Bioactive Potential of Marine Macroalgae from the Central Red Sea (Saudi Arabia) Assessed by High-Throughput Imaging-Based Phenotypic Profiling
Abstract
:1. Introduction
2. Results
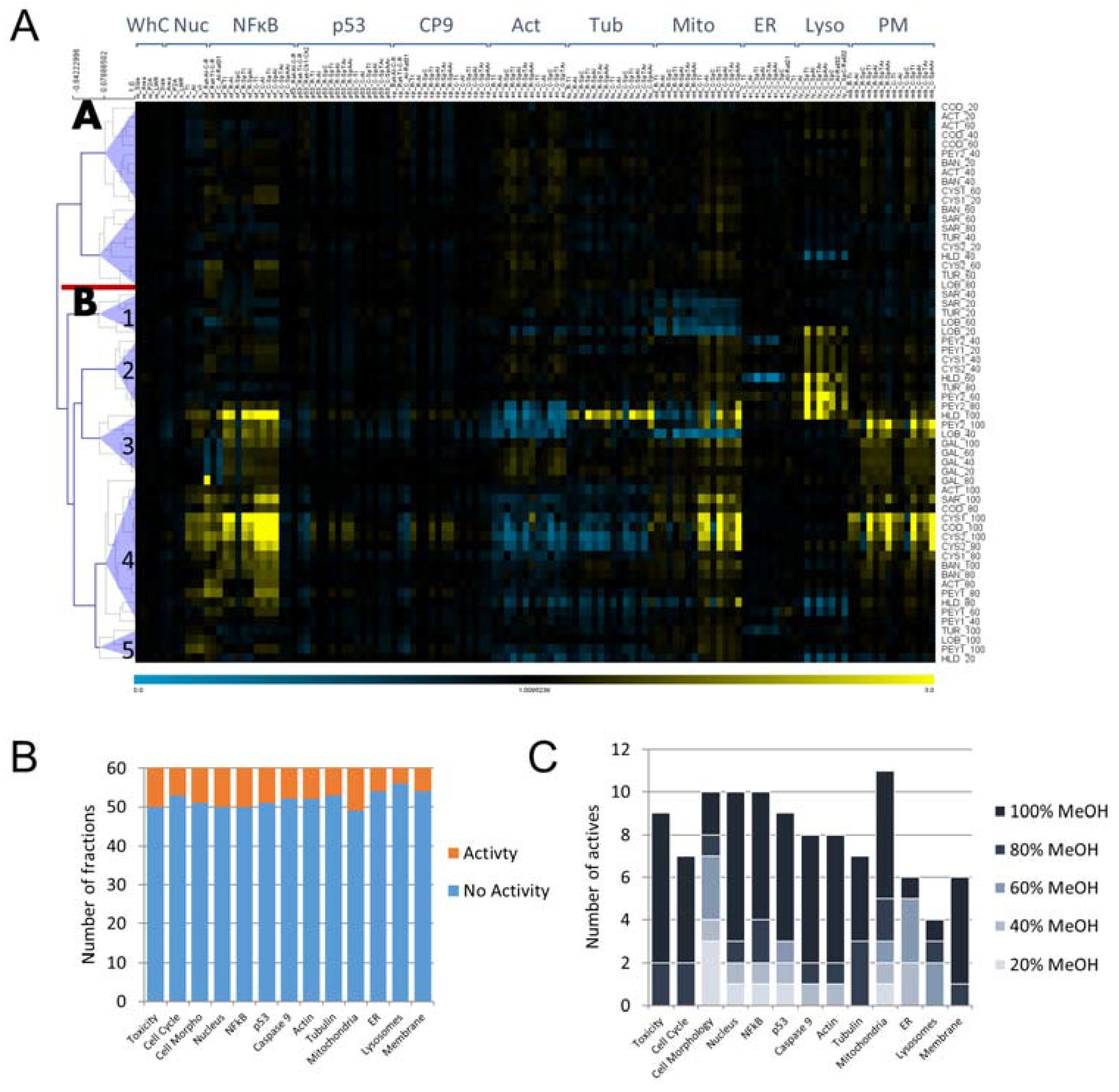
2.1. Cytological Profiling and Clustering of a Pre-Fractionated Algal Extracts Library
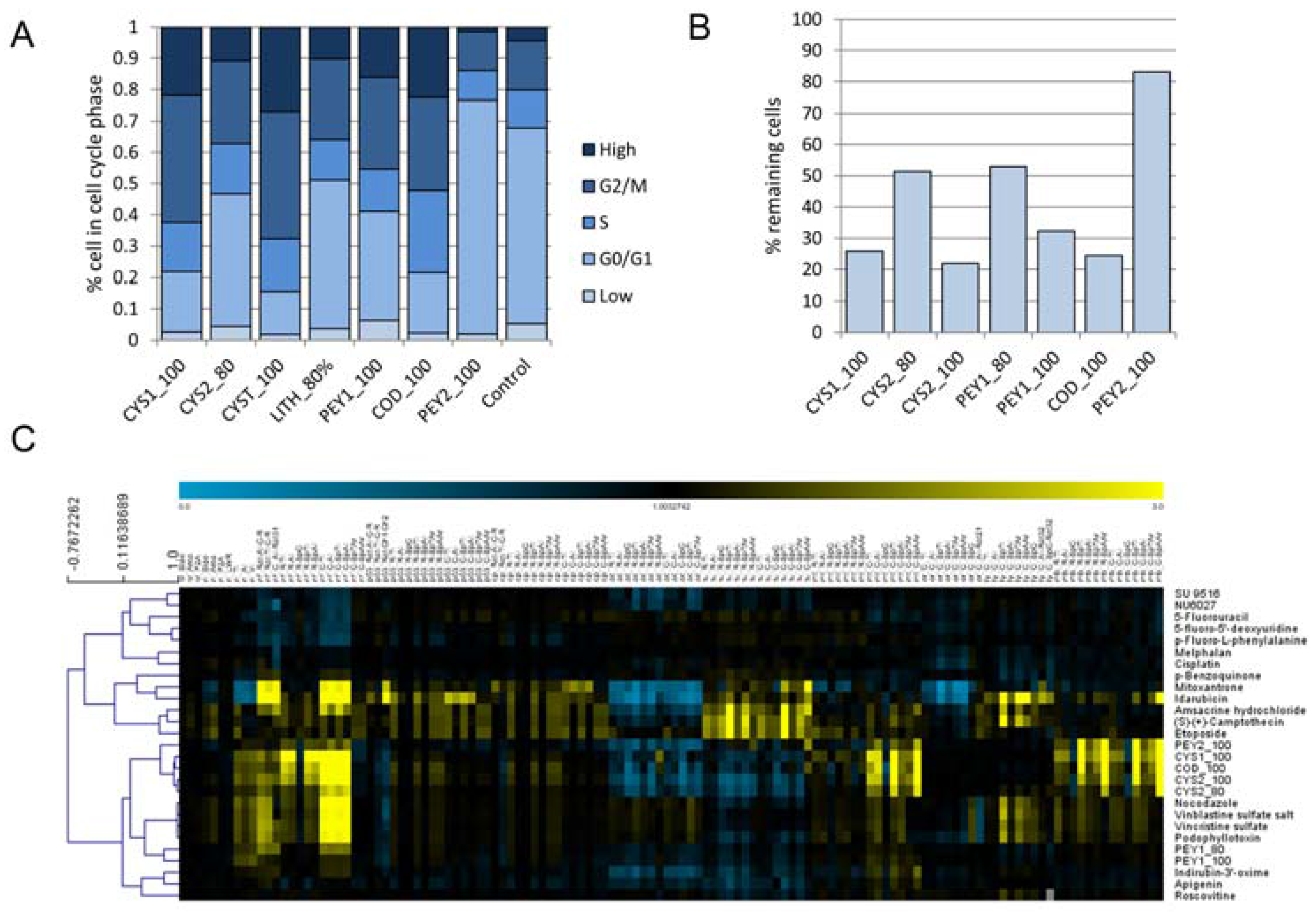
2.2. Cell Cycle Analysis
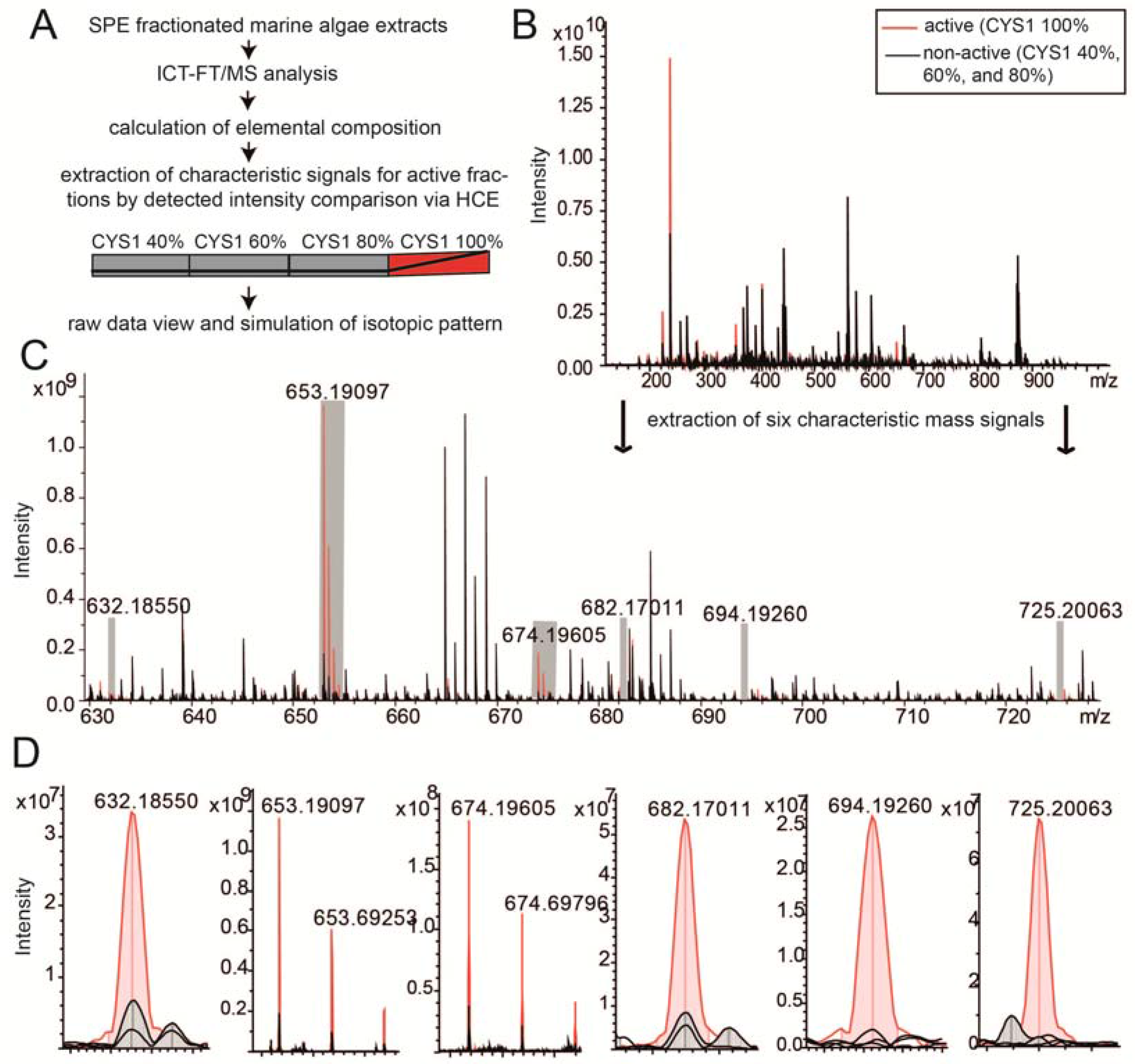
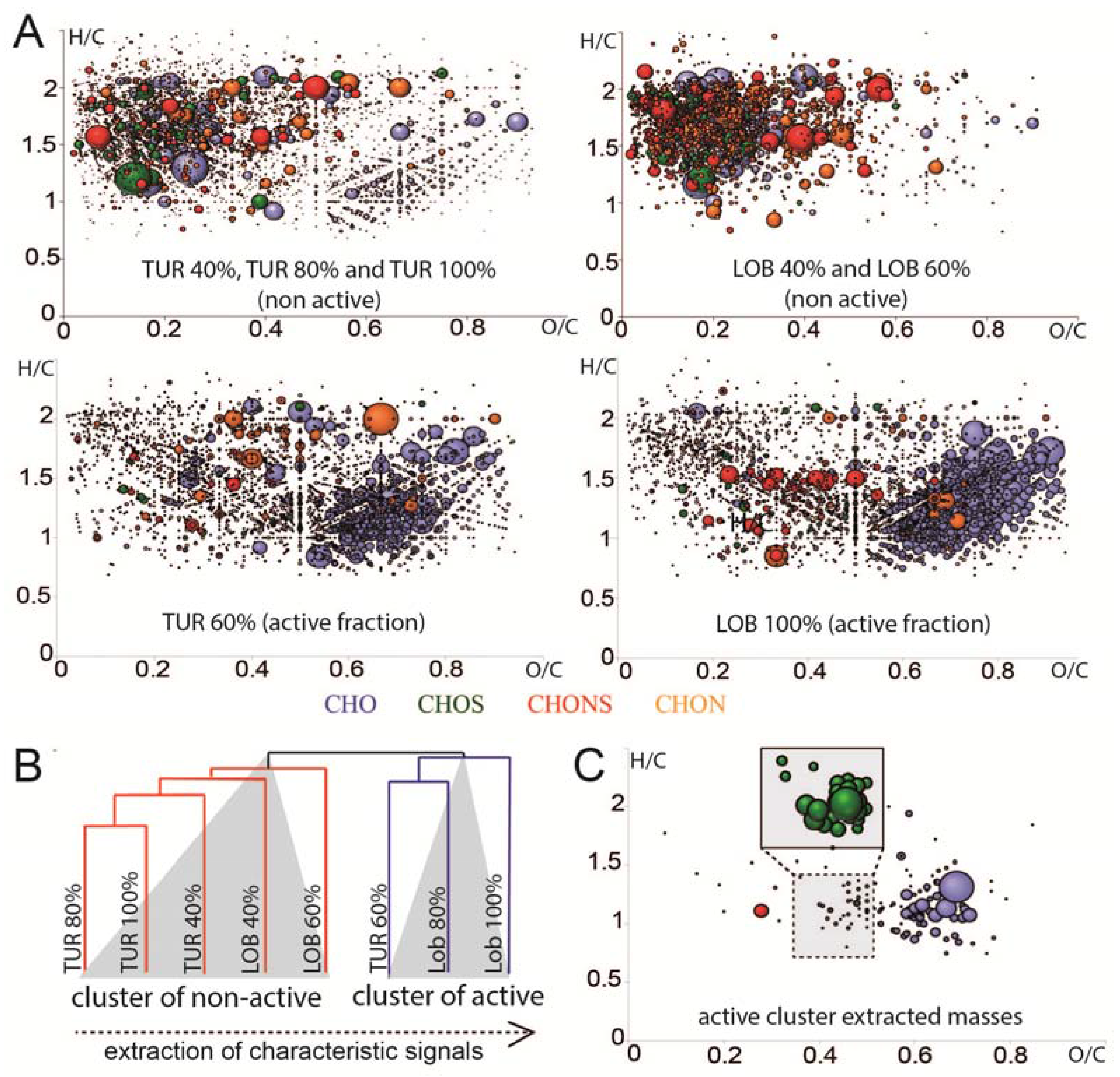
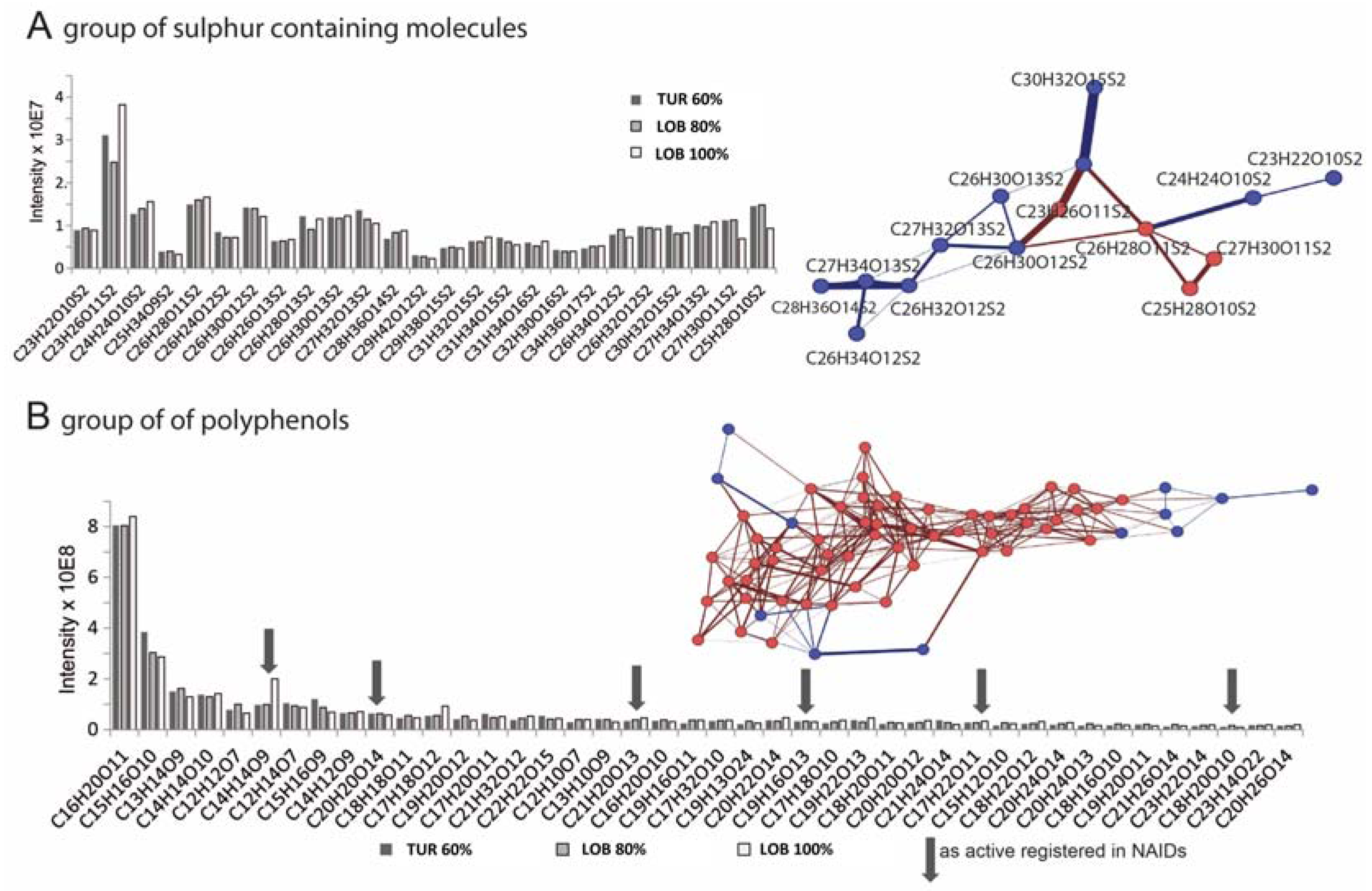
2.3. Chemical Analysis of Cell Cycle Interfering Fractions
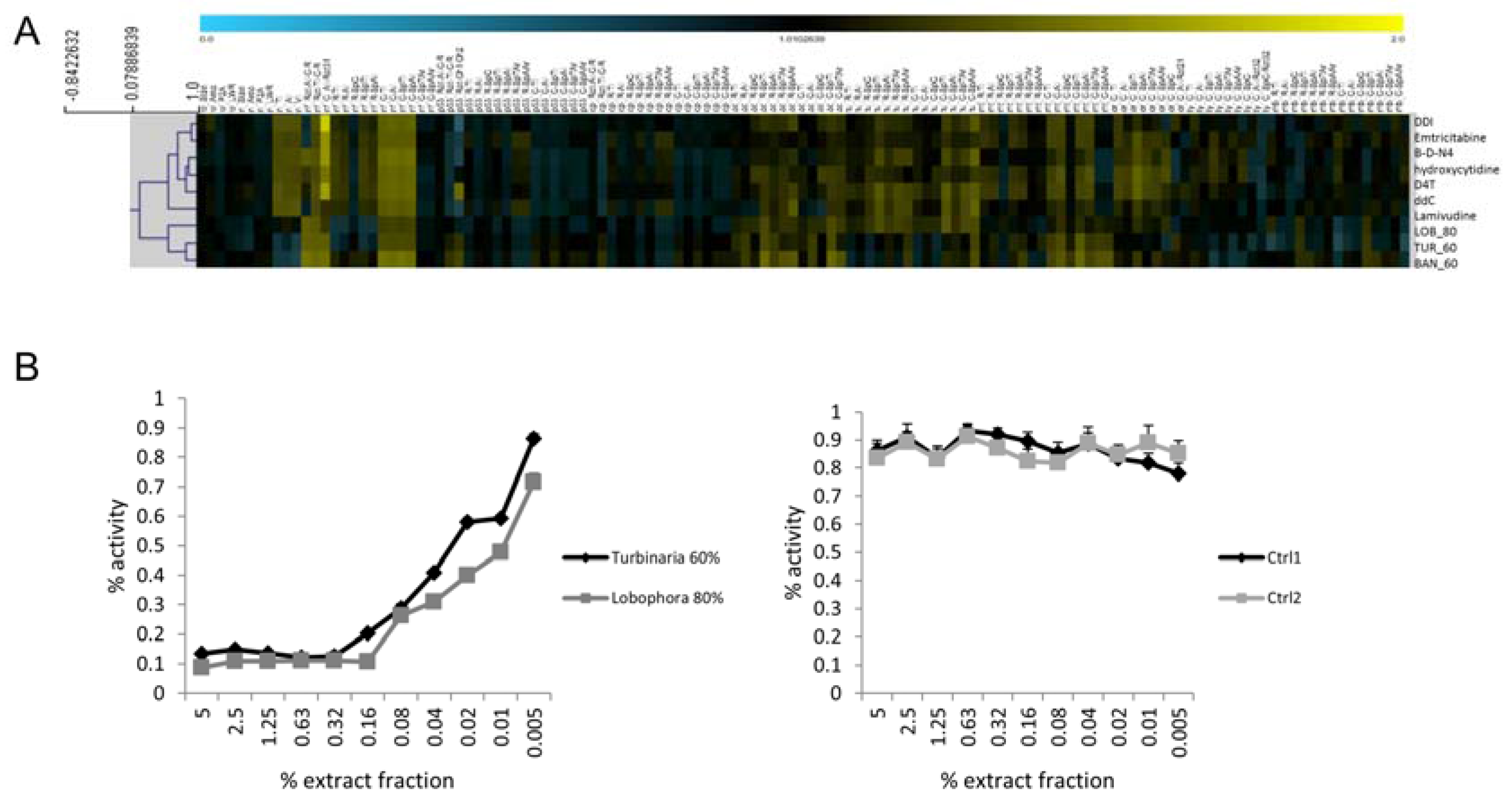
2.4. HIV-1 Reverse Transcriptase Activity
3. Discussion
3.1. Using HCS as a Broad-Spectrum Technology for Bioprospecting
3.2. Bioprospecting of Marine Algae from the Red Sea Reveals Common Active Fractions and Correlative Trends Across Cellular Markers
3.3. HCS Predicts MoAs by Comparative Analysis to Reference Compounds
3.4. Successful In Silico HIV-1 RT Target Predictions via Profile Comparison to Known Compounds
3.5. Deep Chemical Profiling of Active Fractions
4. Materials and Methods
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gerwick, W.H.; Moore, B.S. Lessons from the past and charting the future of marine natural products drug discovery and chemical biology. Chem. Biol. 2012, 19, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L.; Edrada-Ebel, R.; Quinn, R.J. The re-emergence of natural products for drug discovery in the genomics era. Nat. Rev. Drug Discov. 2015, 14, 111–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kijjoa, A.; Sawangwong, P. Drugs and cosmetics from the sea. Mar. Drugs 2004, 2, 73–82. [Google Scholar] [CrossRef]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2014, 31, 160–258. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.C.; Puga, J.; Serodio, J.; Gomes, N.C.; Calado, R. Trends in the discovery of new marine natural products from invertebrates over the last two decades—Where and what are we bioprospecting? PLoS ONE 2012, 7, e30580. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Karadeniz, F. Anti-hiv activity of extracts and compounds from marine algae. Adv. Food Nutr. Res. 2011, 64, 255–265. [Google Scholar] [PubMed]
- Kremb, S.; Helfer, M.; Kraus, B.; Wolff, H.; Wild, C.; Schneider, M.; Voolstra, C.R.; Brack-Werner, R. Aqueous extracts of the marine brown alga Lobophora variegata inhibit HIV-1 infection at the level of virus entry into cells. PLoS ONE 2014, 9, e103895. [Google Scholar] [CrossRef] [PubMed]
