Chemopreventive Effects of Sarcophine-diol on Ultraviolet B-induced Skin Tumor Development in SKH-1 Hairless Mice
Abstract
:1. Introduction
2. Results
SD treatment did not affect body weight gain of SKH-1 mice
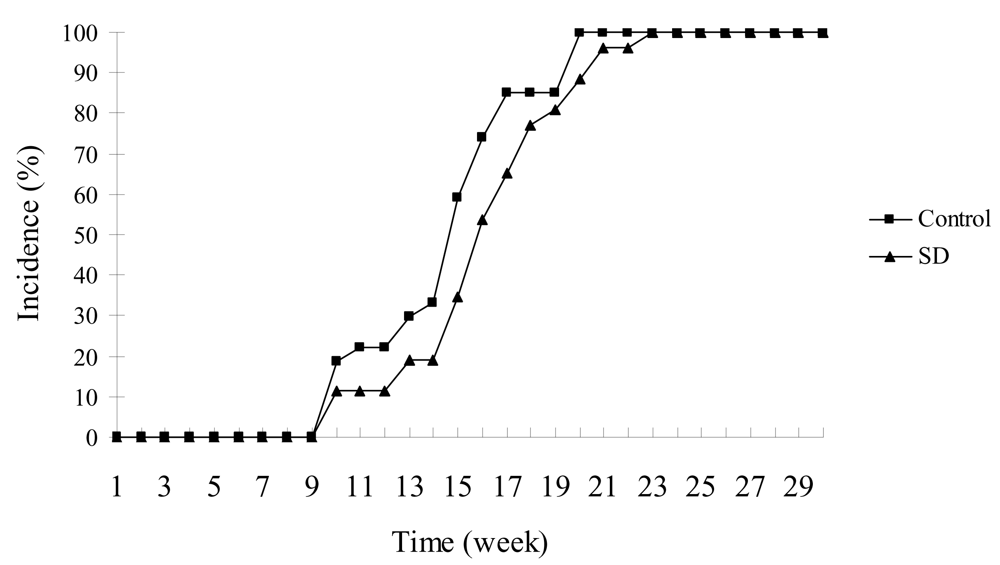
SD treatment did not significantly inhibit the incidence of skin tumors in SKH-1 mice
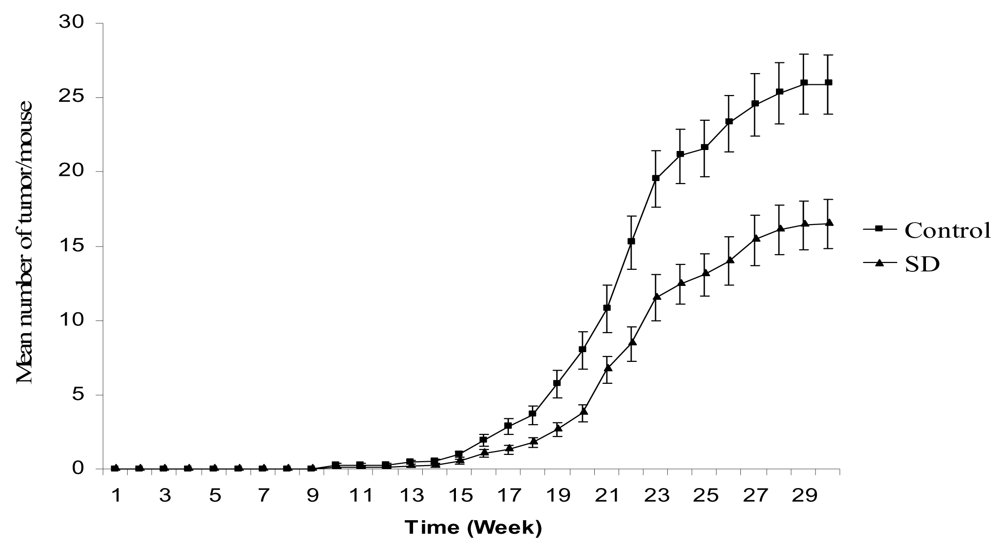
SD treatment inhibited tumor multiplicity in SKH-1 mice
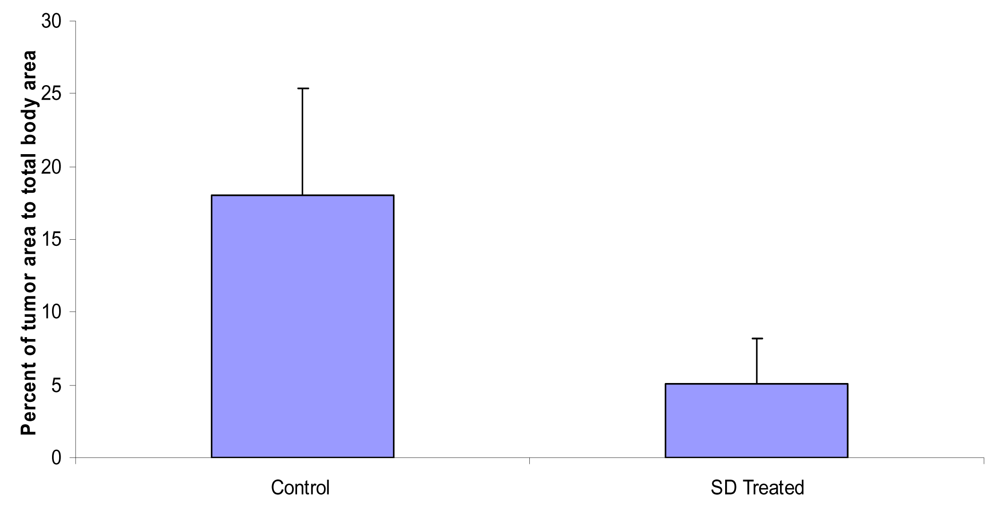
SD treatment inhibited the percent of total tumor area to total back area of SKH-1 mice
UVB radiation induced squamous cell carcinoma in SKH-1 mice
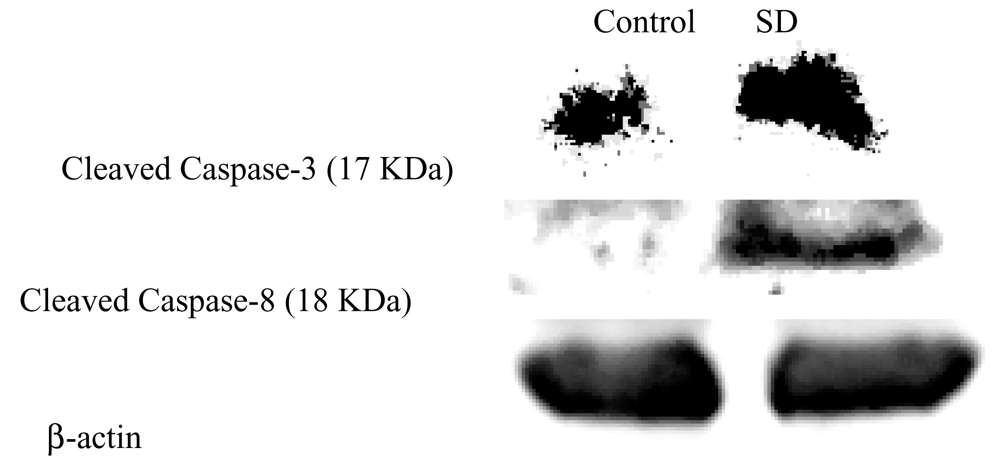
