How “Pharmacoresistant” is Cav2.3, the Major Component of Voltage-Gated R-type Ca2+ Channels?
Abstract
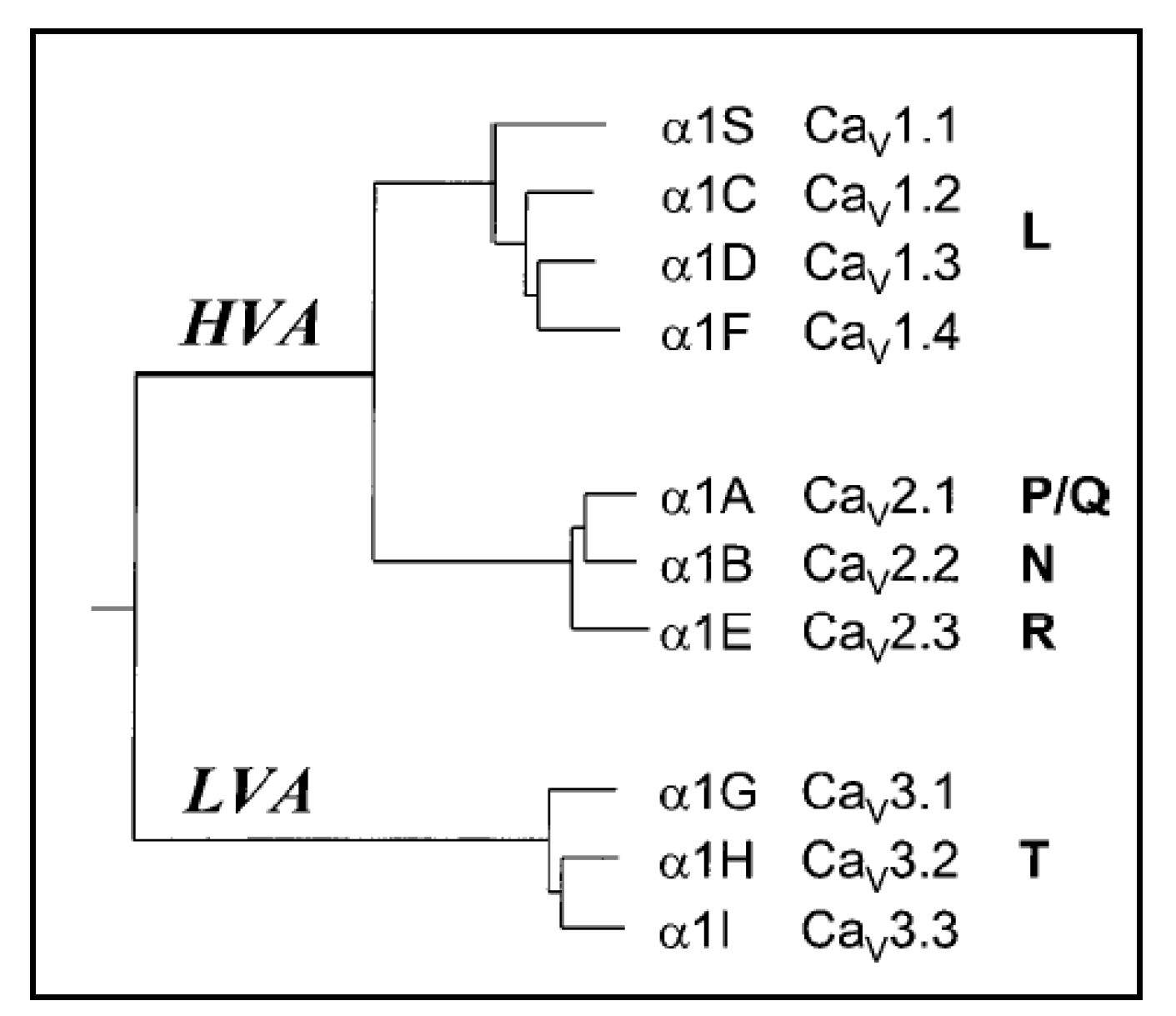
:1. The Cav2.3 Voltage-Gated Ca2+ Channel
| Nomenclature, splice variant | Structure related to alternate exons expressed (+) | Expression (tissue and species) | Ref. | |||
|---|---|---|---|---|---|---|
| Novel terms | Old terms | Exon 19 (57 nts) | Segment (21 nts) in exon 20 | Exon 45 (129 nts) | ||
| Cav2.3a | alpha1E-1 | - | + | - | Rat cerebellum | [100] |
| Cav2.3b | alpha1E-2 | + | - | - | Less important in CNS | [3] |
| Cav2.3c | alpha1E-3 | + | + | - | Dominant in CNS | [3] |
| Cav2.3d | alpha1Ed | + | + | + | Human fetal brain | [4] |
| Cav2.3e | alpha1Ee | - | + | + | Pancreas, kidney, heart | [70,101] |
| Cav2.3f | alpha1Ef | + | - | + | Rat cerebellum | [100] |
| Brain region (mouse) | Major splice variant | Miscellaneous |
|---|---|---|
| Neocortex | Cav2.3c | Minor amounts of Cav2.3e |
| Hippocampus | Cav2.3c | Minor amounts of Cav2.3e |
| Thalamus | Cav2.3c | Substantial amounts of Cav2.3e and Cav2.3f |
| Cerebellum, mesencephalon, medulla oblongata | Cav2.3e | minor amounts of Cav2.3a |
2. Selective and Non-Selective Antagonists of Cav2.3
3. Physiological Functions, in Which Cav2.3 may be Involved, as Deduced from Cav2.3-Deficient Mice
| Substance | Application | IC50 or Kd [µM] | Amount of max. Inhibition | Selectivity | Ref. |
|---|---|---|---|---|---|
| SNX-482 | Peptide toxin | 0.015–0.030 | > 80 % | Cav2.3-prevalent | [107,108,128,129,130] |
| ω-Aga-IVA | Peptide toxin | 0.051 | 80% | non-selective | [116] |
| ω-Aga-IIIA | Peptide toxin | 0.003–0.010 | 100% | non-selective | [107] |
| Ni2+ | Unphysiological | 27.4/303 | 100% | non-selective | [3,131] |
| Cd2+ | Unphysiological | 0.8 | 100% | non-selective | [3] |
| Zn2+ | Trace element | 31.8 | >90% | non-selective | [132] |
| Zn2+ (calibrated) | Trace element | 1.3 | 100% | non-selective | [10] |
| Cu2+ | Trace element | 0.018 | 100% | non-selective | [10] |
| Topiramate | Anticonvulsive | 50.9 | >70% | non-selective | [133] |
| Lamotrigine | Anticonvulsive | >10 | non-selective | [134] | |
| Sipatrigine | Anticonvulsive | 10 | 100% | non-selective | [134] |
| 202W92 | Anticonvulsive | 56 | 100% | [134] | |
| Ethosuximide | Anticonvulsive | 20000 | 100% | non-selective | [135] |
| MPS (α-methylphenylsuccinimide) | Anticonvulsive | 2300 | 100% | [135] | |
| Phenytoin | Anticonvulsive | 360 | 100% | [135] | |
| Phenobarbital | Anticonvulsive | 2700 | >80% | [135] | |
| Pentobarbital | Anticonvulsive | 600 | 100% | [135] | |
| Halothane | Inhalation anaesthetic | [136,137] | |||
| Isoflurane | Inhalation anaesthetic | 206 | 100% | [136,138,139] | |
| Fomocaine | Local anaestetic | 95 | 100% | [140] | |
| Procaine | Local anaestetic | [140] | |||
| Octanol | Organic solvent | 206 | 100% | [135] | |
| (+)-ACN | Steroid anaestetic | 5.3–10.2 | 100% | [141] | |
| (+)-ECN | Steroid anaestetic | 9.9–16.1 | >70% | [141] | |
| Flecainide | Antiarrhythmic | 320 | [140] | ||
| Penfluridol | Antipsychotic | 13 | [140] | ||
| Verapamil | Antihypertensive | 100 | 100% | non-selective | [142] |
| Diltiazem | Antihypertensive | 220 | 100% | non-selective | [4,142] |
| Isradipine | Antihypertensive | 9.1 | 100% | non-selective | [87] |
| Nicardipine | Antihypertensive | 1 | n.t. | non-selective | [116] |
| Mibefradil | Antihypertensive | 0.4/6.5 | 100% | non-selective | [143] |
| Amiloride | Diuretic | 7400 | 100% | non-selective | [135] |
| Ethoxyzolamide | Carboanhydrase inhibitor/anticonvulsive | 1 | 70% | [144] | |
| Eugenol | Analgetic | [145] | |||
| Bisphenol A | Environmental pollutant | 26 | 50% | non-selective | [146] |
Acknowledgments
Conflict of Interest
References
- Niidome, T.; Kim, M.-S.; Friedrich, T.; Mori, Y. Molecular cloning and characterization of a novel calcium channel from rabbit brain. FEBS Lett. 1992, 308, 7–13. [Google Scholar] [CrossRef]
- Soong, T.W.; Stea, A.; Hodson, C.D.; Dubel, S.J.; Vincent, S.R.; Snutch, T.P. Structure and functional expression of a member of the low voltage-activated calcium channel family. Science 1993, 260, 1133–1136. [Google Scholar]
- Williams, M.E.; Marubio, L.M.; Deal, C.R.; Hans, M.; Brust, P.F.; Philipson, L.H.; Miller, R.J.; Johnson, E.C.; Harpold, M.M.; Ellis, S.B. Structure and functional characterization of neuronal α1E calcium channel subtypes. J. Biol. Chem. 1994, 269, 22347–22357. [Google Scholar]
- Schneider, T.; Wei, X.; Olcese, R.; Costantin, J.L.; Neely, A.; Palade, P.; Perez-Reyes, E.; Qin, N.; Zhou, J.; Crawford, G.D.; et al. Molecular analysis and functional expression of the human type E α1 subunit. Receptors Channels 1994, 2, 255–270. [Google Scholar]
- Perez-Reyes, E. Molecular Physiology of Low-Voltage-Activated T-type Calcium Channels. Physiol Rev. 2003, 83, 117–161. [Google Scholar]
- Perez-Reyes, E.; Schneider, T. Calcium channels: Structure, function, and classification. Drug Dev. Res. 1994, 33, 295–318. [Google Scholar] [CrossRef]
- Pereverzev, A.; Leroy, J.; Krieger, A.; Malecot, C.O.; Hescheler, J.; Pfitzer, G.; Klockner, U.; Schneider, T. Alternate Splicing in the Cytosolic II-III Loop and the Carboxy Terminus of Human E-type Voltage-Gated Ca2+ Channels: Electrophysiological Characterization of Isoforms. Mol. Cell Neurosci. 2002, 21, 352–365. [Google Scholar] [CrossRef]