- Witvrouw, M.; De Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen. Pharmacol. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Freile-Pelegrin, Y.; Robledo, D.; Chan-Bacab, M.J.; Ortega-Morales, B.O. Antileishmanial properties of tropical marine algae extracts. Fitoterapia 2008, 79, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Thomas, N.V.; Li, X. Anticancer compounds from marine macroalgae and their application as medicinal foods. Adv. Food Nutr. Res 2011, 64, 213–224. [Google Scholar] [PubMed]
- Medeiros, V.P.; Queiroz, K.C.; Cardoso, M.L.; Monteiro, G.R.; Oliveira, F.W.; Chavante, S.F.; Guimaraes, L.A.; Rocha, H.A.; Leite, E.L. Sulfated galactofucan from Lobophora variegata: Anticoagulant and anti-inflammatory properties. Biochemistry 2008, 73, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Joint United Nations Programme on HIV/AIDS. Global Report: UNAIDS Report on the Global Aids Epidemics 2013; Joint United Nations Programme on HIV/AIDS (UNAIDS): Geneva, Switzerland, 2013. [Google Scholar]
- Huskens, D.; Schols, D. Algal lectins as potential HIV microbicide candidates. Mar. Drugs 2012, 10, 1476–1497. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; O’Keefe, B.R.; Sowder, R.C., 2nd; Bringans, S.; Gardella, R.; Berg, S.; Cochran, P.; Turpin, J.A.; Buckheit, R.W., Jr.; McMahon, J.B.; et al. Isolation and characterization of griffithsin, a novel hiv-inactivating protein, from the red alga Griffithsia sp. J. Biol. Chem. 2005, 280, 9345–9353. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.J.; Yoon, K.D.; Min, S.Y.; Lee, J.S.; Kim, J.H.; Kim, T.G.; Kim, S.H.; Kim, N.G.; Huh, H.; Kim, J. Inhibition of HIV-1 reverse transcriptase and protease by phlorotannins from the brown alga Ecklonia cava. Biol. Pharm. Bull. 2004, 27, 544–547. [Google Scholar] [CrossRef] [PubMed]
- Artan, M.; Li, Y.; Karadeniz, F.; Lee, S.H.; Kim, M.M.; Kim, S.K. Anti-hiv-1 activity of phloroglucinol derivative, 6,6′-bieckol, from ecklonia cava. Bioorg. Med. Chem. 2008, 16, 7921–7926. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, K.C.; Medeiros, V.P.; Queiroz, L.S.; Abreu, L.R.; Rocha, H.A.; Ferreira, C.V.; Juca, M.B.; Aoyama, H.; Leite, E.L. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed. Pharmacother. 2008, 62, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Hebert, J.R.; Fitton, J.H.; Zimba, P.V. Algae—A poor man’s haart? Med. Hypotheses 2004, 62, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Irhimeh, M.R. Dietary algae and HIV/AIDS: Proof of concept clinical data. J. Appl. Phycol. 2012, 24, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Roder, C.; Berumen, M.L.; Bouwmeester, J.; Papathanassiou, E.; Al-Suwailem, A.; Voolstra, C.R. First biological measurements of deep-sea corals from the Red Sea. Sci. Rep. 2013, 3, 2802. [Google Scholar] [CrossRef] [PubMed]
