Acetyl-L-Carnitine and Liposomal Co-Enzyme Q10 Attenuate Hepatic Inflammation, Apoptosis, and Fibrosis Induced by Propionic Acid
Abstract
:1. Introduction
2. Results
2.1. A-CAR and/or L-CoQ10 Retained Liver Function in PRA-Induced Liver Injury
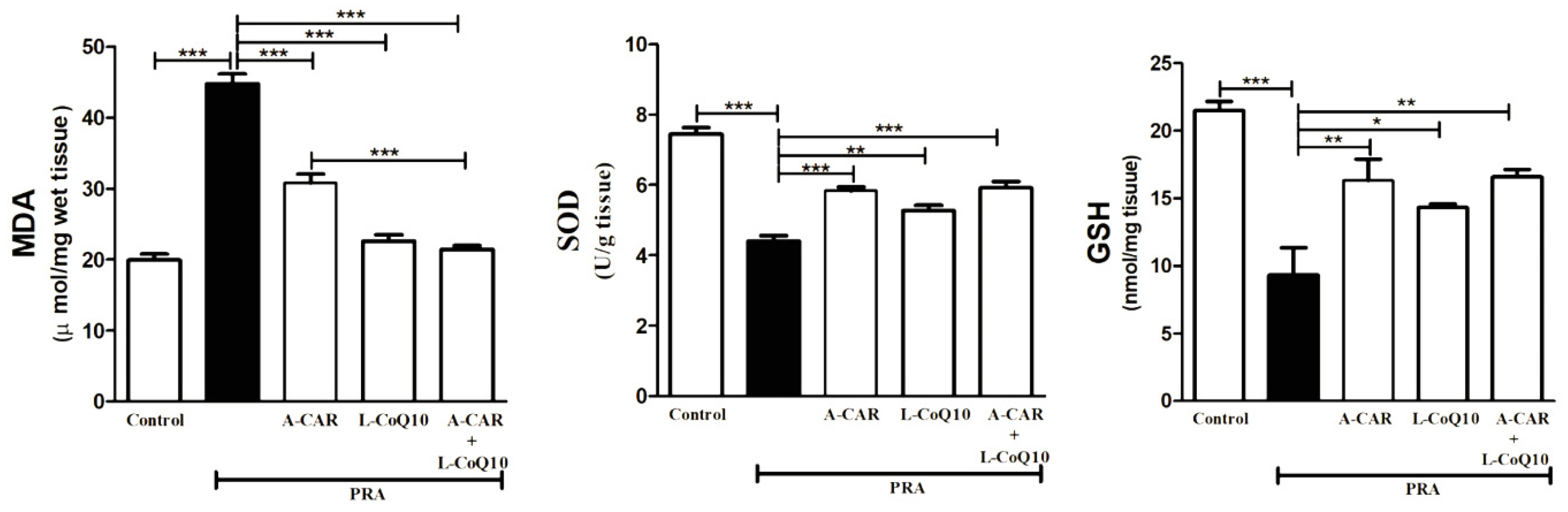
2.2. A-CAR and/or L-CoQ10 Attenuated Oxidative Stress Induced by PRA
2.3. A-CAR and/or L-CoQ10 Diminished the Inflammation and Apoptosis Induced by PRA
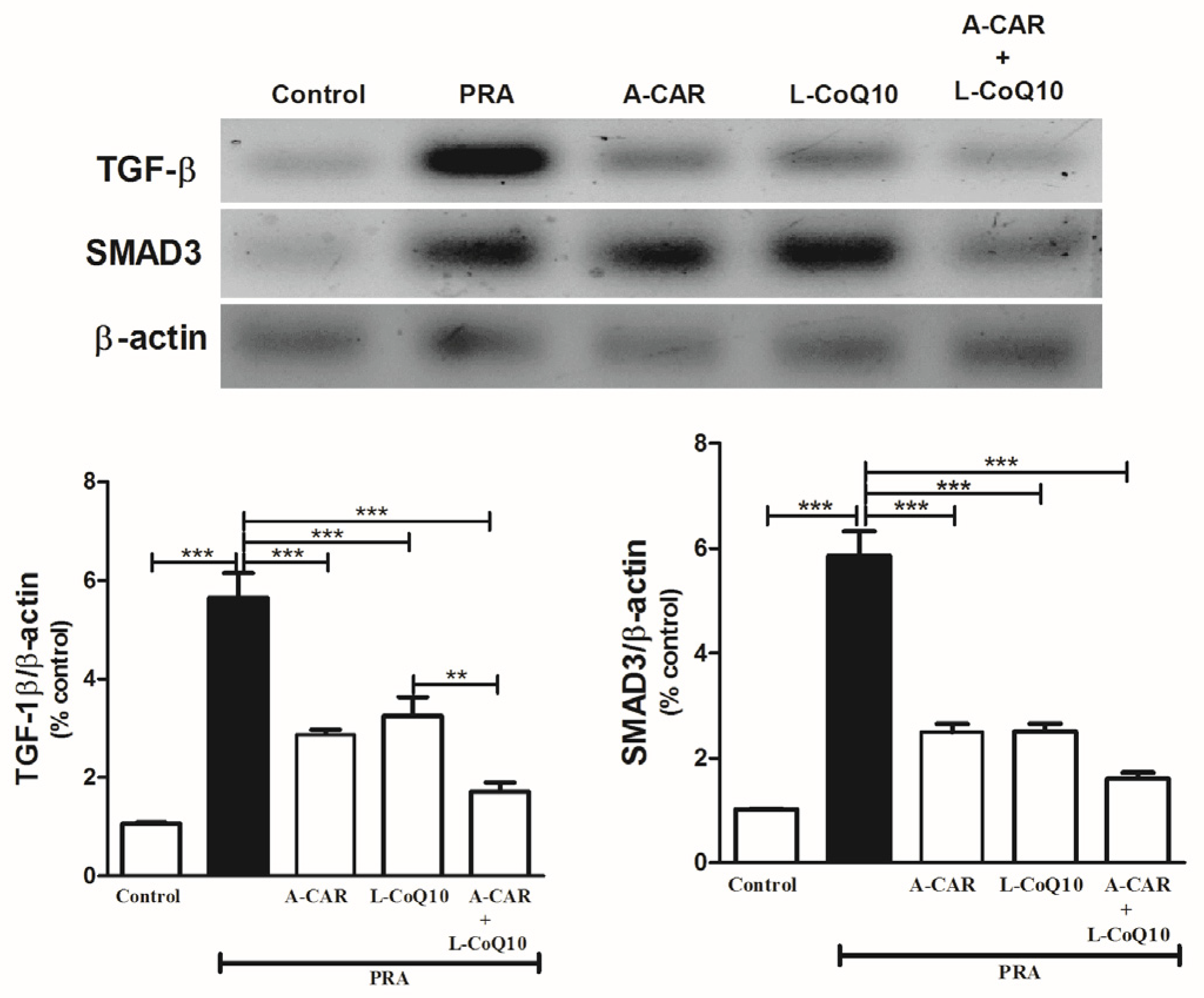
2.4. A-CAR and/or L-CoQ10 Suppressed the Hepatic Expression of TGF-β and SMAD3 Proteins
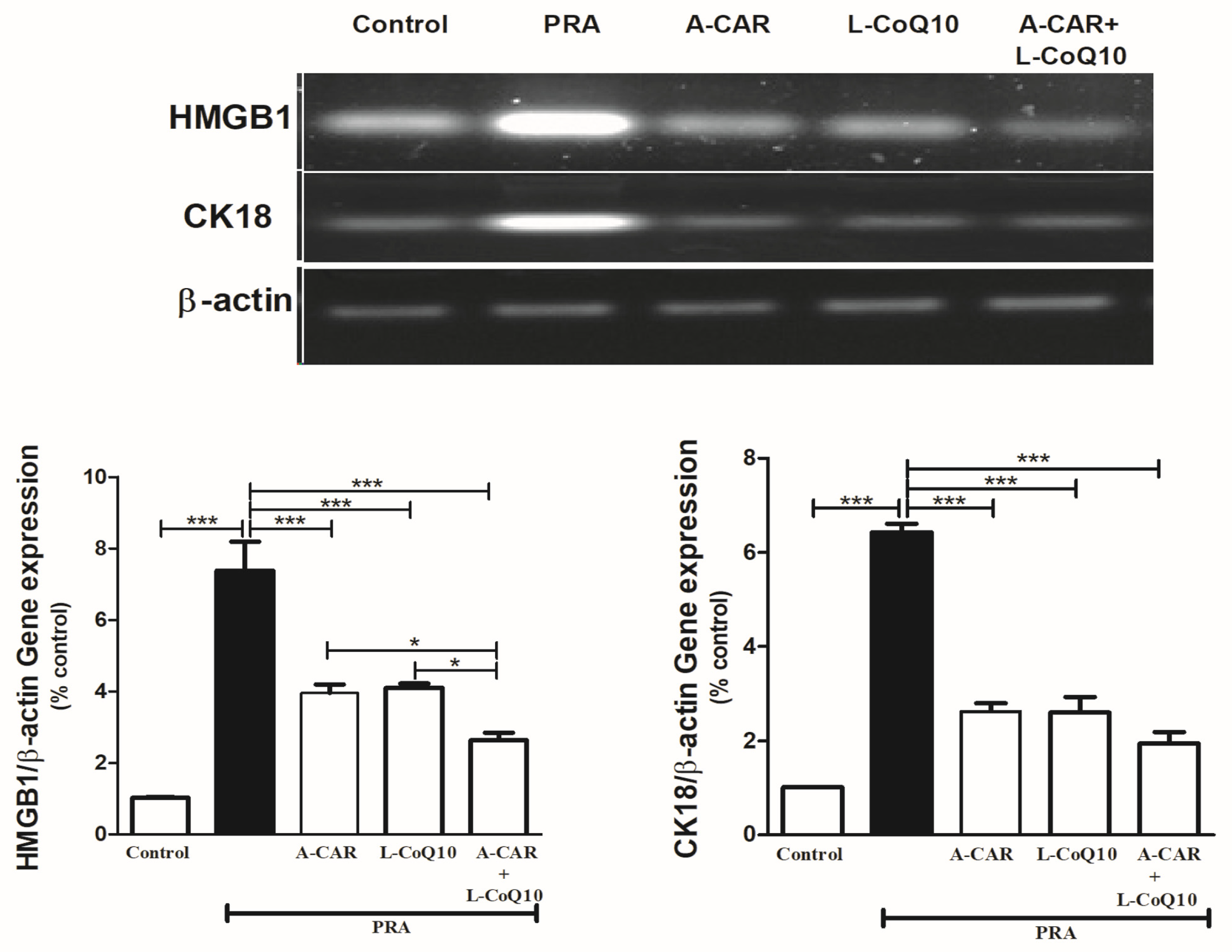
2.5. A-CAR and/or L-CoQ10 Downregulated the Hepatic Expression of HMGB1 and CK18 mRNA
2.6. A-CAR and/or L-CoQ10 Improved the Morphological Changes Induced by PRA
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals
4.3. Experimental Design
4.4. Determination of the Serum Levels of Liver Function Enzymes
4.5. Assessment of Oxidative Stress Biomarkers in Liver Tissue
4.6. Measurement of Inflammatory and Apoptotic Biomarkers in Liver Tissue
4.7. Determination of the Expression of TGF-β1 and SMAD3 Proteins
4.8. Determination of the Expression of HMGB-1 and CK18 mRNA
4.9. Histopathological Analysis
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yun, S.S.; Kim, J.; Lee, S.J.; So, J.S.; Lee, M.Y.; Lee, G.; Lim, H.S.; Kim, M. Naturally occurring benzoic, sorbic, and propionic acid in vegetables. Food Addit. Contam. Part B 2019, 12, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Kim, I.S.; Mun, J.Y. Propionic acid induces dendritic spine loss by MAPK/ERK signaling and dysregulation of autophagic flux. Mol. Brain 2020, 13, 86. [Google Scholar] [CrossRef]
- Loong, C.; Tsen, S.Y.; Ho, X.L.; Raman, M.F.B.; Loke, W.M. Common food antimicrobials: Effects on cellular inflammation and oxidative damage and their estimated occurrence in Singapore. Asia Pac. J. Clin. Nutr. 2018, 27, 113–120. [Google Scholar] [PubMed]
- Al-Daihan, S.; Bhat, R.S. Impact of propionic acid on liver damage in rats. Int. J. Mol. Cell. Med. 2015, 4, 188. [Google Scholar]
