Assessment of Chemical and Physico-Chemical Properties of Cyanobacterial Lipids for Biodiesel Production
Abstract
:1. Introduction
2. Results and Discussion
2.1. Biomass and Lipid Productivity
| Cyanobacterium | Micrograph | Strain source | Biomass productivity | Lipid productivity |
|---|---|---|---|---|
| Scale bar: 20 µm | (mg·L−1·day−1) | (mg·L−1·day−1) | ||
| Microcystis aeruginosa NPCD-1 |  | Sewage treatment
plant of Cidade de Deus, RJ, Brazil | 46.9 | 13.1 |
| Synechococcus sp. PCC7942 |  | Pasteur Culture
Collection, France | 52.7 | 14.2 |
| Chlorogloea sp. CENA170 |  | Mangrove soil from
Cardoso Island, SP, Brazil | 6.8 | 0.9 |
| Trichormus sp. CENA77 |  | Flooded rice field,
SC, Brazil | 30.8 | 7.3 |
| Leptolyngbya sp. CENA104 |  | Sewage treatment
plant of Cajati, SP, Brazil | 3.7 | 0.8 |
2.2. Fatty Acids Profile
| FA | Microcystis aeruginosa NPCD-1 | Synechococcus sp. PCC7942 | Chlorogloea sp. CENA170 | Trichormus sp. CENA77 | Leptolyngbya sp. CENA104 | |
|---|---|---|---|---|---|---|
| Saturated FA (%wt) | ||||||
| C6:0 | Caproic | 0.1 | 0.06 | 0.05 | 0.08 | 0.09 |
| C8:0 | Caprilic | 0.6 | 0.06 | 0.4 | 0.6 | 0.2 |
| C10:0 | Capric | 0.9 | 0.06 | 0.6 | 0.6 | 0.2 |
| C12:0 | Lauric | 13.2 | 0.7 | 8.7 | 9.7 | 3.9 |
| C14:0 | Myristic | 5.2 | 0.7 | 3.4 | 3.9 | 1.7 |
| C15:0 | Pentadecanoic | 0.1 | 0.06 | 0.06 | 0.06 | 0.08 |
| C16:0 | Palmitic | 24.3 | 23.5 | 15.4 | 24.9 | 14.6 |
| C17:0 | Margaric | 0.2 | 0.1 | 0.1 | 0.2 | 0.15 |
| C18:0 | Stearic | 4.9 | 3.6 | 3.8 | 3.4 | 2.8 |
| C20:0 | Arachidic | 0.3 | 0.3 | 0.2 | 0.37 | 0.4 |