- Kamp, M.A.; Krieger, A.; Henry, M.; Hescheler, J.; Weiergräber, M.; Schneider, T. Presynaptic “Cav2.3 containing” E-type Ca2+ channels share dual roles during neurotransmitter release. Eur. J. Neurosci. 2005, 21, 1617–1625. [Google Scholar] [CrossRef]
- Weiergräber, M.; Kamp, M.A.; Radhakrishnan, K.; Hescheler, J.; Schneider, T. The Cav2.3 Voltage-gated calcium channel in epileptogenesis. Shedding new light on an enigmatic channel. Neurosci. Biobehav. Rev. 2006, 30, 1122–1144. [Google Scholar] [CrossRef]
- Shcheglovitov, A.; Vitko, I.; Lazarenko, R.M.; Orestes, P.; Todorovic, S.M.; Perez-Reyes, E. Molecular and biophysical basis of glutamate and trace metal modulation of voltage-gated Ca(v)2.3 calcium channels. J. Gen. Physiol 2012, 139, 219–234. [Google Scholar] [CrossRef]
- Spafford, J.D.; Zamponi, G.W. Functional interactions between presynaptic calcium channels and the neurotransmitter release machinery. Curr. Opin. Neurobiol. 2003, 13, 308–314. [Google Scholar] [CrossRef]
- Parent, L.; Schneider, T.; Moore, C.P.; Talwar, D. Subunit Regulation of the Human Brain α1E Calcium Channel. J. Membrane Biol. 1997, 160, 127–140. [Google Scholar] [CrossRef]
- Nakashima, Y.M.; Todorovic, S.M.; Pereverzev, A.; Hescheler, J.; Schneider, T.; Lingle, C.J. Properties of Ba2+ currents arising from human α1E and α1Eβ3 constructs expressed in HEK293 cells: Physiology, pharmacology, and comparison to native T-type Ba2+ currents. Neuropharmacology 1998, 37, 957–972. [Google Scholar] [CrossRef]
- Berrou, L.; Bernatchez, G.; Parent, L. Molecular Determinants of Inactivation within the I-II Linker of alpha1E (Cav2.3) Calcium Channels. Biophys. J. 2001, 80, 215–228. [Google Scholar] [CrossRef]
- Berrou, L.; Dodier, Y.; Raybaud, A.; Tousignant, A.; Dafi, O.; Pelletier, J.N.; Parent, L. The C-terminal residues in the alpha-interacting domain (AID) helix anchor CaVbeta subunit interaction and modulation of CaV2.3 channels. J. Biol. Chem. 2005, 280, 494–505. [Google Scholar]
- Shakeri, B.; Bourdin, B.; Demers-Giroux, P.O.; Sauve, R.; Parent, L. A quartet of leucine residues in the guanylate kinase domain of CaVbeta determines the plasma membrane density of the CaV2.3 channel. J. Biol. Chem. 2012, 287, 32835–32847. [Google Scholar]
- Flockerzi, V.; Oeken, H.J.; Hofmann, F. Purification of a functional receptor for calcium-channel blockers from rabbit skeletal-muscle microsomes. Eur. J. Biochem. 1986, 161, 217–224. [Google Scholar] [CrossRef]
- Sieber, M.; Nastainczyk, W.; Zubor, V.; Wernet, W.; Hofmann, F. The 165-kDa peptide of the purified skeletal muscle dihydropyridine receptor contains the known regulatory sites of the calcium channel. Eur. J. Biochem. 1987, 167, 117–122. [Google Scholar] [CrossRef]
- Takahashi, M.; Seagar, M.J.; Jones, J.F.; Reber, B.F.; Catterall, W.A. Subunit structure of dihydropyridine-sensitive calcium channels from skeletal muscle. Proc. Natl. Acad. Sci. USA 1987, 84, 5478–5482. [Google Scholar] [CrossRef]
- Striessnig, J.; Knaus, H.G.; Grabner, M.; Moosburger, K.; Seitz, W.; Lietz, H.; Glossmann, H. Photoaffinity labelling of the phenylalkylamine receptor of the skeletal muscle transverse-tubule calcium channel. FEBS Lett. 1987, 212, 247–253. [Google Scholar] [CrossRef]
- Schneider, T.; Hofmann, F. The bovine cardiac receptor for calcium channel blockers is a 195-kDa protein. Eur. J. Biochem. 1988, 174, 369–375. [Google Scholar] [CrossRef]
- Witcher, D.R.; De Waard, M.; Campbell, K.P. Characterization of the purified N-type Ca2+ channel and the cation sensitivity of omega-conotoxin GVIA binding. Neuropharmacology 1993, 32, 1127–1139. [Google Scholar] [CrossRef]
- Witcher, D.R.; de Waard, M.; Sakamoto, J.; Franzini Armstrong, C.; Pragnell, M.; Kahl, S.D.; Campbell, K.P. Subunit identification and reconstitution of the N-type Ca2+ channel complex purified from brain. Science 1993, 261, 486–489. [Google Scholar]
- Guy, H.R.; Conti, F. Pursuing the structure and function of voltage-gated channels. TINS 1990, 13, 201–206. [Google Scholar]
- Doyle, D.A.; Cabral, J.M.; Pfuetzner, R.A.; Kuo, A.L.; Gulbis, J.M.; Cohen, S.L.; Chait, B.T.; MacKinnon, R. The structure of the potassium channel: Molecular basis of K+ conduction and selectivity. Science 1998, 280, 69–77. [Google Scholar] [CrossRef]
- Long, S.B.; Campbell, E.B.; MacKinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 2005, 309, 897–903. [Google Scholar] [CrossRef]
- Payandeh, J.; Scheuer, T.; Zheng, N.; Catterall, W.A. The crystal structure of a voltage-gated sodium channel. Nature 2011, 475, 353–358. [Google Scholar] [CrossRef]
- Payandeh, J.; Gamal El-Din, T.M.; Scheuer, T.; Zheng, N.; Catterall, W.A. Crystal structure of a voltage-gated sodium channel in two potentially inactivated states. Nature 2012, 486, 135–139. [Google Scholar]
- Hofmann, F.; Lacinová, L.; Klugbauer, N. Voltage-dependent calcium channels: From structure to function. Rev. Physiol. Biochem. Pharmacol. 1999, 139, 33–87. [Google Scholar] [CrossRef]
- Zhen, X.G.; Xie, C.; Fitzmaurice, A.; Schoonover, C.E.; Orenstein, E.T.; Yang, J. Functional architecture of the inner pore of a voltage-gated Ca2+ channel. J. Gen. Physiol. 2005, 126, 193–204. [Google Scholar] [CrossRef]
