UV Light Illumination Can Improve the Sensing Properties of LaFeO3 to Acetone Vapor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation
2.2. Vapor Sensing Measurements
2.3. Characterization
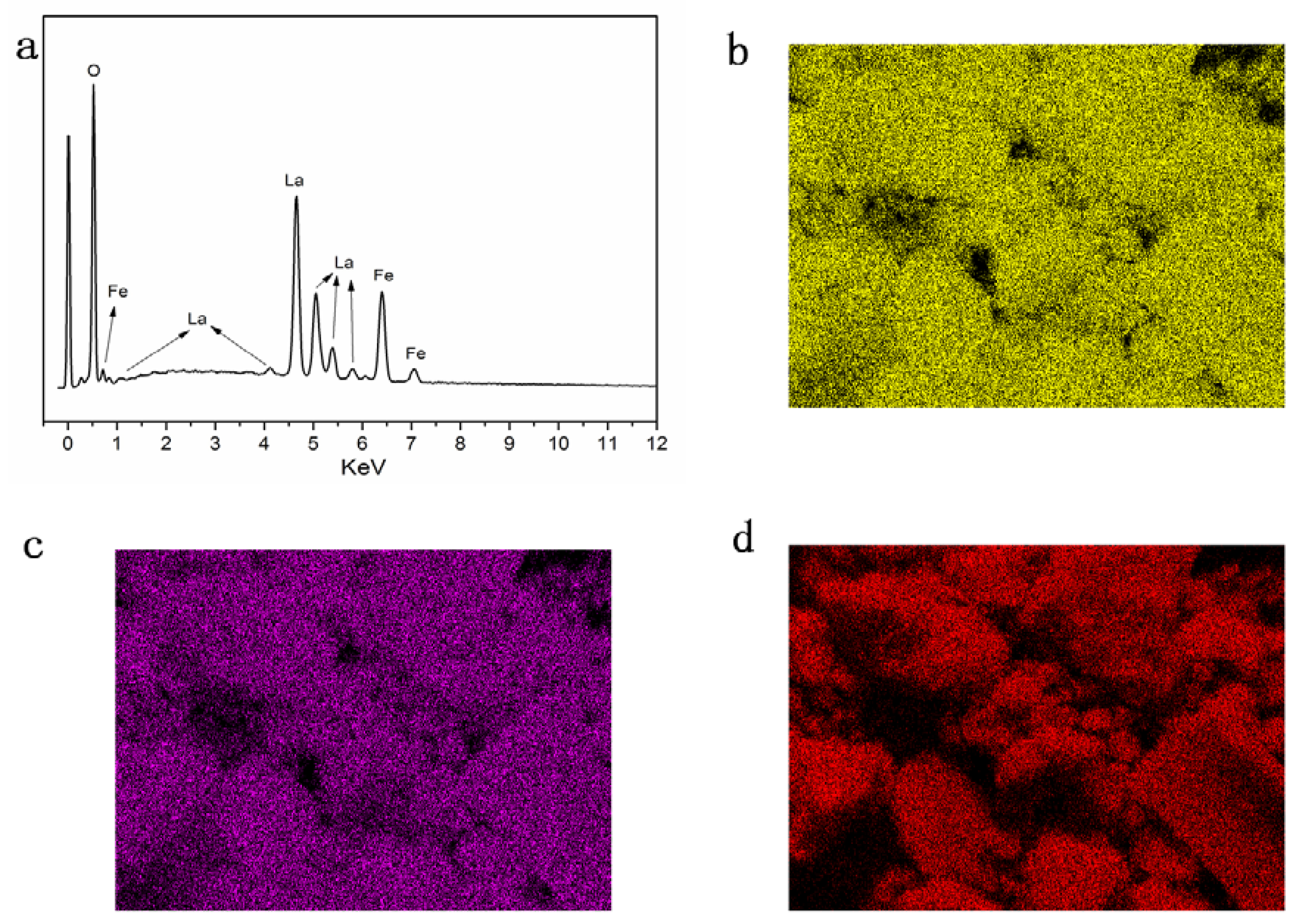
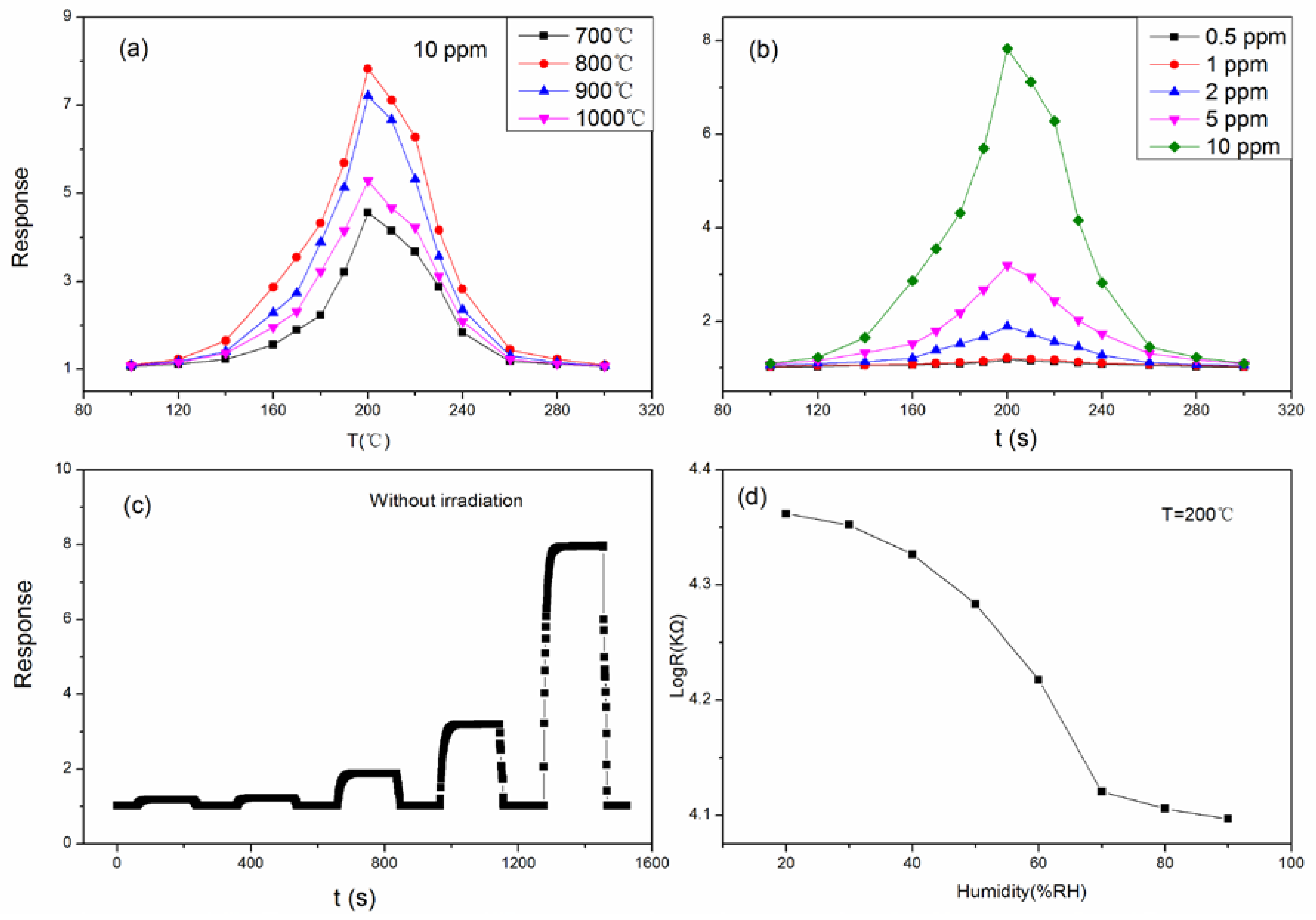
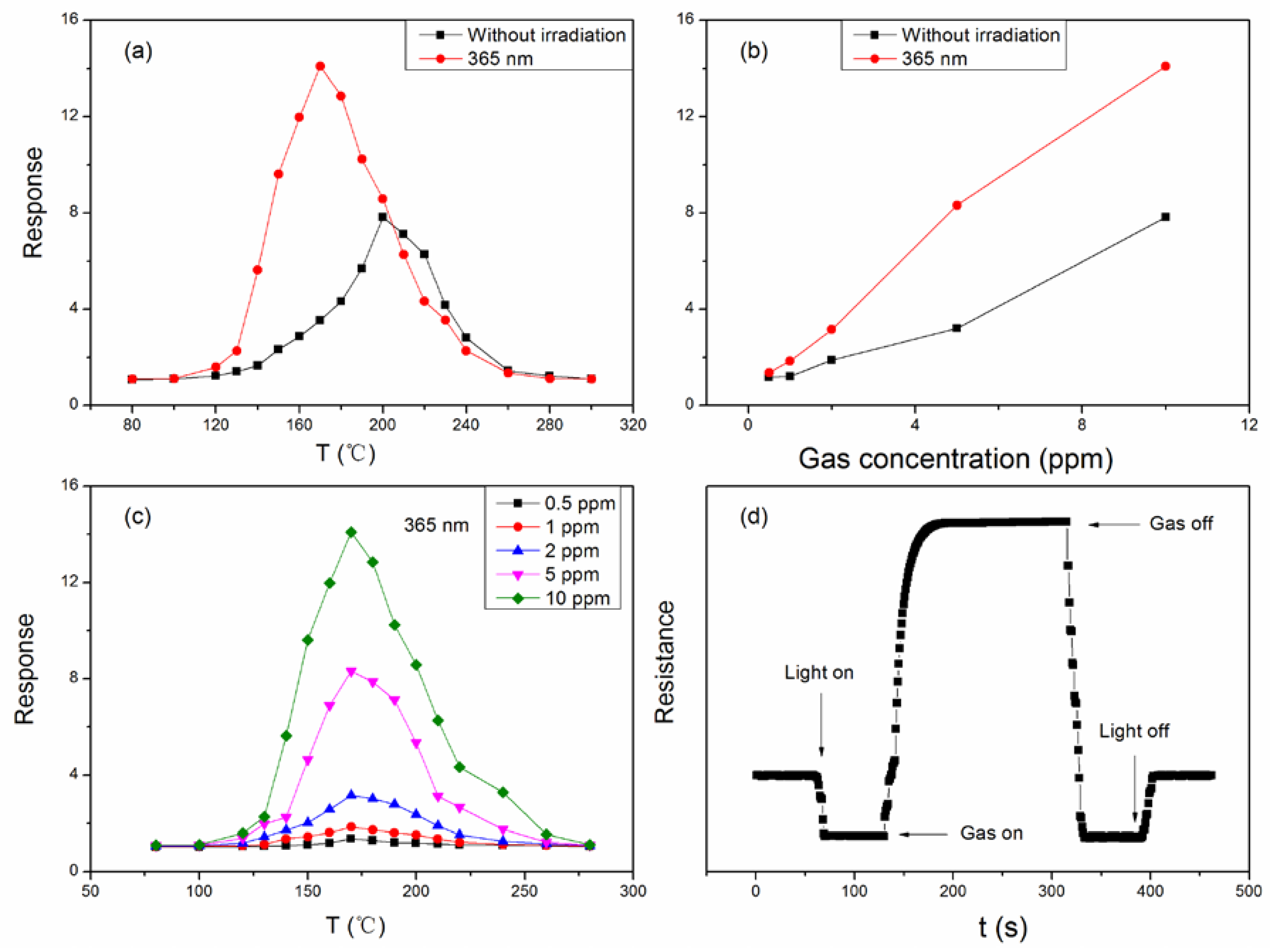
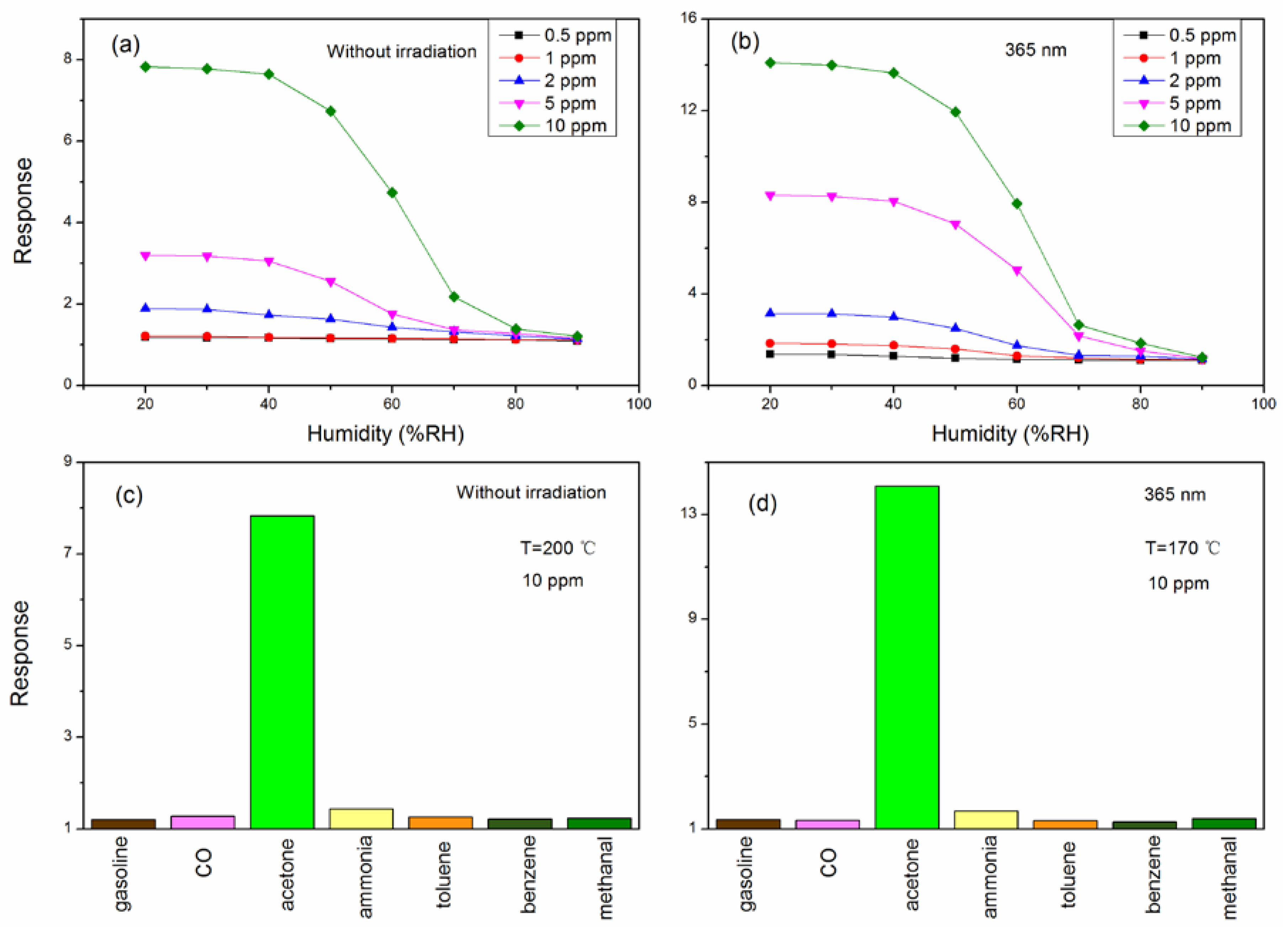
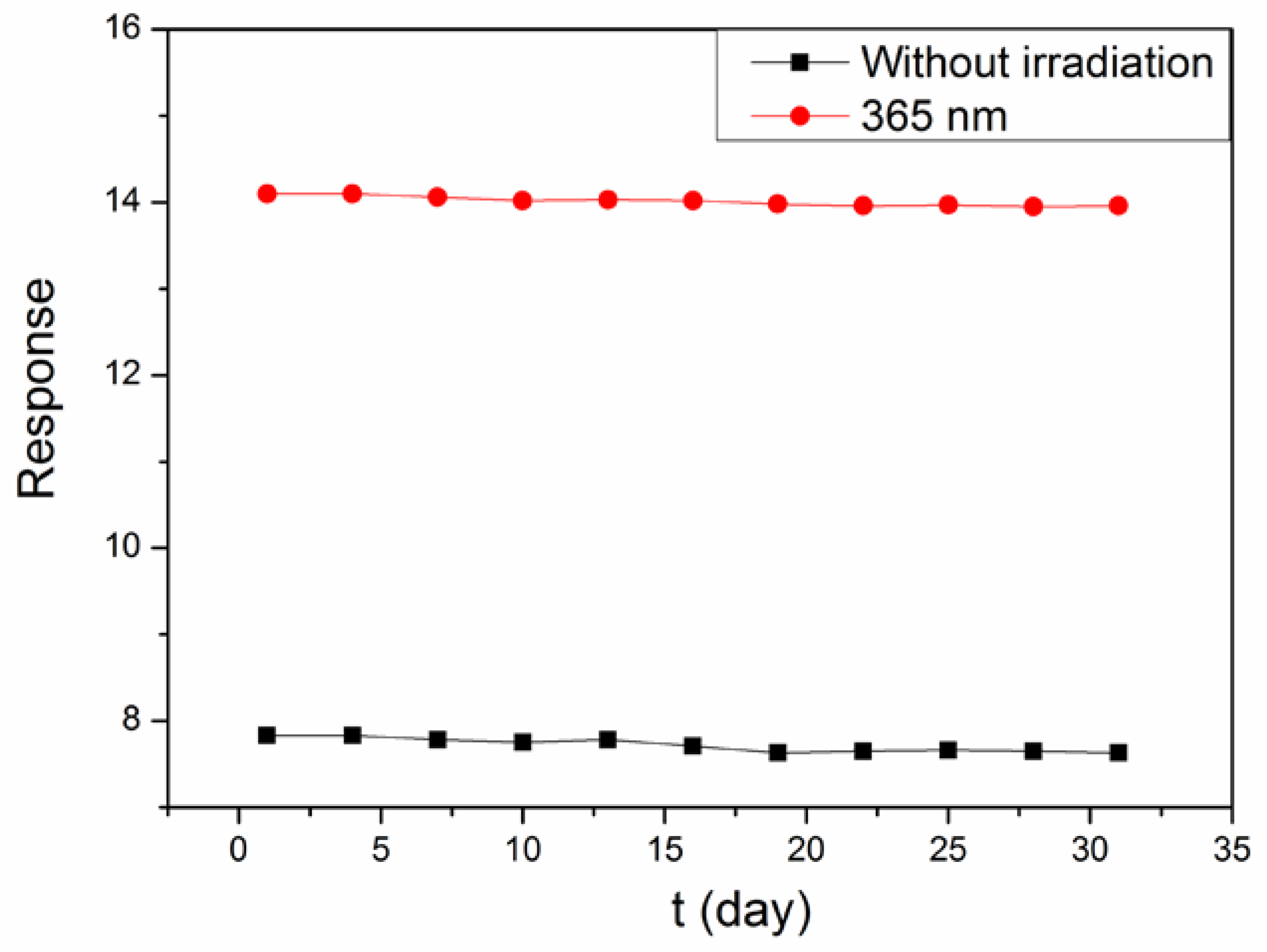
3. Result and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, F.; Guan, Y.; Sun, R.; Liang, X.; Sun, P.; Liu, F.; Lu, G. Mixed potential type acetone sensor using stabilized zirconia and M3V2O8 (M: Zn, Co and Ni) sensing electrode. Sens. Actuators B Chem. 2015, 221, 673–680. [Google Scholar] [CrossRef]
- Wang, C.-C.; Weng, Y.C.; Chou, T.C. Acetone sensor using lead foil as working electrode. Sens. Actuators B Chem. 2007, 122, 591–595. [Google Scholar] [CrossRef]
- Makisimovich, N.; Vorottyntsev, V.; Nikitina, N.; Kaskevich, O.; Karabum, P.; Martynenko, F. Adsorption semiconductor sensor for diabetic ketoacidosis diagnosis. Sens. Actuators B Chem. 1996, 36, 419–421. [Google Scholar] [CrossRef]
- Epifani, M.; Comini, E.; Díaz, R.; Genç, A.; Andreu, T.; Siciliano, P.; Morante, J.R. Acetone sensors based on TiO2 nanocrystals modified with tungsten oxide species. J. Alloy. Compd. 2016, 665, 345–351. [Google Scholar] [CrossRef]
- Bian, H.; Ma, S.; Sun, A.; Xu, X.; Yang, G.; Gao, J.; Zhang, Z.; Zhu, H. Characterization and acetone gas sensing properties of electrospun TiO2 nanorods. Superlattice Microstruct. 2015, 81, 107–113. [Google Scholar] [CrossRef]