| C22:0 | Behenic | 0.2 | 0.3 | 0.2 | 0.15 | 0.3 |
| C24:0 | Lignoceric | 0.1 | 0.2 | 0.1 | 0.10 | 0.15 |
| Total | 50.1 | 29.6 | 33.0 | 44.1 | 24.6 | |
| Monounsaturated FA | ||||||
| C16:1 | Palmitoleic | 1.1 | 3.3 | 5.4 | 1.5 | 3.9 |
| C17:1 | cis-10-Heptadecenoic | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 |
| C18:1 | Oleic | 26.8 | 31.5 | 28.8 | 36.9 | 38.8 |
| C20:1 | Gadoleic | 0.3 | 0.2 | 0.1 | 0.3 | 0.42 |
| C22:1 | Erucic | 2.5 | nd | nd | nd | nd |
| C24:1 | Nervonic | 0.8 | nd | nd | nd | nd |
| Total | 31.6 | 35.1 | 34.4 | 38.8 | 43.3 | |
| Polyunsaturated FA | ||||||
| C18:2 | Linoleic | 12.5 | 30.9 | 29.4 | 10.7 | 26.4 |
| C18:3 | .-Linolenic | 0.8 | 0.5 | 0.27 | nd | 0.4 |
| C18:3 | Linolenic | 1.6 | 2.9 | 1.63 | 5.1 | 4.3 |
| Total | 14.9 | 34.3 | 31.3 | 15.8 | 31.1 | |
| N.I. | Unidentified | 3.4 | 1.0 | 1.3 | 1.3 | 1.0 |
2.3. Cyanobacterial Fatty Acid Composition: Implications for Biodiesel Quality
| Property | M. aeruginosa NPCD-1 | Synechococcus sp. PCC7942 | Chlorogloea sp. CENA170 | Trichormus sp. CENA77 | Leptolyngbya sp. CENA104 |
|---|---|---|---|---|---|
| Iodine value (g I2/100 g) | 57 | 97 | 90 | 68 | 100 |
| Saponification value | 210 | 203 | 210 | 213 | 205 |
| Degree of unsaturation (%) | 60.7 | 103.7 | 97 | 70.4 | 105.5 |
| Long chain saturated factor | 5.7 | 5.2 | 4.1 | 5.0 | 4.0 |
2.4. Characterization of Lipid Materials
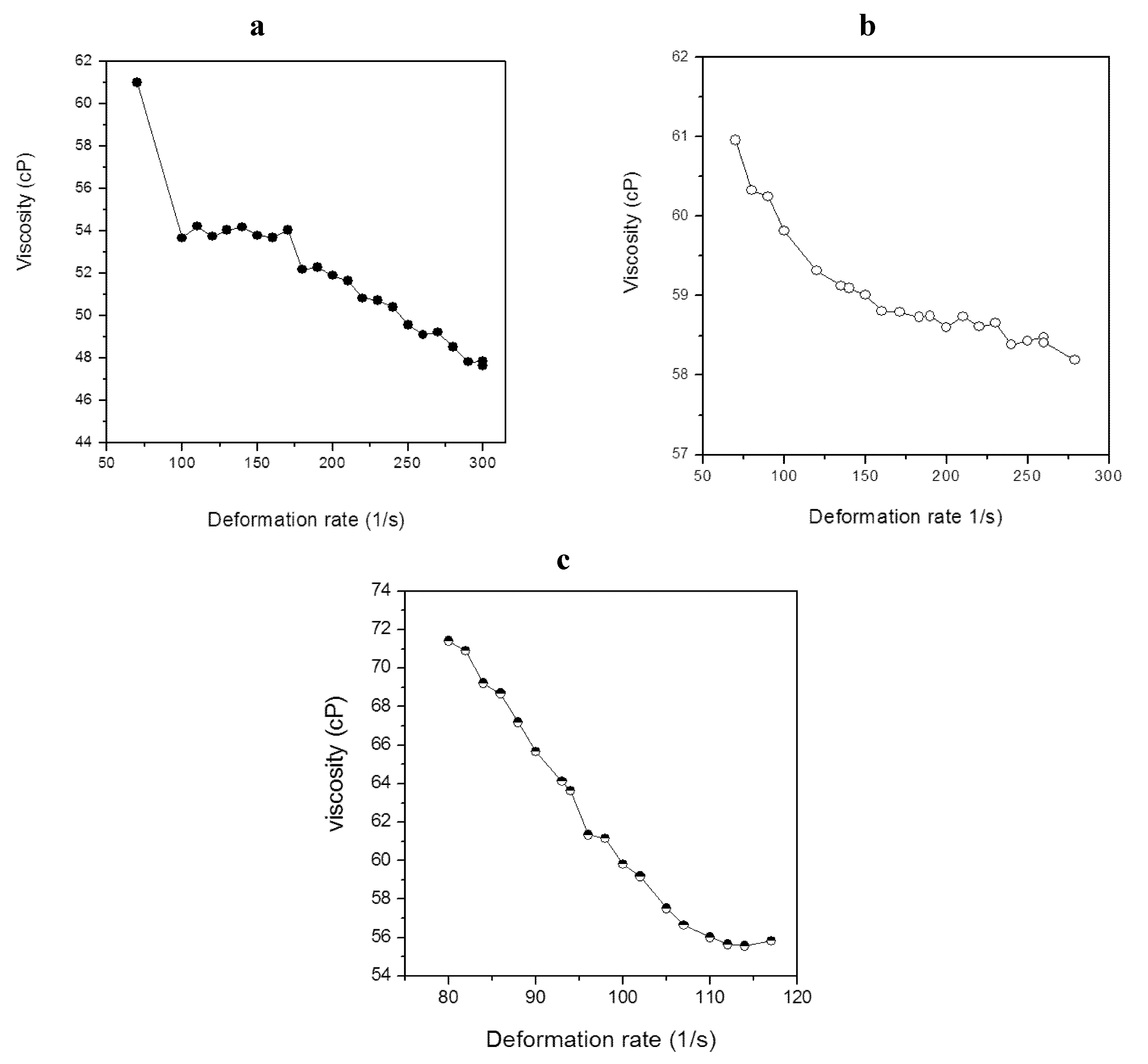
| Strain | Viscosity (cP) | η * |
|---|---|---|
| M. aeruginosa NPCD-1 | 52.7 | 0.84 |
| Trichormus sp. CENA77 | 59.1 | 0.97 |
| Synechococcus sp. PCC7942 | 62.3 | 0.37 |

