Ripasudil as a Potential Therapeutic Agent in Treating Secondary Glaucoma in HTLV-1-Uveitis: An In Vitro Analysis
Abstract
:1. Introduction
2. Results
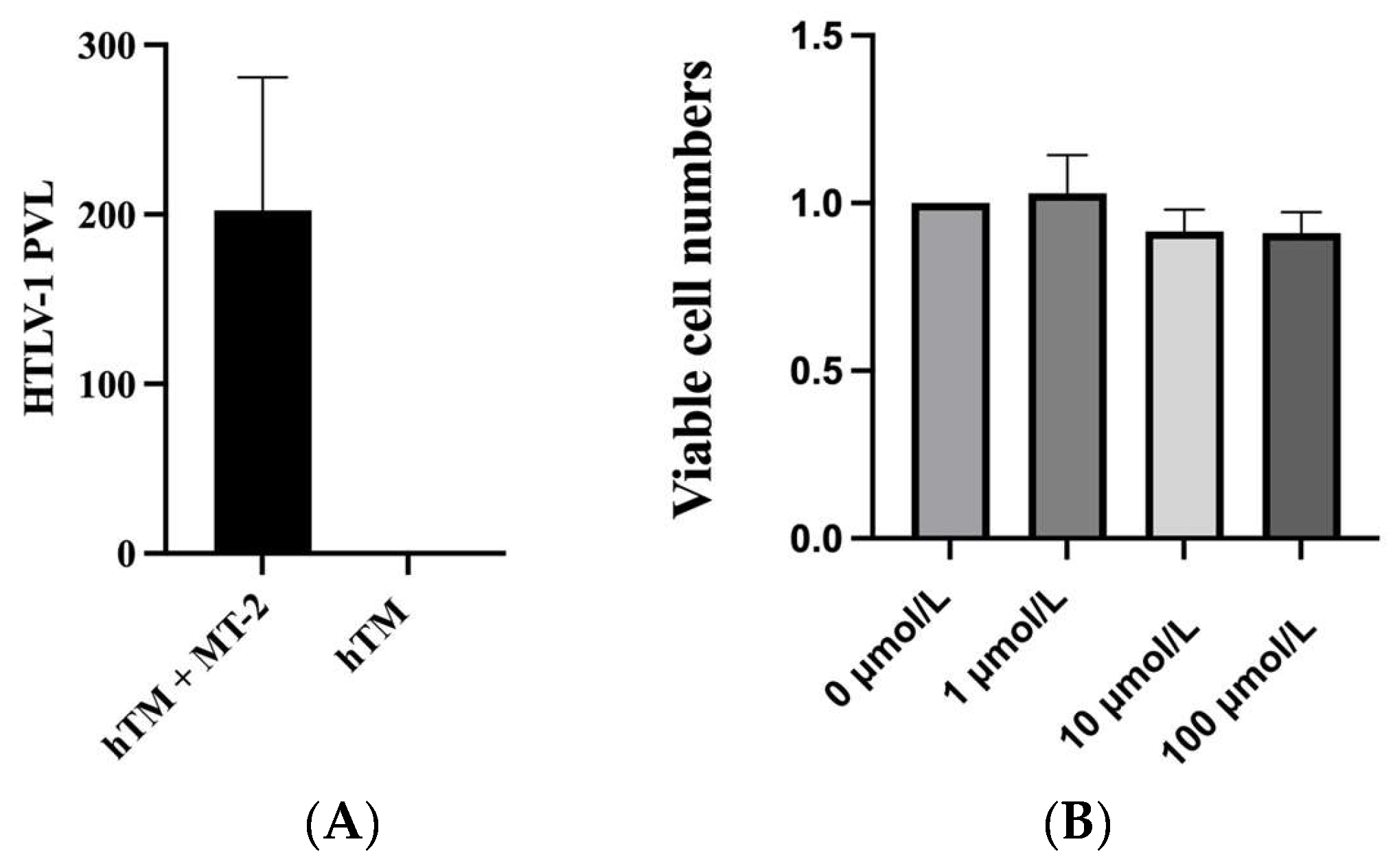
2.1. HTLV-1 Provial Loads after Cocultivation with HTLV-1-Infected Cell
2.2. Effect of Ripasudil on hTM Cell Viability
2.3. Effect of Ripasudil on the Morphology of hTM Cell
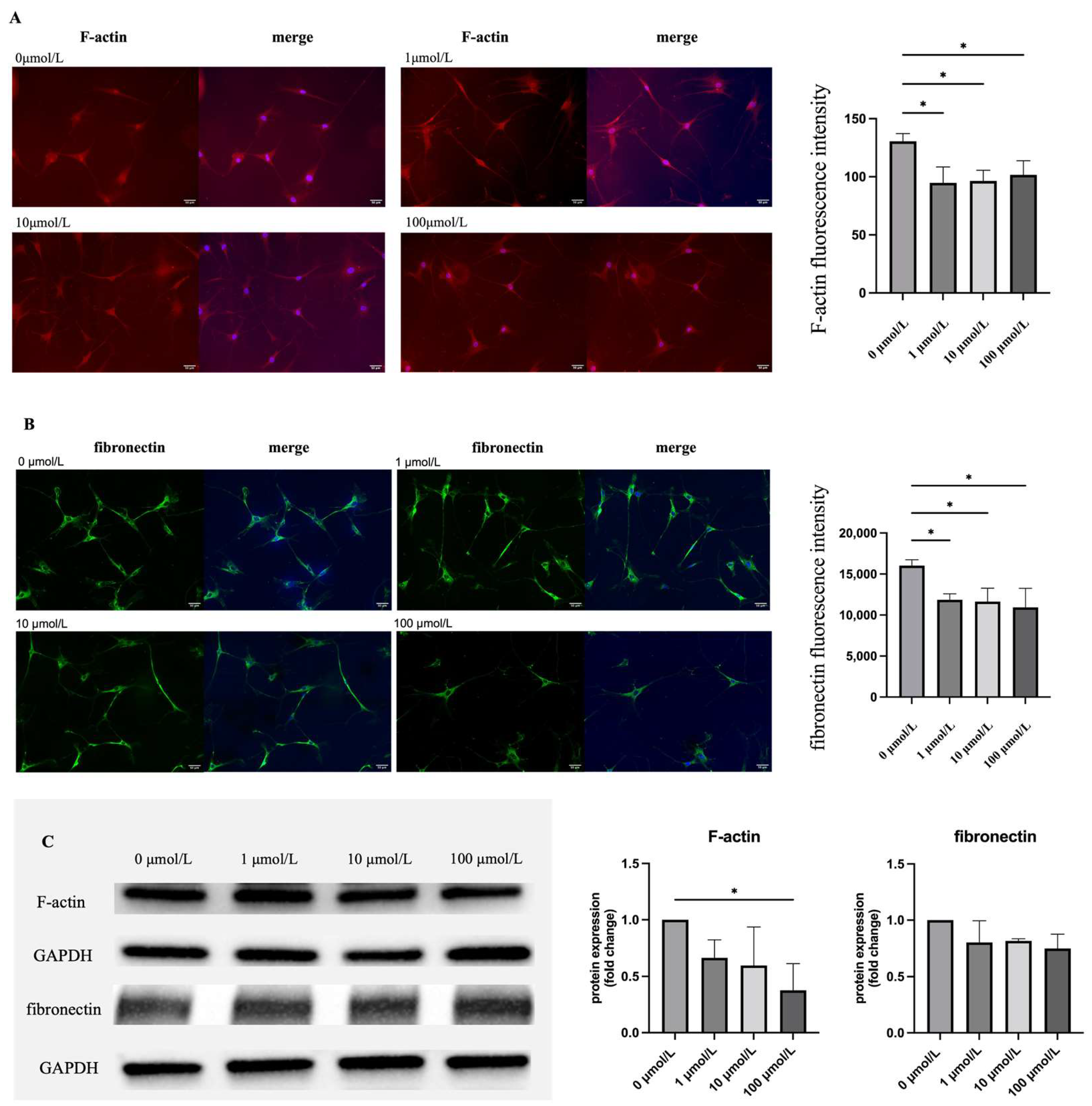
2.4. Effect of Ripasudil on the Cytoskeleton of hTM Cells
2.5. Effect of Ripasudil on Inflammation Cytokines and Chemokines
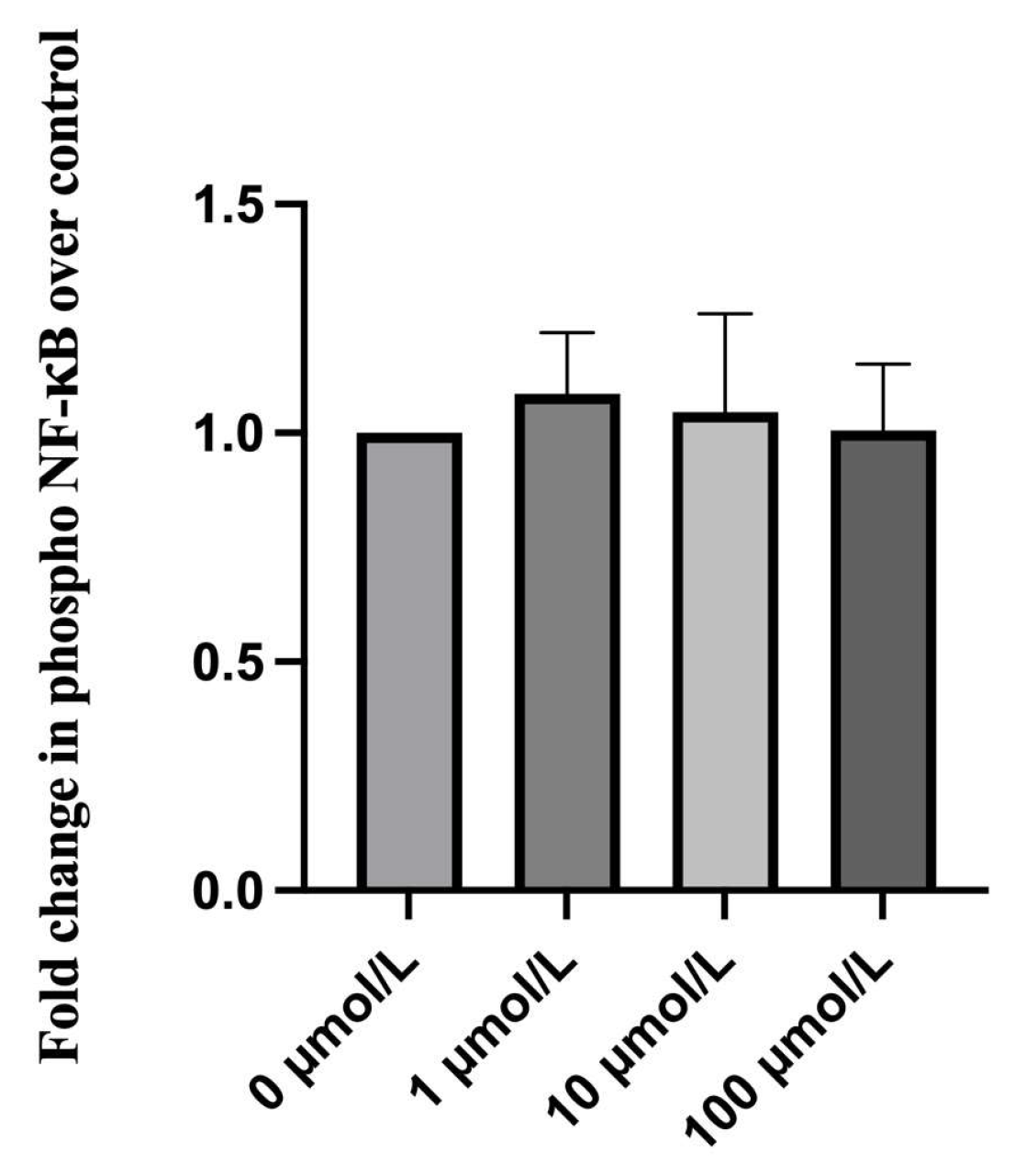
2.6. Effect of Ripasudil on NF-κB Activation
3. Discussion
4. Materials and Methods
4.1. Cell Culture, In Vitro HTLV-1 Infection, and Treatment
4.2. Measurement of HTLV-1 Proviral Loads
4.3. Cell Viability Assay
4.4. Cell Morphology
4.5. Immunofluorescence Assay
4.6. Western Blot
4.7. CBA
4.8. NF-κB Activity
4.9. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tagaya, Y.; Matsuoka, M.; Gallo, R. 40 years of the human T-cell leukemia virus: Past, present, and future. F1000Research 2019, 8, 228. [Google Scholar] [CrossRef] [PubMed]
- Gessain, A.; Cassar, O. Epidemiological Aspects and World Distribution of HTLV-1 Infection. Front. Microbiol. 2012, 3, 388. [Google Scholar] [CrossRef] [PubMed]
- Satake, M.; Iwanaga, M.; Sagara, Y.; Watanabe, T.; Okuma, K.; Hamaguchi, I. Incidence of human T-lymphotropic virus 1 infection in adolescent and adult blood donors in Japan: A nationwide retrospective cohort analysis. Lancet Infect. Dis. 2016, 16, 1246–1254. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, E.; Ono, A.; Hikita, N.; Arima, K.; Mochizuki, M.; Yamaguchi, K.; Tajima, K.; Kiyokawa, H. Estimated prevalence rate of HTLV-I uveitis in Chikugo. Nippon. Ganka Gakkai Zasshi 1998, 102, 327–332. [Google Scholar]
- Kamoi, K.; Watanabe, T.; Uchimaru, K.; Okayama, A.; Kato, S.; Kawamata, T.; Kurozumi-Karube, H.; Horiguchi, N.; Zong, Y.; Yamano, Y.; et al. Updates on HTLV-1 Uveitis. Viruses 2022, 14, 794. [Google Scholar] [CrossRef] [PubMed]