SD treatment induced caspase-3, -8 expressions but not caspase-9 in SKH-1 mice
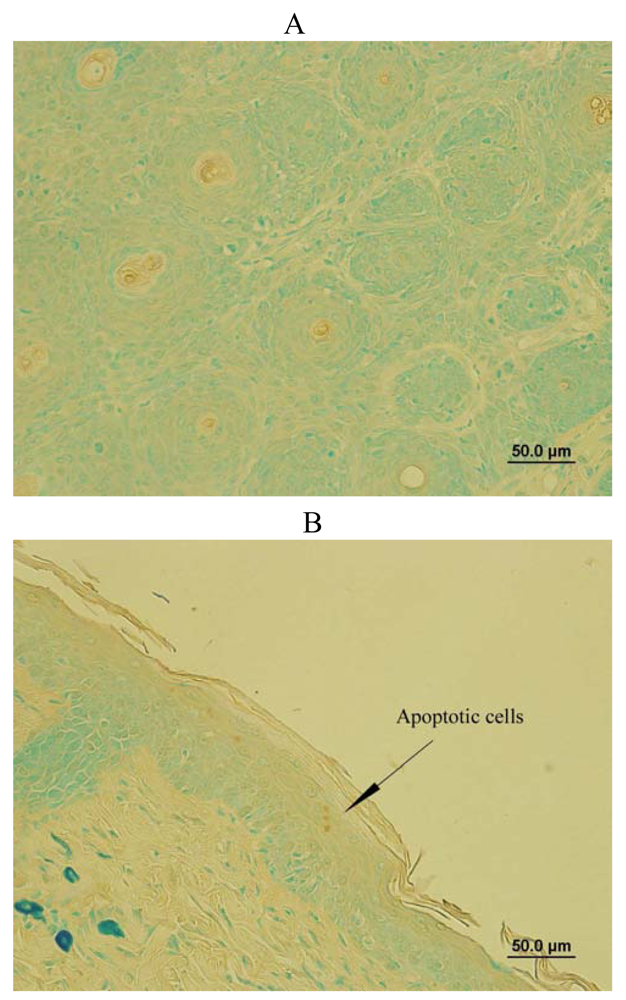
SD treatment increased DNA fragmented apoptotic cells in SKH-1 mice
3. Discussion
4. Experimental Section
Materials and reagents
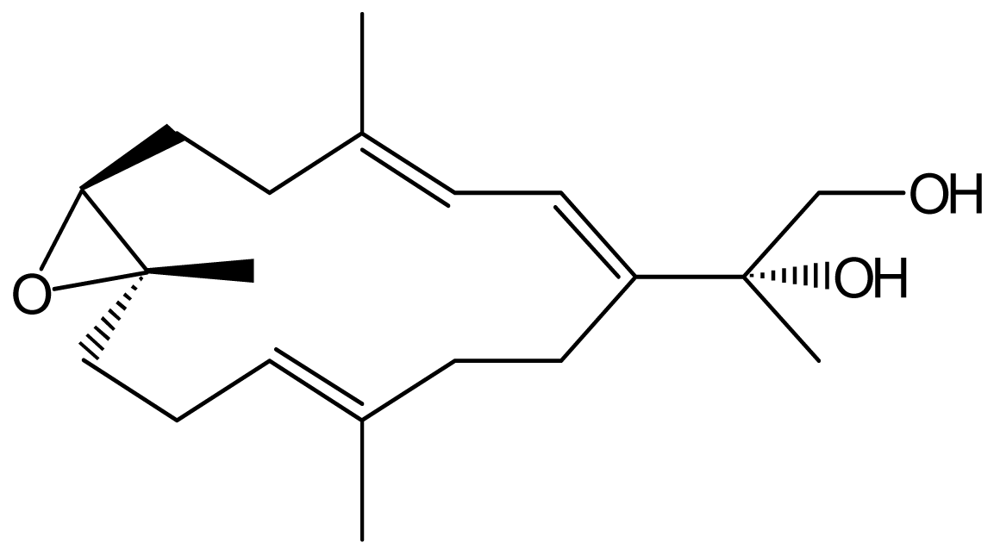
Synthesis of SD
Animals
UVB source
UVB-initiated and UVB-promoted skin tumor development protocol
Effects of SD on tumor area of SKH-1 mice
Histopathological analysis of mouse tumors
Tumorigenesis protocol for mechanistic studies
Preparation of cell lysate for Western blotting
Western blot analysis of caspase -3, -8, and -9 expressions
Determination of DNA fragmented cells on skin sections
Statistical analysis
5. Conclusions
Acknowledgements
References and Notes
- Mantena, SK; Sharma, SD; Katiyar, SK. Berberine inhibits growth, induces G1 arrest and apoptosis in human epidermoid carcinoma A431 cells by regulating Cdki-Cdk-cyclin cascade, disruption of mitochondrial membrane potential and cleavage of caspase 3 and PARP. Carcinogenesis 2006, 27, 2018–2027. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society Publication. Cancer Facts & Figures-2008. Available online: http://www.cancer.org/docroot/STT/content/STT_1x_Cancer_Facts_and_Figures_2008.asp?from=fast.