- Messelmani, T.; Morisseau, L.; Sakai, Y.; Legallais, C.; Le Goff, A.; Leclerc, E.; Jellali, R. Liver organ-on-chip models for toxicity studies and risk assessment. Lab Chip 2022, 22, 2423–2450. [Google Scholar] [CrossRef] [PubMed]
- Bayrak, S.; Aktaş, S.; Altun, Z.; Çakir, Y.; Tütüncü, M.; Özşengezer, S.K.; Yilmaz, O.; Olgun, N. Antioxidant effect of acetyl-l-carnitine against cisplatin-induced cardiotoxicity. J. Int. Med. Res. 2020, 48, 0300060520951393. [Google Scholar] [CrossRef]
- Alhasaniah, A.H. L-carnitine: Nutrition, pathology, and health benefits. Saudi J. Biol. Sci. 2022, 30, 103555. [Google Scholar] [CrossRef]
- Saini, R. Coenzyme Q10: The essential nutrient. J. Pharm. Bioallied Sci. 2011, 3, 466–467. [Google Scholar] [CrossRef]
- Lee, W.-C.; Tsai, T.-H. Preparation and characterization of liposomal coenzyme Q10 for in vivo topical application. Int. J. Pharm. 2010, 395, 78–83. [Google Scholar] [CrossRef]
- MacFabe, D.F.; Cain, D.P.; Rodriguez-Capote, K.; Franklin, A.E.; Hoffman, J.E.; Boon, F.; Taylor, A.R.; Kavaliers, M.; Ossenkopp, K.-P. Neurobiological effects of intraventricular propionic acid in rats: Possible role of short chain fatty acids on the pathogenesis and characteristics of autism spectrum disorders. Behav. Brain Res. 2007, 176, 149–169. [Google Scholar] [CrossRef]
- Lobzhanidze, G.; Lordkipanidze, T.; Zhvania, M.; Japaridze, N.; MacFabe, D.F.; Pochkidze, N.; Gasimov, E.; Rzaev, F. Effect of propionic acid on the morphology of the amygdala in adolescent male rats and their behavior. Micron 2019, 125, 102732. [Google Scholar] [CrossRef] [PubMed]
- Farsi, F.; Mohammadshahi, M.; Alavinejad, P.; Rezazadeh, A.; Zarei, M.; Engali, K.A. Functions of coenzyme Q10 supplementation on liver enzymes, markers of systemic inflammation, and adipokines in patients affected by nonalcoholic fatty liver disease: A double-blind, placebo-controlled, randomized clinical trial. J. Am. Coll. Nutr. 2016, 35, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Hu, C.-M.J.; Fang, R.H.; Zhang, L. Liposome-like nanostructures for drug delivery. J. Mater. Chem. B 2013, 1, 6569–6585. [Google Scholar] [CrossRef]