| Band | Assignment | Lipid from cyanobacteria (this study) | Andiroba oil (cm−1) * |
|---|---|---|---|
| 1 | (=C–H) stretching | 3006 | 3005 |
| 2 | CH2 asymmetry stretching | 2928 | 2923 |
| 3 | CH2 symmetry stretching | 2852 | 2854 |
| 4 | C=O stretching | 1744 | 1741 |
| 5 | CH2 scissors | 1464 | 1463 |
| 6 | C=C–C–O | 1150 | 1040–1290 |
| 7 | CH2 rocking | 724 | 722 |
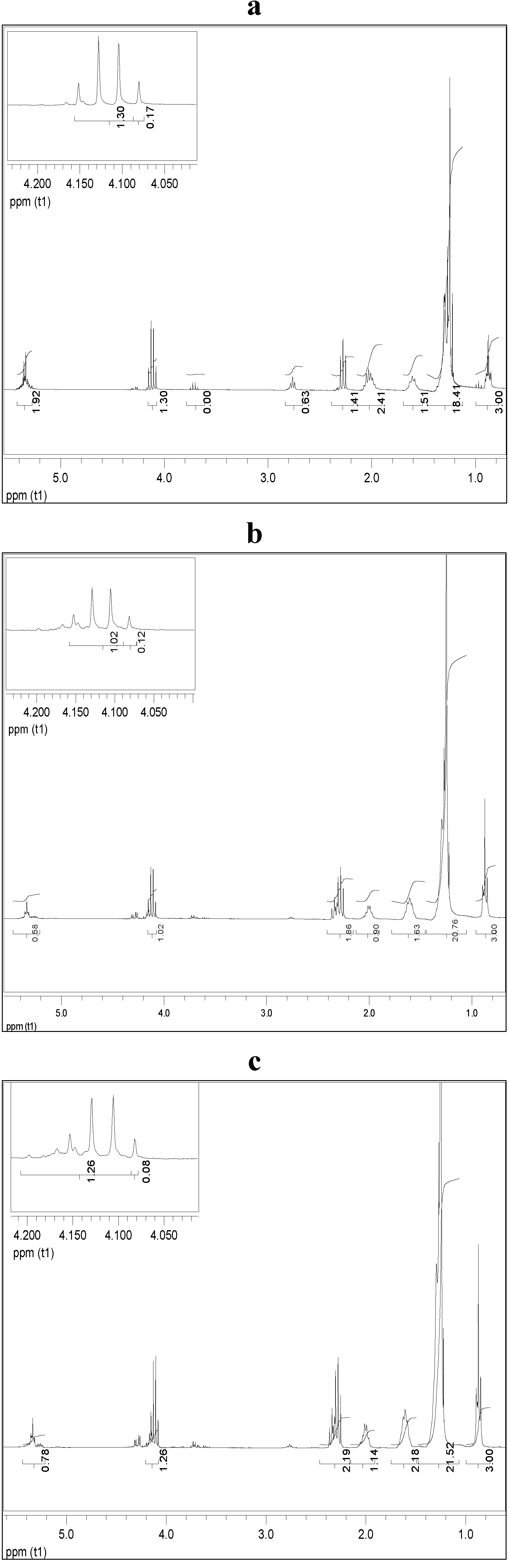
2.5. Ethanolysis of Lipid Feedstocks
3. Experimental Section
3.1. Cyanobacterial Strains and Cultivation
3.2. Total Lipids Extraction and FA Analysis
3.3. Empirical Parameters
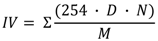
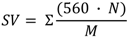
3.4. Viscosity Determination
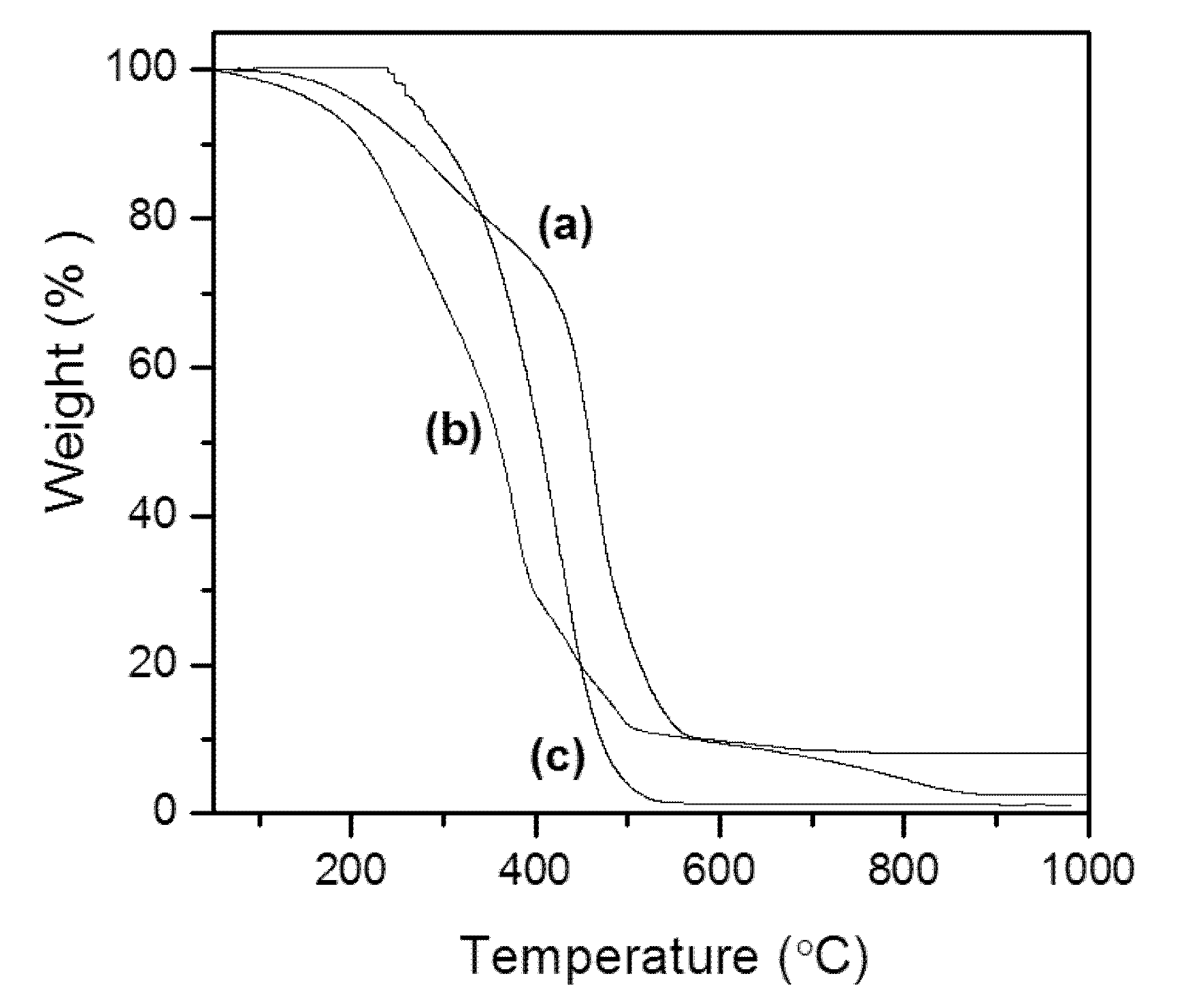
3.5. Thermal Study (TG)
3.6. Infrared Spectroscopy (IR)
3.7. Biodiesel Synthesis
3.8. Downstream Procedure
3.9. Proton Nuclear Magnetic Resonance Spectrometry (1H NMR)
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Jansson, C.; Northen, T. Calcifying cyanobacteria—The potential of biomineralization for carbon capture and storage. Curr. Opin. Biotechnol. 2010, 21, 365–371. [Google Scholar]
- Burja, A.M.; Banaigs, B.; Abou-Mansour, E.; Burgess, J.G.; Wright, P.C. Marine cyanobacteria: A prolific source of natural products. Tetrahedron Lett. 2001, 57, 9347–9377. [Google Scholar] [CrossRef]
- Silva-Stenico, M.E.; Silva, C.S.P.; Lorenzi, A.S.; Shishido, T.K.; Etchegaray, A.; Lira, S.P.; Moraes, L.A.B.; Fiore, M.F. Non-ribosomal peptides produced by Brazilian cyanobacterial isolates with antimicrobial activity. Microbiol. Res. 2011, 166, 161–175. [Google Scholar] [CrossRef]