- Xie, C.; Zhen, X.G.; Yang, J. Localization of the activation gate of a voltage-gated Ca2+ channel. J. Gen. Physiol. 2005, 126, 205–212. [Google Scholar] [CrossRef]
- Raybaud, A.; Baspinar, E.E.; Dionne, F.; Dodier, Y.; Sauve, R.; Parent, L. The role of distal S6 hydrophobic residues in the voltage-dependent gating of CaV2.3 channels. J. Biol. Chem. 2007, 282, 27944–27952. [Google Scholar] [CrossRef]
- Tang, S.; Mikala, G.; Bahinski, A.; Yatani, A.; Varadi, G.; Schwartz, A. Molecular localization of ion selectivity sites within the pore of a human L-type cardiac calcium channel. J. Biol. Chem. 1993, 268, 13026–13029. [Google Scholar]
- Yang, J.; Ellinor, P.T.; Sather, W.A.; Zhang, J.-F.; Tsien, R.W. Molecular determinants of Ca2+ selectivity and ion permeation in L-type Ca2+ channels. Nature 1993, 366, 158–161. [Google Scholar] [CrossRef]
- Kim, M.-S.; Morii, T.; Sun, L.-X.; Imoto, K.; Mori, Y. Structural determinants of ion selectivity in brain calcium channel. FEBS Lett. 1993, 318, 145–148. [Google Scholar] [CrossRef]
- Ellinor, P.T.; Yang, J.; Sather, W.A.; Zhang, J.F.; Tsien, R.W. Ca2+ channel selectivity at a single locus for high-affinity Ca2+ interactions. Neuron 1995, 15, 1121–1132. [Google Scholar] [CrossRef]
- Parent, L.; Gopalakrishnan, M. Glutamate substitution in repeat IV alters divalent and monovalent cation permeation in the heart Ca2+ channel. Biophys. J. 1995, 69, 1801–1813. [Google Scholar] [CrossRef]
- Dirksen, R.T.; Nakai, J.; Gonzalez, A.; Imoto, K.; Beam, K.G. The S5-S6 linker of repeat I is a critical determinant of L-type Ca2+ channel conductance. Biophys. J. 1997, 73, 1402–1409. [Google Scholar] [CrossRef]
- Cibulsky, S.M.; Sather, W.A. Control of ion conduction in L-type Ca2+ channels by the concerted action of S5–6 regions. Biophys. J. 2003, 84, 1709–1719. [Google Scholar] [CrossRef]
- Cibulsky, S.M.; Sather, W.A. The EEEE Locus Is the Sole High-affinity Ca2+ Binding Structure in the Pore of a Voltage-gated Ca2+ Channel Block by Ca2+ Entering from the Intracellular Pore Entrance. J. Gen. Physiol. 2000, 116, 349–362. [Google Scholar] [CrossRef]
- Jiang, Y.; Ruta, V.; Chen, J.; Lee, A.; MacKinnon, R. The principle of gating charge movement in a voltage-dependent K+ channel. Nature 2003, 423, 42–48. [Google Scholar] [CrossRef]
- Lacinova, L. Voltage-dependent calcium channels. Gen. Physiol Biophys. 2005, 24 (Suppl. 1), 1–78. [Google Scholar] [CrossRef]
- Lee, S.Y.; Banerjee, A.; MacKinnon, R. Two separate interfaces between the voltage sensor and pore are required for the function of voltage-dependent K+ channels. PLoS. Biol. 2009, 7, e47. [Google Scholar] [CrossRef]
- Van Petegem, F.; Clark, K.A.; Chatelain, F.C.; Minor, D.L., Jr. Structure of a complex between a voltage-gated calcium channel beta-subunit and an alpha-subunit domain. Nature 2004, 429, 671–675. [Google Scholar] [CrossRef]
- Petegem, F.V.; Chatelain, F.C.; Minor, D.L. Insights into voltage-gated calcium channel regulation from the structure of the Ca(V)1.2 IQ domain-Ca2+/calmodulin complex. Nat. Struct. Mol. Biol. 2005, 12, 1108–1115. [Google Scholar] [CrossRef]
- Kim, E.Y.; Rumpf, C.H.; Fujiwara, Y.; Cooley, E.S.; Van Petegem, F.; Minor, D.L., Jr. Structures of CaV2 Ca2+/CaM-IQ domain complexes reveal binding modes that underlie calcium-dependent inactivation and facilitation. Structure 2008, 16, 1455–1467. [Google Scholar] [CrossRef]
- Dick, I.E.; Tadross, M.R.; Liang, H.; Tay, L.H.; Yang, W.; Yue, D.T. A modular switch for spatial Ca2+ selectivity in the calmodulin regulation of CaV channels. Nature 2008, 451, 830–834. [Google Scholar] [CrossRef]
- Tadross, M.R.; Dick, I.E.; Yue, D.T. Mechanism of local and global Ca2+ sensing by calmodulin in complex with a Ca2+ channel. Cell 2008, 133, 1228–1240. [Google Scholar] [CrossRef]
- Krieger, A.; Radhakrishnan, K.; Pereverzev, A.; Siapich, S.A.; Banat, M.; Kamp, M.A.; Leroy, J.; Klöckner, U.; Hescheler, J.; Weiergräber, M.; Schneider, T. The molecular chaperone hsp70 interacts with the cytosolic II-III loop of the Cav2.3 E-type voltage-gated Ca2+ channel. Cell Physiol Biochem. 2006, 17, 97–110. [Google Scholar] [CrossRef]
- Kamp, M.A.; Shakeri, B.; Tevoufouet, E.E.; Krieger, A.; Henry, M.; Behnke, K.; Herzig, S.; Hescheler, J.; Radhakrishnan, K.; Parent, L.; Schneider, T. The C-terminus of human Ca(v)2.3 voltage-gated calcium channel interacts with alternatively spliced calmodulin-2 expressed in two human cell lines. Biochim. Biophys. Acta 2012, 1824, 1045–1057. [Google Scholar] [CrossRef]
- Radhakrishnan, K.; Kamp, M.A.; Siapich, S.A.; Hescheler, J.; Lüke, M.; Schneider, T. Cav2.3 Ca2+ channel interacts with the G1-subunit of V-ATPase. Cellul. Physiol. Biochem. 2011, 27, 421–432. [Google Scholar] [CrossRef]
- Radhakrishnan, K.; Krieger, A.; Dibué, M.; Hescheler, J.; Schneider, T. APLP1 and Rab5A Interact with the II-III loop of the Voltage-gated Ca2+-channel Cav2.3 and Modulate its Internalization Differently. Cellul. Physiol. Biochem. 2011, 28, 603–612. [Google Scholar] [CrossRef]
- Pragnell, M.; De Waard, M.; Mori, Y.; Tanabe, T.; Snutch, T.P.; Campbell, K.P. Calcium channel β-subunit binds to a conserved motif in the I-II cytoplasmic linker of the α1-subunit. Nature 1994, 368, 67–70. [Google Scholar] [CrossRef]