- Kathirvel, E.; Morgan, K.; French, S.W.; Morgan, T.R. Acetyl-L-carnitine and lipoic acid improve mitochondrial abnormalities and serum levels of liver enzymes in a mouse model of nonalcoholic fatty liver disease. Nutr. Res. 2013, 33, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Chen, X.; Xue, H.; Zhang, P.; Fang, W.; Chen, X.; Ling, W. Coenzyme Q10 attenuates high-fat diet-induced non-alcoholic fatty liver disease through activation of the AMPK pathway. Food Funct. 2019, 10, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Alhusaini, A.; Fadda, L.; Albogami, L.; Alnaim, N.; Sarawi, W.; Mattar, D.; Hasan, I. Liposomal coenzyme Q10 abates inflammation, apoptosis and DNA damage induced by an overdose of paracetamol in rat’s liver. J. King Saud Univ. 2022, 34, 102144. [Google Scholar] [CrossRef]
- Mohamed, D.I.; Khairy, E.; Tawfek, S.S.; Habib, E.K.; Fetouh, M.A. Coenzyme Q10 attenuates lung and liver fibrosis via modulation of autophagy in methotrexate treated rat. Biomed. Pharmacother. 2019, 109, 892–901. [Google Scholar] [CrossRef]
- Oh, H.; Park, C.H.; Jun, D.W. Impact of l-Carnitine Supplementation on Liver Enzyme Normalization in Patients with Chronic Liver Disease: A Meta-Analysis of Randomized Trials. J. Pers. Med. 2022, 12, 1053. [Google Scholar] [CrossRef]
- Khalil, S.R.; Abd-Elhakim, Y.M.; Selim, M.E.; Al-Ayadhi, L.Y. Apitoxin protects rat pups brain from propionic acid-induced oxidative stress: The expression pattern of Bcl-2 and Caspase-3 apoptotic genes. Neurotoxicology 2015, 49, 121–131. [Google Scholar] [CrossRef]
- Erten, F. Lycopene ameliorates propionic acid-induced autism spectrum disorders by inhibiting inflammation and oxidative stress in rats. J. Food Biochem. 2021, 45, e13922. [Google Scholar] [CrossRef]
- Wang, S.; Xu, J.; Zheng, J.; Zhang, X.; Shao, J.; Zhao, L.; Hao, J. Anti-inflammatory and antioxidant effects of acetyl-L-carnitine on atherosclerotic rats. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2020, 26, e920250-1–e920250-11. [Google Scholar] [CrossRef] [PubMed]
- Sifuentes-Franco, S.; Sánchez-Macías, D.C.; Carrillo-Ibarra, S.; Rivera-Valdés, J.J.; Zuñiga, L.Y.; Sánchez-López, V.A. Antioxidant and anti-inflammatory effects of coenzyme Q10 supplementation on infectious diseases. Healthcare 2022, 10, 487. [Google Scholar] [CrossRef] [PubMed]