- Kamoi, K.; Horiguchi, N.; Kurozumi-Karube, H.; Hamaguchi, I.; Yamano, Y.; Uchimaru, K.; Tojo, A.; Watanabe, T.; Ohno-Matsui, K. Horizontal transmission of HTLV-1 causing uveitis. Lancet Infect. Dis. 2021, 21, 578. [Google Scholar] [CrossRef] [PubMed]
- Kamoi, K.; Uchimaru, K.; Tojo, A.; Watanabe, T.; Ohno-Matsui, K. HTLV-1 uveitis and Graves’ disease presenting with sudden onset of blurred vision. Lancet 2022, 399, 60. [Google Scholar] [CrossRef]
- Kamoi, K. Current challenges facing the clinical treatment for HTLV-1 ocular manifestations. Expert Rev. Ophthalmol. 2023, 18, 405–419. [Google Scholar] [CrossRef]
- Kamoi, K. HTLV-1 in Ophthalmology. Front. Microbiol. 2020, 11, 388. [Google Scholar] [CrossRef]
- Kesav, N.; Palestine, A.G.; Kahook, M.Y.; Pantcheva, M.B. Current management of uveitis-associated ocular hypertension and glaucoma. Surv. Ophthalmol. 2020, 65, 397–407. [Google Scholar] [CrossRef]
- Zong, Y.; Kamoi, K.; Ando, N.; Kurozumi-Karube, H.; Ohno-Matsui, K. Mechanism of Secondary Glaucoma Development in HTLV-1 Uveitis. Front. Microbiol. 2022, 13, 738742. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, E.; Nakajima, T.; Minagawa, Y.; Shearer, T.R.; Azuma, M. Contribution of ROCK in contraction of trabecular meshwork: Proposed mechanism for regulating aqueous outflow in monkey and human eyes. J. Pharm. Sci. 2005, 94, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Ohta, M.; Inoue, T.; Mizuno, K.; Isobe, T.; Tanabe, S.; Tanihara, H. Effects of K-115 (Ripasudil), a novel ROCK inhibitor, on trabecular meshwork and Schlemm’s canal endothelial cells. Sci. Rep. 2016, 6, 19640. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.A.; Perkumas, K.M.; Campbell, M.; Farrar, G.J.; Stamer, W.D.; Humphries, P.; O’Callaghan, J.; O’Brien, C.J. Fibrotic Changes to Schlemm’s Canal Endothelial Cells in Glaucoma. Int. J. Mol. Sci. 2021, 22, 9446. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Barua, S.; Tripathi, B.N.; Kumar, N. Role of ROCK signaling in virus replication. Virus Res. 2023, 329, 199105. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Chen, X.; Han, Y.; Li, C.; Chen, P.; Su, S.; Zhang, Y.; Pan, Z. Rho kinase inhibition by fasudil suppresses lipopolysaccharide-induced apoptosis of rat pulmonary microvascular endothelial cells via JNK and p38 MAPK pathway. Biomed. Pharmacother. 2014, 68, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Futakuchi, A.; Inoue, T.; Fujimoto, T.; Inoue-Mochita, M.; Kawai, M.; Tanihara, H. The effects of ripasudil (K-115), a Rho kinase inhibitor, on activation of human conjunctival fibroblasts. Exp. Eye Res. 2016, 149, 107–115. [Google Scholar] [CrossRef]
- Okumura, N.; Okazaki, Y.; Inoue, R.; Kakutani, K.; Nakano, S.; Kinoshita, S.; Koizumi, N. Effect of the Rho-Associated Kinase Inhibitor Eye Drop (Ripasudil) on Corneal Endothelial Wound Healing. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1284–1292. [Google Scholar] [CrossRef]
- Schlötzer-Schrehardt, U.; Zenkel, M.; Strunz, M.; Gießl, A.; Schondorf, H.; da Silva, H.; Schmidt, G.A.; Greiner, M.A.; Okumura, N.; Koizumi, N.; et al. Potential Functional Restoration of Corneal Endothelial Cells in Fuchs Endothelial Corneal Dystrophy by ROCK Inhibitor (Ripasudil). Am. J. Ophthalmol. 2021, 224, 185–199. [Google Scholar] [CrossRef]
- Yang, J.; Ruan, F.; Zheng, Z. Ripasudil Attenuates Lipopolysaccharide (LPS)-Mediated Apoptosis and Inflammation in Pulmonary Microvascular Endothelial Cells via ROCK2/eNOS Signaling. Med. Sci. Monit. 2018, 24, 3212–3219. [Google Scholar] [CrossRef]
- Sato, T.; Kawaji, T. Effects of Ripasudil on Open-Angle Glaucoma after Circumferential Suture Trabeculotomy Ab Interno. J. Clin. Med. 2021, 10, 401. [Google Scholar] [CrossRef]
