The Multifaceted Functions of TRPV4 and Calcium Oscillations in Tissue Repair
Abstract
:1. Introduction
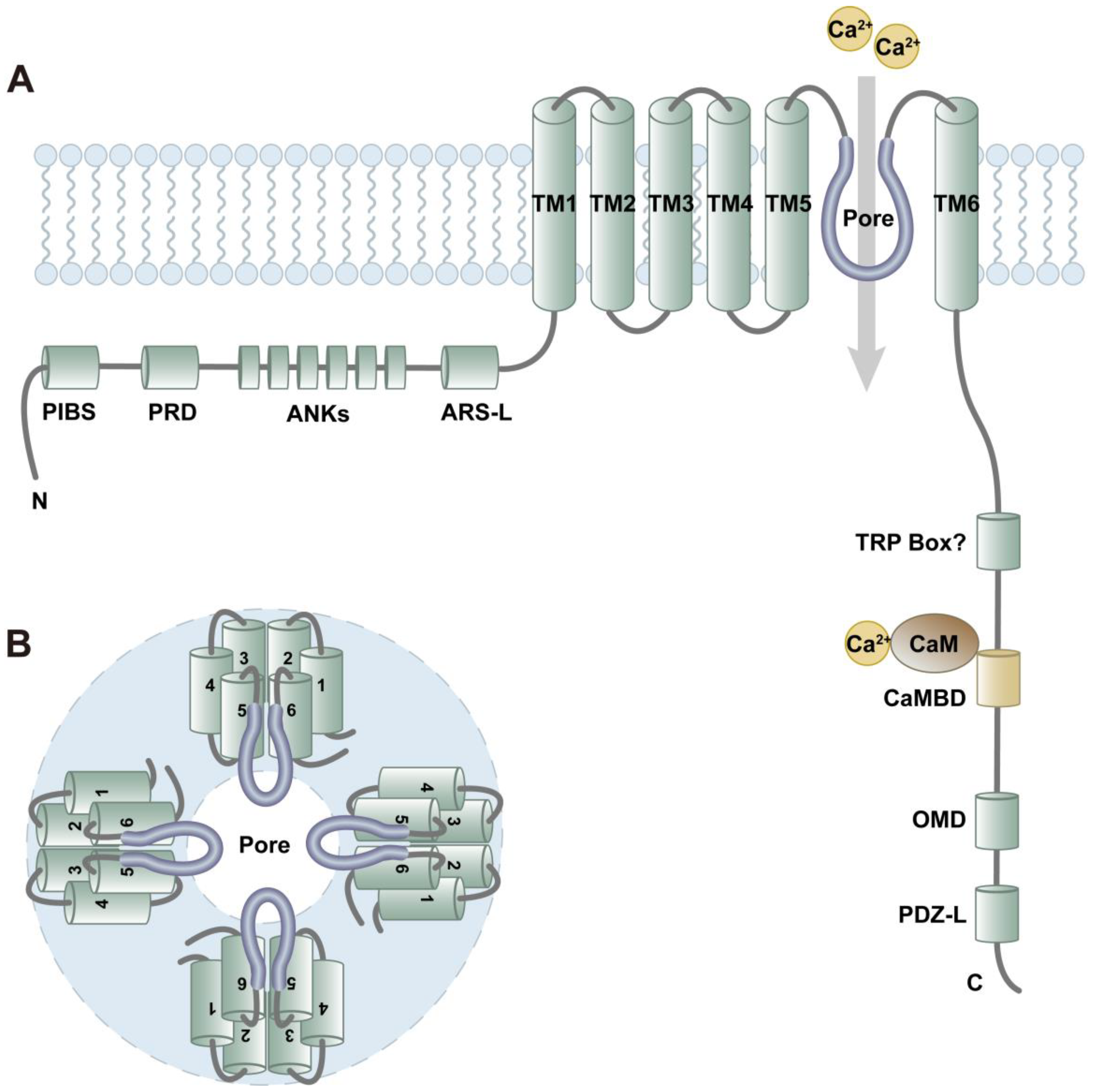
2. Protein and Channel Structure of TRPV4
3. TRPV4 Signaling in Mechanotransduction
4. TRPV4 Triggers Ca2+ Oscillations
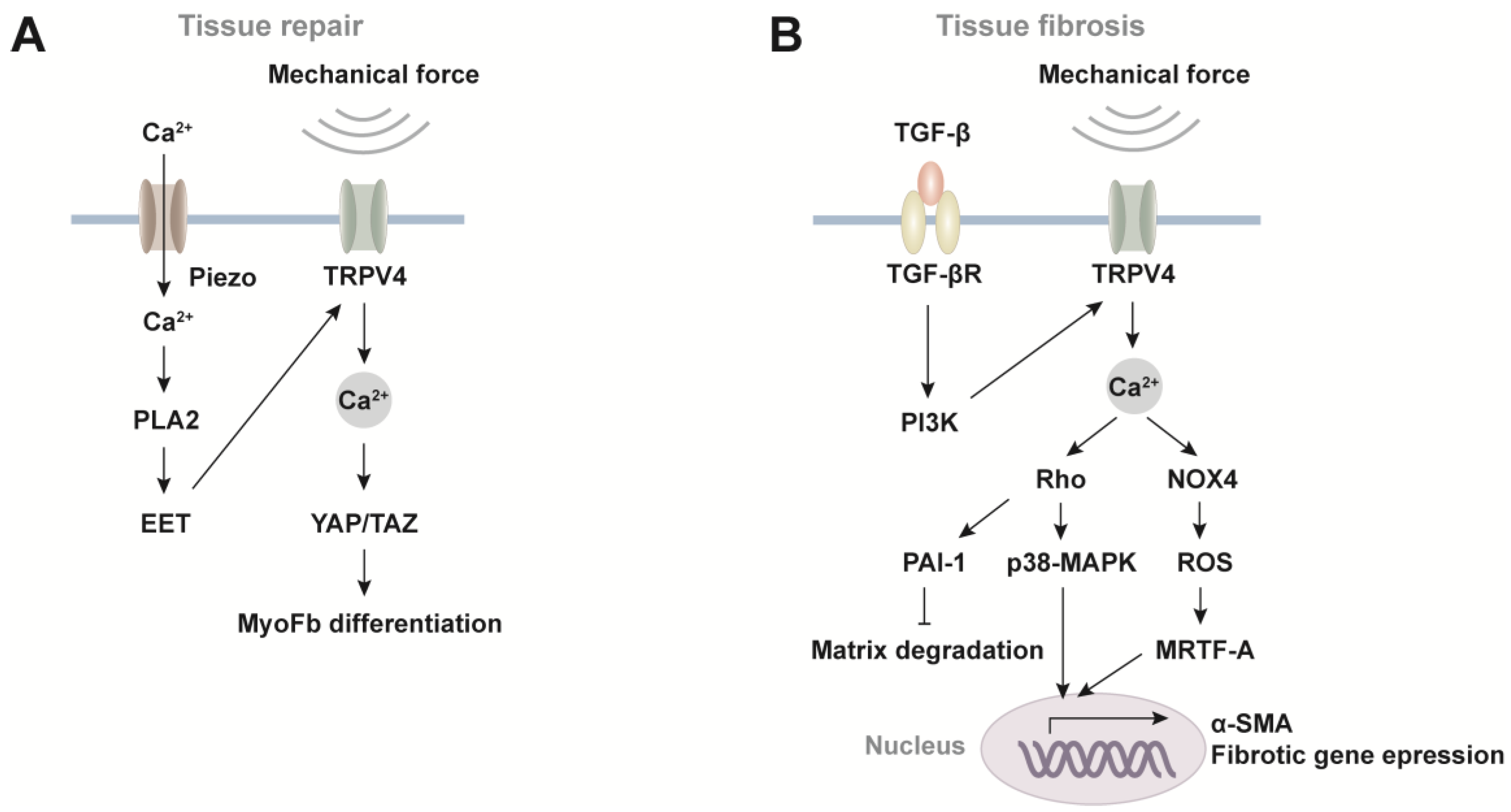
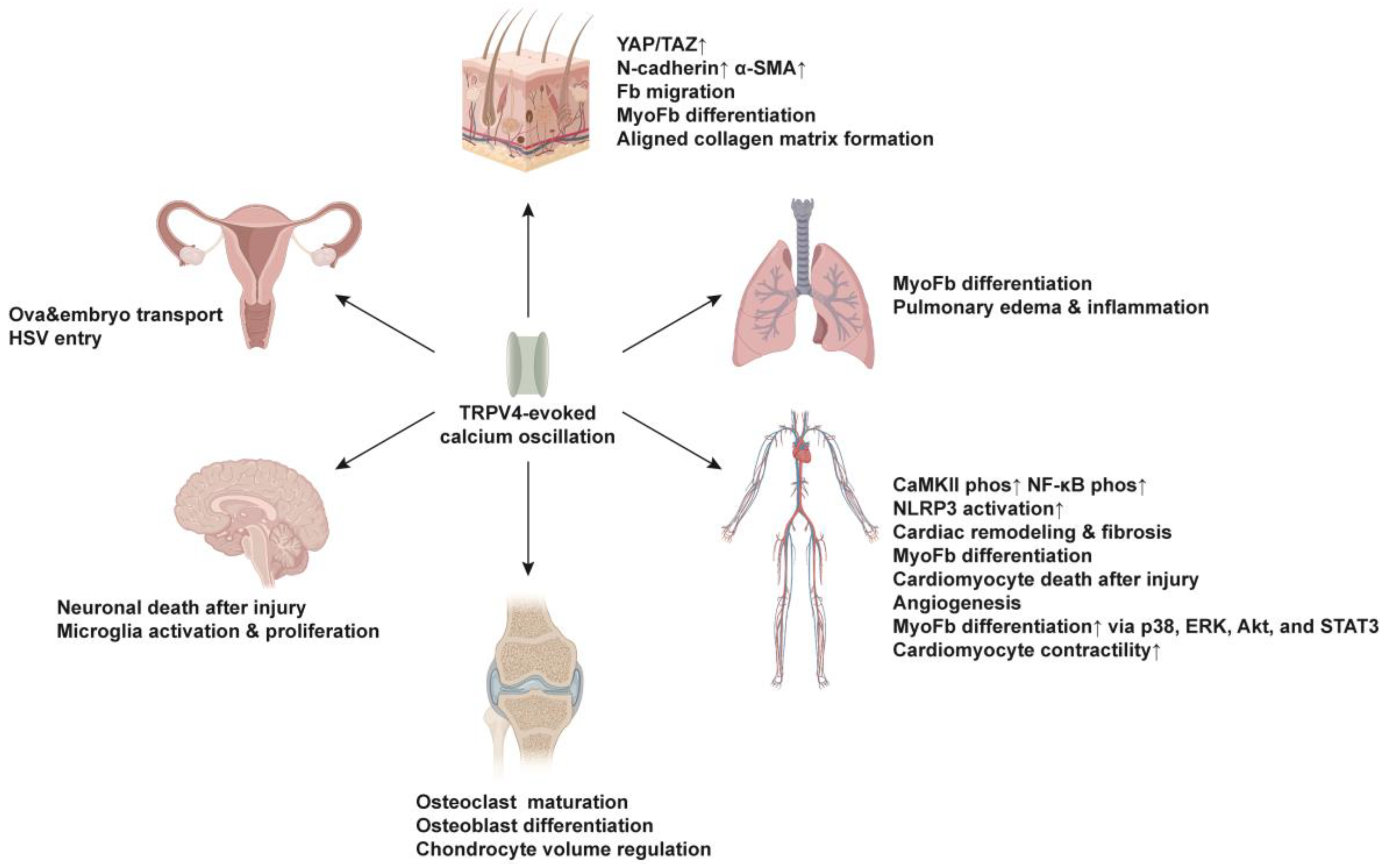
5. Function of TRPV4-Evoked Ca2+ Oscillations in Injury Repair and Fibrosis
5.1. Skin
5.2. Lung
5.3. Cardiovascular System
5.4. Skeletal System
5.5. Nervous System
5.6. Reproductive System
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- White, J.P.; Cibelli, M.; Urban, L.; Nilius, B.; McGeown, J.G.; Nagy, I. TRPV4: Molecular Conductor of a Diverse Orchestra. Physiol. Rev. 2016, 96, 911–973. [Google Scholar] [PubMed]
- Venkatachalam, K.; Montell, C. TRP channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.M. TRP Channels as Potential Drug Targets. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 309–330. [Google Scholar] [PubMed]
- Cosens, D.J.; Manning, A. Abnormal electroretinogram from a Drosophila mutant. Nature 1969, 224, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Montell, C. The TRP superfamily of cation channels. Sci. STKE 2005, 2005, re3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Ma, Y.; Ye, X.; Zhang, N.; Pan, L.; Wang, B. TRP (transient receptor potential) ion channel family: Structures, biological functions and therapeutic interventions for diseases. Signal Transduct. Target. Ther. 2023, 8, 261. [Google Scholar] [PubMed]
- Himmel, N.J.; Cox, D.N. Transient receptor potential channels: Current perspectives on evolution, structure, function and nomenclature. Proc. Biol. Sci. 2020, 287, 20201309. [Google Scholar]
- Rosenbaum, T.; Islas, L.D. Molecular Physiology of TRPV Channels: Controversies and Future Challenges. Annu. Rev. Physiol. 2023, 85, 293–316. [Google Scholar] [CrossRef]
- Ho, C.Y.; Gu, Q.; Lin, Y.S.; Lee, L.Y. Sensitivity of vagal afferent endings to chemical irritants in the rat lung. Respir. Physiol. 2001, 127, 113–124. [Google Scholar] [CrossRef]
- Bohlen, C.J.; Priel, A.; Zhou, S.; King, D.; Siemens, J.; Julius, D. A bivalent tarantula toxin activates the capsaicin receptor, TRPV1, by targeting the outer pore domain. Cell 2010, 141, 834–845. [Google Scholar] [CrossRef]
- Shibasaki, K.; Murayama, N.; Ono, K.; Ishizaki, Y.; Tominaga, M. TRPV2 enhances axon outgrowth through its activation by membrane stretch in developing sensory and motor neurons. J. Neurosci. 2010, 30, 4601–4612. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Biro, T.; Owsianik, G. TRPV3: Time to decipher a poorly understood family member! J. Physiol. 2014, 592, 295–304. [Google Scholar]
- Liedtke, W. TRPV4 plays an evolutionary conserved role in the transduction of osmotic and mechanical stimuli in live animals. J. Physiol. 2005, 567 Pt 1, 53–58. [Google Scholar] [PubMed]
- Liedtke, W.; Choe, Y.; Martí-Renom, M.A.; Bell, A.M.; Denis, C.S.; Sali, A.; Hudspeth, A.J.; Friedman, J.M.; Heller, S. Vanilloid receptor-related osmotically activated channel (VR-OAC), a candidate vertebrate osmoreceptor. Cell 2000, 103, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Strotmann, R.; Harteneck, C.; Nunnenmacher, K.; Schultz, G.; Plant, T.D. OTRPC4, a nonselective cation channel that confers sensitivity to extracellular osmolarity. Nat. Cell Biol. 2000, 2, 695–702. [Google Scholar] [CrossRef]
- Delany, N.S.; Hurle, M.; Facer, P.; Alnadaf, T.; Plumpton, C.; Kinghorn, I.; See, C.G.; Costigan, M.; Anand, P.; Woolf, C.J.; et al. Identification and characterization of a novel human vanilloid receptor-like protein, VRL-2. Physiol. Genom. 2001, 4, 165–174. [Google Scholar] [CrossRef]
- Nilius, B.; Prenen, J.; Wissenbach, U.; Bödding, M.; Droogmans, G. Differential activation of the volume-sensitive cation channel TRP12 (OTRPC4) and volume-regulated anion currents in HEK-293 cells. Pflug. Arch. 2001, 443, 227–233. [Google Scholar]
- Güler, A.D.; Lee, H.; Iida, T.; Shimizu, I.; Tominaga, M.; Caterina, M. Heat-evoked activation of the ion channel, TRPV4. J. Neurosci. 2002, 22, 6408–6414. [Google Scholar] [CrossRef]
- Laing, R.J.; Dhaka, A. ThermoTRPs and Pain. Neuroscientist 2016, 22, 171–187. [Google Scholar] [CrossRef]
