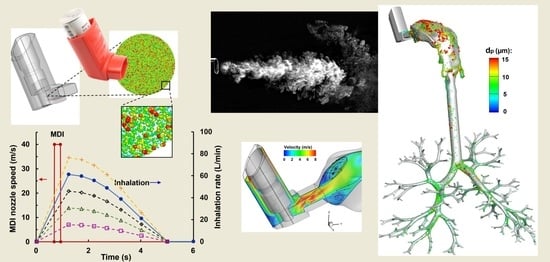
Lower Inspiratory Breathing Depth Enhances Pulmonary Delivery Efficiency of ProAir Sprays
Abstract
:1. Introduction
2. Results
2.1. Model Development for MDI Delivery
2.1.1. MDI-Airway Geometry
2.1.2. Modeling of Spray Aerosols during Actuation
2.1.3. Inhalation Waveforms: Control and Variants
2.1.4. Computational Mesh
2.2. Characterization of MDI Actuation
2.2.1. Imaging of MDI Sprays
2.2.2. Reverse Identification of Orifice Discharge Velocity
2.3. Flow and Aerosol Dynamics in Airway: Control Case (QMax = 60 L/min)
2.3.1. Airflow Dynamics
2.3.2. Spray Droplet Dynamics
2.4. Effects of Breathing Depth
2.4.1. Variation in Airflows
2.4.2. Surface Deposition of MDI Droplets
2.4.3. Deposition Fraction (DF) and Penetration Rate (PR) vs. Time
2.4.4. Deposition Enhancement Factor (DEF)
3. Discussion
4. Materials and Methods
4.1. MDI and Airway Models
4.2. High-Speed Imaging and Image Analysis
4.3. Numerical Methods
4.3.1. Boundary Conditions
4.3.2. Experiment-Based Estimation of the Spray Discharge Speed
4.3.3. Flow and Particle Transport Simulations
5. Conclusions
- The initial velocity of the spray plume from the ProAir-FHA actuator orifice was predicted to be 26 m/s, which matched the measured velocities at 3 and 6 cm from the mouthpiece.
- The LES-Lagrangian predicted spray plume topologies into the open-air agreed well with corresponding high-speed images both temporally and spatially.
- The drug loss in the device itself peaked at 45–60 L/min, while that in the mouth constantly increased with the inhalation depth from 15 to 75 L/min.
- The pulmonary drug delivery efficiency (beyond G9) had a negative relationship with the inhalation rate, with a predicted penetration rate of 11.4% at PIFR of 60 L/min, 30.7% at 30 L/min, and 45.7% at 15 L/min.
- Model cross-validation with existing experiments indicates a high dosimetry sensitivity to initial spray properties (size and velocity) and transient inhalation rate, which should be correctly considered in numerical modeling for accurate dosimetry predictions.
- This study has not reached the most efficient way of inhalation for optimal ProAir delivery yet; however, improved personalized inhalation therapy can be achieved by matching the inhaler type with the patient’s disease and breathing capacity.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Newman, S.P.; Pavia, D.; Clarke, S.W. Simple instructions for using pressurized aerosol bronchodilators. J. R. Soc. Med. 1980, 73, 776–779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laube, B.L.; Janssens, H.M.; de Jongh, F.H.; Devadason, S.G.; Dhand, R.; Diot, P.; Everard, M.L.; Horvath, I.; Navalesi, P.; Voshaar, T.; et al. What the pulmonary specialist should know about the new inhalation therapies. Eur. Respir. J. 2011, 37, 1308–1331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Capstick, T.G.; Clifton, I.J. Inhaler technique and training in people with chronic obstructive pulmonary disease and asthma. Expert Rev. Respir. Med. 2012, 6, 91–101, quiz 102–103. [Google Scholar] [CrossRef] [PubMed]
- Melani, A.S.; Bonavia, M.; Cilenti, V.; Cinti, C.; Lodi, M.; Martucci, P.; Serra, M.; Scichilone, N.; Sestini, P.; Aliani, M.; et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir. Med. 2011, 105, 930–938. [Google Scholar] [CrossRef] [Green Version]