- Sawall, Y.; Al-Sofyani, A.; Banguera-Hinestroza, E.; Voolstra, C.R. Spatio-temporal analyses of symbiodinium physiology of the coral pocillopora verrucosa along large-scale nutrient and temperature gradients in the red sea. PLoS ONE 2014, 9, e103179. [Google Scholar]
- Wahl, M.; Al Sofyani, A.; Saha, M.; Kruse, I.; Lenz, M.; Sawall, Y. Large scale patterns of antimicrofouling defenses in the hard coral Pocillopora verrucosa in an environmental gradient along the saudi arabian coast of the red sea. PLoS ONE 2014, 9, e106573. [Google Scholar] [CrossRef] [PubMed]
- Mhadhebi, L.; Laroche-Clary, A.; Robert, J.; Bouraoui, A. Antioxidant, anti-inflammatory, and antiproliferative activities of organic fractions from the mediterranean brown seaweed Cystoseira sedoides. Can. J. Physiol. Pharmacol. 2011, 89, 911–921. [Google Scholar] [CrossRef] [PubMed]
- Spavieri, J.; Allmendinger, A.; Kaiser, M.; Casey, R.; Hingley-Wilson, S.; Lalvani, A.; Guiry, M.D.; Blunden, G.; Tasdemir, D. Antimycobacterial, antiprotozoal and cytotoxic potential of twenty-one brown algae (phaeophyceae) from british and irish waters. Phytother. Res. 2010, 24, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Suzgec-Selcuk, S.; Mericli, A.H.; Guven, K.C.; Kaiser, M.; Casey, R.; Hingley-Wilson, S.; Lalvani, A.; Tasdemir, D. Evaluation of turkish seaweeds for antiprotozoal, antimycobacterial and cytotoxic activities. Phytother. Res. 2011, 25, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Mitchison, T.J.; Bender, A.; Young, D.W.; Tallarico, J.A. Multi-parameter phenotypic profiling: Using cellular effects to characterize small-molecule compounds. Nat. Rev. Drug Discov. 2009, 8, 567–578. [Google Scholar] [PubMed]
- Bray, M.A.; Singh, S.; Han, H.; Davis, C.T.; Borgeson, B.; Hartland, C.; Kost-Alimova, M.; Gustafsdottir, S.M.; Gibson, C.C.; Carpenter, A.E. Cell painting, a high-content image-based assay for morphological profiling using multiplexed fluorescent dyes. Nat. Protoc. 2016, 11, 1757–1774. [Google Scholar] [CrossRef] [PubMed]