- Hong, K.; Cope, E.L.; DeLalio, L.J.; Marziano, C.; Isakson, B.E.; Sonkusare, S.K. TRPV4 (Transient Receptor Potential Vanilloid 4) Channel-Dependent Negative Feedback Mechanism Regulates G(q) Protein-Coupled Receptor-Induced Vasoconstriction. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 542–554. [Google Scholar] [CrossRef]
- Luo, J.; Qian, A.; Oetjen, L.K.; Yu, W.; Yang, P.; Feng, J.; Xie, Z.; Liu, S.; Yin, S.; Dryn, D.; et al. TRPV4 Channel Signaling in Macrophages Promotes Gastrointestinal Motility via Direct Effects on Smooth Muscle Cells. Immunity 2018, 49, 107–119.e4. [Google Scholar] [PubMed]
- Szabó, I.L.; Lisztes, E.; Béke, G.; Tóth, K.F.; Paus, R.; Oláh, A.; Bíró, T. The Phytocannabinoid (-)-Cannabidiol Operates as a Complex, Differential Modulator of Human Hair Growth: Anti-Inflammatory Submicromolar versus Hair Growth Inhibitory Micromolar Effects. J. Investig. Dermatol. 2020, 140, 484–488.e5. [Google Scholar] [PubMed]
- Szabó, I.L.; Herczeg-Lisztes, E.; Szegedi, A.; Nemes, B.; Paus, R.; Bíró, T.; Szöllősi, A.G. TRPV4 Is Expressed in Human Hair Follicles and Inhibits Hair Growth In Vitro. J. Investig. Dermatol. 2019, 139, 1385–1388. [Google Scholar] [CrossRef] [PubMed]
- She, G.; Hou, M.C.; Zhang, Y.; Zhang, Y.; Wang, Y.; Wang, H.F.; Lai, B.C.; Zhao, W.B.; Du, X.J.; Deng, X.L. Gal-3 (Galectin-3) and K(Ca)3.1 Mediate Heterogeneous Cell Coupling and Myocardial Fibrogenesis Driven by βAR (β-Adrenoceptor) Activation. Hypertension 2020, 75, 393–404. [Google Scholar] [CrossRef]
- Zhan, L.; Li, J. The role of TRPV4 in fibrosis. Gene 2018, 642, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, S.O.; Grove, L.M.; Paruchuri, S.; Southern, B.D.; Abraham, S.; Niese, K.A.; Scheraga, R.G.; Ghosh, S.; Thodeti, C.K.; Zhang, D.X.; et al. TRPV4 mediates myofibroblast differentiation and pulmonary fibrosis in mice. J. Clin. Investig. 2014, 124, 5225–5238. [Google Scholar] [CrossRef] [PubMed]
- Goswami, R.; Cohen, J.; Sharma, S.; Zhang, D.X.; Lafyatis, R.; Bhawan, J.; Rahaman, S.O. TRPV4 ION Channel Is Associated with Scleroderma. J. Investig. Dermatol. 2017, 137, 962–965. [Google Scholar] [CrossRef]
- Saez, F.; Hong, N.J.; Cabral, P.D.; Garvin, J.L. Stretch-Induced Increases in Intracellular Ca Stimulate Thick Ascending Limb O2−) Production and Are Enhanced in Dahl Salt-Sensitive Rats. Hypertension 2020, 75, 431–438. [Google Scholar] [CrossRef]
- Greenstein, A.S.; Kadir, S.; Csato, V.; Sugden, S.A.; Baylie, R.A.; Eisner, D.A.; Nelson, M.T. Disruption of Pressure-Induced Ca2+ Spark Vasoregulation of Resistance Arteries, Rather Than Endothelial Dysfunction, Underlies Obesity-Related Hypertension. Hypertension 2020, 75, 539–548. [Google Scholar] [CrossRef]
- Tang, B.; Wu, J.; Zhu, M.X.; Sun, X.; Liu, J.; Xie, R.; Dong, T.X.; Xiao, Y.; Carethers, J.M.; Yang, S.; et al. VPAC1 couples with TRPV4 channel to promote calcium-dependent gastric cancer progression via a novel autocrine mechanism. Oncogene 2019, 38, 3946–3961. [Google Scholar] [CrossRef]
- Xie, R.; Xu, J.; Xiao, Y.; Wu, J.; Wan, H.; Tang, B.; Liu, J.; Fan, Y.; Wang, S.; Wu, Y.; et al. Calcium Promotes Human Gastric Cancer via a Novel Coupling of Calcium-Sensing Receptor and TRPV4 Channel. Cancer Res. 2017, 77, 6499–6512. [Google Scholar] [PubMed]
- Mascarenhas, N.L.; Wang, Z.; Chang, Y.L.; Di Nardo, A. TRPV4 Mediates Mast Cell Activation in Cathelicidin-Induced Rosacea Inflammation. J. Investig. Dermatol. 2017, 137, 972–975. [Google Scholar] [PubMed]
- Chen, Y.; Moore, C.D.; Zhang, J.Y.; Hall, R.P., 3rd; MacLeod, A.S.; Liedtke, W. TRPV4 Moves toward Center-Fold in Rosacea Pathogenesis. J. Investig. Dermatol. 2017, 137, 801–804. [Google Scholar] [CrossRef]
- Gonzalez-Lopez, A.; Lopez-Alonso, I.; Pickerodt, P.A.; von Haefen, C.; Amado-Rodriguez, L.; Reimann, H.; Niendorf, T.; Kuebler, W.; Albaiceta, G.M.; Francis, R.C.E.; et al. Lung Purinoceptor Activation Triggers Ventilator-Induced Brain Injury. Crit. Care Med. 2019, 47, e911–e918. [Google Scholar] [CrossRef]
- Gorelick, F.; Nathanson, M.H. TRPV4 helps Piezo1 put the squeeze on pancreatic acinar cells. J. Clin. Investig. 2020, 130, 2199–2201. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Romac, J.M.; Shahid, R.A.; Pandol, S.J.; Liedtke, W.; Vigna, S.R.; Liddle, R.A. TRPV4 channel opening mediates pressure-induced pancreatitis initiated by Piezo1 activation. J. Clin. Investig. 2020, 130, 2527–2541. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Paknejad, N.; Maksaev, G.; Sala-Rabanal, M.; Nichols, C.G.; Hite, R.K.; Yuan, P. Cryo-EM and X-ray structures of TRPV4 reveal insight into ion permeation and gating mechanisms. Nat. Struct. Mol. Biol. 2018, 25, 252–260. [Google Scholar]
