Spontaneous Transformation of Biomedical Polymeric Silver Salt into a Nanocomposite: Physical–Chemical and Antimicrobial Properties Dramatically Depend on the Initial Preparation State
Abstract
:1. Introduction
2. Results
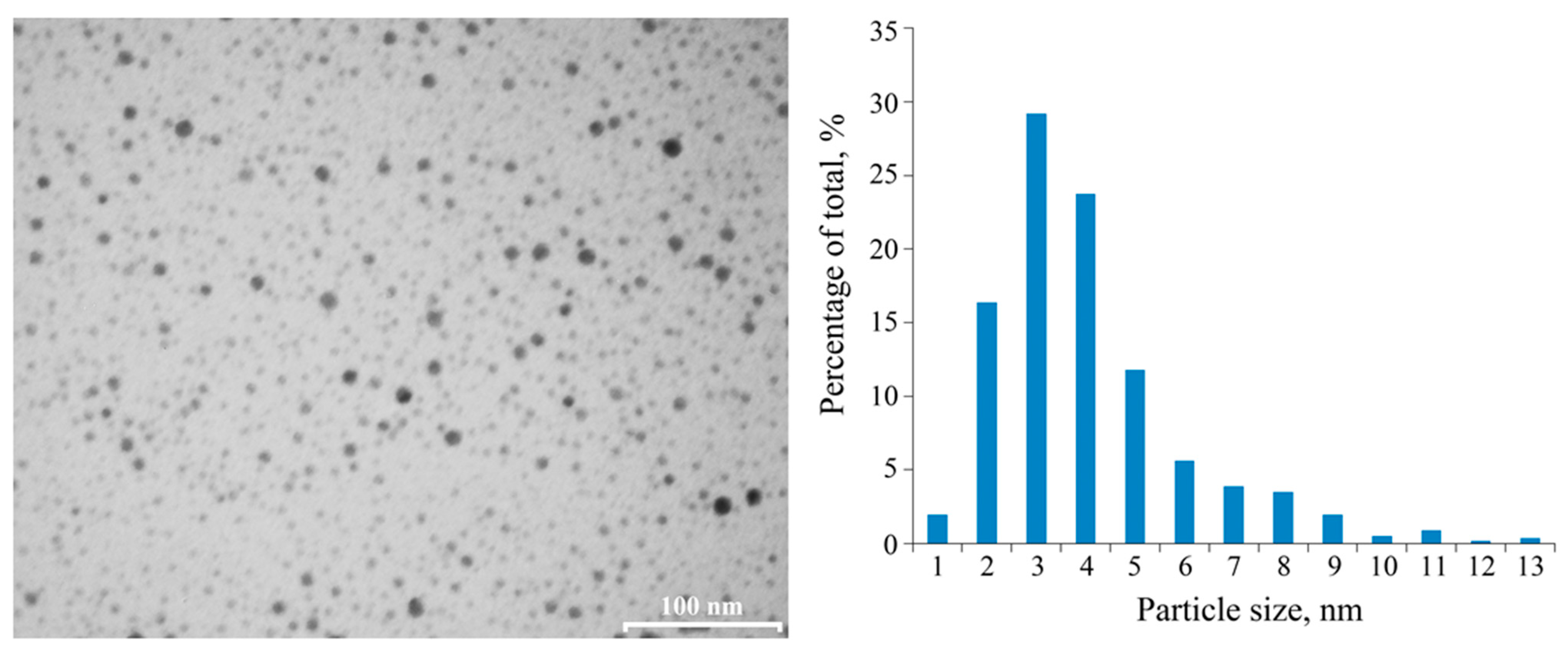
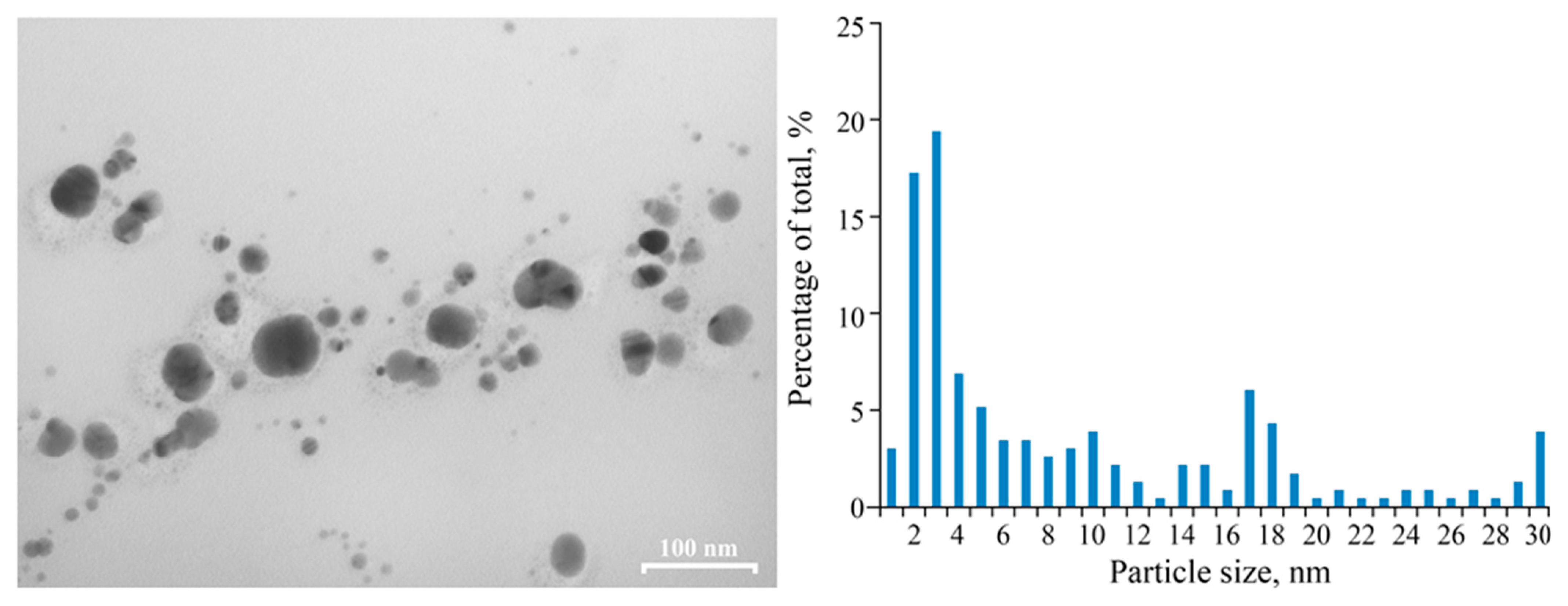
2.1. Structure Features of Freshly Synthesized PAAg
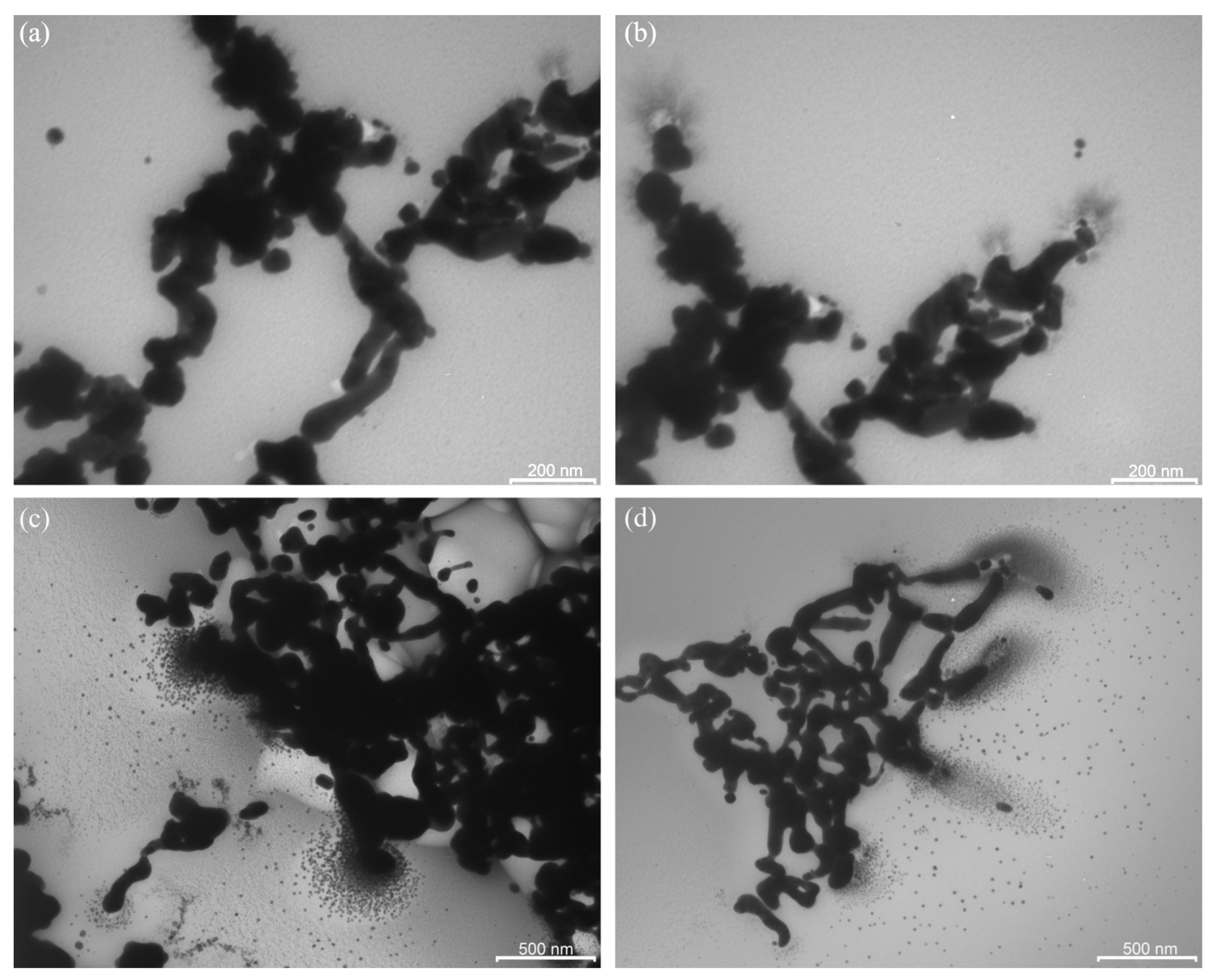
2.2. Solid Film PAAg Stored for One Year
2.3. Stimulated Aging of Solid PAAg
2.4. Aqueous PAAg Solution Stored for One Year
2.5. Antimicrobial Activity of PAAg
3. Discussion
 , in which the unpaired electron interacts with one ά-and one β-proton or to the radical
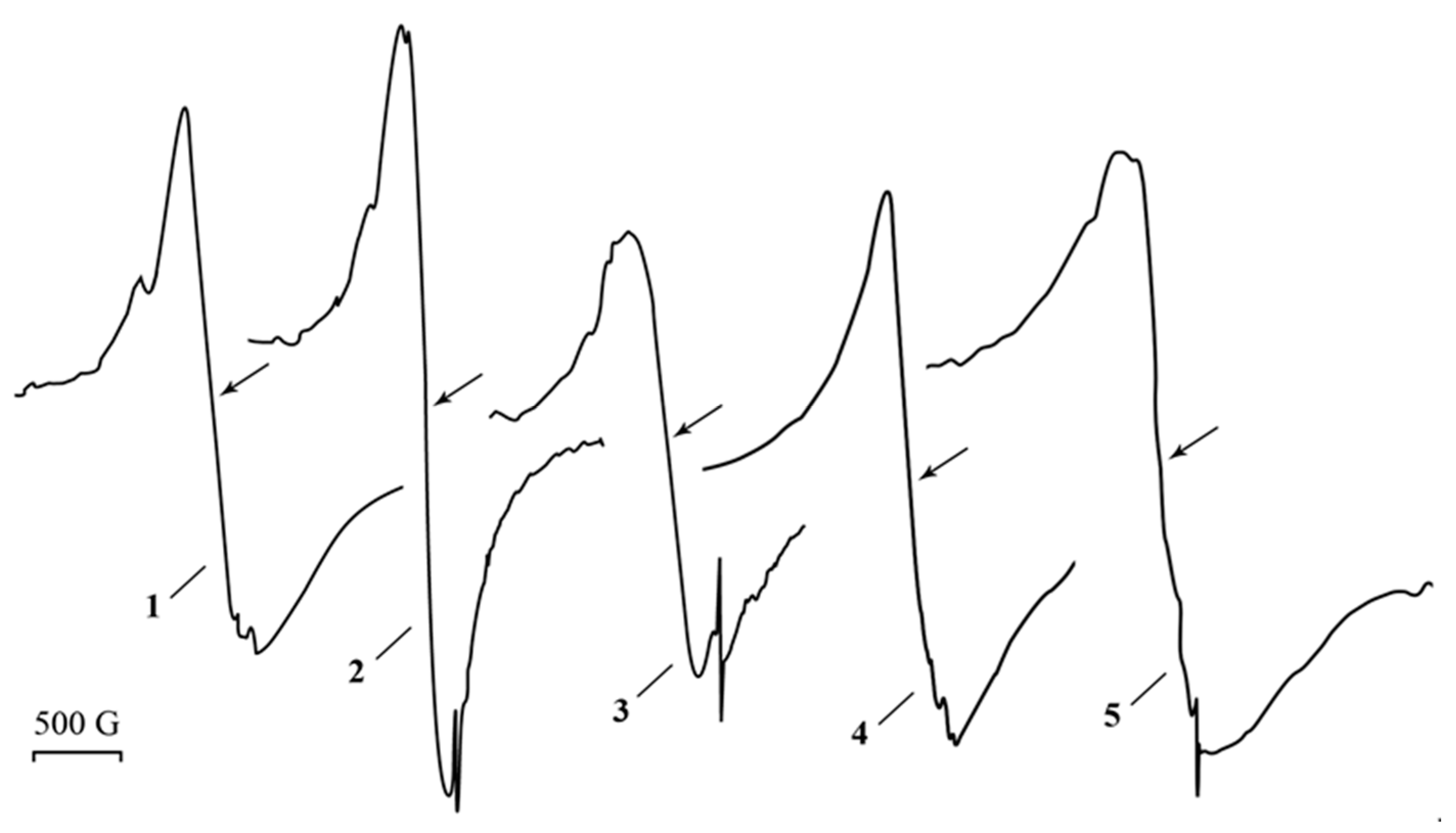
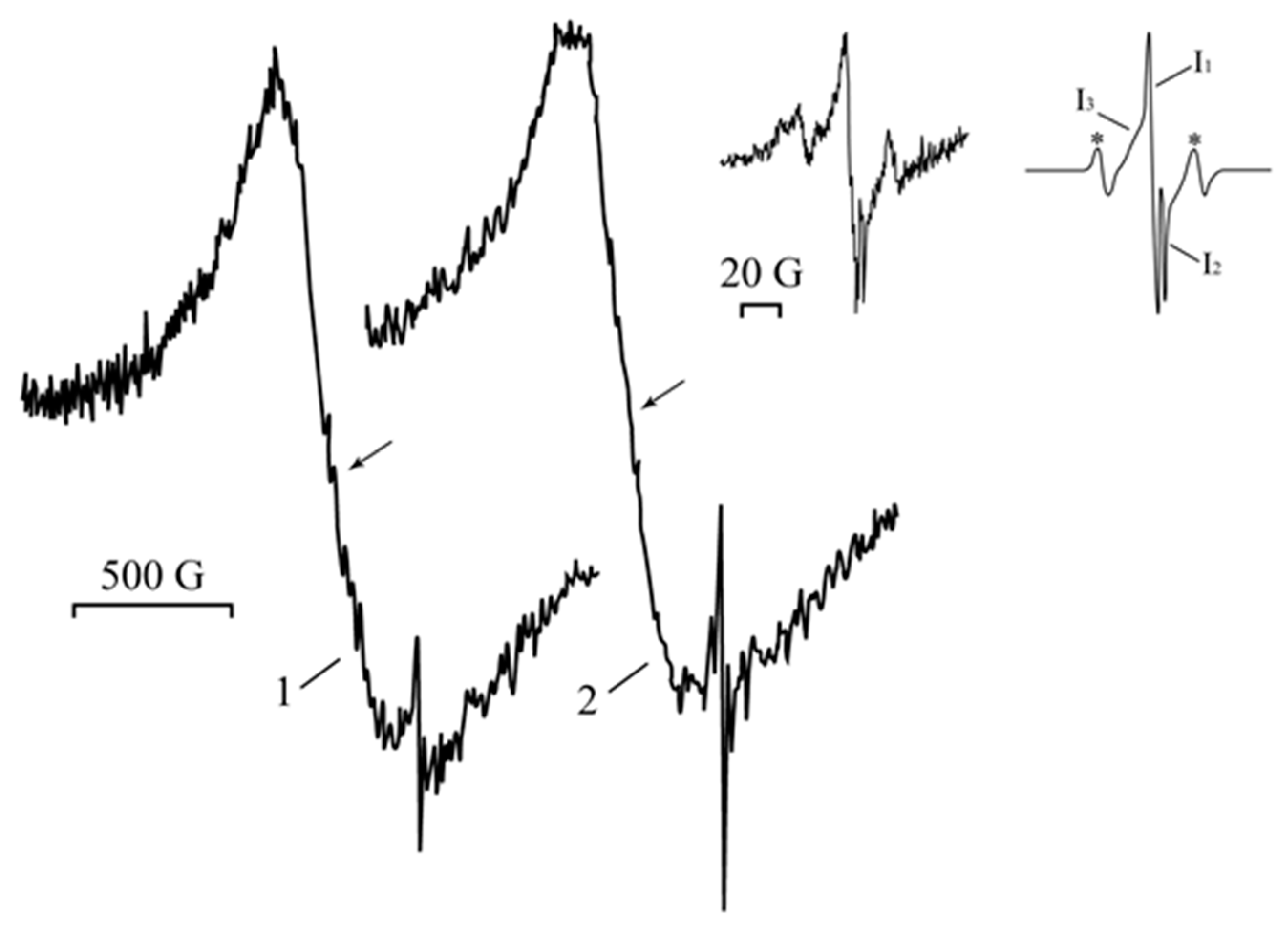