- Caicedo, J.C.; Singh, S.; Carpenter, A.E. Applications in image-based profiling of perturbations. Curr. Opin. Biotechnol. 2016, 39, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Korn, K.; Krausz, E. Cell-based high-content screening of small-molecule libraries. Curr. Opin. Chem. Biol. 2007, 11, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Schulze, C.J.; Bray, W.M.; Woerhmann, M.H.; Stuart, J.; Lokey, R.S.; Linington, R.G. “Function-first” lead discovery: Mode of action profiling of natural product libraries using image-based screening. Chem. Biol. 2013, 20, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Young, D.W.; Bender, A.; Hoyt, J.; McWhinnie, E.; Chirn, G.W.; Tao, C.Y.; Tallarico, J.A.; Labow, M.; Jenkins, J.L.; Mitchison, T.J.; et al. Integrating high-content screening and ligand-target prediction to identify mechanism of action. Nat. Chem. Biol. 2008, 4, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Kremb, S.; Voolstra, C.R. High-resolution phenotypic profiling of natural products-induced effects on the single-cell level. Sci. Rep. 2017, 7, 44472. [Google Scholar] [CrossRef] [PubMed]
- Kurita, K.L.; Glassey, E.; Linington, R.G. Integration of high-content screening and untargeted metabolomics for comprehensive functional annotation of natural product libraries. Proc. Natl. Acad. Sci. USA 2015, 112, 11999–12004. [Google Scholar] [CrossRef] [PubMed]
- Sumiya, E.; Shimogawa, H.; Sasaki, H.; Tsutsumi, M.; Yoshita, K.; Ojika, M.; Suenaga, K.; Uesugi, M. Cell-morphology profiling of a natural product library identifies bisebromoamide and miuraenamide a as actin filament stabilizers. ACS Chem. Biol. 2011, 6, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Hertkorn, N.; Frommberger, M.; Witt, M.; Koch, B.P.; Schmitt-Kopplin, P.; Perdue, E.M. Natural organic matter and the event horizon of mass spectrometry. Anal. Chem. 2008, 80, 8908–8919. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.; Dietz, I.; Tziotis, D.; Moritz, F.; Rupp, J.; Schmitt-Kopplin, P. Molecular cartography in acute Chlamydia pneumoniae infections—A non-targeted metabolomics approach. Anal. Bioanal. Chem. 2013, 405, 5119–5131. [Google Scholar] [CrossRef] [PubMed]
- Tziotis, D.; Hertkorn, N.; Schmitt-Kopplin, P. Kendrick-analogous network visualisation of ion cyclotron resonance fourier transform mass spectra: Improved options for the assignment of elemental compositions and the classification of organic molecular complexity. Eur. J. Mass Spectrom. 2011, 17, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Bugni, T.S.; Harper, M.K.; McCulloch, M.W.; Reppart, J.; Ireland, C.M. Fractionated marine invertebrate extract libraries for drug discovery. Molecules 2008, 13, 1372–1383. [Google Scholar] [CrossRef] [PubMed]