- Garcia-Elias, A.; Mrkonjic, S.; Pardo-Pastor, C.; Inada, H.; Hellmich, U.A.; Rubio-Moscardó, F.; Plata, C.; Gaudet, R.; Vicente, R.; Valverde, M.A. Phosphatidylinositol-4,5-biphosphate-dependent rearrangement of TRPV4 cytosolic tails enables channel activation by physiological stimuli. Proc. Natl. Acad. Sci. USA 2013, 110, 9553–9558. [Google Scholar]
- Cuajungco, M.P.; Grimm, C.; Oshima, K.; D’Hoedt, D.; Nilius, B.; Mensenkamp, A.R.; Bindels, R.J.; Plomann, M.; Heller, S. PACSINs bind to the TRPV4 cation channel. PACSIN 3 modulates the subcellular localization of TRPV4. J. Biol. Chem. 2006, 281, 18753–18762. [Google Scholar] [CrossRef]
- Inada, H.; Procko, E.; Sotomayor, M.; Gaudet, R. Structural and biochemical consequences of disease-causing mutations in the ankyrin repeat domain of the human TRPV4 channel. Biochemistry 2012, 51, 6195–6206. [Google Scholar]
- Nilius, B.; Watanabe, H.; Vriens, J. The TRPV4 channel: Structure-function relationship and promiscuous gating behaviour. Pflug. Arch. 2003, 446, 298–303. [Google Scholar]
- Nilius, B.; Flockerzi, V. Mammalian transient receptor potential (TRP) cation channels. Preface. Handb. Exp. Pharmacol. 2014, 223, v–vi. [Google Scholar] [PubMed]
- Garcia-Elias, A.; Lorenzo, I.M.; Vicente, R.; Valverde, M.A. IP3 receptor binds to and sensitizes TRPV4 channel to osmotic stimuli via a calmodulin-binding site. J. Biol. Chem. 2008, 283, 31284–31288. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.; Müller, M.; Leuner, K.; Jendrach, M. The C-terminal domain of TRPV4 is essential for plasma membrane localization. Mol. Membr. Biol. 2008, 25, 139–151. [Google Scholar]
- Strotmann, R.; Schultz, G.; Plant, T.D. Ca2+-dependent potentiation of the nonselective cation channel TRPV4 is mediated by a C-terminal calmodulin binding site. J. Biol. Chem. 2003, 278, 26541–26549. [Google Scholar] [CrossRef] [PubMed]
- Voets, T.; Prenen, J.; Vriens, J.; Watanabe, H.; Janssens, A.; Wissenbach, U.; Bödding, M.; Droogmans, G.; Nilius, B. Molecular determinants of permeation through the cation channel TRPV4. J. Biol. Chem. 2002, 277, 33704–33710. [Google Scholar]
- Chaigne, S.; Barbeau, S.; Ducret, T.; Guinamard, R.; Benoist, D. Pathophysiological Roles of the TRPV4 Channel in the Heart. Cells 2023, 12, 1654. [Google Scholar] [CrossRef]
- Zhang, M.; Meng, N.; Wang, X.; Chen, W.; Zhang, Q. TRPV4 and PIEZO Channels Mediate the Mechanosensing of Chondrocytes to the Biomechanical Microenvironment. Membranes 2022, 12, 237. [Google Scholar]
- Lee, W.; Leddy, H.A.; Chen, Y.; Lee, S.H.; Zelenski, N.A.; McNulty, A.L.; Wu, J.; Beicker, K.N.; Coles, J.; Zauscher, S.; et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc. Natl. Acad. Sci. USA 2014, 111, E5114-22. [Google Scholar]
- Batan, D.; Peters, D.K.; Schroeder, M.E.; Aguado, B.A.; Young, M.W.; Weiss, R.M.; Anseth, K.S. Hydrogel cultures reveal Transient Receptor Potential Vanilloid 4 regulation of myofibroblast activation and proliferation in valvular interstitial cells. FASEB J. 2022, 36, e22306. [Google Scholar]
- Sharma, S.; Goswami, R.; Zhang, D.X.; Rahaman, S.O. TRPV4 regulates matrix stiffness and TGFβ1-induced epithelial-mesenchymal transition. J. Cell Mol. Med. 2019, 23, 761–774. [Google Scholar] [CrossRef] [PubMed]
- Al-Azzam, N.; Teegala, L.R.; Pokhrel, S.; Ghebreigziabher, S.; Chachkovskyy, T.; Thodeti, S.; Gavilanes, I.; Covington, K.; Thodeti, C.K.; Paruchuri, S. Transient Receptor Potential Vanilloid channel regulates fibroblast differentiation and airway remodeling by modulating redox signals through NADPH Oxidase 4. Sci. Rep. 2020, 10, 9827. [Google Scholar] [CrossRef] [PubMed]
- Gombedza, F.; Kondeti, V.; Al-Azzam, N.; Koppes, S.; Duah, E.; Patil, P.; Hexter, M.; Phillips, D.; Thodeti, C.K.; Paruchuri, S. Mechanosensitive transient receptor potential vanilloid 4 regulates Dermatophagoides farinae-induced airway remodeling via 2 distinct pathways modulating matrix synthesis and degradation. FASEB J. 2017, 31, 1556–1570. [Google Scholar]
- Adapala, R.K.; Katari, V.; Teegala, L.R.; Thodeti, S.; Paruchuri, S.; Thodeti, C.K. TRPV4 Mechanotransduction in Fibrosis. Cells 2021, 10, 3053. [Google Scholar] [CrossRef]