- De Waard, M.; Pragnell, M.; Campbell, K.P. Ca2+ channel regulation by a conserved β subunit domain. Neuron 1994, 13, 495–503. [Google Scholar] [CrossRef]
- Leroy, J.; Pereverzev, A.; Vajna, R.; Qin, N.; Pfitzer, G.; Hescheler, J.; Malecot, C.O.; Schneider, T.; Klockner, U. Ca2+-sensitive regulation of E-type Ca2+ channel activity depends on an arginine-rich region in the cytosolic II-III loop. Eur. J. Neurosci. 2003, 18, 841–855. [Google Scholar] [CrossRef]
- Klöckner, U.; Pereverzev, A.; Leroy, J.; Krieger, A.; Vajna, R.; Hescheler, J.; Pfitzer, G.; Malecot, C.O.; Schneider, T. The cytosolic II-III loop of Cav2.3 provides an essential determinant for the phorbol ester-mediated stimulation of E-type Ca2+ channel activity. Eur. J. Neurosci. 2004, 19, 2659–2668. [Google Scholar] [CrossRef]
- Mehrke, G.; Pereverzev, A.; Grabsch, H.; Hescheler, J.; Schneider, T. Receptor Mediated Modulation of Recombinant Neuronal Class E Calcium Channels. FEBS Lett. 1997, 408, 261–270. [Google Scholar] [CrossRef]
- Meza, U.; Bannister, R.; Melliti, K.; Adams, B. Biphasic, Opposing Modulation of Cloned Neuronal α1E Ca Channels by Distinct Signaling Pathways Coupled to M2 Muscarinic Acetylcholine Receptors. J. Neurosci. 1999, 19, 6806–6817. [Google Scholar]
- Melliti, K.; Meza, U.; Adams, B. Muscarinic stimulation of α1E Ca Channels is selectively blocked by the effector antagonist function of RGS2 and phsopholipase C-β1. J. Neurosci. 2000, 20, 7167–7173. [Google Scholar]
- Bannister, R.A.; Melliti, K.; Adams, B.A. Differential Modulation of CaV2.3 Ca2+ Channels by G{alpha}q/11-Coupled Muscarinic Receptors. Mol. Pharmacol. 2004, 65, 381–388. [Google Scholar] [CrossRef]
- Kamatchi, G.L.; Franke, R.; Lynch, C., III; Sando, J.J. Identification of sites responsible for potentiation of type 2.3 calcium currents by acetyl-beta-methylcholine. J. Biol. Chem. 2004, 279, 4102–4109. [Google Scholar]
- Tai, C.; Kuzmiski, J.B.; MacVicar, B.A. Muscarinic enhancement of R-type calcium currents in hippocampal CA1 pyramidal neurons. J. Neurosci. 2006, 26, 6249–6258. [Google Scholar] [CrossRef]
- Liang, H.; DeMaria, C.D.; Erickson, M.G.; Mori, M.X.; Alseikhan, B.A.; Yue, D.T. Unified Mechanisms of Ca2+ Regulation across the Ca2+ Channel Family. Neuron 2003, 39, 951–960. [Google Scholar] [CrossRef]
- Weiergräber, M.; Henry, M.; Krieger, A.; Kamp, M.A.; Radhakrishnan, K.; Hescheler, J.; Schneider, T. Altered seizure susceptibility in mice lacking the Cav2.3 E-type Ca2+ channel. Epilepsia 2006, 47, 839–850. [Google Scholar] [CrossRef]
- Sochivko, D.; Chen, J.; Becker, A.; Beck, H. Blocker-resistant Ca2+ currents in rat CA1 hippocampal pyramidal neurons. Neuroscience 2003, 116, 629–638. [Google Scholar] [CrossRef]
- Sochivko, D.; Pereverzev, A.; Smyth, N.; Gissel, C.; Schneider, T.; Beck, H. The α1E calcium channel subunit underlies R-type calcium current in hippocampal and cortical pyramidal neurons. J. Physiol. 2002, 542, 699–710. [Google Scholar] [CrossRef]
- Dietrich, D.; Kirschstein, T.; Kukley, M.; Pereverzev, A.; von der Brelie, C.; Schneider, T.; Beck, H. Functional specialization of presynaptic Cav2.3 Ca2+ channels. Neuron 2003, 39, 483–496. [Google Scholar] [CrossRef]
- Osanai, M.; Saegusa, H.; Kazuno, A.A.; Nagayama, S.; Hu, Q.; Zong, S.; Murakoshi, T.; Tanabe, T. Altered cerebellar function in mice lacking CaV2.3 Ca2+ channel. Biochem. Biophys. Res. Commun. 2006, 344, 920–925. [Google Scholar] [CrossRef]
- Han, W.; Saegusa, H.; Zong, S.; Tanabe, T. Altered cocaine effects in mice lacking Ca(v)2.3 (alpha(1E)) calcium channel. Biochem. Biophys. Res. Commun. 2002, 299, 299–304. [Google Scholar] [CrossRef]
- Vajna, R.; Schramm, M.; Pereverzev, A.; Arnhold, S.; Grabsch, H.; Klöckner, U.; Perez-Reyes, E.; Hescheler, J.; Schneider, T. New Isoform of the Neuronal Ca2+ Channel α1E Subunit in Islets of Langerhans, and Kidney. Distribution of Voltage-Gated Ca2+ Channel α1 Subunits in Cell Lines and Tissues. Eur. J. Biochem. 1998, 257, 274–285. [Google Scholar]
- Vajna, R.; Klöckner, U.; Pereverzev, A.; Weiergräber, M.; Chen, X.H.; Miljanich, G.; Klugbauer, N.; Hescheler, J.; Perez-Reyes, E.; Schneider, T. Functional coupling between 'R-type' calcium channels and insulin secretion in the insulinoma cell line INS-1. Eur. J. Biochem. 2001, 268, 1066–1075. [Google Scholar] [CrossRef]
- Pereverzev, A.; Mikhna, M.; Vajna, R.; Gissel, C.; Henry, M.; Weiergräber, M.; Hescheler, J.; Smyth, N.; Schneider, T. Disturbances in glucose-tolerance, insulin-release and stress-induced hyperglycemia upon disruption of the Cav2.3 (α1E) subunit of voltage-gated Ca2+ channels. Mol. Endocrinol. 2002, 16, 884–895. [Google Scholar] [CrossRef]
- Pereverzev, A.; Vajna, R.; Pfitzer, G.; Hescheler, J.; Klockner, U.; Schneider, T. Reduction of insulin secretion in the insulinoma cell line INS-1 by overexpression of a Ca(v)2.3 (alpha1E) calcium channel antisense cassette. Eur. J. Endocrinol. 2002, 146, 881–889. [Google Scholar] [CrossRef]
- Pereverzev, A.; Salehi, A.; Mikhna, M.; Renstrom, E.; Hescheler, J.; Weiergraber, M.; Smyth, N.; Schneider, T. The ablation of the Ca(v)2.3/E-type voltage-gated Ca2+ channel causes a mild phenotype despite an altered glucose induced glucagon response in isolated islets of Langerhans. Eur. J. Pharmacol. 2005, 511, 65–72. [Google Scholar] [CrossRef]