- Karatay, S.E.; Donmez, G. Microbial oil production from thermophile cyanobacteria for biodiesel production. Appl. Energ. 2011, 88, 3632–3635. [Google Scholar] [CrossRef]
- Costa, J.A.V.; Morais, M.G. The role of biochemical engineering in the production of biofuels from microalgae. Bioresour. Technol. 2011, 102, 2–9. [Google Scholar] [CrossRef]
- Taher, H.; Al-Zuhair, S.; Al-Marzouqi, A.H.; Haik, Y.; Farid, M.M. A review of enzymatic transesterification of microalgal oil-based biodiesel using supercritical technology. Enzyme Res. 2011, 2011, 468292. [Google Scholar] [CrossRef]
- Rittmann, B.E. Opportunities for renewable bioenergy using microorganisms. Biotechnol. Bioeng. 2008, 2, 203–212. [Google Scholar]
- Balasubramanian, L.; Subramanian, G.; Nazeer, T.T.; Simpson, H.S.; Rahuman, S.T.; Raju, P. Cyanobacteria cultivation in industrial wastewaters and biodiesel production from their biomass: A review. Biotechnol. Appl. Biochem. 2012, 59, 220–225. [Google Scholar]
- Vargas, M.A.; Rodriguez, H.; Moreno, J.; Olivares, H.; Delcampo, J.A.; Rivas, J.; Guerrero, M.G. Biochemical composition and fatty acid content of filamentous nitrogen-fixing cyanobacteria. J. Phycol. 1998, 34, 812–817. [Google Scholar] [CrossRef]
- Murata, N.; Wada, H.; Gombos, Z. Modes of fatty acid desaturation in cyanobacteria. Plant Cell Physiol. 1993, 33, 933–941. [Google Scholar]
- Hoffmann, L.; Komárek, J.; Kastovsky, J. System of cyanoprokaryotes (cyanobacteria)—State in 2004. Algol. Stud. 2005, 117, 95–115. [Google Scholar] [CrossRef]