- Tanihara, H.; Kakuda, T.; Sano, T.; Kanno, T.; Kurihara, Y. Long-Term Intraocular Pressure-Lowering Effects and Adverse Events of Ripasudil in Patients with Glaucoma or Ocular Hypertension over 24 Months. Adv. Ther. 2022, 39, 1659–1677. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Ohta, M.; Isobe, T.; Nakamura, Y.; Mizuno, K. Additive Intraocular Pressure-Lowering Effects of Ripasudil with Glaucoma Therapeutic Agents in Rabbits and Monkeys. J. Ophthalmol. 2017, 2017, 7079645. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Li, Q.; Zhang, Y.; Gao, X.; Li, H.; Yuan, Z. Ripasudil alleviated the inflammation of RPE cells by targeting the miR-136-5p/ROCK/NLRP3 pathway. BMC Ophthalmol. 2020, 20, 134. [Google Scholar] [CrossRef] [PubMed]
- Yanai, R.; Uchi, S.H.; Kobayashi, M.; Nagai, T.; Teranishi, S.; Wakuta, M.; Kimura, K. Efficacy of ripasudil in reducing intraocular pressure and medication score for ocular hypertension with inflammation and corticosteroid. Int. J. Ophthalmol. 2023, 16, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Li, Z.; Mahesh, S.P.; Kurup, S.K.; Giam, C.Z.; Nussenblatt, R.B. HTLV-1 infection of human retinal pigment epithelial cells and inhibition of viral infection by an antibody to ICAM-1. Investig. Ophthalmol. Vis. Sci. 2006, 47, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Garnock-Jones, K.P. Ripasudil: First global approval. Drugs 2014, 74, 2211–2215. [Google Scholar] [CrossRef]
- Yuan, Y.; Call, M.K.; Yuan, Y.; Zhang, Y.; Fischesser, K.; Liu, C.Y.; Kao, W.W. Dexamethasone induces cross-linked actin networks in trabecular meshwork cells through noncanonical wnt signaling. Investig. Ophthalmol. Vis. Sci. 2013, 54, 6502–6509. [Google Scholar] [CrossRef]
- Hoare, M.J.; Grierson, I.; Brotchie, D.; Pollock, N.; Cracknell, K.; Clark, A.F. Cross-linked actin networks (CLANs) in the trabecular meshwork of the normal and glaucomatous human eye in situ. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1255–1263. [Google Scholar] [CrossRef]