- Bugni, T.S.; Richards, B.; Bhoite, L.; Cimbora, D.; Harper, M.K.; Ireland, C.M. Marine natural product libraries for high-throughput screening and rapid drug discovery. J. Nat. Prod. 2008, 71, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
- Ayyad, S.E.; Abdel-Halim, O.B.; Shier, W.T.; Hoye, T.R. Cytotoxic hydroazulene diterpenes from the brown alga cystoseira myrica. Z. Naturforsch. C 2003, 58, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.D.; Lee, Y.; Kang, S.H.; Kim, G.Y.; Kim, H.S.; Hyun, J.W. Cytotoxic effect of clerosterol isolated from Codium fragile on A2058 human melanoma cells. Mar. Drugs 2013, 11, 418–430. [Google Scholar] [CrossRef] [PubMed]
- Spavieri, J.; Kaiser, M.; Casey, R.; Hingley-Wilson, S.; Lalvani, A.; Blunden, G.; Tasdemir, D. Antiprotozoal, antimycobacterial and cytotoxic potential of some british green algae. Phytother. Res. 2010, 24, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.J.; Low, J.; Blosser, W.; Dowless, M.; Engler, T.A.; Stancato, L.F. A robust high-content imaging approach for probing the mechanism of action and phenotypic outcomes of cell-cycle modulators. Mol. Cancer Ther. 2011, 10, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Thangam, R.; Senthilkumar, D.; Suresh, V.; Sathuvan, M.; Sivasubramanian, S.; Pazhanichamy, K.; Gorlagunta, P.K.; Kannan, S.; Gunasekaran, P.; Rengasamy, R.; et al. Induction of ros-dependent mitochondria-mediated intrinsic apoptosis in mda-mb-231 cells by glycoprotein from codium decorticatum. J. Agric. Food Chem. 2014, 62, 3410–3421. [Google Scholar] [CrossRef] [PubMed]
- Khanavi, M.; Nabavi, M.; Sadati, N.; Shams Ardekani, M.; Sohrabipour, J.; Nabavi, S.M.; Ghaeli, P.; Ostad, S.N. Cytotoxic activity of some marine brown algae against cancer cell lines. Biol. Res. 2010, 43, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Guner, A.; Koksal, C.; Erel, S.B.; Kayalar, H.; Nalbantsoy, A.; Sukatar, A.; Karabay Yavasoglu, N.U. Antimicrobial and antioxidant activities with acute toxicity, cytotoxicity and mutagenicity of Cystoseira compressa (esper) gerloff & nizamuddin from the coast of urla (izmir, turkey). Cytotechnology 2015, 67, 135–143. [Google Scholar] [PubMed]
- Garcia-Dominguez, P.; Lepore, I.; Erb, C.; Gronemeyer, H.; Altucci, L.; Alvarez, R.; de Lera, A.R. Total synthesis of the proposed structures of the DNA methyl transferase inhibitors peyssonenynes, and structural revision of peyssonenyne b. Org. Biomol. Chem. 2011, 9, 6979–6987. [Google Scholar] [CrossRef] [PubMed]