- Bhowmik, B.; Hazra, A.; Dutta, K.; Bhattacharyya, P. Repeatability and Stability of Room-Temperature Acetone Sensor Based on Nanotubes: Influence of Stoichiometry Variation. IEEE Trans. Device Mater. Reliab. 2014, 14, 961–967. [Google Scholar] [CrossRef]
- Wang, L.; Lou, Z.; Fei, T.; Zhang, T. Enhanced acetone sensing performances of hierarchical hollow Au-loaded NiO hybrid structures. Sens. Actuators B Chem. 2012, 161, 178–183. [Google Scholar] [CrossRef]
- Wang, C.; Liu, J.; Yang, Q.; Sun, P.; Gao, Y.; Liu, F.; Zheng, J.; Lu, G. Ultrasensitive and low detection limit of acetone gas sensor based on W-doped NiO hierarchical nanostructure. Sens. Actuators B Chem. 2015, 220, 59–67. [Google Scholar] [CrossRef]
- Wei, S.; Zhou, M.; Du, W. Improved acetone sensing properties of ZnO hollow nanofibers by single capillary electrospinning. Sens. Actuators B Chem. 2011, 160, 753–759. [Google Scholar] [CrossRef]
- An, D.; Tong, X.; Liu, J.; Wang, Q.; Zhou, Q.; Dong, J.; Li, Y. Template-free hydrothermal synthesis of ZnO micro/nano-materials and their application in acetone sensing properties. Superlattice Microstruct. 2015, 77, 1–11. [Google Scholar] [CrossRef]
- Al-Hardan, N.H.; Abdullah, M.J.; Aziz, A.A. Performance of Cr-doped ZnO for acetone sensing. Appl. Surf. Sci. 2013, 270, 480–485. [Google Scholar] [CrossRef]
