh AG, Novoyatlova US, Bazhenov SV, Stepanova EA, Khrulnova SA, Gnuchikh EY, Kotova VY, Kudryavtseva AA, Bermeshev MV, Manukhov IV. Constructing of Bacillus subtilis-Based Lux-Biosensors with the Use of Stress-Inducible Promoters. International Journal of Molecular Sciences. 2021; 22(17):9571. https://doi.org/10.3390/ijms22179571
Chicago/Turabian StyleKessenikh, Andrew G., Uliana S. Novoyatlova, Sergey V. Bazhenov, Eugeniya A. Stepanova, Svetlana A. Khrulnova, Eugeny Yu. Gnuchikh, Vera Yu. Kotova, Anna A. Kudryavtseva, Maxim V. Bermeshev, and Ilya V. Manukhov. 2021. "Constructing of Bacillus subtilis-Based Lux-Biosensors with the Use of Stress-Inducible Promoters" International Journal of Molecular Sciences 22, no. 17: 9571. https://doi.org/10.3390/ijms22179571






