The Anti-Caries Effects of a Novel Peptide on Dentine Caries: An In Vitro Study
Abstract
:1. Introduction
2. Results
2.1. Effect of GAPI Peptide on Biofilm
2.2. Effects of GAPI Peptide on Hard Tissue
3. Discussion
4. Materials and Methods
4.1. Synthesisation of Peptide
4.2. Preparation of Dentine Blocks with Artificial Carious Lesions
4.3. Experimental Treatment
4.4. Antibiofilm Effect of GAPI Peptide
4.4.1. Biofilm Viability
4.4.2. Biofilm Kinetics
4.4.3. Biofilm Morphology
4.5. Effect of GAPI Peptide on Hard Tissue
4.5.1. Lesion Depth and Mineral Loss
4.5.2. Chemical Structure
4.5.3. Surface Morphology and Cross-Sectional Morphology
4.5.4. Crystal Characteristics
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Caufield, P.W.; Li, Y.; Dasanayake, A. Dental caries: An infectious and transmissible disease. Compend. Contin. Educ. Dent. 2005, 26, 10–16. [Google Scholar] [PubMed]
- Machiulskiene, V.; Campus, G.; Carvalho, J.C.; Dige, I.; Ekstrand, K.R.; Jablonski-Momeni, A.; Maltz, M.; Manton, D.J.; Martignon, S.; Martinez-Mier, E.A.; et al. Terminology of Dental Caries and Dental Caries Management: Consensus Report of a Workshop Organized by ORCA and Cariology Research Group of IADR. Caries Res. 2020, 54, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Daliri, E.B.-M.; Tyagi, A.; Oh, D.-H. Cariogenic Biofilm: Pathology-Related Phenotypes and Targeted Therapy. Microorganisms 2021, 9, 1311. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.; Xiao, J.; Klein, M.I.; Jeon, J.G. Exopolysaccharides Produced by Streptococcus mutans Glucosyltransferases Modulate the Establishment of Microcolonies within Multispecies Biofilms. J. Bacteriol. 2010, 192, 3024–3032. [Google Scholar] [CrossRef]
- Shellis, R.P.; Barbour, M.E.; Jones, S.B.; Addy, M. Effects of pH and acid concentration on erosive dissolution of enamel, dentine, and compressed hydroxyapatite. Eur. J. Oral Sci. 2010, 118, 475–482. [Google Scholar] [CrossRef]
- Takahashi, N.; Nyvad, B. The Role of Bacteria in the Caries Process: Ecological perspectives. J. Dent. Res. 2011, 90, 294–303. [Google Scholar] [CrossRef]
- Federation, F.W.D. FDI policy statement on Minimal Intervention Dentistry (MID) for managing dental caries: Adopted by the General Assembly: September 2016, Poznan, Poland. Int. Dent. J. 2017, 67, 6–7. [Google Scholar] [CrossRef]
- Zhang, O.L.; Niu, J.Y.; Yin, I.X.; Yu, O.Y.; Mei, M.L.; Chu, C.H. Bioactive Materials for Caries Management: A Literature Review. Dent. J. 2023, 11, 59. [Google Scholar] [CrossRef]
- Zhang, O.L.; Niu, J.Y.; Yin, I.X.; Yu, O.Y.; Mei, M.L.; Chu, C.H. Growing Global Research Interest in Antimicrobial Peptides for Caries Management: A Bibliometric Analysis. J. Funct. Biomater. 2022, 13, 210. [Google Scholar] [CrossRef]
- Kumar, P.; Kizhakkedathu, J.N.; Straus, S.K. Antimicrobial Peptides: Diversity, Mechanism of Action and Strategies to Improve the Activity and Biocompatibility In Vivo. Biomolecules 2018, 8, 4. [Google Scholar] [CrossRef]
- Lei, J.; Sun, L.; Huang, S.; Zhu, C.; Li, P.; He, J.; Mackey, V.; Coy, D.H.; He, Q. The antimicrobial peptides and their potential clinical applications. Am. J. Transl. Res. 2019, 11, 3919–3931. [Google Scholar] [PubMed]
- Mai, S.; Mauger, M.T.; Niu, L.-N.; Barnes, J.B.; Kao, S.; Bergeron, B.E.; Ling, J.-Q.; Tay, F.R. Potential applications of antimicrobial peptides and their mimics in combating caries and pulpal infections. Acta Biomater. 2017, 49, 16–35. [Google Scholar] [CrossRef]