, in which the unpaired electron interacts with one ά-and one β-proton or to the radical  [60,61]. Singlet I3 (ΔH = 12.00 G; g = 2.0043) can be assigned to radicals -CH2-ĊH-, which are formed via the decarboxylation reaction of polyacrylic acid [50]. Signal I1 can be referred to the small Ag0 silver nanoparticles of about 1–2 nm [48,62,63]. The singlet I2 (ΔH = 1.55 G; g = 2.0005) relates to acyl radicals from the polyacrylic matrix [64,65]. Moreover, the comparison of the changes in the EPR spectra upon heating and UV irradiation clearly shows that the narrow signals (g = 2.0038) in Figure 2, and I1 (g = 2.0043) on inset (Figure 6) are close for both processes and correspond to the nature of the narrow weak signal in the spectrum of the freshly prepared silver polyacrylate (g = 2.0038). Thus, such EPR singlet signal in all three cases can be attributed to the silver nanoparticles.
[60,61]. Singlet I3 (ΔH = 12.00 G; g = 2.0043) can be assigned to radicals -CH2-ĊH-, which are formed via the decarboxylation reaction of polyacrylic acid [50]. Signal I1 can be referred to the small Ag0 silver nanoparticles of about 1–2 nm [48,62,63]. The singlet I2 (ΔH = 1.55 G; g = 2.0005) relates to acyl radicals from the polyacrylic matrix [64,65]. Moreover, the comparison of the changes in the EPR spectra upon heating and UV irradiation clearly shows that the narrow signals (g = 2.0038) in Figure 2, and I1 (g = 2.0043) on inset (Figure 6) are close for both processes and correspond to the nature of the narrow weak signal in the spectrum of the freshly prepared silver polyacrylate (g = 2.0038). Thus, such EPR singlet signal in all three cases can be attributed to the silver nanoparticles. and/or
and/or  (triplet), as well as -CH2-ĊH-(I3)) or products of this matrix reduction (
(triplet), as well as -CH2-ĊH-(I3)) or products of this matrix reduction (  (I2)). Simultaneous formation of these radicals can be rationalized by synchronous participation of the polyacrylate molecules in the redox processes on opposite poles of permanently polarized silver nanoparticles, which in this case can be considered as plasmon–polariton-stimulated electrochemical nanocells (Figure 11a). Reduction of the polyacrylate molecules can occur on the negative poles of these redox-nanocells, while oxidation can take place on the positive poles. Radical products of such a conjugated redox-process involving intensively discussed plasmon-stimulated photocatalysis on nanoparticles (see, for example in Refs. [66,67]) are here observed by the EPR method.