- Adapala, R.K.; Kanugula, A.K.; Paruchuri, S.; Chilian, W.M.; Thodeti, C.K. TRPV4 deletion protects heart from myocardial infarction-induced adverse remodeling via modulation of cardiac fibroblast differentiation. Basic. Res. Cardiol. 2020, 115, 14. [Google Scholar] [CrossRef]
- Ahn, M.S.; Eom, Y.W.; Oh, J.E.; Cha, S.K.; Park, K.S.; Son, J.W.; Lee, J.W.; Youn, Y.J.; Ahn, S.G.; Kim, J.Y.; et al. Transient receptor potential channel TRPV4 mediates TGF-β1-induced differentiation of human ventricular fibroblasts. Cardiol. J. 2020, 27, 162–170. [Google Scholar] [PubMed]
- Parekh, A.B. Decoding cytosolic Ca2+ oscillations. Trends Biochem. Sci. 2011, 36, 78–87. [Google Scholar]
- Janssen, L.J.; Farkas, L.; Rahman, T.; Kolb, M.R. ATP stimulates Ca2+-waves and gene expression in cultured human pulmonary fibroblasts. Int. J. Biochem. Cell Biol. 2009, 41, 2477–2484. [Google Scholar] [CrossRef]
- Dunn, K.M.; Hill-Eubanks, D.C.; Liedtke, W.B.; Nelson, M.T. TRPV4 channels stimulate Ca2+-induced Ca2+ release in astrocytic endfeet and amplify neurovascular coupling responses. Proc. Natl. Acad. Sci. USA 2013, 110, 6157–6162. [Google Scholar]
- Lv, M.; Zhou, Y.; Chen, X.; Han, L.; Wang, L.; Lu, X.L. Calcium signaling of in situ chondrocytes in articular cartilage under compressive loading: Roles of calcium sources and cell membrane ion channels. J. Orthop. Res. 2018, 36, 730–738. [Google Scholar] [CrossRef]
- Mukherjee, S.; Duan, F.; Kolb, M.R.; Janssen, L.J. Platelet derived growth factor-evoked Ca2+ wave and matrix gene expression through phospholipase C in human pulmonary fibroblast. Int. J. Biochem. Cell Biol. 2013, 45, 1516–1524. [Google Scholar]
- Logan, A.; Pell, V.R.; Shaffer, K.J.; Evans, C.; Stanley, N.J.; Robb, E.L.; Prime, T.A.; Chouchani, E.T.; Cocheme, H.M.; Fearnley, I.M.; et al. Assessing the Mitochondrial Membrane Potential in Cells and In Vivo using Targeted Click Chemistry and Mass Spectrometry. Cell Metab. 2016, 23, 379–385. [Google Scholar]
- Sack, M.N. Mitochondrial depolarization and the role of uncoupling proteins in ischemia tolerance. Cardiovasc. Res. 2006, 72, 210–219. [Google Scholar] [PubMed]
- Zhang, X.; Lee, M.D.; Buckley, C.; Wilson, C.; McCarron, J.G. Mitochondria regulate TRPV4-mediated release of ATP. Br. J. Pharmacol. 2022, 179, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Xu, S.; Zhang, J.; Li, J.; Shen, Q. Cascade Amplifiers of Intracellular Reactive Oxygen Species Based on Mitochondria-Targeted Core-Shell ZnO-TPP@D/H Nanorods for Breast Cancer Therapy. ACS Appl. Mater. Interfaces 2018, 10, 38749–38759. [Google Scholar] [CrossRef] [PubMed]
- Bhat, E.A.; Sajjad, N. Human Pannexin 1 channel: Insight in structure-function mechanism and its potential physiological roles. Mol. Cell Biochem. 2021, 476, 1529–1540. [Google Scholar] [CrossRef] [PubMed]
- Moesta, A.K.; Li, X.Y.; Smyth, M.J. Targeting CD39 in cancer. Nat. Rev. Immunol. 2020, 20, 739–755. [Google Scholar] [CrossRef]
- Domenici, M.R.; Ferrante, A.; Martire, A.; Chiodi, V.; Pepponi, R.; Tebano, M.T.; Popoli, P. Adenosine A(2A) receptor as potential therapeutic target in neuropsychiatric disorders. Pharmacol. Res. 2019, 147, 104338. [Google Scholar] [CrossRef]
- Fernanda da Rocha, L.; Sérgio José Macedo, J.; Murilo Luiz, C.; Adair Roberto Soares, S. Pharmacology of Adenosine Receptors and Their Signaling Role in Immunity and Inflammation. In Pharmacology and Therapeutics; Gowder, S.G.T., Ed.; IntechOpen: Rijeka, Croatia, 2014; Chapter 4. [Google Scholar]
- Shibasaki, K.; Sugio, S.; Takao, K.; Yamanaka, A.; Miyakawa, T.; Tominaga, M.; Ishizaki, Y. TRPV4 activation at the physiological temperature is a critical determinant of neuronal excitability and behavior. Pflug. Arch. 2015, 467, 2495–2507. [Google Scholar] [CrossRef]
- Shibasaki, K.; Suzuki, M.; Mizuno, A.; Tominaga, M. Effects of body temperature on neural activity in the hippocampus: Regulation of resting membrane potentials by transient receptor potential vanilloid 4. J. Neurosci. 2007, 27, 1566–1575. [Google Scholar] [CrossRef]
- Li, L.; Qu, W.; Zhou, L.; Lu, Z.; Jie, P.; Chen, L.; Chen, L. Activation of Transient Receptor Potential Vanilloid 4 Increases NMDA-Activated Current in Hippocampal Pyramidal Neurons. Front. Cell Neurosci. 2013, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Hong, Z.; Tian, Y.; Qi, M.; Li, Y.; Du, Y.; Chen, L.; Liu, W.; Chen, L. Transient Receptor Potential Vanilloid 4 Inhibits gamma-Aminobutyric Acid-Activated Current in Hippocampal Pyramidal Neurons. Front. Mol. Neurosci. 2016, 9, 77. [Google Scholar] [CrossRef] [PubMed]