- Somlyo, A.P.; Somlyo, A.V. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: Modulated by G proteins, kinases, and myosin phosphatase. Physiol. Rev. 2003, 83, 1325–1358. [Google Scholar] [CrossRef]
- Rao, V.P.; Epstein, D.L. Rho GTPase/Rho kinase inhibition as a novel target for the treatment of glaucoma. BioDrugs 2007, 21, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yang, X.; Fang, J.; Zhang, Y.; Zhu, W.; Yang, X. Rho-Associated Protein Kinase Inhibitor Treatment Promotes Proliferation and Phagocytosis in Trabecular Meshwork Cells. Front. Pharmacol. 2020, 11, 302. [Google Scholar] [CrossRef] [PubMed]
- Diao, Y.M.; Hong, J. Rho-associated protein kinase inhibitor, Y-27632, significantly enhances cell adhesion and induces a delay in G1 to S phase transition in rabbit corneal endothelial cells. Mol. Med. Rep. 2015, 12, 1951–1956. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, Q.; Yang, Y.; Guo, X.; Lian, R.; Li, S.; Wang, C.; Zhang, S.; Chen, J. The effects of ROCK inhibitor Y-27632 on injectable spheroids of bovine corneal endothelial cells. Cell. Reprogramming 2015, 17, 77–87. [Google Scholar] [CrossRef]
- Inomata, T.; Fujimoto, K.; Okumura, Y.; Zhu, J.; Fujio, K.; Shokirova, H.; Miura, M.; Okano, M.; Funaki, T.; Sung, J.; et al. Novel immunotherapeutic effects of topically administered ripasudil (K-115) on corneal allograft survival. Sci. Rep. 2020, 10, 19817. [Google Scholar] [CrossRef] [PubMed]
- Arima, M.; Nakao, S.; Yamaguchi, M.; Feng, H.; Fujii, Y.; Shibata, K.; Wada, I.; Kaizu, Y.; Ahmadieh, H.; Ishibashi, T.; et al. Claudin-5 Redistribution Induced by Inflammation Leads to Anti-VEGF-Resistant Diabetic Macular Edema. Diabetes 2020, 69, 981–999. [Google Scholar] [CrossRef] [PubMed]
- Kuchtey, J.; Rezaei, K.A.; Jaru-Ampornpan, P.; Sternberg, P., Jr.; Kuchtey, R.W. Multiplex cytokine analysis reveals elevated concentration of interleukin-8 in glaucomatous aqueous humor. Investig. Ophthalmol. Vis. Sci. 2010, 51, 6441–6447. [Google Scholar] [CrossRef]