- Peng, C.; Guo, J.; Yang, W.; Shi, C.; Liu, M.; Zheng, Y.; Xu, J.; Chen, P.; Huang, T.; Yang, Y. Synthesis of three-dimensional flower-like hierarchical ZnO nanostructure and its enhanced acetone gas sensing properties. J. Alloy. Compd. 2016, 654, 371–378. [Google Scholar] [CrossRef]
- Rajgure, A.V.; Patil, J.Y.; Pawar, R.C.; Lee, C.S.; Suryavanshi, S.S. Aqueous chemical route deposition of nanocrystalline ZnO thin films as acetone sensor: Effect of molarity. Ceram. Int. 2013, 39, 87–92. [Google Scholar] [CrossRef]
- Zhang, Z.; Wen, Z.; Ye, Z.; Zhu, L. Gas sensors based on ultrathin porous Co3O4 nanosheets to detect acetone at low temperature. RSC Adv. 2015, 5, 59976–59982. [Google Scholar] [CrossRef]
- Su, C.; Liu, C.; Liu, L.; Ni, M.; Li, H.; Bo, X.; Liu, L.; Chi, X. Excellent acetone sensing properties of Sm-doped alpha-Fe2O3. Appl. Surf. Sci. 2014, 314, 931–935. [Google Scholar] [CrossRef]
- Shan, H.; Liu, C.; Liu, L.; Li, S.; Wang, L.; Zhang, X.; Bo, X.; Chi, X. Highly sensitive acetone sensors based on La-doped alpha-Fe2O3 nanotubes. Sens. Actuators B Chem. 2013, 184, 243–247. [Google Scholar] [CrossRef]
- Kim, S.; Park, S.; Park, S.; Lee, C. Acetone sensing of Au and Pd-decorated WO3 nanorod sensors. Sens. Actuators B Chem. 2015, 209, 180–185. [Google Scholar] [CrossRef]
- Chen, D.; Hou, X.; Li, T.; Yin, L.; Fan, B.; Wang, H.; Li, X.; Xu, H.; Lu, H.; Zhang, R.; et al. Effects of morphologies on acetone-sensing properties of tungsten trioxide nanocrystals. Sens. Actuators B Chem. 2011, 153, 373–381. [Google Scholar] [CrossRef]