- Qi, M.; Wu, C.; Wang, Z.; Zhou, L.; Men, C.; Du, Y.; Huang, S.; Chen, L.; Chen, L. Transient Receptor Potential Vanilloid 4 Activation-Induced Increase in Glycine-Activated Current in Mouse Hippocampal Pyramidal Neurons. Cell Physiol. Biochem. 2018, 45, 1084–1096. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kolb, M.R.; Duan, F.; Janssen, L.J. Transforming growth factor-β evokes Ca2+ waves and enhances gene expression in human pulmonary fibroblasts. Am. J. Respir. Cell Mol. Biol. 2012, 46, 757–764. [Google Scholar]
- Rahman, M.; Mukherjee, S.; Sheng, W.; Nilius, B.; Janssen, L.J. Electrophysiological characterization of voltage-dependent calcium currents and TRPV4 currents in human pulmonary fibroblasts. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 310, L603–L614. [Google Scholar] [CrossRef]
- Shen, J.; Tu, L.; Chen, D.; Tan, T.; Wang, Y.; Wang, S. TRPV4 channels stimulate Ca2+-induced Ca2+ release in mouse neurons and trigger endoplasmic reticulum stress after intracerebral hemorrhage. Brain Res. Bull. 2019, 146, 143–152. [Google Scholar] [CrossRef]
- De Koninck, P.; Schulman, H. Sensitivity of CaM kinase II to the frequency of Ca2+ oscillations. Science 1998, 279, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wu, X.; Du, G.; Chen, W.; Zhang, Q. Substrate stiffness-dependent regulatory volume decrease and calcium signaling in chondrocytes. Acta Biochim. Biophys. Sin. 2022, 54, 113–125. [Google Scholar] [CrossRef]
- Yarishkin, O.; Phuong, T.T.T.; Vazquez-Chona, F.; Bertrand, J.; van Battenburg-Sherwood, J.; Redmon, S.N.; Rudzitis, C.N.; Lakk, M.; Baumann, J.M.; Freichel, M.; et al. Emergent Temporal Signaling in Human Trabecular Meshwork Cells: Role of TRPV4-TRPM4 Interactions. Front. Immunol. 2022, 13, 805076. [Google Scholar] [CrossRef]
- Che, H.; Yue, J.; Tse, H.F.; Li, G.R. Functional TRPV and TRPM channels in human preadipocytes. Pflug. Arch. 2014, 466, 947–959. [Google Scholar] [CrossRef]
- Gilchrist, C.L.; Leddy, H.A.; Kaye, L.; Case, N.D.; Rothenberg, K.E.; Little, D.; Liedtke, W.; Hoffman, B.D.; Guilak, F. TRPV4-mediated calcium signaling in mesenchymal stem cells regulates aligned collagen matrix formation and vinculin tension. Proc. Natl. Acad. Sci. USA 2019, 116, 1992–1997. [Google Scholar] [PubMed]
- Haywood, N.; Ta, H.Q.; Zhang, A.; Charles, E.J.; Rotar, E.; Noona, S.T.; Salmon, M.; Daneva, Z.; Sonkusare, S.K.; Laubach, V.E. Endothelial Transient Receptor Potential Vanilloid 4 Channels Mediate Lung Ischemia-Reperfusion Injury. Ann. Thorac. Surg. 2022, 113, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Ottolini, M.; Hong, K.; Cope, E.L.; Daneva, Z.; DeLalio, L.J.; Sokolowski, J.D.; Marziano, C.; Nguyen, N.Y.; Altschmied, J.; Haendeler, J.; et al. Local Peroxynitrite Impairs Endothelial Transient Receptor Potential Vanilloid 4 Channels and Elevates Blood Pressure in Obesity. Circulation 2020, 141, 1318–1333. [Google Scholar] [PubMed]
- Hatano, N.; Itoh, Y.; Muraki, K. Cardiac fibroblasts have functional TRPV4 activated by 4alpha-phorbol 12,13-didecanoate. Life Sci. 2009, 85, 808–814. [Google Scholar] [CrossRef]
- Liao, J.; Wu, Q.; Qian, C.; Zhao, N.; Zhao, Z.; Lu, K.; Zhang, S.; Dong, Q.; Chen, L.; Li, Q.; et al. TRPV4 blockade suppresses atrial fibrillation in sterile pericarditis rats. JCI Insight 2020, 5, e137528. [Google Scholar] [CrossRef]
- Adapala, R.K.; Thoppil, R.J.; Luther, D.J.; Paruchuri, S.; Meszaros, J.G.; Chilian, W.M.; Thodeti, C.K. TRPV4 channels mediate cardiac fibroblast differentiation by integrating mechanical and soluble signals. J. Mol. Cell Cardiol. 2013, 54, 45–52. [Google Scholar] [CrossRef]
- Che, H.; Xiao, G.S.; Sun, H.Y.; Wang, Y.; Li, G.R. Functional TRPV2 and TRPV4 channels in human cardiac c-kit(+) progenitor cells. J. Cell Mol. Med. 2016, 20, 1118–1127. [Google Scholar]
- Balducci, V.; Faris, P.; Balbi, C.; Costa, A.; Negri, S.; Rosti, V.; Bollini, S.; Moccia, F. The human amniotic fluid stem cell secretome triggers intracellular Ca2+ oscillations, NF-κB nuclear translocation and tube formation in human endothelial colony-forming cells. J. Cell Mol. Med. 2021, 25, 8074–8086. [Google Scholar] [CrossRef]
