Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria
Abstract
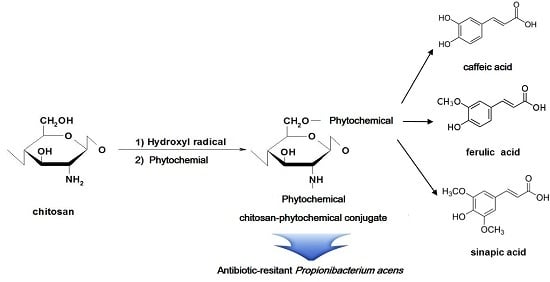
:1. Introduction
2. Results
2.1. Antibacterial Effect of Chitosan-Phytochemical Conjugates against Acne-Related Bacteria
2.2. Antibiotic Resistance of Acne-Related Bacteria against Commercial Antibiotics
2.3. Synergistic Antibacterial Effect between CCA and Antibiotics against Acne-Related Bacteria
3. Discussion
4. Materials and Methods
4.1. Preparation of Chitosan-Phytochemical Conjugates
4.2. Bacterial Strains and Culture Conditions
4.3. Determination of Minimum Inhibitory Concentration (MIC) and Fractional Inhibitory Concentration (FIC) Index
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Charakida, A.; Seaton, E.D.; Charakida, M.; Mouser, P.; Avgerinos, A.; Chu, A.C. Phototherapy in the treatment of acne vulgaris. Am. J. Clin. Dermatol. 2004, 5, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Huang, Y.Y.; Hamblin, M.R. Photodynamic therapy for localized infections—State of the art. Photodiagn. Photodyn. Ther. 2009, 6, 170–188. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, N.; Satoh, K.; Ono, M. In vitro evaluation of antibacterial, anticollagenase, and antioxidant activities of hop components (Humulus lupulus) addressing acne vulgaris. Phytomedicine 2009, 16, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Bae, H.J.; Kim, S.J.; Choi, I.S. In vitro antibacterial and anti-inflammatory properties of seaweed extracts against acne inducing bacteria, Propionibacterium acnes. J. Environ. Biol. 2011, 32, 313–318. [Google Scholar] [PubMed]
- Lee, J.H.; Eom, S.H.; Lee, E.H.; Jung, Y.J.; Kim, H.J.; Jo, M.R.; Son, K.T.; Lee, H.J.; Kim, J.H.; Lee, M.S.; et al. In vitro antibacterial and synergistic effect of phlorotannins isolated from edible brown seaweed Eisenia bicyclis against acne-related bacteria. Algae 2014, 29, 47–55. [Google Scholar] [CrossRef]
- Gollnick, H.; Cunliffe, W.; Berson, D.; Dreno, B.; Finlay, A.; Leyden, J.J.; Shalita, A.R.; Thiboutot, D. Management of acne: A report from a global alliance to improve outcomes in acne. J. Am. Acad. Dermatol. 2003, 49, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Han, S.M.; Lee, K.G.; Yeo, J.H.; Baek, H.J.; Park, K. Antibacterial and anti-inflammatory effects of honeybee (Apis mellifera) venom against acne-inducing bacteria. J. Med. Plants Res. 2010, 4, 459–464. [Google Scholar]
- Kim, J.Y.; Oh, T.H.; Kim, B.J.; Kim, S.S.; Lee, N.H.; Hyun, C.G. Chemical composition and anti-inflammatory effects of essential oil from Farfugium japonicum flower. J. Oleo Sci. 2008, 57, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Hatwar, B.; Nayak, S. Herbal plants and Propionibacterium acnes: An overview. Int. J. Biomed. Res. 2011, 2, 486–498. [Google Scholar] [CrossRef]
- Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of ten essential oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 cancer cells. Molecules 2010, 15, 3200–3210. [Google Scholar] [CrossRef] [PubMed]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, E.; Winnicka, K. Stability of chitosan-a challenge for pharmaceutical and biomedical applications. Mar. Drugs 2015, 13, 1819–1846. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.S.; Kim, S.K.; Ahn, C.B.; Je, J.Y. Preparation, characterization, and antioxidant properties of gallic acid-grafted-chitosans. Carbohydr. Polym. 2011, 83, 1617–1622. [Google Scholar] [CrossRef]