- Zhang, Q.-Y.; Yan, Z.-B.; Meng, Y.-M.; Hong, X.-Y.; Shao, G.; Ma, J.-J.; Cheng, X.-R.; Liu, J.; Kang, J.; Fu, C.-Y. Antimicrobial peptides: Mechanism of action, activity and clinical potential. Mil. Med. Res. 2021, 8, 48. [Google Scholar] [CrossRef]
- Niu, J.Y.; Yin, I.X.; Wu, W.K.K.; Li, Q.-L.; Mei, M.L.; Chu, C.H. Efficacy of the dual-action GA-KR12 peptide for remineralising initial enamel caries: An in vitro study. Clin. Oral Investig. 2022, 26, 2441–2451. [Google Scholar] [CrossRef] [PubMed]
- Pfalzgraff, A.; Brandenburg, K.; Weindl, G. Antimicrobial Peptides and Their Therapeutic Potential for Bacterial Skin Infections and Wounds. Front. Pharmacol. 2018, 9, 281. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shen, Y.; Haapasalo, M. Antibiofilm peptides against oral biofilms. J. Oral Microbiol. 2017, 9, 1327308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.-Y.; Fang, Z.-H.; Li, Q.-L.; Cao, C.Y. A tooth-binding antimicrobial peptide to prevent the formation of dental biofilm. J. Mater. Sci. Mater. Med. 2019, 30, 45. [Google Scholar] [CrossRef]
- Guo, L.; McLean, J.S.; Yang, Y.; Eckert, R.; Kaplan, C.W.; Kyme, P.; Sheikh, O.; Varnum, B.; Lux, R.; Shi, W.; et al. Precision-guided antimicrobial peptide as a targeted modulator of human microbial ecology. Proc. Natl. Acad. Sci. USA 2015, 112, 7569–7574. [Google Scholar] [CrossRef]
- Namburu, J.R.; Sanosh, A.B.R.; Poosarla, C.S.; Manthapuri, S.; Pinnaka, M.; Baddam, V.R.R. Streptococcus mutans-Specific Antimicrobial Peptide C16G2-Mediated Caries Prevention: A Review. Front. Dent. 2022, 19, 17. [Google Scholar] [CrossRef]
- Zhang, O.L.; Niu, J.Y.; Yin, I.X.; Yu, O.Y.; Mei, M.L.; Chu, C.H. Antibacterial Properties of the Antimicrobial Peptide Gallic Acid-Polyphemusin I (GAPI). Antibiotics 2023, 12, 1350. [Google Scholar] [CrossRef]
- Goldberg, M.; Kulkarni, A.B.; Young, M.; Boskey, A. Dentin structure composition and mineralization. Front. Biosci. 2011, E3, 711–735. [Google Scholar] [CrossRef] [PubMed]
- Mazzoni, A.; Tjäderhane, L.; Checchi, V.; Di Lenarda, R.; Salo, T.; Tay, F.; Pashley, D.; Breschi, L. Role of Dentin MMPs in Caries Progression and Bond Stability. J. Dent. Res. 2014, 94, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Raheem, N.; Straus, S.K. Mechanisms of Action for Antimicrobial Peptides with Antibacterial and Antibiofilm Functions. Front. Microbiol. 2019, 10, 2866. [Google Scholar] [CrossRef] [PubMed]
- Edwards, I.A.; Elliott, A.G.; Kavanagh, A.M.; Zuegg, J.; Blaskovich, M.A.T.; Cooper, M.A. Contribution of Amphipathicity and Hydrophobicity to the Antimicrobial Activity and Cytotoxicity of β-Hairpin Peptides. ACS Infect. Dis. 2016, 2, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Amiss, A.S.; von Pein, J.B.; Webb, J.R.; Condon, N.D.; Harvey, P.J.; Phan, M.-D.; Schembri, M.A.; Currie, B.J.; Sweet, M.J.; Craik, D.J.; et al. Modified horseshoe crab peptides target and kill bacteria inside host cells. Cell. Mol. Life Sci. 2021, 79, 38. [Google Scholar] [CrossRef]
- Guidotti, G.; Brambilla, L.; Rossi, D. Cell-Penetrating Peptides: From Basic Research to Clinics. Trends Pharmacol. Sci. 2017, 38, 406–424. [Google Scholar] [CrossRef]
- González-Cabezas, C.; Fernández, C. Recent Advances in Remineralization Therapies for Caries Lesions. Adv. Dent. Res. 2018, 29, 55–59. [Google Scholar] [CrossRef]
 ) and damage to bacterial cell membranes (
) and damage to bacterial cell membranes ( ).
).
 ) and damage to bacterial cell membranes (
) and damage to bacterial cell membranes ( ).
).