- Tomer, V.K.; Singh, K.; Kaur, H.; Shorie, M.; Sabherwal, P. Rapid acetone detection using indium loaded WO3/SnO2 nanohybrid sensor. Sens. Actuators B Chem. 2017, 253, 703–713. [Google Scholar] [CrossRef]
- Malik, R.; Tomer, V.K.; Dankwort, T.; Misha, Y.K.; Kienle, L. Cubic mesoporous Pd–WO3 loaded graphitic carbon nitride (g-CN) nanohybrids: Highly sensitive and temperature dependent VOC sensors. J. Mater. Chem. A 2018, 6, 10718–10730. [Google Scholar] [CrossRef]
- Mishra, R.K.; Kushwaha, A.; Sahay, P.P. Cr-induced modifications in the structural, photoluminescence and acetone-sensing behaviour of hydrothermally synthesised SnO2 nanoparticles. J. Exp. Nanosci. 2014, 10, 1042–1056. [Google Scholar] [CrossRef]
- Jin, W.X.; Ma, S.Y.; Sun, A.M.; Luo, J.; Cheng, L.; Li, W.Q.; Tie, Z.Z.; Jiang, X.H.; Wang, T.T. Synthesis of hierarchical SnO2 nanoflowers and their high gas-sensing properties. Mater. Lett. 2015, 143, 283–286. [Google Scholar] [CrossRef]
- Punginsang, M.; Wisitsoraat, A.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Effects of cobalt doping on nitric oxide, acetone and ethanol sensing performances of FSP-made SnO2 nanoparticles. Sens. Actuators B Chem. 2015, 210, 589–601. [Google Scholar] [CrossRef]