(I2)). Simultaneous formation of these radicals can be rationalized by synchronous participation of the polyacrylate molecules in the redox processes on opposite poles of permanently polarized silver nanoparticles, which in this case can be considered as plasmon–polariton-stimulated electrochemical nanocells (Figure 11a). Reduction of the polyacrylate molecules can occur on the negative poles of these redox-nanocells, while oxidation can take place on the positive poles. Radical products of such a conjugated redox-process involving intensively discussed plasmon-stimulated photocatalysis on nanoparticles (see, for example in Refs. [66,67]) are here observed by the EPR method.4. Materials and Methods
4.1. Materials
4.2. Physical–Chemical Measurements
4.3. Antimicrobial and Fungicidal Activity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cao, F.; Huang, T.; Wang, Y.; Liu, F.; Chen, L.; Ling, J.; Sun, J. Novel lanthanide-polymer complexes for dye-free dual modal probes for MRI and fluorescence imaging. Polym. Chem. 2015, 6, 7949–7957. [Google Scholar] [CrossRef]
- Shamsi, M.H.; Kraatz, H.-B. Interactions of metal ions with DNA and some applications. J. Inorg. Organomet. Polym. 2018, 23, 4–23. [Google Scholar] [CrossRef]
- Tikhonov, N.I.; Khutsishvili, S.S.; Larina, L.I.; Pozdnyakov, A.S.; Emel’yanov, A.I.; Prozorova, G.F.; Vashchenko, A.V.; Vakul’skaya, T.I. Silver polymer complexes as precursors of nanocomposites based on polymers of 1-vinyl-1,2,4-triazole. J. Mol. Struct. 2019, 1180, 272–279. [Google Scholar] [CrossRef]
- Artem’ev, A.V.; Vysotskaya, O.V.; Oparina, L.A.; Bogomyakov, A.S.; Khutsishvili, S.S.; Sterkhova, I.V.; Ovcharenko, V.I.; Trofimov, B.A. New heterospin chain-polymers based on Cu (hfac) 2 complex with TEMPO derivatives bearing b-(oxy) acrylate moiety: Synthesis, structural and magnetic properties. Polyhedron 2016, 119, 293–299. [Google Scholar] [CrossRef]
- Zalesskiy, S.S.; Ananikov, V.P. Pd2(dba)3 as a precursor of soluble metal complexes and nanoparticles: Determination of palladium active species for catalysis and synthesis. Organometallics 2012, 31, 2302–2309. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as antibacterial agent: Ion, nanoparticle, and metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef]
- Maillard, J.-Y.; Hartemann, P. Silver as an antimicrobial: Facts and gaps in knowledge. Crit. Rev. Microbiol. 2013, 39, 373–383. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X. Macromolecular agents with antimicrobial potentialities: A drive to combat antimicrobial resistance. Int. J. Biol. Macromol. 2017, 103, 554–574. [Google Scholar] [CrossRef]
- Ferreres, G.; Pérez-Rafael, S.; Torrent-Burgués, J.; Tzanov, T. Hyaluronic acid derivative molecular weight-dependent synthesis and antimicrobial effect of hybrid silver nanoparticles. Int. J. Mol. Sci. 2021, 22, 13428. [Google Scholar] [CrossRef]
- Sellami, H.; Khan, S.A.; Ahmad, I.; Alarfaj, A.A.; Hirad, A.H.; Al-Sabri, A.E. Green synthesis of silver nanoparticles using Olea europaea leaf extract for their enhanced antibacterial, antioxidant, cytotoxic and biocompatibility applications. Int. J. Mol. Sci. 2021, 22, 12562. [Google Scholar] [CrossRef] [PubMed]
- Voronkov, M.G.; Lopyrev, V.A.; Antonik, L.M.; Abzaeva, K.A.; Kogan, A.S.; Grigoriev, E.G.; Fadeeva, T.V.; Marchenko, V.I. Argacryl as New Antiseptic and Hemostatic Agent. RU 2220982 C2, 10 October 2004. [Google Scholar]
- Abzaeva, K.A.; Zelenkov, L.E. Modern topical hemostatic agents and unique representatives of their new generation. Russ. Chem. Bull. 2015, 6, 1233–1239. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Abzaeva, K.A.; Zhilitskaya, L.V.; Ostrovskaya, L.A.; Korman, D.B.; Fomina, M.M.; Blyukhterova, N.V.; Rykova, V.A. Antitumor Agent, Related to Group of Metal-Organic Derivatives of Polyacrylic Acid. RU 2372091 C1, 10 November 2009. [Google Scholar]
- Abzaeva, K.A.; Voronkov, M.G.; Zhilitskaya, L.V.; Ostrovskaya, L.A.; Fomina, M.M.; Blyukhterova, N.V.; Rykova, V.A. Antitumor effect of polymetalloacrylates, a new generation of haemostatics. Pharm. Chem. J. 2012, 46, 207–209. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Platonova, A.T.; Annenkova, V.Z.; Kononchuk, G.M.; Kazimirovskaya, V.B.; Ugrjumova, G.S.; Annenkova, V.M. Local Hemostatic. U.S. Patent 4215106A, 29 July 1980. SU Patent 698622 A1, 25 November 1979. [Google Scholar]
- Wang, S.Y. Water Soluble Antimicrobial Polyacrylate Silver Salt. U.S. Patent 2016/0309721 A1, 27 October 2016. [Google Scholar]
- White, J.R. Polymer aging: Physics, chemistry or enginering? Time to reflect. ComptesRendus.Chim. 2006, 9, 1396–1408. [Google Scholar] [CrossRef]
- Cangialosi, D.; Boucher, V.M.; Alegría, A.; Colmenero, J. Physical aging in polymers and polymer nanocomposites: Recent results and open questions. Soft Matter. 2013, 9, 8619–8630. [Google Scholar] [CrossRef]
- Kreibig, U.; Vollmer, M. Optical Properties of Metal Clusters; Springer: New York, NY, USA, 1995. [Google Scholar]