- Eom, S.H.; Kang, S.K.; Lee, D.S.; Myeong, J.I.; Lee, J.H.; Kim, H.W.; Kim, K.H.; Je, J.Y.; Jung, W.K.; Kim, Y.M. Synergistic antibacterial effect and antibacterial action mode of chitosan-ferulic acid conjugate against methicillin-resistant Staphylococcus aureus. J. Microbiol. Biotechnol. 2016, 26, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Ryu, B.; Je, J.Y.; Kim, S.K. Diethylaminoethyl chitosan induces apoptosis in HeLa cells via activation caspase-3 and p53 expression. Carbohydr. Polym. 2011, 84, 571–578. [Google Scholar] [CrossRef]
- Satheeshababu, B.K.; Shivakumar, K.L. Synthesis of conjugated chitosan and its effect on drug permeation from transdermal patches. Indian J. Pharm. Sci. 2013, 75, 162. [Google Scholar] [PubMed]
- Alves, N.M.; Mano, J.F. Chitosan derivatives obtained by chemical modifications for biomedical and environmental applications. Int. J. Biol. Macromol. 2008, 43, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Je, J.Y. Preparation of chitosan-catechin conjugates and their antioxidant activity. J. Chit. Chit. 2012, 17, 123–128. [Google Scholar]
- Xie, W.; Xu, P.; Liu, Q. Antioxidant activity of water-soluble chitosan derivatives. Bioorg. Med. Chem. Lett. 2011, 11, 1699–1701. [Google Scholar] [CrossRef]
- Yoon, H.J.; Moon, M.E.; Park, H.S.; Im, S.Y.; Kim, Y.H. Chitosan oligosaccharide (COS) inhibits LPS-induced inflammatory effects in RAW 264.7 macrophage cells. Biochem. Biophys. Res. Commun. 2007, 358, 954–959. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; Woo, J.Y.; Ahn, C.B.; Je, J.Y. Chitosan-hydroxycinnamic acid conjugates: Preparation, antioxidant and antimicrobial activity. Food Chem. 2014, 148, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Doshi, P.; Adsule, P.; Banerjee, K.; Oulkar, D. Phenolic compounds, antioxidant activity and insulinotropic effect of extracts prepared from grape (Vitis vinifera L.) byproducts. J. Food Sci. Technol. 2015, 52, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Moo-Huchin, V.M.; Moo-Huchin, M.I.; Estrada-León, R.J.; Cuevas-Glory, L.; Estrada-Mota, I.A.; Ortiz-Vázquez, E.; Betancur-Anconad, D.; Sauri-Duch, E. Antioxidant compounds, antioxidant activity and phenolic content in peel from three tropical fruits from Yucatan, Mexico. Food Chem. 2015, 166, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Mazurova, J.; Kukla, R.; Rozkot, M.; Lustykova, A.; Slehova, E.; Sleha, R.; Lipensky, J.; Opletal, L. Use of natural substances for boar semen decontamination. Vet. Med. 2015, 60, 235–247. [Google Scholar] [CrossRef]
- Lee, S.M.; Liu, K.H.; Liu, Y.Y.; Chang, Y.P.; Lin, C.C.; Chen, Y.S. Chitosonic® acid as a novel cosmetic ingredient: Evaluation of its antimicrobial, antioxidant and hydration activities. Materials 2013, 6, 1391–1402. [Google Scholar] [CrossRef]
- Tsai, T.H.; Tsai, T.H.; Wu, W.H.; Tseng, J.T.P.; Tsai, P.J. In vitro antimicrobial and anti-inflammatory effects of herbs against Propionibacterium acnes. Food Chem. 2010, 119, 964–968. [Google Scholar] [CrossRef]
- Lee, D.S.; Cho, Y.S.; Je, J.Y. Antioxidant and antibacterial activities of chitosan-phloroglucinol conjugate. Fish. Aquat. Sci. 2013, 16, 229–235. [Google Scholar] [CrossRef]
- Kim, D.H.; Je, J.Y. Antimicrobial activity of gallic acid-grafted-chitosan against fish pathogens. J. Carbohydr. Chem. 2015, 34, 163–171. [Google Scholar] [CrossRef]