- Jing, X.; Li, D.Q.; Olofsson, C.S.; Salehi, A.; Surve, V.V.; Caballero, J.; Ivarsson, R.; Lundquist, I.; Pereverzev, A.; Schneider, T.; Rorsman, P.; Renstrom, E. Ca(V)2.3 calcium channels control second-phase insulin release. J. Clin. Invest. 2005, 115, 146–154. [Google Scholar]
- Wang, G.; Dayanithi, G.; Newcomb, R.; Lemos, J.R. An R-Type Ca2+ Current in Neurohypophysial Terminals Preferentially Regulates Oxytocin Secretion. J. Neurosci. 1999, 19, 9235–9241. [Google Scholar]
- Ortiz-Miranda, S.; Dayanithi, G.; Custer, E.; Treistman, S.N.; Lemos, J.R. Micro-opioid receptor preferentially inhibits oxytocin release from neurohypophysial terminals by blocking R-type Ca2+ channels. J. Neuroendocrinol. 2005, 17, 583–590. [Google Scholar] [CrossRef]
- Albillos, A.; Neher, E.; Moser, T. R-type Ca2+ channels are coupled to the rapid component of secretion in mouse adrenal slice chromaffin cells. J. Neurosci. 2000, 20, 8323–8330. [Google Scholar]
- Mergler, S.; Wiedenmann, B.; Prada, J. R-type Ca2+-channel activity is associated with chromogranin A secretion in human neuroendocrine tumor BON cells. J. Membr. Biol. 2003, 194, 177–186. [Google Scholar] [CrossRef]
- Zhang, Q.; Bengtsson, M.; Partridge, C.; Salehi, A.; Braun, M.; Cox, R.; Eliasson, L.; Johnson, P.R.; Renström, E.; Schneider, T.; Berggren, P.-O.; Gopel, S.; Ashcroft, F.M.; Rorsman, P. R-type calcium-channel-evoked CICR regulates glucose-induced somatostatin secretion. Nat. Cell Biol. 2007, 9, 453–460. [Google Scholar] [CrossRef]
- Watanabe, M.; Sakuma, Y.; Kato, M. High expression of the R-type voltage-gated Ca2+ channel and its involvement in Ca2+-dependent gonadotropin-releasing hormone release in GT1-7 cells. Endocrinology 2004, 145, 2375–2383. [Google Scholar] [CrossRef]
- Grabsch, H.; Pereverzev, A.; Weiergräber, M.; Schramm, M.; Henry, M.; Vajna, R.; Beattie, R.E.; Volsen, S.G.; Klöckner, U.; Hescheler, J.; Schneider, T. Immunohistochemical detection of α1E voltage-gated Ca2+ channel isoforms in cerebellum, INS-1 cells, and neuroendocrine cells of the digestive system. J. Histochem. Cytochem. 1999, 47, 981–993. [Google Scholar] [CrossRef]
- Matsuda, Y.; Saegusa, H.; Zong, S.; Noda, T.; Tanabe, T. Mice Lacking Cav2.3 (α1E) Calcium Channel Exhibit Hyperglycemia. Biochem. Biophys. Res. Commun. 2001, 289, 791–795. [Google Scholar] [CrossRef]
- Holmkvist, J.; Tojjar, D.; Almgren, P.; Lyssenko, V.; Lindgren, C.M.; Isomaa, B.; Tuomi, T.; Berglund, G.; Renstrom, E.; Groop, L. Polymorphisms in the gene encoding the voltage-dependent Ca2+ channel Ca (V)2.3 (CACNA1E) are associated with type 2 diabetes and impaired insulin secretion. Diabetologia 2007, 50, 2467–2475. [Google Scholar] [CrossRef]
- Muller, Y.L.; Hanson, R.L.; Zimmerman, C.; Harper, I.; Sutherland, J.; Kobes, S.; Knowler, W.C.; Bogardus, C.; Baier, L.J. Variants in the Cav2.3 (alpha1E) Subunit of Voltage-Activated Ca2+ Channels are Associated with Insulin Resistance and Type 2 Diabetes in Pima Indians. Diabetes 2007, 56, 3089–3094. [Google Scholar] [CrossRef]
- Weiergräber, M.; Pereverzev, A.; Vajna, R.; Henry, M.; Schramm, M.; Nastainczyk, W.; Grabsch, H.; Schneider, T. Immunodetection of α1E voltage-gated Ca2+ channel in chromogranin-positive muscle cells of rat heart, and in distal tubules of human kidney. J. Histochem. Cytochem. 2000, 48, 807–819. [Google Scholar] [CrossRef]
- Lu, Z.-L.; Pereverzev, A.; Liu, H.-L.; Weiergraber, M.; Henry, M.; Krieger, A.; Smyth, N.; Hescheler, J.; Schneider, T. Arrhythmia in isolated prenatal hearts after ablation of the Cav2.3 (α1E) subunit of voltage-gated Ca2+ channels. Cell. Physiol. Biochem. 2004, 14, 11–22. [Google Scholar] [CrossRef]
- Weiergräber, M.; Henry, M.; Südkamp, M.; De Vivie, E.R.; Hescheler, J.; Schneider, T. Ablation of Cav2.3/E-type voltage-gated calcium channel results in cardiac arrhythmia and altered autonomic control within the murine cardiovascular system. Basic Res. Cardiol. 2005, 100, 1–13. [Google Scholar] [CrossRef]
- Natrajan, R.; Little, S.E.; Reis-Filho, J.S.; Hing, L.; Messahel, B.; Grundy, P.E.; Dome, J.S.; Schneider, T.; Vujanic, G.M.; Pritchard-Jones, K.; Jones, C. Amplification and overexpression of CACNA1E correlates with relapse in favorable histology Wilms’ tumors. Clin. Cancer Res. 2006, 12, 7284–7293. [Google Scholar] [CrossRef]
- Wennemuth, G.; Westenbroek, R.E.; Xu, T.; Hille, B.; Babcock, D.F. Cav2.2 and Cav2,3 (N- and R-type) Ca2+ channels in depolarization-evoked entry of Ca2+ into mouse sperm. J. Biol. Chem. 2000, 275, 21210–21217. [Google Scholar]
- Carlson, A.E.; Westenbroek, R.E.; Quill, T.; Ren, D.; Clapham, D.E.; Hille, B.; Garbers, D.L.; Babcock, D.F. CatSper1 required for evoked Ca2+ entry and control of flagellar function in sperm. Proc. Natl. Acad. Sci. USA 2003, 100, 14864–14868. [Google Scholar] [CrossRef]
- Lievano, A.; Santi, C.M.; Serrano, C.J.; Trevino, C.L.; Bellve, A.R.; Hernandez-Cruz, A.; Darszon, A. T-type Ca2+ channels and alpha1E expression in spermatogenic cells, and their possible relevance to the sperm acrosome reaction. FEBS Lett. 1996, 388, 150–154. [Google Scholar] [CrossRef]
- Sakata, Y.; Saegusa, H.; Zong, S.Q.; Osanai, M.; Murakoshi, T.; Shimizu, Y.; Noda, T.; Aso, T.; Tanabe, T. Cav2.3 (α1E) Ca2+ channel participates in the control of sperm function. FEBS Lett. 2002, 516, 229–233. [Google Scholar] [CrossRef]