- Khutsishvili, S.S.; Vakul’skaya, T.I.; Aleksandrova, G.P.; Sukhov, B.G. Strong stabilization properties of humic substance matrixes for silver bionanocomposites. Micro Nano Lett. 2017, 12, 418–421. [Google Scholar] [CrossRef]
- Travan, A.; Marsich, E.; Donati, I.; Paoletti, S. Silver nanocomposites and their biomedical applications. In Nanotechnologies for the Life Sciences; Kumar, C.S.S.R., Ed.; John Wiley and Sons: Baton Rouge, LA, USA, 2012; Volume 10, p. 81. [Google Scholar]
- Cioffi, N.; Rai, M. Nano-Antimicrobials: Progress and Prospects; Springer: New York, NY, USA, 2012. [Google Scholar]
- Eckhardt, S.; Brunetto, P.S.; Gagnon, J.; Priebe, M.; Giese, B.; Fromm, K.M. Nanobio silver: Its interactions with peptides and bacteria, and its uses in medicine. Chem. Rev. 2013, 113, 4708–4754. [Google Scholar] [CrossRef]
- Alarcon, E.; Griffith, M.; Udekwu, K.I. Silver Nanoparticle Applications: In the Fabrication and Design of Medical and Biosensing; Springer International Publishing: Cham, Switzerland, 2015. [Google Scholar]
- Shurygina, I.A.; Sukhov, B.G.; Fadeeva, T.V.; Umanets, V.A.; Shurygin, M.G.; Ganenko, T.V.; Kostyro, Y.A.; Grigoriev, E.G.; Trofimov, B.A. Bactericidal action of Ag(0)-antithrombotic sulfated arabinogalactan nanocomposite: Coevolution of initial nanocomposite and living microbial cell to a novel non-living nanocomposite. Nanomed. NBM 2011, 7, 827–833. [Google Scholar] [CrossRef]
- Shurygina, I.A.; Shurygin, M.G.; Sukhov, B.G. Nanobiocomposites of metals as antimicrobial agents. In Antibiotic Resistance: Mechanisms and New Antimicronial Approaches; Kon, K., Rai, M., Eds.; Academic Press: London, UK, 2016; p. 167. [Google Scholar]
- Regiel-Futyra, A.; Dąbrowski, J.M.; Mazuryk, O.; Śpiewak, K.; Kyzioł, A.; Pucelik, B.; Brindell, M.; Stochel, G. Bioinorganic antimicrobial strategies in the resistance era. Coord. Chem. Rev. 2017, 351, 76–117. [Google Scholar] [CrossRef]
- Khutsishvili, S.S.; Lesnichaya, M.V.; Vakul’skaya, T.I.; Dolmaa, G.; Aleksandrova, G.P.; Rakevich, A.L.; Sukhov, B.G. Humic-based bionanocomposites containing stable paramagnetic gold nanoparticles for prospective use in pharmaceuticals. Spectrosc. Lett. 2018, 51, 169–173. [Google Scholar] [CrossRef]
- Togashi, T.; Saito, K.; Matsuda, Y.; Sato, I.; Kon, H.; Uruma, K.; Ishizaki, M.; Kanaizuka, K.; Sakamoto, M.; Ohya, N.; et al. Synthesis of water-dispersible silver nanoparticles by thermal decomposition of water-soluble silver oxalate precursors. J. Nanosci. Nanotechnol. 2014, 14, 6022–6027. [Google Scholar] [CrossRef] [PubMed]
- Tetsu, Y.; Hideo, G.; Kunihito, K. Cationic silver nanoparticles dispersed in water prepared from insoluble salts. Chem. Lett. 2003, 32, 194–195. [Google Scholar] [CrossRef]
- Pozdnyakov, A.S.; Emel’yanov, A.I.; Kuznetsova, N.P.; Ermakova, T.G.; Korzhova, S.A.; Khutsishvili, S.S.; Vakul’skaya, T.I.; Prozorova, G.F. Synthesis and characterization of silver containing nanocomposites based on 1-vinyl-1,2,4-triazole and acrylonitrile copolymer. J. Nanomater. 2019, 2019, 4895192. [Google Scholar] [CrossRef]
- Burchard, W. Solution properties of branched macromolecules. Adv. Polym. Sci. 1999, 143, 113–194. [Google Scholar] [CrossRef]
- Pospíšil, J.; Horák, Z.; Pilař, J.; Billingham, N.C.; Zweifel, H.; Nešpůrek, S. Influence of testing conditions on the performance and durability of polymer stabilisers in thermal oxidation. Polym. Degrad. Stab. 2003, 82, 145–162. [Google Scholar] [CrossRef]
- Kockott, D. Natural and artificial weathering of polymers. Polym. Degrad. Stab. 1989, 25, 181–208. [Google Scholar] [CrossRef]
- Lund, C.E.; Nielsen, C.J. Optical properties in the UV and visible spectral region of organic acids relevant to tropospheric aerosols. Atmos. Chem. Phys. 2004, 4, 1759–1769. [Google Scholar] [CrossRef]
- Ershov, B.G.; Sukkov, N.L.; Kiselel, A.K.; Lonow, G.F. A silver clusters: Optical absorption and ESR spectra; Structure and calculation of electron transitions. Russ. Chem. Bull. 1996, 45, 545–549. [Google Scholar] [CrossRef]
- Ershov, B.G.; Janata, E.; Henglein, A.; Fojtik, A. Silver atoms and clusters in aqueous solution: Absorption spectra and the particle growth in the absence of stabilizing Ag+ ions. J. Phys. Chem. 1993, 97, 4589–4594. [Google Scholar] [CrossRef]
- Ershov, B.G. Short-lived metal clusters in aqeous solutions: Formation, identification and properties. Russ. Chem. Bull. 1999, 48, 1–15. [Google Scholar] [CrossRef]
- Harb, M.; Rabilloud, F.; Simon, D.; Rydlo, A.; Lecoultre, S.; Conus, F.; Rodrigues, V.; Félix, C. Optical absorption of small silver clusters: Agn (n = 4–22). J. Chem. Phys. 2008, 129, 194108. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.K.; Sehested, J.; Nielsen, O.J.; Wallington, T.J.; Guschin, A.; Hurley, M.D. Kinetics and mechanism of the reactions of 2,3-butadione with F and Cl atoms, UV absorption spectra of CH3C(O)C(O)CH2·and CH3C(O)C(O)CH2O2·radicals, and atmospheric fate of CH3C(O)C(O)CH2O·radicals. J. Phys. Chem. A 1998, 102, 8913–8923. [Google Scholar] [CrossRef]