- Singkammo, S.; Wisitsoraat, A.; Sriprachuabwong, C.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Electrolytically Exfoliated Graphene-Loaded Flame-Made Ni-Doped SnO2 Composite Film for Acetone Sensing. ACS Appl. Mater. Interfaces 2015, 7, 3077–3092. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.B.; Patil, P.P.; More, M.A. Acetone vapour sensing characteristics of cobalt-doped SnO2 thin films. Sens. Actuators B Chem. 2007, 125, 126–130. [Google Scholar] [CrossRef]
- Tomer, V.K.; Malik, R.; Kailasam, K. Near-Room-Temperature Ethanol Detection Using Ag-Loaded Mesoporous Carbon Nitrides. ACS Omega 2017, 2, 3658–3668. [Google Scholar] [CrossRef]
- Wu, Z.L.; Zhang, R.; Zhao, M.; Fang, S.M.; Han, Z.X.; Hu, J.F.; Wang, K.Y. Effect of Pd doping on the acetone-sensing properties of NdFeO3. Int. J. Miner. Metall. Mater. 2012, 19, 141–145. [Google Scholar] [CrossRef]
- Liu, X.; Hu, J.; Cheng, B.; Qin, H.; Jiang, M. Acetone gas sensing properties of SmFe1-xMgxO3 perovskite oxides. Sens. Actuators B Chem. 2008, 134, 483–487. [Google Scholar] [CrossRef]
- Zhang, P.; Qin, H.; Lv, W.; Zhang, H.; Hu, J. Gas sensors based on ytterbium ferrites nanocrystalline powders for detecting acetone with low concentrations. Sens. Actuators B Chem. 2017, 246, 9–19. [Google Scholar] [CrossRef]
- Yang, M.; Huo, L.; Zhao, H.; Gao, S.; Rong, Z. Electrical properties and acetone-sensing characteristics of LaNi1−xTixO3 perovskite system prepared by amorphous citrate decomposition. Sens. Actuators B Chem. 2009, 143, 111–118. [Google Scholar] [CrossRef]