- Da Rós, P.C.M.; Silva, C.S.P.; Silva-Stenico, M.E.; Fiore, M.F.; de Castro, H.F. Microcystis aeruginosa lipids as feedstock for biodiesel synthesis by enzymatic route. J. Mol. Catal. B Enzym. 2012, 84, 177–182. [Google Scholar] [CrossRef]
- Bruno, L.; di Pippo, F.; Antonaroli, S.; Gismondi, A.; Valentini, C.; Albertano, P. Characterization of biofilm-forming cyanobacteria for biomass and lipid production. J. Appl. Microb. 2012, 113, 1052–1064. [Google Scholar] [CrossRef] [Green Version]
- Sharathchandra, K.; Rajashekhar, M. Total lipid and fatty acid composition in some freshwater cyanobacteria. J. Algal Biomass Util. 2011, 2, 83–97. [Google Scholar]
- Francisco, E.C.; Neves, D.B.; Jacob-Lopes, E.; Franco, T.T. Microalgae as feedstock for biodiesel production: Carbon dioxide sequestration, lipid production and biofuel quality. J. Chem. Technol. Biotechnol. 2010, 85, 395–403. [Google Scholar] [CrossRef]
- Rodolfi, L.; Zittelli, G.C.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar]
- Gouveia, L.; Oliveira, A.C. Microalgae as a raw material for biofuels production. J. Ind. Microbiol. Biotechnol. 2009, 36, 269–274. [Google Scholar] [CrossRef]
- Mandal, S.; Mallick, N. Microalga Scenedesmus obliquus as a potential source for biodiesel production. Appl. Microbiol. Biotechnol. 2009, 84, 281–291. [Google Scholar] [CrossRef]
- Griffiths, M.J.; Harrison, S.T.L. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J. Appl. Phycol. 2009, 21, 493–507. [Google Scholar] [CrossRef]