- Zou, Y.; Zhang, M.; Wu, Q.; Zhao, N.; Chen, M.; Yang, C.; Du, Y.; Han, B. Activation of transient receptor potential vanilloid 4 is involved in pressure overload-induced cardiac hypertrophy. eLife 2022, 11, e74519. [Google Scholar] [CrossRef]
- Peana, D.; Polo-Parada, L.; Domeier, T.L. Arrhythmogenesis in the aged heart following ischaemia-reperfusion: Role of transient receptor potential vanilloid 4. Cardiovasc. Res. 2022, 118, 1126–1137. [Google Scholar]
- Veteto, A.B.; Peana, D.; Lambert, M.D.; McDonald, K.S.; Domeier, T.L. Transient receptor potential vanilloid-4 contributes to stretch-induced hypercontractility and time-dependent dysfunction in the aged heart. Cardiovasc. Res. 2020, 116, 1887–1896. [Google Scholar] [PubMed]
- Jones, J.L.; Peana, D.; Veteto, A.B.; Lambert, M.D.; Nourian, Z.; Karasseva, N.G.; Hill, M.A.; Lindman, B.R.; Baines, C.P.; Krenz, M.; et al. TRPV4 increases cardiomyocyte calcium cycling and contractility yet contributes to damage in the aged heart following hypoosmotic stress. Cardiovasc. Res. 2019, 115, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Gao, S.; Chen, Y.; Bao, H.; Li, Z.T.; Yao, Q.P.; Liu, J.T.; Wang, Y.; Qi, Y.X. Platelet-derived microvesicles induce calcium oscillations and promote VSMC migration via TRPV4. Theranostics 2021, 11, 2410–2423. [Google Scholar] [PubMed]
- Chen, Y.L.; Daneva, Z.; Kuppusamy, M.; Ottolini, M.; Baker, T.M.; Klimentova, E.; Shah, S.A.; Sokolowski, J.D.; Park, M.S.; Sonkusare, S.K. Novel Smooth Muscle Ca2+-Signaling Nanodomains in Blood Pressure Regulation. Circulation 2022, 146, 548–564. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Bian, X.; Liu, C.; Wang, S.; Guo, M.; Tao, Y.; Huo, B. STIM1 and TRPV4 regulate fluid flow-induced calcium oscillation at early and late stages of osteoclast differentiation. Cell Calcium 2018, 71, 45–52. [Google Scholar] [CrossRef]
- Williams, K.M.; Leser, J.M.; Gould, N.R.; Joca, H.C.; Lyons, J.S.; Khairallah, R.J.; Ward, C.W.; Stains, J.P. TRPV4 calcium influx controls sclerostin protein loss independent of purinergic calcium oscillations. Bone 2020, 136, 115356. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Notomi, T.; Miyajima, D.; Mizoguchi, F.; Hayata, T.; Nakamoto, T.; Hanyu, R.; Kamolratanakul, P.; Mizuno, A.; Suzuki, M.; et al. Osteoblastic differentiation enhances expression of TRPV4 that is required for calcium oscillation induced by mechanical force. Bone 2013, 54, 172–178. [Google Scholar] [PubMed]
- Masuyama, R.; Vriens, J.; Voets, T.; Karashima, Y.; Owsianik, G.; Vennekens, R.; Lieben, L.; Torrekens, S.; Moermans, K.; Vanden Bosch, A.; et al. TRPV4-mediated calcium influx regulates terminal differentiation of osteoclasts. Cell Metab. 2008, 8, 257–265. [Google Scholar] [CrossRef]
- Du, G.; Li, L.; Zhang, X.; Liu, J.; Hao, J.; Zhu, J.; Wu, H.; Chen, W.; Zhang, Q. Roles of TRPV4 and piezo channels in stretch-evoked Ca2+ response in chondrocytes. Exp. Biol. Med. 2020, 245, 180–189. [Google Scholar] [CrossRef]
- Kim, M.K.; Ramachandran, R.; Séguin, C.A. Spatiotemporal and functional characterisation of transient receptor potential vanilloid 4 (TRPV4) in the murine intervertebral disc. Eur. Cell Mater. 2021, 41, 194–203. [Google Scholar] [CrossRef]
- Alessandri-Haber, N.; Dina, O.A.; Joseph, E.K.; Reichling, D.; Levine, J.D. A transient receptor potential vanilloid 4-dependent mechanism of hyperalgesia is engaged by concerted action of inflammatory mediators. J. Neurosci. 2006, 26, 3864–3874. [Google Scholar] [CrossRef]
- Kochukov, M.Y.; McNearney, T.A.; Yin, H.; Zhang, L.; Ma, F.; Ponomareva, L.; Abshire, S.; Westlund, K.N. Tumor necrosis factor-alpha (TNF-alpha) enhances functional thermal and chemical responses of TRP cation channels in human synoviocytes. Mol. Pain. 2009, 5, 49. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Du, L.; Liu, S.; Lan, Z.; Zang, K.; Feng, J.; Zhao, Y.; Yang, X.; Xie, Z.; Wang, P.L.; et al. A TRPV4-dependent neuroimmune axis in the spinal cord promotes neuropathic pain. J. Clin. Investig. 2023, 133, e161507. [Google Scholar] [CrossRef] [PubMed]
- Butenko, O.; Dzamba, D.; Benesova, J.; Honsa, P.; Benfenati, V.; Rusnakova, V.; Ferroni, S.; Anderova, M. The increased activity of TRPV4 channel in the astrocytes of the adult rat hippocampus after cerebral hypoxia/ischemia. PLoS ONE 2012, 7, e39959. [Google Scholar] [CrossRef] [PubMed]