- Chrystyn, H.; van der Palen, J.; Sharma, R.; Barnes, N.; Delafont, B.; Mahajan, A.; Thomas, M. Device errors in asthma and COPD: Systematic literature review and meta-analysis. NPJ Prim. Care Respir. Med. 2017, 27, 22. [Google Scholar] [CrossRef]
- Allen, S.C.; Prior, A. What determines whether an elderly patient can use a metered dose inhaler correctly? Br. J. Dis. Chest 1986, 80, 45–49. [Google Scholar] [CrossRef]
- Tobin, M.J. Use of bronchodilator aerosols. Arch. Intern. Med. 1985, 145, 1659–1663. [Google Scholar] [CrossRef]
- Biswas, R.; Hanania, N.A.; Sabharwal, A. Factors determining in vitro lung deposition of albuterol aerosol delivered by ventolin metered-dose inhaler. J. Aerosol Med. Pulm. Drug Deliv. 2017, 30, 256–266. [Google Scholar] [CrossRef]
- Smyth, H.; Hickey, A.J.; Brace, G.; Barbour, T.; Gallion, J.; Grove, J. Spray pattern analysis for metered dose inhalers I: Orifice size, particle size, and droplet motion correlations. Drug Dev. Ind. Pharm. 2006, 32, 1033–1041. [Google Scholar] [CrossRef]
- Worth Longest, P.; Hindle, M.; Das Choudhuri, S. Effects of generation time on spray aerosol transport and deposition in models of the mouth-throat geometry. J. Aerosol Med. Pulm. Drug Deliv. 2009, 22, 67–83. [Google Scholar] [CrossRef]
- Fadl, A.; Wang, J.; Yang, P.; Zhang, Z.; Cheng, Y.S. Balloon-based in vitro MDI aerosol deposition experiments on the effects of mouthpiece diameter. Inhal. Toxicol. 2007, 19, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.C.; Breysse, P.N.; Laube, B.L.; Swift, D.L. Mouthpiece diameter affects deposition efficiency in cast models of the human oral airways. J. Aerosol Med. Off. J. Int. Soc. Aerosols Med. 2001, 14, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gilbertson, K.; Finlay, W.H. In vivo-in vitro comparison of deposition in three mouth-throat models with Qvar and Turbuhaler inhalers. J. Aerosol Med. Off. J. Int. Soc. Aerosols Med. 2007, 20, 227–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yousefi, M.; Inthavong, K.; Tu, J. Effect of Pressurized Metered Dose Inhaler Spray Characteristics and Particle Size Distribution on Drug Delivery Efficiency. J. Aerosol Med. Pulm. Drug Deliv. 2017, 30, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Kleinstreuer, C.; Shi, H.; Zhang, Z. Computational analyses of a pressurized metered dose inhaler and a new drug-aerosol targeting methodology. J. Aerosol Med. Off. J. Int. Soc. Aerosols Med. 2007, 20, 294–309. [Google Scholar] [CrossRef]
- Longest, P.W.; Tian, G.; Walenga, R.L.; Hindle, M. Comparing MDI and DPI aerosol deposition using in vitro experiments and a new stochastic individual path (SIP) model of the conducting airways. Pharm. Res. 2012, 29, 1670–1688. [Google Scholar] [CrossRef]
- Xi, J.; Talaat, M.; Si, X.A.; Chandra, S. The application of statistical shape modeling for lung morphology in aerosol inhalation dosimetry. J. Aerosol Sci. 2021, 151, 105623. [Google Scholar] [CrossRef]
- Talaat, M.; Si, X.A.; Dong, H.; Xi, J. Leveraging statistical shape modeling in computational respiratory dynamics: Nanomedicine delivery in remodeled airways. Comput. Methods Programs Biomed. 2021, 204, 106079. [Google Scholar] [CrossRef]
- Talaat, M.; Si, X.; Xi, J. Effect of MDI actuation timing on inhalation dosimetry in a human respiratory tract model. Pharmaceuticals 2022, 15, 61. [Google Scholar] [CrossRef]