- Gunstone, F.D.; Harwood, J.L.; Dijkstra, A.J. The Lipid Handbook; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Moreira, A.B.R.; Perez, V.H.; Zanin, G.M.; de Castro, H.F. Biodiesel synthesis by enzymatic transesterification of palm oil with ethanol using lipases from several sources immobilized on silica-PVA composite. Energy Fuels 2007, 21, 3689–3694. [Google Scholar] [CrossRef]
- Da Rós, P.C.M.; Silva, G.A.M.; Mendes, A.A.; Santos, J.C.; de Castro, H.F. Evaluation of the catalytic properties of Burkholderia cepacia lipase immobilized on non-commercial matrices to be used in biodiesel synthesis from different feedstocks. Bioresour. Technol. 2010, 101, 5508–5516. [Google Scholar] [CrossRef]
- Knothe, G. Dependence of biodiesel fuel properties on the structures of fatty acid alkyl esters. Fuel Process. Technol. 2005, 86, 1059–1070. [Google Scholar] [CrossRef]
- Ramos, M.J.; Fernández, C.M.; Casas, A.; Rodríguez, L.; Pérez, Á. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour. Technol. 2009, 100, 261–268. [Google Scholar] [CrossRef]
- UNE-EN 14214, European Standard. In Automotive Fuels. Fatty Acid Methyl Esters (FAME) for Diesel Engines. Requirements and Test Methods; CEN-European Committee for Standardization: Brussels, Belgium, 2003.
- ASTM D6751-02, Standard Specification for Biodiesel Fuel (B100) Blend Stock for Distillate Fuels; ASTM International: West Conshohocken, PA, USA, 2002.
- RESOLUÇÃO ANP N° 14, DE 11.5.2012-DOU 18.5.2012. Available online: http://nxt.anp.gov.br/nxt/gateway.dll/leg/resolucoes_anp/2012/maio/ranp%2014%20-%202012.xml (accessed on 20 June 2013).