- Eilert-Olsen, M.; Hjukse, J.B.; Thoren, A.E.; Tang, W.; Enger, R.; Jensen, V.; Pettersen, K.H.; Nagelhus, E.A. Astroglial endfeet exhibit distinct Ca2+ signals during hypoosmotic conditions. Glia 2019, 67, 2399–2409. [Google Scholar] [PubMed]
- Jung, C.; Fernández-Dueñas, V.; Plata, C.; Garcia-Elias, A.; Ciruela, F.; Fernández-Fernández, J.M.; Valverde, M.A. Functional coupling of GABA(A/B) receptors and the channel TRPV4 mediates rapid progesterone signaling in the oviduct. Sci. Signal 2018, 11, eaam6558. [Google Scholar] [CrossRef]
- Jiang, P.; Li, S.S.; Xu, X.F.; Yang, C.; Cheng, C.; Wang, J.S.; Zhou, P.Z.; Liu, S.W. TRPV4 channel is involved in HSV-2 infection in human vaginal epithelial cells through triggering Ca2+ oscillation. Acta Pharmacol. Sin. 2023, 44, 811–821. [Google Scholar] [PubMed]
- Jiang, D.; Christ, S.; Correa-Gallegos, D.; Ramesh, P.; Kalgudde Gopal, S.; Wannemacher, J.; Mayr, C.H.; Lupperger, V.; Yu, Q.; Ye, H.; et al. Injury triggers fascia fibroblast collective cell migration to drive scar formation through N-cadherin. Nat. Commun. 2020, 11, 5653. [Google Scholar] [CrossRef]
- Wan, L.; Jiang, D.; Correa-Gallegos, D.; Ramesh, P.; Zhao, J.; Ye, H.; Zhu, S.; Wannemacher, J.; Volz, T.; Rinkevich, Y. Connexin43 gap junction drives fascia mobilization and repair of deep skin wounds. Matrix Biol. 2021, 97, 58–71. [Google Scholar] [CrossRef]
- Rajendran, V.; Ramesh, P.; Dai, R.; Kalgudde Gopal, S.; Ye, H.; Machens, H.G.; Adler, H.; Jiang, D.; Rinkevich, Y. Therapeutic Silencing of p120 in Fascia Fibroblasts Ameliorates Tissue Repair. J. Investig. Dermatol. 2023, 143, 854–863.e4. [Google Scholar]
- Toumpanakis, D.; Chatzianastasiou, A.; Vassilakopoulou, V.; Mizi, E.; Dettoraki, M.; Perlikos, F.; Giatra, G.; Mikos, N.; Theocharis, S.; Vassilakopoulos, T. TRPV4 Inhibition Exerts Protective Effects Against Resistive Breathing Induced Lung Injury. Int. J. Chron. Obs. Pulmon Dis. 2022, 17, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Nam, M.H.; Park, H.J.; Seo, Y.K. Reduction of Osteoclastic Differentiation of Raw 264.7 Cells by EMF Exposure through TRPV4 and p-CREB Pathway. Int. J. Mol. Sci. 2023, 24, 3058. [Google Scholar] [CrossRef]
- Hurd, L.; Kirwin, S.M.; Boggs, M.; Mackenzie, W.G.; Bober, M.B.; Funanage, V.L.; Duncan, R.L. A mutation in TRPV4 results in altered chondrocyte calcium signaling in severe metatropic dysplasia. Am. J. Med. Genet. A 2015, 167, 2286–2293. [Google Scholar] [CrossRef]
- O’Conor, C.J.; Ramalingam, S.; Zelenski, N.A.; Benefield, H.C.; Rigo, I.; Little, D.; Wu, C.L.; Chen, D.; Liedtke, W.; McNulty, A.L.; et al. Cartilage-Specific Knockout of the Mechanosensory Ion Channel TRPV4 Decreases Age-Related Osteoarthritis. Sci. Rep. 2016, 6, 29053. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.Y.; Li, M.Y.; Li, Y.T.; Ning, J.Y.; Gou, X.C.; Shi, J.; Li, Y.Q. Expression and functional characterization of transient receptor potential vanilloid 4 in the dorsal root ganglion and spinal cord of diabetic rats with mechanical allodynia. Brain Res. Bull. 2020, 162, 30–39. [Google Scholar] [PubMed]
- Jang, Y.; Jung, J.; Kim, H.; Oh, J.; Jeon, J.H.; Jung, S.; Kim, K.T.; Cho, H.; Yang, D.J.; Kim, S.M.; et al. Axonal neuropathy-associated TRPV4 regulates neurotrophic factor-derived axonal growth. J. Biol. Chem. 2012, 287, 6014–6024. [Google Scholar]
- Rodrigues, P.; Ruviaro, N.A.; Trevisan, G. TRPV4 Role in Neuropathic Pain Mechanisms in Rodents. Antioxidants 2022, 12, 24. [Google Scholar] [CrossRef]
- Jie, P.; Lu, Z.; Hong, Z.; Li, L.; Zhou, L.; Li, Y.; Zhou, R.; Zhou, Y.; Du, Y.; Chen, L.; et al. Activation of Transient Receptor Potential Vanilloid 4 is Involved in Neuronal Injury in Middle Cerebral Artery Occlusion in Mice. Mol. Neurobiol. 2016, 53, 8–17. [Google Scholar] [CrossRef]
- Liedtke, W.; Friedman, J.M. Abnormal osmotic regulation in trpv4-/- mice. Proc. Natl. Acad. Sci. USA 2003, 100, 13698–13703. [Google Scholar]
- Koide, M.; Wellman, G.C. Activation of TRPV4 channels does not mediate inversion of neurovascular coupling after SAH. Acta Neurochir. Suppl. 2015, 120, 111–116. [Google Scholar]

| Organ | Species | Tissue/Cell Type | Non-Excitable/Excitable | State of TRPV4 | Physiological/Pathological Function | References |
|---|---|---|---|---|---|---|
| Skin | mouse | skin fibroblasts | non-excitable | activation | nuclear translocation of YAP/TAZ, N-cadherin and α-SMA↑, myofibroblasts differentiation | [51] |