- Chen, T.; Zhou, Z.; Wang, Y. Surfactant CATB-assisted generation and gas-sensing characteristics of LnFeO3 (Ln=La, Sm, Eu) materials. Sens. Actuators B Chem. 2009, 143, 124–131. [Google Scholar] [CrossRef]
- Liu, X.; Ji, H.; Gu, Y.; Xu, M. Preparation and acetone sensitive characteristics of nano LaFeO3 semiconductor thin films by polymerization complex method. Mater. Sci. Eng. B Adv. 2006, 133, 98–101. [Google Scholar] [CrossRef]
- Song, P.; Zhang, H.; Han, D.; Li, J.; Yang, Z.; Wang, Q. Preparation of biomorphic porous LaFeO3 by sorghum straw biotemplate method and its acetone sensing properties. Sens. Actuators B Chem. 2014, 196, 140–146. [Google Scholar] [CrossRef]
- Fan, K.; Qin, H.; Zhang, Z.; Sun, L.; Sun, L.; Hu, J. Gas sensing properties of nanocrystalline La0.75Ba0.25FeO3 thick-film sensors. Sens. Actuators B Chem. 2012, 171, 302–308. [Google Scholar] [CrossRef]
- Shi, C.M.; Qin, H.W.; Zhao, M.; Wang, X.F.; Li, L.; Hu, J.F. Investigation on electrical transport, CO sensing characteristics and mechanism for nanocrystalline La1−xCaxFeO3 sensors. Sens. Actuators B Chem. 2014, 190, 25–31. [Google Scholar] [CrossRef]





| Author | Materials | To | C | S | Ref. |
|---|---|---|---|---|---|
| Epifani et al. | TiO2 | 400 | 100 | 1.2 | [4] |
| Bian et al. | TiO2 | 500 | 10 | 9 | [5] |
| Bhowmik et al. | TiO2 | 270 | 10 | 1.136 | [6] |
| Wang et al. | Au-doped NiO | 240 | 20 | 7.6 | [7] |
| Wang et al. | W-doped NiO | 250 | 100 | 198.1 | [8] |
| Wei et al. | ZnO | 220 | 1 | 7.1 | [9] |
| An et al. | ZnO | 220 | 100 | 6.0 | [10] |