- De Nevers, N. Fluid Mechanics for Chemical Engineers, 3rd ed; McGraw-Hill: Boston, MA, USA, 2005. [Google Scholar]
- Silva, G.A.M.; da Rós, P.C.M.; Souza, L.T.A.; Costa, A.P.O.; de Castro, H.F. Physico-chemical, spectroscopical and thermal characterization of biodiesel obtained by enzymatic route as a tool to select the most efficient immobilized lipase. Braz. J. Chem. Eng. 2012, 29, 39–47. [Google Scholar] [CrossRef] [Green Version]
- Miranda, T.; Esteban, A.; Rojas, S.; Montero, I.; Ruiz, A. Combustion analysis of different olive residues. Int. J. Mol. Sci. 2008, 9, 512–525. [Google Scholar] [CrossRef]
- Marcilla, A.; Gómez-Siurana, A.; Gomis, C.; Chápuli, E.; Catalá, M.C.; Valdés, F.J. Characterization of microalgal species through TGA/FTIR analysis: Application to Nannochloropsis sp. Thermochim. Acta 2009, 484, 41–47. [Google Scholar] [CrossRef]
- Carvalho, A.K.F. Synthesis of Biodiesel by Transesterification by Ethylic Route: A Comparison of Heterogeneous Catalysts. M.Sc Dissertation, Engineering School of Lorena-USP, Lorena, Brazil, 2011. [Google Scholar]
- Dean, A.P.; Sigee, D.C.; Estrada, B.; Pittman, J.K. Using FTIR spectroscopy for rapid determination of lipid accumulation response to nitrogen limitation in freshwater microalgae. Bioresour. Technol. 2010, 101, 4499–4507. [Google Scholar] [CrossRef]
- Albuquerque, M.L.S.; Guedes, I.; Alcantara, J., Jr.; Moreira, S.G.C. Infrared absorption spectra of Buriti (Mauritia flexuosa L.) oil. Vibrat. Spectrosc. 2003, 33, 127–131. [Google Scholar]
- Kaplan, M.; Davidson, G.; Poliakoff, M. Capillary supercritical fluid chromatography-Fourier transform infrared spectroscopy study of triglycerides and the qualitative analysis of normal and “unsaturated” cheeses. J. Chromatogr. 1994, 673, 231–237. [Google Scholar]
- Pavia, D.L.; Lampman, G.M.; Kriz, G.S. Introduction to Spectroscopy, 2nd ed; Saunders College Publishing: Orlando, FL, USA, 1996. [Google Scholar]
- Porphy, S.J.; Farid, M.M. Feasibility study for production of biofuel and chemicals from marine microalgae Nannochloropsis sp. based on basic mass and energy analysis. ISRN Renew. Energy 2012, 2012, 156824. [Google Scholar] [CrossRef]
- Allen, M.B. Simple conditions for growth of unicellular blue-green algae on plates. J. Phycol. 1968, 4, 1–4. [Google Scholar]
- Gorham, P.R.; Mclahlan, J.R.; Hammer, V.T.; Kim, W.K. Isolation and culture of toxic strains of Anabaena flos-aquae (Lyngb.) de Bréb. Verh. Int. Ver. Theor. Angew Limnol 1964, 15, 796–804. [Google Scholar]
- Castenholz, R.W. Culturing methods for cyanobacteria. Methods Enzymol. 1988, 167, 68–95. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method for total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar]
- American Oil Chemists’ Society. In Official Methods and Recommended Practices of the AOCS, 5th ed; AOCS Press: Boulder, CO, USA, 2004.
- Paiva, E.J.M.; Silva, M.L.C.P.; Barboza, J.C.S.; Oliveira, P.C.; Castro, H.F.; Giordani, D.S. Non-edible babassu oil as a new source for energy production—A feasibility transesterification survey assisted by ultrasound. Ultrason. Sonochem. 2013, 20, 833–838. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rós, P.C.M.D.; Silva, C.S.P.; Silva-Stenico, M.E.; Fiore, M.F.; Castro, H.F.D. Assessment of Chemical and Physico-Chemical Properties of Cyanobacterial Lipids for Biodiesel Production. Mar. Drugs 2013, 11, 2365-2381. https://doi.org/10.3390/md11072365
Rós PCMD, Silva CSP, Silva-Stenico ME, Fiore MF, Castro HFD. Assessment of Chemical and Physico-Chemical Properties of Cyanobacterial Lipids for Biodiesel Production. Marine Drugs. 2013; 11(7):2365-2381. https://doi.org/10.3390/md11072365
Chicago/Turabian StyleRós, Patrícia C. M. Da, Caroline S. P. Silva, Maria E. Silva-Stenico, Marli F. Fiore, and Heizir F. De Castro. 2013. "Assessment of Chemical and Physico-Chemical Properties of Cyanobacterial Lipids for Biodiesel Production" Marine Drugs 11, no. 7: 2365-2381. https://doi.org/10.3390/md11072365