- Kamphuis, W.; Hendriksen, H. Expression patterns of voltage-dependent calcium channel α1 subunits (α1A-α1E) mRNA in rat retina. Mol. Brain Res. 1998, 55, 209–220. [Google Scholar] [CrossRef]
- Lüke, M.; Henry, M.; Lingohr, T.; Maghsoodian, M.; Hescheler, J.; Sickel, w.; Schneider, T. A Ni2+-sensitive component of the ERG-b-wave from the isolated bovine retina is related to E-type voltage-gated Ca2+ channels. Graefes Arch. Clin. Exp. Ophthalmol. 2005, 243, 933–941. [Google Scholar] [CrossRef]
- Siapich, S.A.; Banat, M.; Albanna, W.; Hescheler, J.; Lüke, M.; Schneider, T. Antagonists of ionotropic gamma-aminobutyric acid receptors impair the NiCl2-mediated stimulation of the electroretinogramm b-wave amplitude from the isolated superfused vertebrate retina. Acta Ophthalmol. 2009, 87, 854–865. [Google Scholar] [CrossRef]
- Siapich, S.A.; Wrubel, H.; Albanna, W.; Hescheler, J.; Weiergräber, M.; Lüke, M.; Schneider, T. Effect of ZnCl2 and chelation of zinc ions by N,N-diethyldithiocarbamate (DEDTC) on the ERG b-wave amplitude from the isolated and superfused vertebrate retina. Curr. Eye Res. 2010, 35, 322–334. [Google Scholar] [CrossRef]
- Yokoyama, C.T.; Westenbroek, R.E.; Hell, J.W.; Soong, T.W.; Snutch, T.P.; Catterall, W.A. Biochemical properties and subcellular distribution of the neuronal class E calcium channel α1 subunit. J. Neurosci. 1995, 15, 6419–6432. [Google Scholar]
- Brenowitz, S.D.; Regehr, W.G. “Resistant” channels reluctantly reveal their roles. Neuron 2003, 39, 391–394. [Google Scholar] [CrossRef]
- Schramm, M.; Vajna, R.; Pereverzev, A.; Tottene, A.; Klöckner, U.; Pietrobon, D.; Hescheler, J.; Schneider, T. Isoforms of α1E voltage-gated calcium channels in rat cerebellar granule cells—Detection of major calcium channel α1-transcripts by reverse transcription-polymerase chain reaction. Neuroscience 1999, 92, 565–575. [Google Scholar] [CrossRef]
- Galetin, T.; Tevoufouet, E.E.; Sandmeyer, J.; Matthes, J.; Nguemo, F.; Hescheler, J.; Weiergräber, M.; Schneider, T. Pharmacoresistant CaV2.3 (E-/R-type) voltage-gated calcium channels influence heart rate dynamics and contribute to cardiac impulse conduction. Cell Biochem. Funct. 2012. [Google Scholar] [CrossRef]
- Olcese, R.; Qin, N.; Schneider, T.; Neely, A.; Wei, X.; Stefani, E.; Birnbaumer, L. The amino terminus of a calcium channel β subunit sets rates of channel inactivation independently of the subunit's effect on activation. Neuron 1994, 13, 1433–1438. [Google Scholar] [CrossRef]
- Perez-Reyes, E.; Schneider, T. Molecular biology of calcium channels. Kidney Int. 1995, 48, 1111–1124. [Google Scholar] [CrossRef]
- Ellinor, P.T.; Zhang, J.F.; Randall, A.D.; Zhou, M.; Schwarz, T.L.; Tsien, R.W.; Horne, W.A. Functional expression of a rapidly inactivating neuronal calcium channel. Nature 1993, 363, 455–458. [Google Scholar] [CrossRef]
- Zhang, J.-F.; Randall, A.D.; Ellinor, P.T.; Horne, W.A.; Sather, W.A.; Tanabe, T.; Schwarz, T.L.; Tsien, R.W. Distinctive pharmacology and kinetics of cloned neuronal Ca2+ channels and their possible counterparts in mammalian CNS neurons. Neuropharmacology 1993, 32, 1075–1088. [Google Scholar] [CrossRef]
- Tanabe, T.; Takeshima, H.; Mikami, A.; Flockerzi, V.; Takahashi, H.; Kangawa, K.; Kojima, M.; Matsuo, H.; Hirose, T.; Numa, S. Primary structure of the receptor for calcium *channel* blockers from skeletal muscle. Nature 1987, 328, 313–318. [Google Scholar] [CrossRef]
- Newcomb, R.; Szoke, B.; Palma, A.; Wang, G.; Chen, X.H.; Hopkins, W.; Cong, R.; Miller, J.; Urge, L.; Tarczy-Hornoch, K.; Loo, J.A.; Dooley, D.J.; Nadasdi, L.; Tsien, R.W.; Lemos, J.; Miljanich, G. Selective peptide antagonist of the class E calcium channel from the venom of the tarantula Hysterocrates gigas. Biochemistry 1998, 37, 15353–15362. [Google Scholar] [CrossRef]
- Bourinet, E.; Stotz, S.C.; Spaetgens, R.L.; Dayanithi, G.; Lemos, J.; Nargeot, J.; Zamponi, G.W. Interaction of SNX482 with Domains III and IV Inhibits Activation Gating of alpha1E (CaV2.3) Calcium Channels. Biophys. J. 2001, 81, 79–88. [Google Scholar] [CrossRef]
- Tottene, A.; Volsen, S.; Pietrobon, D. alpha1E Subunits Form the Pore of Three Cerebellar R-Type Calcium Channels with Different Pharmacological and Permeation Properties. J. Neurosci. 2000, 20, 171–178. [Google Scholar]
- Wilson, S.M.; Toth, P.T.; Oh, S.B.; Gillard, S.E.; Volsen, S.; Ren, D.; Philipson, L.H.; Lee, E.C.; Fletcher, C.F.; Tessarollo, L.; Copeland, N.G.; Jenkins, N.A.; Miller, R.J. The Status of Voltage-Dependent Calcium Channels in alpha1E Knock-Out Mice. J. Neurosci. 2000, 20, 8566–8571. [Google Scholar]
- Neelands, T.R.; King, A.P.; Macdonald, R.L. Functional expression of L-, N-, P/Q-, and R-type calcium channels in the human NT2-N cell line. J. Neurophysiol. 2000, 84, 2933–2944. [Google Scholar]
- Striessnig, J.; Koschak, A. Exploring the function and pharmacotherapeutic potential of voltage-gated Ca2+ channels with gene knockout models. Channels (Austin.) 2008, 2, 233–251. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Koh, J.Y.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Suh, S.W.; Silva, D.; Frederickson, C.J.; Thompson, R.B. Importance of zinc in the central nervous system: The zinc-containing neuron. J. Nutr. 2000, 130, 1471S–1483S. [Google Scholar]