- Yapar, K.; Kart, A.; Karapehlivan, M.; Atakisi, O.; Tunca, R.; Erginsoy, S.; Citil, M. Hepatoprotective effect of L-carnitine against acute acetaminophen toxicity in mice. Exp. Toxicol. Pathol. 2007, 59, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Sepand, M.R.; Razavi-Azarkhiavi, K.; Omidi, A.; Zirak, M.R.; Sabzevari, S.; Kazemi, A.R.; Sabzevari, O. Effect of acetyl-l-carnitine on antioxidant status, lipid peroxidation, and oxidative damage of arsenic in rat. Biol. Trace Elem. Res. 2016, 171, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Abdelli, L.S.; Samsam, A.; Naser, S.A. Propionic acid induces gliosis and neuro-inflammation through modulation of PTEN/AKT pathway in autism spectrum disorder. Sci. Rep. 2019, 9, 8824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bodaghi-Namileh, V.; Sepand, M.R.; Omidi, A.; Aghsami, M.; Seyednejad, S.A.; Kasirzadeh, S.; Sabzevari, O. Acetyl-l-carnitine attenuates arsenic-induced liver injury by abrogation of mitochondrial dysfunction, inflammation, and apoptosis in rats. Environ. Toxicol. Pharmacol. 2018, 58, 11–20. [Google Scholar] [CrossRef]
- Mazdeh, M.; Abolfathi, P.; Sabetghadam, M.; Mohammadi, Y.; Mehrpooya, M. Clinical evidence of acetyl-l-carnitine efficacy in the treatment of acute ischemic stroke: A pilot clinical trial. Oxid. Med. Cell. Longev. 2022, 2022, 2493053. [Google Scholar] [CrossRef]
- Baci, D.; Bruno, A.; Bassani, B.; Tramacere, M.; Mortara, L.; Albini, A.; Noonan, D.M. Acetyl-l-carnitine is an anti-angiogenic agent targeting the VEGFR2 and CXCR4 pathways. Cancer Lett. 2018, 429, 100–116. [Google Scholar] [CrossRef]
- Albogami, S. The Potential Inhibitory Role of Acetyl-L-Carnitine on Proliferation, Migration, and Gene Expression in HepG2 and HT29 Human Adenocarcinoma Cell Lines. Curr. Issues Mol. Biol. 2023, 45, 2393–2408. [Google Scholar] [CrossRef]
- Farsi, F.; Heshmati, J.; Keshtkar, A.; Irandoost, P.; Alamdari, N.M.; Akbari, A.; Janani, L.; Morshedzadeh, N.; Vafa, M. Can coenzyme Q10 supplementation effectively reduce human tumor necrosis factor-α and interleukin-6 levels in chronic inflammatory diseases? A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2019, 148, 104290. [Google Scholar] [CrossRef]