- McPhail, K.L.; France, D.; Cornell-Kennon, S.; Gerwick, W.H. Peyssonenynes a and b, novel enediyne oxylipins with DNA methyl transferase inhibitory activity from the red marine alga Peyssonnelia caulifera. J. Nat. Prod. 2004, 67, 1010–1013. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.S.; Engel, S.; Smith, B.A.; Fairchild, C.R.; Aalbersberg, W.; Hay, M.E.; Kubanek, J. Structure and biological evaluation of novel cytotoxic sterol glycosides from the marine red alga Peyssonnelia sp. Bioorg. Med. Chem. 2010, 18, 8264–8269. [Google Scholar] [CrossRef] [PubMed]
- Raja, V.J.; Lim, K.H.; Leong, C.O.; Kam, T.S.; Bradshaw, T.D. Novel antitumour indole alkaloid, jerantinine a, evokes potent G2/m cell cycle arrest targeting microtubules. Investig. New Drugs 2014, 32, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Yoo, Y.H. Taxol-induced growth arrest and apoptosis is associated with the upregulation of the Cdk inhibitor, p21WAF1/CIP1, in human breast cancer cells. Oncol. Rep. 2012, 28, 2163–2169. [Google Scholar] [CrossRef] [PubMed]
- Fourest-Lieuvin, A.; Peris, L.; Gache, V.; Garcia-Saez, I.; Juillan-Binard, C.; Lantez, V.; Job, D. Microtubule regulation in mitosis: Tubulin phosphorylation by the cyclin-dependent kinase Cdk1. Mol. Biol. Cell 2006, 17, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Wali, V.B.; Bachawal, S.V.; Sylvester, P.W. Combined treatment of gamma-tocotrienol with statins induce mammary tumor cell cycle arrest in g1. Exp. Biol. Med. 2009, 234, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Lim do, Y.; Cho, H.J.; Seon, M.R.; Kim, J.K.; Lee, B.Y.; Park, J.H. Piceatannol, a natural stilbene from grapes, induces G1 cell cycle arrest in androgen-insensitive DU145 human prostate cancer cells via the inhibition of CDK activity. Cancer Lett. 2009, 285, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Mu, C.; Jia, P.; Yan, Z.; Liu, X.; Li, X.; Liu, H. Quercetin induces cell cycle G1 arrest through elevating Cdk inhibitors p21 and p27 in human hepatoma cell line (HEPG2). Methods Find. Exp. Clin. Pharmacol. 2007, 29, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Chen, W.J.; Lin-Shiau, S.Y.; Ho, C.T.; Lin, J.K. Tangeretin induces cell-cycle G1 arrest through inhibiting cyclin-dependent kinases 2 and 4 activities as well as elevating CDK inhibitors p21 and p27 in human colorectal carcinoma cells. Carcinogenesis 2002, 23, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Kim, G.Y.; Hyun, J.W.; Kim, N.D.; Kim, C.G.; Kim, W.J.; Yoo, Y.H.; Choi, Y.H. 7,8-dihydroxyflavone induces G1 arrest of the cell cycle in U937 human monocytic leukemia cells via induction of the Cdk inhibitor p27 and downregulation of pRB phosphorylation. Oncol. Rep. 2012, 28, 353–357. [Google Scholar] [PubMed]