- Naylor, MF; Boyd, A; Smith, DW; Cameron, GS; Hubbard, D; Neldner, KH. High sun protection factor sunscreens in the uppression of actinic neoplasia. Arch Dermatol 1995, 131, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, N; Irby, C; Katiyar, SK; Elmets, CA. Photoprotective effects of green tea polyphenols. Photodermatol Photoimmunol Photomed 2007, 23, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Koh, HK. Preventive strategies and research for ultraviolet-associated cancer. Environ Health Perspect 1995, 103, 255–257. [Google Scholar] [PubMed]
- Learn, DB; Beasley, DG; Giddens, LD; Beard, J; Stanfield, JW; Roberts, LK. Minimum doses of ultraviolet radiation required to induce murine skin edema and immunosuppression are different and depend on the ultraviolet emission spectrum of the source. Photochem Photobiol 1995, 62, 1066–1075. [Google Scholar] [PubMed]
- Soehnge, H; Ouhtit, A; Ananthaswamy, HN. Mechanisms of induction of skin cancer by UV radiation. Front Biosci 1997, 2, 538–551. [Google Scholar]
- Kraemer, KH. Sunlight and skin cancer: another link revealed. Proc Natl Acad Sci 1997, 94, 11–14. [Google Scholar] [CrossRef] [PubMed]
- De Gruijl, FR; Forbes, PD. UV-induced skin cancer in a hairless mouse model. BioEssays 1995, 17, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Forbes, PD. Relevance of animal models of photocarcinogenesis to humans. Photochem Photobiol 1996, 63 , 357–362. [Google Scholar] [PubMed]
- Willis, I; Menter, JM; Whyte, HJ. The rapid induction of cancers in the hairless mouse utilizing the principle of photoaugmentation. J Invest Dematol 1981, 76, 404–408. [Google Scholar] [CrossRef]
- Bowden, GT. Prevention of non-melanoma skin cancer by targeting ultraviolet-B-light signaling. Nat Rev Cancer 2004, 4, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Haefner, B. Drugs from the deep: marine natural products as drug candidates. Drug Discov Today 2003, 8, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, H; Khalifa, S; Konoshima, T; Zjawiony, JK. An improved synthesis of 7, 8-epoxy-1, 3, 11-cembratriene-15R (α), 16-diol, a cembranoid of marine origin with a potent cancer chemopreventive activity. Mar Drugs 2002, 2, 1–7. [Google Scholar]
- Fahmy, H; Zjawiony, JK; Konoshima, T; Tokuda, H; Khan, S; Khalifa, S. Potent skin cancer chemopreventing activity of some novel semi-synthesis cembranoids from marine sources. Mar Drugs 2006, 4, 1–9. [Google Scholar] [CrossRef]
- Kundoor, V; Zhang, X; Khalifa, S; Fahmy, H; Dwivedi, C. A possible mechanism of action of the chemopreventive effects of sarcotriol on skin tumor development in CD-1 mice. Mar Drugs 2006, 4, 274–285. [Google Scholar]
- Zhang, X; Kundoor, V; Khalifa, S; Zeman, D; Fahmy, H; Dwivedi, C. Chemopreventive effects of sarcophine-diol on skin tumor development in CD-1 mice. Cancer Lett 2007, 253, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Kundoor, V; Zhang, X; Bommareddy, A; Khalifa, S; Fahmy, H; Dwivedi, C. Chemopreventive effects of sarcotriol on ultraviolet B-induced skin tumor development in SKH-1 hairless mice. Mar Drugs 2007, 5, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M; Agarwal, C; Singh, RP; Guan, X; Dwivedi, C; Agarwal, R. Skin cancer chemopreventive agent, α-santalol, induces apoptotic death of human epidermoid carcinoma A431 cells via caspase activation together with dissipation of mitochondrial membrane potential and cytochrome c release. Carcinogenesis 2005, 26, 369–380. [Google Scholar] [PubMed]