- Cox, R.A.; Munk, J.; Nielsen, O.J.; Pagsberg, P.; Ratajczak, E. Ultraviolet absorption spectra and kinetics of acetonyl and acetonylperoxy radicals. Chem. Phys. Lett. 1990, 173, 206–210. [Google Scholar] [CrossRef]
- Lide, D.R.; Milne, G.W.A. Handbook of Data on Organic Compounds; CRC Press: Boca Raton, FL, USA, 1993; Volume 1. [Google Scholar]
- Smirnov, A. EPR Studies of Nanomaterials. In Multifrequency Electron Paramagnetic Resonance; Misra, S.K., Ed.; Willey-VCH: Verlag, Germany, 2011; pp. 825–843. [Google Scholar]
- Khutsishvili, S.S.; Toidze, P.; Donadze, M.; Gabrichidze, M.; Makhaldiani, N.; Agladze, T. Structural surface features of paramagnetic multifunctional nanohybrids based on silver oleic acid. J. Clust. Sci. 2021, 32, 1351–1359. [Google Scholar] [CrossRef]
- Ganenko, T.V.; Tantsyrev, A.P.; Sapozhnikov, A.N.; Khutsishvili, S.S.; Vakul’skaya, T.I.; Fadeeva, T.V.; Sukhov, B.G.; Trofimov, B.A. Nanocomposites of silver with arabinogalactan sulfate: Preparation, structure, and antimicrobial activity. Russ. J. Gen. Chem. 2015, 85, 477–484. [Google Scholar] [CrossRef]
- Eichelbaum, M.; Rademann, K.; Hoell, A.; Tatchev, D.M.; Weigel, W.; Stößer, R.; Pacchioni, G. Photoluminescence of atomic gold and silver particles in soda-lime silicate glasses. Nanotechnology 2008, 19, 135701. [Google Scholar] [CrossRef]
- Shabatina, T.I.; Mascetti, J.; Ogden, J.S.; Sergeev, G.B. Competitive cryochemical reactions of transition metal atoms, clusters and nanosized particles. Russ. Chem. Rev. 2007, 76, 1123–1137. [Google Scholar] [CrossRef]
- Khutsishvili, S.S.; Vakul’skaya, T.I.; Aleksandrova, G.P.; Sukhov, B.G. Stabilized silver nanoparticles and clusters Agn of humic-based bioactive nanocomposites. J. Clust. Sci. 2017, 28, 3067–3074. [Google Scholar] [CrossRef]
- Zezin, A.N.; Fel’dman, V.I.; Egorov, A.V.; Shmakova, N.A. Formation of radicals in irradiated complexes of polyacrylate anions and alkyltrimethylammonium cations. Dokl. Chem. 2003, 390, 158–161. [Google Scholar] [CrossRef]
- Halas, N.J.; Lal, S.; Chang, W.-S.; Link, S.; Nordlander, P. Plasmons in strongly coupled metallic nanostructures. Chem. Rev. 2011, 111, 3913–3961. [Google Scholar] [CrossRef]
- Gilbert, B.C.; Hodges, G.R.; Smith, J.R.L.; Taylor, P. Photo-decarboxylation of substituted alkylcarboxylic acids brought about by visible light and iron(III) tetra(2-N-methylpyridyl)-porphyrin in aqueous solution. J. Chem. Soc. 1996, 2, 519–524. [Google Scholar] [CrossRef]
- Benneu, F.; Monod, P. Conduction-electron spin resonance in cold-worked Al, Cu, and Ag: The spin-flip cross section of dislocations. Phys. Rev. B 1976, 13, 3424–3430. [Google Scholar] [CrossRef]
- Benneu, F.; Monod, P. The Elliott relation in pure metals. Phys. Rev. B 1978, 18, 2422–2425. [Google Scholar] [CrossRef]
- Garitaonandia, J.S.; Insausti, M.; Goikolea, E.; Suzuki, M.; Cashion, J.D.; Kawamura, N.; Ohsawa, H.; de Muro, I.G.; Suzuki, K.; Plazaola, F.; et al. Chemically induced permanent magnetism in Au, Ag, and Cu nanoparticles: Localization of the magnetism by element selective techniques. Nano Lett. 2008, 8, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Caudillo, R.; Gao, X.; Escudero, R.; José-Yacaman, M.; Goodenough, J.B. Ferromagnetic behavior of carbon nanospheres encapsulating silver nanoparticles. Phys. Rev. B 2006, 74, 214418. [Google Scholar] [CrossRef]
- Kawabata, A. Electronic properties of fine metallic particles. III. E.S.R. absorption line shape. J. Phys. Soc. Jpn. 1970, 29, 902–911. [Google Scholar] [CrossRef]
- Claus, P.; Bruckner, A.; Mohr, C.; Hofmeister, H. Supported gold nanoparticles from quantum dot to mesoscopic size scale: Effect of electronic and structural properties on catalytic hydrogenation of conjugated functional groups. J. Am. Chem. Soc. 2000, 122, 11430–11439. [Google Scholar] [CrossRef]
- Michalik, J.; Brown, D.; Yu, J.-S.; Danilczuk, M.; Kim, J.Y.; Kevan, L. Conduction electron paramagnetic resonance of metal nanoparticles in AlMCM-41 aluminosilica mesoporous molecular sieves. J. Phys. Chem. 2001, 3, 1705–1708. [Google Scholar] [CrossRef]
- Saraev, V.V.; Alsarsur, I.A.; Annenkov, V.V.; Danilovtseva, E.N. Structure of free radicals formed under X-ray irradiation of organic polymeric acid. Russ. J. Appl. Chem. 2001, 74, 1585–1589. [Google Scholar] [CrossRef]
- Hildenbrand, K.; Schulte-Frohlinde, D. Time-resolved EPR studies on the reaction rates of peroxyl radicals of poly(acrylic acid) and of calf thymus DNA with glutathione. Re-examination of a rate constant for DNA. Int. J. Radiat. Boil. 1997, 71, 377–385. [Google Scholar] [CrossRef]
- Moon, H.R.; Kim, J.H.; Suh, M.P. Redox-active porousorganic framework producing silver nanoparticles from AgI ions at room temperature. Angew. Chem. Int. Ed. 2005, 44, 1261–1265. [Google Scholar] [CrossRef]
- Khutsishvili, S.S.; Tikhonov, N.I.; Pavlov, D.V.; Vakul’skaya, T.I.; Penzik, M.V.; Kozlov, A.N.; Lesnichaya, M.V.; Aleksandrova, G.P.; Sukhov, B.G. Gold- and silver-containing bionanocomposites based on humic substances extracted from coals: A thermal analysis study. J. Therm. Anal. Calorim. 2019, 137, 1181–1188. [Google Scholar] [CrossRef]