- Shin, D.Y.; Sung Kang, H.; Kim, G.Y.; Kim, W.J.; Yoo, Y.H.; Choi, Y.H. Decitabine, a DNA methyltransferases inhibitor, induces cell cycle arrest at G2/m phase through p53-independent pathway in human cancer cells. Biomed. Pharmacother. 2013, 67, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.; Chen, Z.F.; Liang, Q.C.; Du, W.; Chen, H.M.; Su, W.Y.; Chen, G.Q.; Han, Z.G.; Fang, J.Y. Inhibition of DNA methyltransferase induces G2 cell cycle arrest and apoptosis in human colorectal cancer cells via inhibition of JAK2/STAT3/STAT5 signalling. J. Cell Mol. Med. 2009, 13, 3668–3679. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, T.; Koch, B.; Hertkorn, N.; Kattner, G. A simple and efficient method for the solid-phase extraction of dissolved organic matter (spe-dom) from seawater. Limnol. Oceanogr. Methods 2008, 6, 230–235. [Google Scholar] [CrossRef]
- Jha, B.; Kavita, K.; Westphal, J.; Hartmann, A.; Schmitt-Kopplin, P. Quorum sensing inhibition by Asparagopsis taxiformis, a marine macro alga: Separation of the compound that interrupts bacterial communication. Mar. Drugs 2013, 11, 253–265. [Google Scholar] [CrossRef] [PubMed]
| Species | Code | Phylum | Order | Family | Taxonomy ID | Location | Depth |
|---|---|---|---|---|---|---|---|
| Sargassum ilicifolium | SAR | Phaeophyceae | Fucales | Sargassaceae | 246892 | AFH, inner | 0.5 m |
| Turbinaria turbinata | TUR | Phaeophyceae | Fucales | Sargassaceae | 91044 | AFH, inner | 0.5 m |
| Cystoseira foeniculacea | CYS1 | Phaeophyceae | Fucales | Sargassaceae | 590109 | AFH, inner | 0.5 m |
| Cystoseira myrica | CYS2 | Phaeophyceae | Fucales | Sargassaceae | 590108 | AFH, inner | 0.5 m |
| Lobophora variegata | LOB | Phaeophyceae | Dictyotales | Dictyotaceae | 157001 | IFS, outer | 10–20 m |
| Peyssonnelia squamaria | PEY1 | Rhodophyta | Gigartinales | Peyssonneliaceae | 339596 | AFH, outer | 10–20 m |
| Peyssonnelia capensis | PEY2 | Rhodophyta | Gigartinales | Peyssonneliaceae | 367055 | AFH, outer | 10–20 m |
| Hildenbrandia sp. | HLD | Rhodophyta | Hildenbrandiales | Hildenbrandiaceae | AFH, outer | 10–20 m | |
| Bangia sp. | BAN | Rhodophyta | Bangiales | Bangiaceae | Mangrove | 0.5 m | |
| Actinotrichia fragilis | ACT | Rhodophyta | Nemaliales | Galaxauraceae | 268562 | AFH, outer | 10–20 m |
| Galaxaura rugosa | GAL | Rhodophyta | Nemaliales | Galaxauraceae | 268570 | AFH, outer | 10–20 m |