- Meynadier, J.; Alirezai, M. Systemic antibiotics for acne. Dermatology 1998, 196, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; Eom, S.H.; Kim, Y.M.; Kim, H.S.; Yim, M.J.; Lee, S.H.; Kim, D.H.; Je, J.Y. Antibacterial and synergic effects of gallic acid-grafted-chitosan with β-lactams against methicillin-resistant Staphylococcus aureus (MRSA). Can. J. Microbiol. 2014, 60, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Oprica, C.; Emtestam, L.; Lapins, J.; Borglund, E.; Nyberg, F.; Stenlund, K.; Lundeberg, L.; Sillerström, E.; Nord, C.E. Antibiotic-resistant Propionibacterium acnes on the skin of patients with moderate to severe acne in Stockholm. Anaerobe 2004, 10, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Soussy, C.J.; Cluzel, R.; Courvalin, P. Definition and Determination of in vitro antibiotic susceptibility breakpoints for bacteria in France. Eur. J. Clin. Microbiol. Infect. Dis. 1994, 13, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.H.; Park, J.H.; Yu, D.U.; Choi, J.I.; Choi, J.D.; Lee, M.S.; Kim, Y.M. Antimicrobial activity of brown alga Eisenia bicyclis against methicillin-resistant Staphylococcus aureus. Fish. Aquat. Sci. 2011, 14, 251–256. [Google Scholar] [CrossRef]
- Kim, K.H.; Eom, S.H.; Kim, H.J.; Lee, D.S.; Nshimiyumukiza, O.; Kim, S.D.; Kim, Y.M.; Lee, M.S. Antifungal and synergistic effects of an ethyl acetate extract of the edible brown seaweed Eisenia bicyclis against Candida species. Fish. Aquat. Sci. 2014, 17, 209–214. [Google Scholar] [CrossRef]
- Eom, S.H.; Santos, J.A.; Kim, J.H.; Jung, W.K.; Kim, D.H.; Kim, Y.M. In vitro antibacterial and synergistic activity of an Ecklonia cava extract against antibiotic-resistant Streptococcus parauberis. Fish. Aquat. Sci. 2015, 18, 241–247. [Google Scholar]
- Eom, S.H.; Jung, Y.J.; Lee, D.S.; Yim, M.J.; Kim, H.S.; Lee, S.H.; Myeong, J.I.; Lee, J.H.; Kim, H.W.; Kim, K.H.; et al. Studies on antimicrobial activity of Poncirus trifoliata ethyl extract fraction against methicillin-resistant Staphylococcus aureus and to elucidate its antibacterial mechanism. J. Environ. Biol. 2016, 37, 129–134. [Google Scholar] [PubMed]
- Nshmiyumukiza, O.; Kang, S.K.; Kim, H.J.; Lee, E.H.; Han, H.N.; Kim, Y.H.; Kim, D.H.; Kim, J.H.; Eom, S.H.; Kim, Y.M. Synergistic antibacterial activity of Ecklonia cava (Phaeophyceae: Laminariales) against Listeria monocytogenes (Bacillales: Listeriaceae). Fish. Aquat. Sci. 2015, 18, 1–6. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, J.H.; Kim, D.H.; Kim, S.H.; Kim, H.R.; Kim, Y.M. Synergistic antimicrobial effect of Sargassum serratifolium (C. Agardh) C. Agardh extract against human skin pathogens. Korean J. Food Sci. Technol. 2016, 48, 241–246. [Google Scholar] [CrossRef]
- Champer, J.; Patel, J.; Fernando, N.; Salehi, E.; Wong, V.; Kim, J. Chitosan against cutaneous pathogens. AMB Express 2013, 3, 37. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Bonomo, R.A.; Szabo, D. Mechanisms of mutidrug rsistance in Acinetobacter species and Pseudomonas aeruginosa. Clin. Infect. Dis. 2006, 43, S49–S56. [Google Scholar] [CrossRef] [PubMed]
- Eaton, P.; Fernandes, J.C.; Pereira, E.; Pintado, M.E.; Malcata, F.X. Atomic force microscopy study of the antibacterial effects of chitosans on Escherichia coli and Staphylococcus aureus. Ultramicroscopy 2008, 108, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Z.S.; Cooper, S.L. Interactions between dendrimer biocides and bacterial membranes. Biomaterials 2002, 23, 3359–3368. [Google Scholar] [CrossRef]