- Mathie, A.; Sutton, G.L.; Clarke, C.E.; Veale, E.L. Zinc and copper: Pharmacological probes and endogenous modulators of neuronal excitability. Pharmacol. Ther. 2006, 111, 567–583. [Google Scholar] [CrossRef]
- Stephens, G.J.; Page, K.M.; Burley, J.R.; Berrow, N.S.; Dolphin, A.C. Functional expression at rat brain cloned α1E calcium channels in COS-7 cells. Pflugers Arch. 1997, 433, 523–532. [Google Scholar] [CrossRef]
- Bergquist, F.; Nissbrandt, H. Influence of R-type (Cav2.3) and T-type (Cav3.1–3.3) antagonists on nigral somatodendritic dopamine release measured by microdialysis. Neuroscience 2003, 120, 757–764. [Google Scholar] [CrossRef]
- Gasparini, S.; Kasyanov, A.M.; Pietrobon, D.; Voronin, L.L.; Cherubini, E. Presynaptic R-type calcium channels contribute to fast excitatory synaptic transmission in the rat hippocampus. J. Neurosci. 2001, 21, 8715–8721. [Google Scholar]
- Breustedt, J.; Vogt, K.E.; Miller, R.J.; Nicoll, R.A.; Schmitz, D. Alpha1E-containing Ca2+ channels are involved in synaptic plasticity. Proc. Natl. Acad. Sci. USA 2003, 100, 12450–12455. [Google Scholar]
- Kubota, M.; Murakoshi, T.; Saegusa, H.; Kazuno, A.; Zong, S.; Hu, Q.; Noda, T.; Tanabe, T. Intact LTP and Fear Memory but Impaired Spatial Memory in Mice Lacking Cav2.3 (α1E) Channel. Biochem. Biophys. Res. Commun. 2001, 282, 242–248. [Google Scholar] [CrossRef]
- Isomura, Y.; Fujiwara-Tsukamoto, Y.; Imanishi, M.; Nambu, A.; Takada, M. Distance-dependent Ni2+-sensitivity of synaptic plasticity in apical dendrites of hippocampal CA1 pyramidal cells. J. Neurophysiol. 2002, 87, 1169–1174. [Google Scholar]
- Bloodgood, B.L.; Sabatini, B.L. Nonlinear regulation of unitary synaptic signals by CaV2.3 voltage-sensitive calcium channels located in dendritic spines. Neuron 2007, 53, 249–260. [Google Scholar] [CrossRef]
- Stackman, R.W.; Hammond, R.S.; Linardatos, E.; Gerlach, A.; Maylie, J.; Adelman, J.P.; Tzounopoulos, T. Small conductance Ca2+-activated K+ channels modulate synaptic plasticity and memory encoding. J. Neurosci. 2002, 22, 10163–10171. [Google Scholar]
- Ngo-Anh, T.J.; Bloodgood, B.L.; Lin, M.; Sabatini, B.L.; Maylie, J.; Adelman, J.P. SK channels and NMDA receptors form a Ca2+-mediated feedback loop in dendritic spines. Nat. Neurosci. 2005, 8, 642–649. [Google Scholar] [CrossRef]
- Bloodgood, B.L.; Sabatini, B.L. Regulation of synaptic signalling by postsynaptic, non-glutamate receptor ion channels. J. Physiol. 2008, 586, 1475–1480. [Google Scholar] [CrossRef]
- Metz, A.E.; Jarsky, T.; Martina, M.; Spruston, N. R-type calcium channels contribute to afterdepolarization and bursting in hippocampal CA1 pyramidal neurons. J. Neurosci. 2005, 25, 5763–5773. [Google Scholar] [CrossRef]
- Wrubel, H. Untersuchungen zur Hemmung des spannungsabhängigen Cav2.3-Calciumkanals mittels Patch-Clamp-Technik an stabil transfizierten HEK-293-Zellen. Ph.D. Thesis, Universität zu Köln, Cologne, Germany, 2009. [Google Scholar]
- Arroyo, G.; Aldea, M.; Fuentealba, J.; Albillos, A.; García, A.G. SNX482 selectively blocks P/Q Ca2+ channels and delays the inactivation of Na+ channels of chromaffin cells. Eur. J. Pharmacol. 2003, 475, 11–18. [Google Scholar] [CrossRef]
- Khosravani, H.; Zamponi, G.W. Voltage-gated calcium channels and idiopathic generalized epilepsies. Physiol Rev. 2006, 86, 941–966. [Google Scholar] [CrossRef]
- Dai, G.; Haedo, R.J.; Warren, V.A.; Ratliff, K.S.; Bugianesi, R.M.; Rush, A.; Williams, M.E.; Herrington, J.; Smith, M.M.; McManus, O.B.; Swensen, A.M. A high-throughput assay for evaluating state dependence and subtype selectivity of Cav2 calcium channel inhibitors. Assay. Drug Dev. Technol. 2008, 6, 195–212. [Google Scholar] [CrossRef]
- Zamponi, G.W.; Bourinet, E.; Snutch, T.P. Nickel Block of a Family of Neuronal Calcium Channels: Subtype- and Subunit-Dependent Action at Multiple Sites. J. Membrane Biol. 1996, 151, 77–90. [Google Scholar] [CrossRef]
- Sun, H.S.; Hui, K.; Lee, D.W.; Feng, Z.P. Zn2+ sensitivity of high- and low-voltage activated calcium channels. Biophys. J. 2007, 93, 1175–1183. [Google Scholar] [CrossRef]
- Kuzmiski, J.B.; Barr, W.; Zamponi, G.W.; MacVicar, B.A. Topiramate inhibits the initiation of plateau potentials in CA1 neurons by depressing R-type calcium channels. Epilepsia 2005, 46, 481–489. [Google Scholar] [CrossRef]
- Hainsworth, A.H.; McNaughton, N.C.; Pereverzev, A.; Schneider, T.; Randall, A.D. Actions of sipatrigine, 202W92 and lamotrigine on R-type and T-type Ca2+ channel currents. Eur. J. Pharmacol. 2003, 467, 77–80. [Google Scholar] [CrossRef]
- Nakashima, Y.M.; Todorovic, S.M.; Covey, D.F.; Lingle, C.J. The anesthetic steroid (+)-3α-hydroxy-5α-androstane-17β-carbonitrile blocks N-, Q-, and R-type, but not L- and P- type, high voltage-activated Ca2+ current in hippocampal and dorsal root ganglion neurons of the rat. Mol. Pharmacol. 1998, 54, 559–568. [Google Scholar]
- Kamatchi, G.L.; Chan, C.K.; Snutch, T.; Durieux, M.E.; Lynch, C., III. Volatile anesthetic inhibition of neuronal Ca channel currents expressed in Xenopus oocytes. Brain Res. 1999, 831, 85–96. [Google Scholar] [CrossRef]