| Codium fragile | COD | Chlorophyta | Bryopsidales | Codiaceae | 3133 | AFH, inner | 8 m |
| Experimental Data | NIAIDS Database | |||||
|---|---|---|---|---|---|---|
| m/z | Intensity | S/N | Assigned Molecular Formula | EC50 (Cell) | Target | Compound Class |
| 457.111 | 1.49 × 106 | 5.8 | C21 H22 O10 | 20 μg/mL | Rnases H, gp120 | Polyphenol, Flavonoids |
| 297.097 | 1.56 × 106 | 8.7 | C14 H16 O7 | 45.9 μM | Polyphenol, | |
| 211.060 | 4.17 × 106 | 45.9 | C10 H10 O5 | inactive | Polyphenol, | |
| 447.127 | 4.28 × 106 | 10.9 | C20 H24 O10 | >46.8 μM | Polyphenol, Cycloalkanes | |
| 473.142 | 5.85 × 106 | 22.2 | C22 H26 O10 | inactive | Polyphenol, Biphenyls | |
| 367.175 | 7.59 × 106 | 49.1 | C19 H26 O7 | >3.4 μM | Polyphenol, | |
| 303.011 | 7.72 × 106 | 28.1 | C12 H8 O8 | inactive | Polyphenol, Cage Compounds | |
| 419.095 | 1.22 × 107 | 44.9 | C18 H20 O10 | >2.63 μM | Polyphenol, | |
| 425.106 | 2.94 × 107 | 75.8 | C17 H22 O11 | 12.4 μM | Polyphenol, | |
| 475.049 | 3.18 × 107 | 89.6 | C19 H16 O13 | 8 μg/mL cell | IN | Polyphenol, Pyrrolidines |
| 289.032 | 3.61 × 107 | 170.8 | C12 H10 O7 | inactive | Polyphenol, Carboxylic Acid Derivatives | |
| 503.080 | 3.95 × 107 | 106.2 | C21 H20 O13 | 4 µg/mL | Polyphenol, Flavonoids | |
| 499.179 | 4.49 × 107 | 120.4 | C21 H32 O12 | >75.6 μM | Polyphenol, | |
| 507.075 | 6.10 × 107 | 171.1 | C20 H20 O14 | 50 μM | RT | Polyphenol, Tannins |
| 363.069 | 9.22 × 107 | 397.6 | C15 H16 O9 | inactive | Polyphenol, Benzopyrans | |
| 349.053 | 1.32 × 108 | 718.3 | C14 H14 O9 | 18 μM | RT | Polyphenol, Tannins |
| 268.070 | 6.62 × 106 | 77.3 | C9 H12 F N3 O4 | >20 μM, 0028 μM | RT | Pyrimidine Nucleosides |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kremb, S.; Müller, C.; Schmitt-Kopplin, P.; Voolstra, C.R. Bioactive Potential of Marine Macroalgae from the Central Red Sea (Saudi Arabia) Assessed by High-Throughput Imaging-Based Phenotypic Profiling. Mar. Drugs 2017, 15, 80. https://doi.org/10.3390/md15030080
Kremb S, Müller C, Schmitt-Kopplin P, Voolstra CR. Bioactive Potential of Marine Macroalgae from the Central Red Sea (Saudi Arabia) Assessed by High-Throughput Imaging-Based Phenotypic Profiling. Marine Drugs. 2017; 15(3):80. https://doi.org/10.3390/md15030080
Chicago/Turabian StyleKremb, Stephan, Constanze Müller, Philippe Schmitt-Kopplin, and Christian R. Voolstra. 2017. "Bioactive Potential of Marine Macroalgae from the Central Red Sea (Saudi Arabia) Assessed by High-Throughput Imaging-Based Phenotypic Profiling" Marine Drugs 15, no. 3: 80. https://doi.org/10.3390/md15030080