- Dludla, P.V.; Orlando, P.; Silvestri, S.; Marcheggiani, F.; Cirilli, I.; Nyambuya, T.M.; Mxinwa, V.; Mokgalaboni, K.; Nkambule, B.B.; Johnson, R.; et al. Coenzyme Q10 Supplementation Improves Adipokine Levels and Alleviates Inflammation and Lipid Peroxidation in Conditions of Metabolic Syndrome: A Meta-Analysis of Randomized Controlled Trials. Int. J. Mol Sci. 2020, 21, 3247. [Google Scholar] [CrossRef]
- Sharafkhah, M.; Mosayebi, G.; Massoudifar, A.; Seddigh, S.H.; Abdolrazaghnejad, A.; Alamdara, M.A.; Mokarian, P.; Mohammadbeigi, A.; Ebrahimi-Monfared, M. Does the Serum Expression Level of High-Mobility Group Box 1 (HMGB1) in Multiple Sclerosis Patients have a Relationship with Physical and Psychological Status? A 12-Month Follow-Up Study on Newly Diagnosed MS Patients. Neurol. India 2022, 70, 238–248. [Google Scholar] [PubMed]
- Techarang, T.; Jariyapong, P.; Viriyavejakul, P.; Punsawad, C. High mobility group box-1 (HMGB-1) and its receptors in the pathogenesis of malaria-associated acute lung injury/acute respiratory distress syndrome in a mouse model. Heliyon 2021, 7, e08589. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.-A.; Chen, H.; Nie, H.; Zheng, B.; Gong, Q. HMGB1: An overview of its roles in the pathogenesis of liver disease. J. Leukoc. Biol. 2021, 110, 987–998. [Google Scholar] [CrossRef]
- Zhang, C.; Dong, H.; Chen, F.; Wang, Y.; Ma, J.; Wang, G. The HMGB1-RAGE/TLR-TNF-α signaling pathway may contribute to kidney injury induced by hypoxia. Exp. Ther. Med. 2019, 17, 17–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kannan, K.; Jain, S.K. Oxidative stress and apoptosis. Pathophysiology 2000, 7, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.P.; Huang, S.H.; De Silva, R.; Tan, B.K.H.; Zhu, Y.Z. Oxidative stress: Apoptosis in neuronal injury. Curr. Alzheimer Res. 2006, 3, 327–337. [Google Scholar] [CrossRef]
- Ozturk, N.; Ozturk, G.; Cerrah, S.; Atamanalp, S.S.; Gul, M.A.; Aksungur, N.; Bakan, N.; Bakan, E. Evaluation of liver function by means of serum cytokeratin 18 and hepatocyte growth factor levels in patients with obstructive jaundice. Acta Chir. Belg. 2018, 118, 167–171. [Google Scholar] [CrossRef]