- Kamatchi, G.L.; Tiwari, S.N.; Durieux, M.E.; Lynch, C., III. Effects of volatile anesthetics on the direct and indirect protein kinase C-mediated enhancement of alpha1E-type Ca2+ current in Xenopus oocytes. J. Pharmacol. Exp. Ther. 2000, 293, 360–369. [Google Scholar]
- Kamatchi, G.L.; Tiwari, S.N.; Durieux, M.E.; Lynch, C., III. Effects of volatile anesthetics on the direct and indirect protein kinase C-mediated enhancement of alpha1E-type Ca2+ current in Xenopus oocytes. J. Pharmacol. Exp. Ther. 2000, 293, 360–369. [Google Scholar]
- Joksovic, P.M.; Weiergraber, M.; Lee, W.; Struck, H.; Schneider, T.; Todorovic, S.M. Isoflurane-sensitive presynaptic R-type calcium channels contribute to inhibitory synaptic transmission in the rat thalamus. J. Neurosci. 2009, 29, 1434–1445. [Google Scholar] [CrossRef]
- Zamponi, G.W.; Soong, T.W.; Bourinet, E.; Snutch, T.P. β Subunit Coexpression and the α1 Subunit Domain I-II Linker Affect Piperidine Block of Neuronal Calcium Channels. J. Neurosci. 1996, 16, 2430–2443. [Google Scholar]
- Nakashima, Y.M.; Pereverzev, A.; Schneider, T.; Covey, D.F.; Lingle, C.J. Blockade of Ba2+ current through human α1E channels by two steroid analogs, (+)-ACN and (+)-ECN. Neuropharmacology 1999, 38, 843–855. [Google Scholar] [CrossRef]
- Cai, D.; Mulle, J.G.; Yue, D.T. Inhibition of recombinant Ca2+ channels by benzothiazepines and phenylalkylamines: Class-specific pharmacology and underlying molecular determinants. Mol. Pharmacol. 1997, 51, 872–881. [Google Scholar]
- Jimenez, C.; Bourinet, E.; Leuranguer, V.; Richard, S.; Snutch, T.P.; Nargeot, J. Determinants of voltage-dependent inactivation affect Mibefradil block of calcium channels. Neuropharmacology 2000, 39, 1–10. [Google Scholar] [CrossRef]
- McNaughton, N.C.; Davies, C.H.; Randall, A. Inhibition of alpha1E Ca2+ channels by carbonic anhydrase inhibitors. J. Pharmacol. Sci. 2004, 95, 240–247. [Google Scholar] [CrossRef]
- Chung, G.; Rhee, J.N.; Jung, S.J.; Kim, J.S.; Oh, S.B. Modulation of CaV2.3 calcium channel currents by eugenol. J. Dent. Res. 2008, 87, 137–141. [Google Scholar] [CrossRef]
- Deutschmann, A.; Hans, M.; Meyer, R.; Haberlein, H.; Swandulla, D. Bisphenol A inhibits voltage-activated Ca2+ channels in vitro: Mechanisms and structural requirements. Mol. Pharmacol. 2013, 83, 501–511. [Google Scholar] [CrossRef]
- Benquet, P.; Le Guen, J.; Pichon, Y.; Tiaho, F. Differential involvement of Ca2+ channels in survival and neurite outgrowth of cultured embryonic cockroach brain neurons. J. Neurophysiol. 2002, 88, 1475–1490. [Google Scholar]
- Benquet, P.; Pichon, Y.; Tiaho, F. In vitro development of P- and R-like calcium currents in insect (Periplaneta americana) embryonic brain neurons. Neurosci. Lett. 2004, 365, 228–232. [Google Scholar] [CrossRef]
- Falk, T.; Muller, Y.L.; Yool, A.J. Differential expression of three classes of voltage-gated Ca2+ channels during maturation of the rat cerebellum in vitro. Dev. Brain Res. 1999, 115, 161–170. [Google Scholar] [CrossRef]
- Iwasaki, S.; Momiyama, A.; Uchitel, O.D.; Takahashi, T. Developmental changes in calcium channel types mediating central synaptic transmission. J. Neurosci. 2000, 20, 59–65. [Google Scholar]
- Saegusa, H.; Kurihara, T.; Zong, S.Q.; Minowa, O.; Kazuno, A.A.; Han, W.H.; Matsuda, Y.; Yamanaka, H.; Osanai, M.; Noda, T.; Tanabe, T. Altered pain responses in mice lacking α1E subunit of the voltage-dependent Ca2+ channel. Proc. Natl. Acad. Sci. USA 2000, 97, 6132–6137. [Google Scholar] [CrossRef]
- Fang, Z.; Park, C.K.; Li, H.Y.; Kim, H.Y.; Park, S.H.; Jung, S.J.; Kim, J.S.; Monteil, A.; Oh, S.B.; Miller, R.J. Molecular basis of Ca(v)2.3 calcium channels in rat nociceptive neurons. J. Biol. Chem. 2007, 282, 4757–4764. [Google Scholar]
- Lee, S.C.; Choi, S.; Lee, T.; Kim, H.L.; Chin, H.; Shin, H.S. Molecular basis of R-type calcium channels in central amygdala neurons of the mouse. Proc. Natl. Acad. Sci. USA 2002, 99, 3276–3281. [Google Scholar] [CrossRef]
- Rorsman, P.; Eliasson, L.; Kanno, T.; Zhang, Q.; Gopel, S. Electrophysiology of pancreatic beta-cells in intact mouse islets of Langerhans. Prog. Biophys. Mol. Biol. 2011, 107, 224–235. [Google Scholar] [CrossRef]
- Ishiguro, M.; Wellman, T.L.; Honda, A.; Russell, S.R.; Tranmer, B.I.; Wellman, G.C. Emergence of a R-type Ca2+ channel (CaV2.3) contributes to cerebral artery constriction after subarachnoid hemorrhage. Circ. Res. 2005, 96, 419–426. [Google Scholar] [CrossRef]
- Wang, F.; Yin, Y.H.; Jia, F.; Jiang, J.Y. Antagonism of R-Type Calcium Channels Significantly Improves Cerebral Blood Flow after Subarachnoid Hemorrhage in Rats. J. Neurotrauma 2010, 27, 1723–1732. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schneider, T.; Dibué, M.; Hescheler, J. How “Pharmacoresistant” is Cav2.3, the Major Component of Voltage-Gated R-type Ca2+ Channels? Pharmaceuticals 2013, 6, 759-776. https://doi.org/10.3390/ph6060759
Schneider T, Dibué M, Hescheler J. How “Pharmacoresistant” is Cav2.3, the Major Component of Voltage-Gated R-type Ca2+ Channels? Pharmaceuticals. 2013; 6(6):759-776. https://doi.org/10.3390/ph6060759
Chicago/Turabian StyleSchneider, Toni, Maxine Dibué, and Jürgen Hescheler. 2013. "How “Pharmacoresistant” is Cav2.3, the Major Component of Voltage-Gated R-type Ca2+ Channels?" Pharmaceuticals 6, no. 6: 759-776. https://doi.org/10.3390/ph6060759