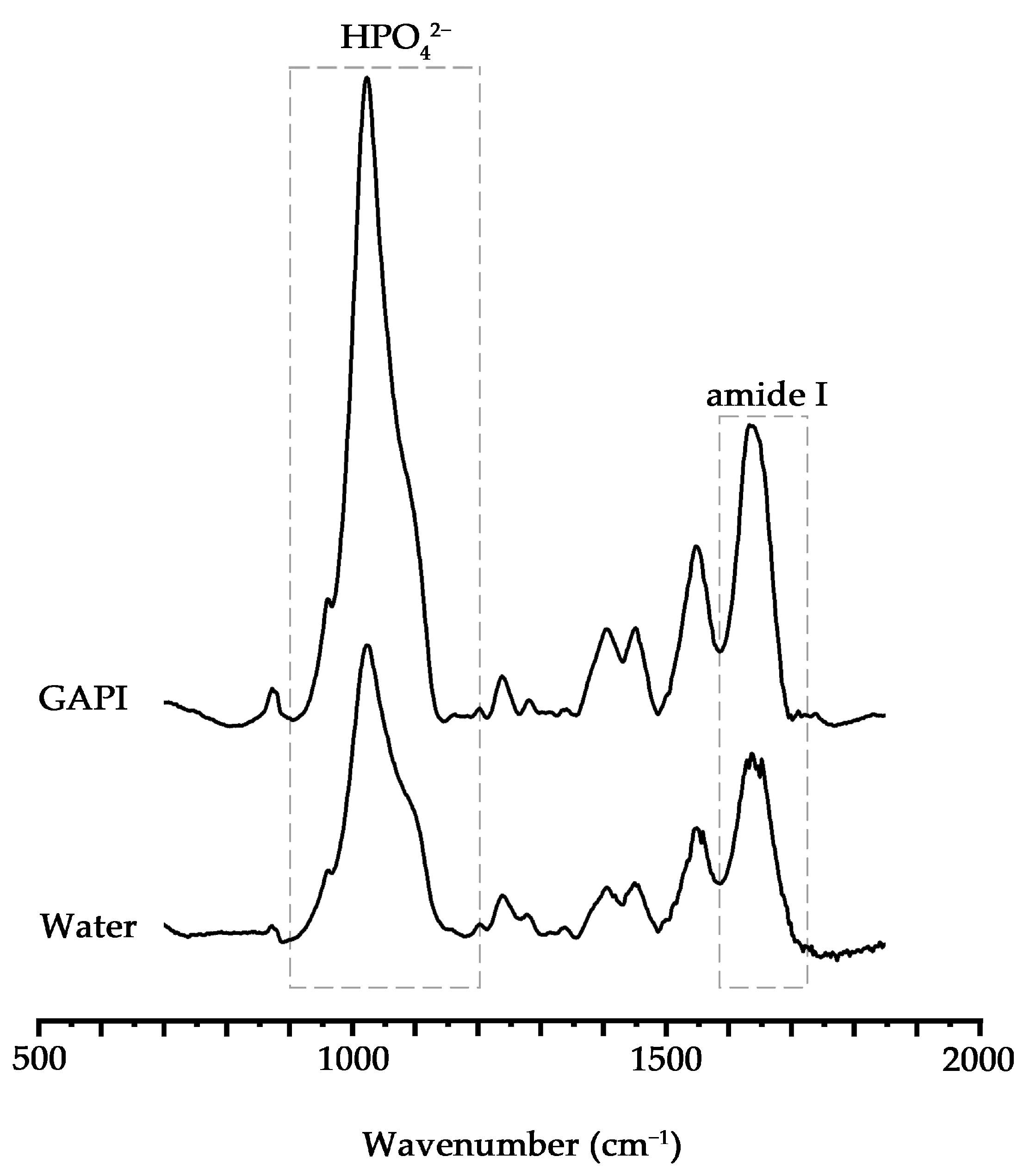
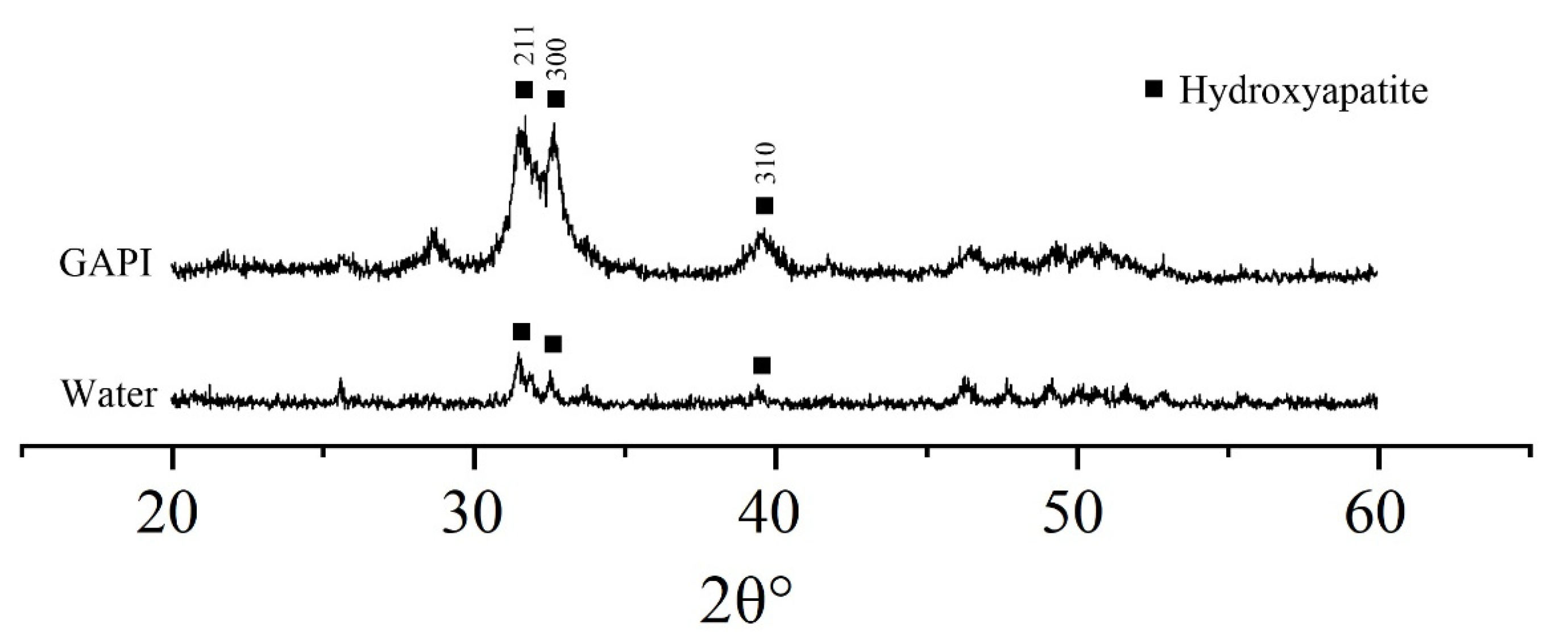
 ) indicating remineralisation. In contrast, the dentin surface appeared rough with serious exposure of collagen fibres in the control group.
) indicating remineralisation. In contrast, the dentin surface appeared rough with serious exposure of collagen fibres in the control group.
 ) indicating remineralisation. In contrast, the dentin surface appeared rough with serious exposure of collagen fibres in the control group.
) indicating remineralisation. In contrast, the dentin surface appeared rough with serious exposure of collagen fibres in the control group.


| Group | GAPI | Water | p Value |
|---|---|---|---|
| S. mutans biofilm assessment | |||
| Viability: Dead-to-live ratio (n = 8) | 0.77 ± 0.13 | 0.37 ± 0.09 | <0.001 |
| Kinetics: Log10 CFU (n = 8) | 7.45 ± 0.32 | 8.74 ± 0.50 | <0.001 |
| Carious dentine lesion assessment | |||
| Lesion depth, µm (n = 8) | 151 ± 18 | 214 ± 15 | <0.001 |
| Mineral loss, gHAcm−3 (n = 8) | 0.91 ± 0.07 | 1.01 ± 0.07 | 0.01 |