- Chatgilialoglu, C.; Crich, D.; Komatsu, M.; Ryu, I. Chemistry of acyl radicals. Chem. Rev. 1999, 99, 1991–2069. [Google Scholar] [CrossRef] [PubMed]
- Conte, M.; Miyamura, H.; Kobayashi, S.; Chechik, V. Enhanced acyl radical formation in the Au nanoparticle-catalysed aldehyde oxidation. Chem. Commun. 2010, 46, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, Y.L.; Liu, R.-S.; Tsai, D.P. Plasmonic photocatalysis. Rep. Prog. Phys. 2013, 76, 046401. [Google Scholar] [CrossRef] [PubMed]
- Baffoua, G.; Quidant, R. Nanoplasmonics for chemistry. Chem. Soc. Rev. 2014, 43, 3898–3907. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.Y.; et al. Antimicrobial effects of silver nanoparticles. Nanomed. NBM 2007, 3, 95–101. [Google Scholar] [CrossRef]
- Lesnichaya, M.V.; Sukhov, B.G.; Aleksandrova, G.P.; Gasilova, E.R.; Vakul’skaya, T.I.; Khutsishvili, S.S.; Sapozhnikov, A.N.; Klimenkov, I.V.; Trofimov, B.A. Chiroplasmonic magnetic gold nanocomposites produced by one-step aqueous method using κ-carrageenan. Carbohydr. Polym. 2017, 175, 18–26. [Google Scholar] [CrossRef]
- Schärtl, W. Light Scattering from Polymer Solutions and Nanoparticle Dispersions; Springer-Verlag Kreibig: Berlin, Germany, 2007. [Google Scholar]
- Fisenko, V.P.; Arzamastsev, E.V.; Babayan, E.A. Guidance for Experimental (Pre-Clinical) Investigation of New Pharmacological Compounds; U.S. Department of Health and Human Services, Food and Drug Administration: Rockville, MD, USA, 2000; pp. 1–398. [Google Scholar]
- ISO 20776-1:2006; Clinical Laboratory Testing and in Vitro Diagnostic Test Systems—Susceptibility Testing of Infectious Agents and Evaluation of Performance of Antimicrobial Susceptibility Test Devices—Part 1. Reference Method Fortesting the in Vitro Activity of Antimicrobial Agents Against Rapidly Growing Aerobic Bacteria Involved in Infectious Diseases. ISO: Geneva, Switzerland, 2006.






| Microorganisms | MIC and MBC/MFC, μg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 500 | 250 | 125 | 62.5 | 31.25 | 15.6 | 7.8 | 3.9 | 1.9 | 0.95 | |
| E. coli ATCC 25922 | −−/−− | −−/−− | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| P. aeruginosa ATCC 27853 | −−/−− | −−/−− | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| K. pneumoniae ATCC 700603 (EBSL) | −−/−− | −−/−− | −−/−− | −−/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| S. aureus ATCC 25923 | −−/−− | −−/−− | −−/−− | −−/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| S. aureus ATCC 25213 | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| E. faecalis ATCC 29212 | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| C. albicans ATCC 90028 | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| Microorganisms | MIC and MBC/MFC, μg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 500 | 250 | 125 | 62.5 | 31.25 | 15.6 | 7.8 | 3.9 | 1.9 | 0.95 | |
| E. coli ATCC 25922 | −−/−− | −−/−− | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| P. aeruginosa ATCC 27853 | −−/−− | −−/−− | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| K. pneumoniae ATCC 700603 (EBSL) | −−/−− | −−/−− | −−/−− | −−/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| S. aureus ATCC 25923 | −−/−− | −−/−− | −−/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| S. aureus ATCC 25213 | −−/−− | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| E. faecalis ATCC 29212 | −−/−− | −−/−− | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
| C. albicans ATCC 90028 | −−/−− | −−/−− | −−/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + | + +/+ + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abzaeva, K.A.; Sukhov, B.G.; Khutsishvili, S.S.; Tarabukina, E.B.; Zelenkov, L.E.; Nevezhina, A.V.; Fadeeva, T.V. Spontaneous Transformation of Biomedical Polymeric Silver Salt into a Nanocomposite: Physical–Chemical and Antimicrobial Properties Dramatically Depend on the Initial Preparation State. Int. J. Mol. Sci. 2022, 23, 10963. https://doi.org/10.3390/ijms231810963
Abzaeva KA, Sukhov BG, Khutsishvili SS, Tarabukina EB, Zelenkov LE, Nevezhina AV, Fadeeva TV. Spontaneous Transformation of Biomedical Polymeric Silver Salt into a Nanocomposite: Physical–Chemical and Antimicrobial Properties Dramatically Depend on the Initial Preparation State. International Journal of Molecular Sciences. 2022; 23(18):10963. https://doi.org/10.3390/ijms231810963
Chicago/Turabian StyleAbzaeva, Klavdia A., Boris G. Sukhov, Spartak S. Khutsishvili, Elena B. Tarabukina, Lev E. Zelenkov, Anna V. Nevezhina, and Tat’yana V. Fadeeva. 2022. "Spontaneous Transformation of Biomedical Polymeric Silver Salt into a Nanocomposite: Physical–Chemical and Antimicrobial Properties Dramatically Depend on the Initial Preparation State" International Journal of Molecular Sciences 23, no. 18: 10963. https://doi.org/10.3390/ijms231810963