- Bratoeva, K.; Nikolova, S.; Merdzhanova, A.; Stoyanov, G.S.; Dimitrova, E.; Kashlov, J.; Conev, N.; Radanova, M. Association between serum CK-18 levels and the degree of liver damage in fructose-induced metabolic syndrome. Metab. Syndr. Relat. Disord. 2018, 16, 350–357. [Google Scholar] [CrossRef]
- Alhusaini, A.; Sarawi, W.; Mattar, D.; Abo-Hamad, A.; Almogren, R.; Alhumaidan, S.; Alsultan, E.; Alsaif, S.; Hasan, I.; Hassanein, E.; et al. Acetyl-L-carnitine and/or liposomal co-enzyme Q10 prevent propionic acid-induced neurotoxicity by modulating oxidative tissue injury, inflammation, and ALDH1A1-RA-RARα signaling in rats. Biomed. Pharmacother. 2022, 153, 113360. [Google Scholar] [CrossRef]
- Di Cesare Mannelli, L.; Ghelardini, C.; Calvani, M.; Nicolai, R.; Mosconi, L.; Toscano, A.; Pacini, A.; Bartolini, A. Neuroprotective effects of acetyl-L-carnitine on neuropathic pain and apoptosis: A role for the nicotinic receptor. J. Neurosci. Res. 2009, 87, 200–207. [Google Scholar] [CrossRef]
- Li, X.; Zhan, J.; Hou, Y.; Hou, Y.; Chen, S.; Luo, D.; Luan, J.; Wang, L.; Lin, D. Coenzyme Q10 regulation of apoptosis and oxidative stress in H2O2 induced BMSC death by modulating the Nrf-2/NQO-1 signaling pathway and its application in a model of spinal cord injury. Oxidative Med. Cell. Longev. 2019, 2019, 6493081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamed, H.A.; Said, R.S. Coenzyme Q10 attenuates inflammation and fibrosis implicated in radiation enteropathy through suppression of NF-kB/TGF-β/MMP-9 pathways. Int. Immunopharmacol. 2021, 92, 107347. [Google Scholar] [CrossRef] [PubMed]
- Tanwar, S.; Rhodes, F.; Srivastava, A.; Trembling, P.M.; Rosenberg, W.M. Inflammation and fibrosis in chronic liver diseases including non-alcoholic fatty liver disease and hepatitis C. World J. Gastroenterol. 2020, 26, 109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Chang, X.; Wang, H.; Liu, Y.; Wang, X.; Wu, M.; Zhan, H.; Li, S.; Sun, Y. TGF-β1 mediated Smad signaling pathway and EMT in hepatic fibrosis induced by Nano NiO in vivo and in vitro. Environ. Toxicol. 2020, 35, 419–429. [Google Scholar] [CrossRef]