| Al-Hardan et al. | Cr-doped ZnO | 400 | 500 | 90 | [11] |
| Peng et al. | ZnO | 300 | 100 | 18.6 | [12] |
| Rajgure et al. | ZnO | 350 | 2000 | 92 | [13] |
| Zhang et al. | Co3O4 | 150 | 10 | 1.7 | [14] |
| Su et al. | Sm-doped α-Fe2O3 | 240 | 0.5 | 2.3 | [15] |
| Shan et al. | La-doped α-Fe2O3 | 240 | 50 | 26 | [16] |
| Kim et al. | WO3 with both Pd and Au | 300 | 200 | 152.4 | [17] |
| Chen et al. | WO3 | 300 | 2 | 2 | [18] |
| Tomer et al. | WO3-SnO2 | 200 | 50 | 31.3 | [19] |
| Malik et | Pd-WO3 | 200 | 25 | 21.3 | [20] |
| Mishra et al. | SnO2 | 250 | 10 | 42 | [21] |
| Jin et al. | SnO2 | 260 | 25 | 40 | [22] |
| Punginsang et al. | Co-doped SnO2 | 250 | 20 | 36.9 | [23] |
| Singkammo et al. | Ni-Doped SnO2 | 350 | 200 | 54.2 | [24] |
| Patil et al. | Co-doped SnO2 | 270 | 60 | 32 | [25] |
| Tomer et | Ag-CN | 250 | 50 | 16.1 | [26] |
| Wu et al. | NdFeO3 | 120 | 50 | 300 | [27] |
| Liu et al. | SmFe0.9Mg0.1O3 | 260 | 300 | 353 | [28] |
| Zhang et al. | Ca-doped YbFeO3 | 230 | 0.1 | 1.72 | [29] |
| Yang et al. | LaNi0.5Ti0.5O3 | 350 | 5 | 29.3 | [30] |
| Chen et al. | SmFeO3 | 250 | 380 | 2.6 | [31] |
| Liu et al. | LaFeO3 | 400 | 80 | 204 | [32] |
| Song et al. | LaFeO3 | 240 | 200 | 12.2 | [33] |
| Fan et al. | La0.75Ba0.25FeO3 | 240 | 50 | 17 | [34] |
| LaFeO3 | 200 | 0.5 | 1.18 | Present work | |
| LaFeO3 | 200 | 1 | 1.22 | Present work | |
| LaFeO3 | 200 | 2 | 1.89 | Present work | |
| LaFeO3 | 200 | 5 | 3.2 | Present work | |