- Liu, X.; Doub, W.H.; Guo, C. Evaluation of metered dose inhaler spray velocities using phase Doppler anemometry (PDA). Int. J. Pharm. 2012, 423, 235–239. [Google Scholar] [CrossRef]
- Longest, P.W.; Vinchurkar, S. Effects of mesh style and grid convergence on particle deposition in bifurcating airway models with comparisons to experimental data. Med. Eng. Phys. 2007, 29, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.H.; Cheng, Y.S.; Yeh, H.C.; Swift, D.L. Measurements of airway dimensions and calculation of mass transfer characteristics of the human oral passage. J. Biomech. Eng. 1997, 119, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Longest, P.W. Transport and deposition of micro-aerosols in realistic and simplified models of the oral airway. Ann. Biomed. Eng. 2007, 35, 560–581. [Google Scholar] [CrossRef]
- Zhang, Y.; Finlay, W.H. Experimental measurements of particle deposition in three proximal lung bifurcation models with an idealized mouth-throat. J. Aerosol Med. Off. J. Int. Soc. Aerosols Med. 2005, 18, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Sun, J.; Cheng, Y.S. Comparison of deposition in the USP and physical mouth-throat models with solid and liquid particles. J. Aerosol Med. Pulm. Drug Deliv. 2011, 24, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Roche, N.; Dekhuijzen, P.N. The evolution of pressurized metered-dose inhalers from early to modern devices. J. Aerosol Med. Pulm. Drug Deliv. 2016, 29, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Usmani, O.S.; Lavorini, F.; Marshall, J.; Dunlop, W.C.N.; Heron, L.; Farrington, E.; Dekhuijzen, R. Critical inhaler errors in asthma and COPD: A systematic review of impact on health outcomes. Respir. Res. 2018, 19, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, C.; Ahmadi, G. Particle deposition with thermophoresis in laminar and turbulent duct flows. Aerosol Sci. Technol. 1998, 29, 525–546. [Google Scholar] [CrossRef]
- Yin, Z.-Q.; Li, X.-F.; Bao, F.-B.; Tu, C.-X.; Gao, X.-Y. Thermophoresis and Brownian motion effects on nanoparticle deposition inside a 90° square bend tube. Aerosol Air Qual. Res. 2018, 18, 1746–1755. [Google Scholar] [CrossRef] [Green Version]
- Xi, J.; Kim, J.; Si, X.A.; Zhou, Y. Hygroscopic aerosol deposition in the human upper respiratory tract under various thermo-humidity conditions. J. Environ. Sci. Health A 2013, 48, 1790–1805. [Google Scholar] [CrossRef]
- Stein, S.W.; Myrdal, P.B. The Relative Influence of Atomization and Evaporation on Metered Dose Inhaler Drug Delivery Efficiency. Aerosol Sci. Technol. 2006, 40, 335–347. [Google Scholar] [CrossRef] [Green Version]
- Xi, J.; Eddie Yuan, J.; Alshaiba, M.; Cheng, D.; Firlit, Z.; Johnson, A.; Nolan, A.; Su, W.-C. Design and testing of electric-guided delivery of charged particles to the olfactory region: Experimental and numerical studies. Curr. Drug Deliv. 2016, 13, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Yuan, J.E.; Si, X.A.; Hasbany, J. Numerical optimization of targeted delivery of charged nanoparticles to the ostiomeatal complex for treatment of rhinosinusitis. Int. J. Nanomed. 2015, 10, 4847–4861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azhdarzadeh, M.; Olfert, J.S.; Vehring, R.; Finlay, W.H. Effect of electrostatic charge on deposition of uniformly charged monodisperse particles in the nasal extrathoracic airways of an infant. J. Aerosol Med. Pulm. Drug Deliv. 2015, 28, 30–34. [Google Scholar] [CrossRef]