| Hydrogen phosphate-to-amide I ratio (n = 8) | 2.92 ± 0.82 | 1.83 ± 0.73 | 0.014 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, O.L.; Niu, J.Y.; Yu, O.Y.; Yin, I.X.; Mei, M.L.; Chu, C.H. The Anti-Caries Effects of a Novel Peptide on Dentine Caries: An In Vitro Study. Int. J. Mol. Sci. 2023, 24, 14076. https://doi.org/10.3390/ijms241814076
Zhang OL, Niu JY, Yu OY, Yin IX, Mei ML, Chu CH. The Anti-Caries Effects of a Novel Peptide on Dentine Caries: An In Vitro Study. International Journal of Molecular Sciences. 2023; 24(18):14076. https://doi.org/10.3390/ijms241814076
Chicago/Turabian StyleZhang, Olivia Lili, John Yun Niu, Ollie Yiru Yu, Iris Xiaoxue Yin, May Lei Mei, and Chun Hung Chu. 2023. "The Anti-Caries Effects of a Novel Peptide on Dentine Caries: An In Vitro Study" International Journal of Molecular Sciences 24, no. 18: 14076. https://doi.org/10.3390/ijms241814076
APA StyleZhang, O. L., Niu, J. Y., Yu, O. Y., Yin, I. X., Mei, M. L., & Chu, C. H. (2023). The Anti-Caries Effects of a Novel Peptide on Dentine Caries: An In Vitro Study. International Journal of Molecular Sciences, 24(18), 14076. https://doi.org/10.3390/ijms241814076