| LaFeO3 | 200 | 10 | 7.83 | Present work | |
| LaFeO3 under 365 nm | 180 | 0.5 | 1.37 | Present work | |
| LaFeO3 under 365 nm | 180 | 1 | 1.85 | Present work | |
| LaFeO3 under 365 nm | 180 | 2 | 3.16 | Present work | |
| LaFeO3 under 365 nm | 180 | 5 | 8.32 | Present work | |
| LaFeO3 under 365 nm | 180 | 10 | 14.1 | Present work |
| Number | Condition | Gender | Age | FPG (mmol/L) |
|---|---|---|---|---|
| A | Healthy | man | 52 | 4.5 |
| B | Healthy | woman | 52 | 5.3 |
| C | diabetic | man | 52 | 7.8 |
| D | diabetic | woman | 52 | 9.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Qin, H.; Gao, C.; Zhou, G.; Chen, Y.; Hu, J. UV Light Illumination Can Improve the Sensing Properties of LaFeO3 to Acetone Vapor. Sensors 2018, 18, 1990. https://doi.org/10.3390/s18071990
Zhang H, Qin H, Gao C, Zhou G, Chen Y, Hu J. UV Light Illumination Can Improve the Sensing Properties of LaFeO3 to Acetone Vapor. Sensors. 2018; 18(7):1990. https://doi.org/10.3390/s18071990
Chicago/Turabian StyleZhang, Heng, Hongwei Qin, Chengyong Gao, Guangjun Zhou, Yanping Chen, and Jifan Hu. 2018. "UV Light Illumination Can Improve the Sensing Properties of LaFeO3 to Acetone Vapor" Sensors 18, no. 7: 1990. https://doi.org/10.3390/s18071990
APA StyleZhang, H., Qin, H., Gao, C., Zhou, G., Chen, Y., & Hu, J. (2018). UV Light Illumination Can Improve the Sensing Properties of LaFeO3 to Acetone Vapor. Sensors, 18(7), 1990. https://doi.org/10.3390/s18071990