- Sánchez-Maldonado, A.F.; Schieber, A.; Gänzle, M.G. Structure-function relationships of the antibacterial activity of phenolic acids and their metabolism by lactic acid bacteria. J. Appl. Microbiol. 2011, 111, 1176–1184. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Je, J.Y.; Kim, Y.M. Anti-inflammatory effects of chitosan–phytochemical conjugates against Propioinbacterium acnes-induced inflammation. Korean J. Fish. Aquat. Sci. 2016, 49, 589–593. [Google Scholar]
- Fan, Y.; Hao, F.; Wang, W.; Lu, Y.; He, L.; Wang, G.; Chen, W. Multicenter cross-sectional observational study of antibiotic resistance and the genotypes of Propionibacterium acnes isolated from Chinese patients with acne vulgaris. J. Dermatol. 2015, 43, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically Approved Standard, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2006. [Google Scholar]
- Norden, C.W.; Wentzel, H.; Keleti, E. Comparison of techniques for measurement of in vitro antibiotic synergism. J. Infect. Dis. 1979, 140, 629–633. [Google Scholar] [CrossRef] [PubMed]
| Strain | MIC (μg/mL) | |||
|---|---|---|---|---|
| CCA 1 | CFA 2 | CSA 3 | Unmodified Chitosan | |
| Staphylococcus aureus KCTC 1927 | 8 | 16 | 16 | 16 |
| Staphylococcus epidermidis KCTC 1370 | 64 | 64 | 64 | 64 |
| Pseudomonas aeruginosa KCTC 1637 | 16 | 32 | 32 | 32 |
| Propionibacterium acnes KCTC 3314 | 256 | 256 | 256 | 512 |
| P. acnes isolate 2874 | 256 | 256 | 256 | 512 |
| P. acnes isolate 2875 | 128 | 256 | 256 | 256 |
| Strain | MIC (μg/mL) | ||
|---|---|---|---|
| Erythromycin | Lincomycin | Tetracycline | |
| Staphylococcus aureus KCTC 1927 | 2 | 4 | 0.5 |
| Staphylococcus epidermidis KCTC 1370 | 0.125 | 0.25 | 2 |
| Pseudomonas aeruginosa KCTC 1637 | 16 | 64 | 0.125 |
| Propionibacterium acnes KCTC 3314 | 1024 | 1024 | 32 |
| P. acnes isolate 2874 | 1024 | 1024 | 16 |
| P. acnes isolate 2875 | 0.125 | 1 | 0.125 |
| Soussy’s MIC breakpoints 1 | 1–4 1 | 2–8 1 | 4–8 1 |
| Strains | Test Compound | MIC (μg/mL) | Median ∑FIC 1 | ∑FICmax 2 | ∑FICmin 3 | Minimum Concentration for Observing Synergy |
|---|---|---|---|---|---|---|
| P. acnes KCTC 3314 | CCA | 256 | 0.533 | 1.016 | 0.188 | 64.0 |
| Tetracycline | 32 | 4.0 | ||||
| P. acnes isolate 2874 | CCA | 256 | 0.502 | 1.004 | 0.375 | 64.0 |
| Tetracycline | 16 | 4.0 | ||||
| P. acnes KCTC 3314 | CCA | 256 | 0.502 | 1.004 | 0.188 | 32.0 |
| Erythromycin | 1024 | 64.0 | ||||
| P. acnes isolate 2874 | CCA | 256 | 0.502 | 1.016 | 0.313 | 64.0 |
| Erythromycin | 1024 | 128.0 | ||||
| Pseudomonas aeruginosa KCTC 1637 | CCA | 16 | 0.5 | 1.063 | 0.266 | 4.0 |
| Erythromycin | 16 | 0.5 | ||||
| P. acnes KCTC 3314 | CCA | 256 | 0.504 | 1.016 | 0.375 | 64.0 |
| Lincomycin | 1024 | 256.0 | ||||
| P. acnes isolate 2874 | CCA | 256 | 0.502 | 1.016 | 0.313 | 64.0 |
| Lincomycin | 1024 | 256.0 | ||||
| P. aeruginosa KCTC 1637 | CCA | 16 | 0.375 | 1.063 | 0.266 | 4.0 |
| Lincomycin | 64 | 16.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.-H.; Yu, D.; Eom, S.-H.; Kim, S.-H.; Oh, J.; Jung, W.; Kim, Y.-M. Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria. Mar. Drugs 2017, 15, 167. https://doi.org/10.3390/md15060167
Kim J-H, Yu D, Eom S-H, Kim S-H, Oh J, Jung W, Kim Y-M. Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria. Marine Drugs. 2017; 15(6):167. https://doi.org/10.3390/md15060167
Chicago/Turabian StyleKim, Ji-Hoon, Daeung Yu, Sung-Hwan Eom, Song-Hee Kim, Junghwan Oh, Won‐Kyo Jung, and Young-Mog Kim. 2017. "Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria" Marine Drugs 15, no. 6: 167. https://doi.org/10.3390/md15060167