- Paudel, R.; Raj, K.; Gupta, Y.K.; Singh, S. Oxiracetam and zinc ameliorates autism-like symptoms in propionic acid model of rats. Neurotox. Res. 2020, 37, 815–826. [Google Scholar] [CrossRef]
- Bielefeld, E.C.; Coling, D.; Chen, G.-D.; Henderson, D. Multiple dosing strategies with acetyl L-carnitine (ALCAR) fail to alter age-related hearing loss in the Fischer 344/NHsd rat. J. Negat. Results Biomed. 2008, 7, 4. [Google Scholar] [CrossRef] [Green Version]
- El-Sheikh, A.A.K.; Morsy, M.A.; Mahmoud, M.M.; Rifaai, R.A.; Abdelrahman, A.M. Effect of coenzyme-Q10 on doxorubicin-induced nephrotoxicity in rats. Adv. Pharmacol. Sci. 2012, 2012, 981461. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Marklund, S. A novel superoxide dismutase of high molecular weight from bovine liver. Acta Chem. Scand. 1973, 27, 1458. [Google Scholar] [CrossRef] [PubMed]



| HMGB-1 | Forward |
| 5′-TTGTCCACACACCCTGCATA-3′ | |
| Reverse | |
| 3′-AATTGATCACTCCTTGCTTTGCT-5′ | |
| CK18 | Forward |
| 5′-GAGACGTACAGTCCAGTCCTTGG-3′ | |
| Reverse | |
| 3′-CCACCTCCCTCAGGCTGTT-5′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhusaini, A.M.; Alsoghayer, R.; Alhushan, L.; Alanazi, A.M.; Hasan, I.H. Acetyl-L-Carnitine and Liposomal Co-Enzyme Q10 Attenuate Hepatic Inflammation, Apoptosis, and Fibrosis Induced by Propionic Acid. Int. J. Mol. Sci. 2023, 24, 11519. https://doi.org/10.3390/ijms241411519
Alhusaini AM, Alsoghayer R, Alhushan L, Alanazi AM, Hasan IH. Acetyl-L-Carnitine and Liposomal Co-Enzyme Q10 Attenuate Hepatic Inflammation, Apoptosis, and Fibrosis Induced by Propionic Acid. International Journal of Molecular Sciences. 2023; 24(14):11519. https://doi.org/10.3390/ijms241411519
Chicago/Turabian StyleAlhusaini, Ahlam M., Rahaf Alsoghayer, Lina Alhushan, Abeer M. Alanazi, and Iman H. Hasan. 2023. "Acetyl-L-Carnitine and Liposomal Co-Enzyme Q10 Attenuate Hepatic Inflammation, Apoptosis, and Fibrosis Induced by Propionic Acid" International Journal of Molecular Sciences 24, no. 14: 11519. https://doi.org/10.3390/ijms241411519