| human | preadipocytes | non-excitable | activation | phosphorylation of Akt kinase↓, adipogenesis↓ | [81] | |
| human | mesenchymal stem cells | non-excitable | activation | aligned collagen matrix assembly | [82] | |
| Lung | human | lung fibroblasts | non-excitable | activation | ECM genes↑, collagen and fibronectin↑, phosphorylation of SMAD-2 protein↑ | [75,76] |
| mouse | lung fibroblasts | non-excitable | activation | differentiation of myofibroblasts↑, stiffer matrices, pulmonary fibrosis↑ | [26] | |
| mouse | lung endothelial cells | non-excitable | activation | vascular permeability↑, lung inflammation and edema↑ | [83,84] | |
| Cardiovascular system | mouse | cardiac fibroblasts | non-excitable | inhibition | fibrosis↓ after myocardial infarction | [55] |
| rat, human | cardiac fibroblasts | non-excitable | activation | via p38 and ERK MAPK pathways, fibroblasts differentiation↑, myofibroblasts↑ | [56,85] | |
| rat | cardiac fibroblasts | non-excitable | inhibition | p38, Akt, and STAT3 signaling↓ | [86,87] | |
| human | cardiac c-kit+ progenitor cells | non-excitable | inhibition | migration↓ | [88] | |
| human | endothelial colony-forming cells | non-excitable | activation | nuclear translocation of a Ca2+-sensitive transcription factor p65 NF-κB, angiogenesis↑ | [89] | |
| mouse, rat, human | cardiomyocytes | excitable | activation | CaMKII phosphorylation↑, NF-κB phosphorylation↑, NLRP3 activation↑ | [90] | |
| mouse | cardiomyocytes | excitable | activation | contractility of cardiomyocytes↑ | [91,92] | |
| mouse | cardiomyocytes | excitable | inhibition | Ca2+ overload↓ | [93] | |
| rat | vascular smooth muscle cells | excitable | activation | vascular remodeling after injury, modulation of the cell migration | [94] | |
| mouse, human | vascular smooth muscle cells | excitable | inhibition | α1 adrenergic receptor↑, hypertension↓ | [95] | |
| Skeletal system | mouse | mature osteoclasts | non-excitable | activation | maturation and function of osteoclasts | [96] |
| mouse | osteocytes | non-excitable | activation | bone homeostasis, regulation of Sost, Sp7, Tnfrsf11b | [97] | |
| mouse | osteoblasts | non-excitable | activation | osteoblastic differentiation, regulation of Alpl, Sp7, Dmp1 and Bglap | [98] | |
| mouse | osteoclast | non-excitable | activation | osteoclast differentiation | [99] | |
| mouse | chondrocytes | non-excitable | activation | major mechanical sensor | [79,100] | |
| mouse | annulus fibrosus cells | non-excitable | activation | regulator of perception of mechanical stimulation in intervertebral discs of spine | [101] | |
| human | synovial cells | non-excitable | activation | cell sensitivity↑ to noxious thermal changes and hypoosmotic stress | [102,103] | |
| Nervous system | mouse | microglia | non-excitable | activation | microglia activation and proliferation, promoting functional and structural plasticity in excitatory spinal cord neurons via lipocalin-2 | [104] |
| rat | astrocytes | excitable | activation | promoting proliferation that protects neurons, initiates repair, and correlates with astrogliosis | [105] | |
| mouse | subpial endfeet of astrocytes | excitable | activation | hypoosmotic changes in cerebrospinal fluid | [106] | |
| Reproductive system | mouse | oviduct | non-excitable | activation | ciliary beat frequency | [107] |
| human | vaginal epithelial cells | non-excitable | activation | facilitating the entry of Herpes simplex virus (HSV) | [108] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, D.; Guo, R.; Dai, R.; Knoedler, S.; Tao, J.; Machens, H.-G.; Rinkevich, Y. The Multifaceted Functions of TRPV4 and Calcium Oscillations in Tissue Repair. Int. J. Mol. Sci. 2024, 25, 1179. https://doi.org/10.3390/ijms25021179
Jiang D, Guo R, Dai R, Knoedler S, Tao J, Machens H-G, Rinkevich Y. The Multifaceted Functions of TRPV4 and Calcium Oscillations in Tissue Repair. International Journal of Molecular Sciences. 2024; 25(2):1179. https://doi.org/10.3390/ijms25021179
Chicago/Turabian StyleJiang, Dongsheng, Ruiji Guo, Ruoxuan Dai, Samuel Knoedler, Jin Tao, Hans-Günther Machens, and Yuval Rinkevich. 2024. "The Multifaceted Functions of TRPV4 and Calcium Oscillations in Tissue Repair" International Journal of Molecular Sciences 25, no. 2: 1179. https://doi.org/10.3390/ijms25021179