- Chono, I.; Miyazaki, D.; Miyake, H.; Komatsu, N.; Ehara, F.; Nagase, D.; Kawamoto, Y.; Shimizu, Y.; Ideta, R.; Inoue, Y. High interleukin-8 level in aqueous humor is associated with poor prognosis in eyes with open angle glaucoma and neovascular glaucoma. Sci. Rep. 2018, 8, 14533. [Google Scholar] [CrossRef]
- Yang, L.; Herrera, J.; Gilbertsen, A.; Xia, H.; Smith, K.; Benyumov, A.; Bitterman, P.B.; Henke, C.A. IL-8 mediates idiopathic pulmonary fibrosis mesenchymal progenitor cell fibrogenicity. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L127–L136. [Google Scholar] [CrossRef]
- Ulhaq, Z.S.; Istifiani, L.A.; Pamungkas, S.A. Evaluation of systemic IL-6 trans-signalling in patients with primary open-angle glaucoma. J. Fr. Ophtalmol. 2023, 46, 622–629. [Google Scholar] [CrossRef]
- Ulhaq, Z.S.; Soraya, G.V.; Hasan, Y.T.N.; Rachma, L.N.; Rachmawati, E.; Shodry, S.; Kusuma, M.A.S. Serum IL-6/IL-10 ratio as a biomarker for the diagnosis and severity assessment of primary-open angle glaucoma. Eur. J. Ophthalmol. 2022, 32, 2259–2264. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Xing, Y.Q.; Chen, Z.; Ma, X.C.; Lu, Q. Association between interleukin-10 genetic polymorphisms and risk of primary open angle glaucoma in a Chinese Han population: A case-control study. Int. J. Ophthalmol. 2019, 12, 1605–1611. [Google Scholar] [CrossRef]
- Micera, A.; Quaranta, L.; Esposito, G.; Floriani, I.; Pocobelli, A.; Saccà, S.C.; Riva, I.; Manni, G.; Oddone, F. Differential Protein Expression Profiles in Glaucomatous Trabecular Meshwork: An Evaluation Study on a Small Primary Open Angle Glaucoma Population. Adv. Ther. 2016, 33, 252–267. [Google Scholar] [CrossRef] [PubMed]