- Koullapis, P.G.; Kassinos, S.C.; Bivolarova, M.P.; Melikov, A.K. Particle deposition in a realistic geometry of the human conducting airways: Effects of inlet velocity profile, inhalation flowrate and electrostatic charge. J. Biomech. 2016, 49, 2201–2212. [Google Scholar] [CrossRef] [Green Version]
- Triep, M.; Brücker, C. Three-dimensional nature of the glottal jet. J. Acoust. Soc. Am. 2010, 127, 1537–1547. [Google Scholar] [CrossRef]
- Bailly, L.; Cochereau, T.; Orgéas, L.; Henrich Bernardoni, N.; Rolland du Roscoat, S.; McLeer-Florin, A.; Robert, Y.; Laval, X.; Laurencin, T.; Chaffanjon, P.; et al. 3D multiscale imaging of human vocal folds using synchrotron X-ray microtomography in phase retrieval mode. Sci. Rep. 2018, 8, 14003. [Google Scholar] [CrossRef]
- Zhao, J.; Feng, Y.; Fromen, C.A. Glottis motion effects on the particle transport and deposition in a subject-specific mouth-to-trachea model: A CFPD study. Comput. Biol. Med. 2020, 116, 103532. [Google Scholar] [CrossRef]
- Cheng, Y.S.; Zhou, Y.; Chen, B.T. Particle deposition in a cast of human oral airways. Aerosol Sci. Technol. 1999, 31, 286–300. [Google Scholar] [CrossRef]
- Kitaoka, H. The origin of frequency dependence of respiratory resistance: An airflow simulation study using a 4D pulmonary lobule model. Respirology 2011, 16, 517–522. [Google Scholar] [CrossRef]
- Kitaoka, H.; Nieman, G.F.; Fujino, Y.; Carney, D.; DiRocco, J.; Kawase, I. A 4-dimensional model of the alveolar structure. J. Physiol. Sci. 2007, 57, 175–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitaoka, H.; Takaki, R.; Suki, B. A three-dimensional model of the human airway tree. J. Appl. Physiol. 1985, 87, 2207–2217. [Google Scholar] [CrossRef] [PubMed]
- Kitaoka, H.; Tamura, S.; Takaki, R. A three-dimensional model of the human pulmonary acinus. J. Appl. Physiol. 2000, 88, 2260–2268. [Google Scholar] [CrossRef]
- Ochowiak, M.; Włodarczak, S.; Krupińska, A.; Matuszak, M. Particle image velocimetry based on matlab and PIVlab for testing flow disturbing elements. In Proceedings of the Advances in Design, Simulation and Manufacturing IV, Cham, Switzerland, 8–11 June 2021; pp. 268–276. [Google Scholar]
- Nicoud, F.; Ducros, F. Subgrid-scale stress modelling based on the square of the velocity gradient tensor. Flow Turbul. Combust. 1999, 62, 183–200. [Google Scholar] [CrossRef]
- Longest, P.W.; Xi, J. Effectiveness of direct Lagrangian tracking models for simulating nanoparticle deposition in the upper airways. Aerosol Sci. Technol. 2007, 41, 380–397. [Google Scholar] [CrossRef]














Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talaat, M.; Si, X.A.; Xi, J. Lower Inspiratory Breathing Depth Enhances Pulmonary Delivery Efficiency of ProAir Sprays. Pharmaceuticals 2022, 15, 706. https://doi.org/10.3390/ph15060706
Talaat M, Si XA, Xi J. Lower Inspiratory Breathing Depth Enhances Pulmonary Delivery Efficiency of ProAir Sprays. Pharmaceuticals. 2022; 15(6):706. https://doi.org/10.3390/ph15060706
Chicago/Turabian StyleTalaat, Mohamed, Xiuhua April Si, and Jinxiang Xi. 2022. "Lower Inspiratory Breathing Depth Enhances Pulmonary Delivery Efficiency of ProAir Sprays" Pharmaceuticals 15, no. 6: 706. https://doi.org/10.3390/ph15060706
APA StyleTalaat, M., Si, X. A., & Xi, J. (2022). Lower Inspiratory Breathing Depth Enhances Pulmonary Delivery Efficiency of ProAir Sprays. Pharmaceuticals, 15(6), 706. https://doi.org/10.3390/ph15060706