- Atanasovska Velkovska, M.; Goričar, K.; Blagus, T.; Dolžan, V.; Cvenkel, B. Association of Genetic Polymorphisms in Oxidative Stress and Inflammation Pathways with Glaucoma Risk and Phenotype. J. Clin. Med. 2021, 10, 1148. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.Y.; Zhang, Y.J.; Zhao, S.Z. Puerarin regulates neovascular glaucoma through pigment epithelium-derived growth factor-induced NF-κB signaling pathway. Mol. Med. Rep. 2018, 17, 7866–7874. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, H.; Roberts, A.L.; McDowell, C.M. Nuclear factor-kappa beta signaling is required for transforming growth factor Beta-2 induced ocular hypertension. Exp. Eye Res. 2020, 191, 107920. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, Q.; Shi, L.; Zhou, X.; Wu, W.; Wang, X.; Wang, L.; Wu, Z. Baicalin prevents fibrosis of human trabecular meshwork cells via inhibiting the MyD88/NF-κB pathway. Eur. J. Pharmacol. 2023, 938, 175425. [Google Scholar] [CrossRef]
- Zhao, H.; Sun, X.; Tong, J. Role of ROCK/NF-κB/AQP8 signaling in ethanol-induced intestinal epithelial barrier dysfunction. Mol. Med. Rep. 2020, 22, 2253–2262. [Google Scholar] [CrossRef]
- Xie, X.; Peng, J.; Chang, X.; Huang, K.; Huang, J.; Wang, S.; Shen, X.; Liu, P.; Huang, H. Activation of RhoA/ROCK regulates NF-κB signaling pathway in experimental diabetic nephropathy. Mol. Cell Endocrinol. 2013, 369, 86–97. [Google Scholar] [CrossRef]
- Uchida, T.; Honjo, M.; Yamagishi, R.; Aihara, M. The Anti-Inflammatory Effect of Ripasudil (K-115), a Rho Kinase (ROCK) Inhibitor, on Endotoxin-Induced Uveitis in Rats. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5584–5593. [Google Scholar] [CrossRef]
- Shafiei, M.; Mozhgani, S.-H. Th17/IL-17 Axis in HTLV-1-Associated Myelopathy Tropical Spastic Paraparesis and Multiple Sclerosis: Novel Insights into the Immunity During HAMTSP. Mol. Neurobiol. 2023, 60, 3839–3854. [Google Scholar] [CrossRef] [PubMed]
- Ohsugi, T.; Kumasaka, T. Low CD4/CD8 T-cell ratio associated with inflammatory arthropathy in human T-cell leukemia virus type I Tax transgenic mice. PLoS ONE 2011, 6, e18518. [Google Scholar] [CrossRef] [PubMed]
- Yarishkin, O.; Phuong, T.T.T.; Križaj, D. Trabecular Meshwork TREK-1 Channels Function as Polymodal Integrators of Pressure and pH. Investig. Ophthalmol. Vis. Sci. 2019, 60, 2294–2303. [Google Scholar] [CrossRef] [PubMed]
- Fukui, S.; Nakamura, H.; Takahashi, Y.; Iwamoto, N.; Hasegawa, H.; Yanagihara, K.; Nakamura, T.; Okayama, A.; Kawakami, A. Tumor necrosis factor alpha inhibitors have no effect on a human T-lymphotropic virus type-I (HTLV-I)-infected cell line from patients with HTLV-I-associated myelopathy. BMC Immunol. 2017, 18, 7. [Google Scholar] [CrossRef] [PubMed]
- Uchida, M.; Kamoi, K.; Ando, N.; Wei, C.; Karube, H.; Ohno-Matsui, K. Safety of Infliximab for the Eye Under Human T-Cell Leukemia Virus Type 1 Infectious Conditions in vitro. Front. Microbiol. 2019, 10, 2148. [Google Scholar] [CrossRef] [PubMed]
- Kurozumi-Karube, H.; Kamoi, K.; Ando, N.; Uchida, M.; Hamaguchi, I.; Ohno-Matsui, K. In vitro Evaluation of the Safety of Adalimumab for the Eye Under HTLV-1 Infection Status: A Preliminary Study. Front. Microbiol. 2020, 11, 522579. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, M.; Kamoi, K.; Zong, Y.; Zhang, J.; Zou, Y.; Ohno-Matsui, K. Ripasudil as a Potential Therapeutic Agent in Treating Secondary Glaucoma in HTLV-1-Uveitis: An In Vitro Analysis. Int. J. Mol. Sci. 2024, 25, 3229. https://doi.org/10.3390/ijms25063229
Yang M, Kamoi K, Zong Y, Zhang J, Zou Y, Ohno-Matsui K. Ripasudil as a Potential Therapeutic Agent in Treating Secondary Glaucoma in HTLV-1-Uveitis: An In Vitro Analysis. International Journal of Molecular Sciences. 2024; 25(6):3229. https://doi.org/10.3390/ijms25063229
Chicago/Turabian StyleYang, Mingming, Koju Kamoi, Yuan Zong, Jing Zhang, Yaru Zou, and Kyoko Ohno-Matsui. 2024. "Ripasudil as a Potential Therapeutic Agent in Treating Secondary Glaucoma in HTLV-1-Uveitis: An In Vitro Analysis" International Journal of Molecular Sciences 25, no. 6: 3229. https://doi.org/10.3390/ijms25063229