- Katsuyama, I; Fahmy, H; Zjawiony, JK; Khalifa, S; Kilada, RW; Konoshima, T; Takasaki, M; Tokuda, H. Semisythesis of new sarcophine derivatives with chemopreventive activity. J Nat Prod 2002, 65, 1809–1814. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, C; Valluri, HB; Guan, X; Agarwal, R. Chemopreventive effects of α-santalol on ultraviolet B radiation-induced skin tumor development in SKH-1 hairless mice. Carcinogenesis 2006, 27, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- Vermes, I; Haanen, C; Reutelingsperger, C. Flow cytometry of apoptotic cell death. J Immunol Methods 2000, 243, 167–190. [Google Scholar] [CrossRef] [PubMed]
- Schwartzman, R; Cidlowski, JA. Apoptosis: the biochemistry and molecular biology of programmed cell death. Endocr Rev 1993, 14, 133–151. [Google Scholar] [PubMed]
- Ne’eman, I; Fishelson, L; Kashman, Y. Sarcophine-a new toxin from the soft coral Sarcophyton glaucum (Alcyonaria). Toxicon 1974, 12, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Sawant, S; Youssef, D; Mayer, A; Sylvester, P; Wali, V; Arant, M; Sayed, KE. Anticancer and anti-inflammatory sulfur-containing semisynthetic derivatives of sarcophine. Chem Pharm Bull 2006, 54, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Arasada, BL; Bommareddy, A; Zhang, X; Bremmon, K; Dwivedi, C. Effects of α-santalol on proapoptotic caspases and p53 expression in UVB irradiated mouse skin. Anticancer Res 2008, 28, 129–132. [Google Scholar] [PubMed]
- Meeran, SM; Mantena, SK; Elmets, CA; Katiyar, SK. (−)-Epigallocatechin-3-gallate prevents photocarcinogenesis in mice through interleukin-12-dependent DNA repair. Cancer Res 2006, 66, 5512–5520. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, KS; Korman, NJ; Mukhtar, H; Agarwal, R. Protective effects of silymarin against photocarcinogenesis in a mouse skin model. J Natl Cancer Inst 1997, 89, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Dhanalakshmi, S; Mallikarjuna, GU; Singh, RP; Agarwal, R. Silibinin prevents ultraviolet radiation-caused skin damages in SKH-1 hairless mice via a decrease in thymine dimer positive cells and an up-regulation of p53-p21/Cip1 in epidermis. Carcinogenesis 2004, 25, 1459–1465. [Google Scholar] [CrossRef] [PubMed]
- Debatin, K. Apoptosis pathway in cancer and cancer therapy. Cancer Immunol Immunother 2004, 53, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Brady, HJ. Apoptosis and leukaemia. Br J Haematol 2003, 123, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Ghobrial, IM; Witzig, TE; Adjei, AA. Targeting apoptosis pathways in cancer therapy. CA-Cancer J Clin 2005, 55, 178–194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X; Bommareddy, A; Chen, W; Khalifa, S; Kaushik, RS; Fahmy, H; Dwivedi, C. Sarcophine-diol, a chemopreventive agent of skin cancer, inhibits cell growth and induces apoptosis through extrinsic pathway in human epidermoid carcinoma A431 cells. Transl Oncol 2009, 2, 21–30. [Google Scholar] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, X.; Bommareddy, A.; Chen, W.; Hildreth, M.B.; Kaushik, R.S.; Zeman, D.; Khalifa, S.; Fahmy, H.; Dwivedi, C. Chemopreventive Effects of Sarcophine-diol on Ultraviolet B-induced Skin Tumor Development in SKH-1 Hairless Mice. Mar. Drugs 2009, 7, 153-165. https://doi.org/10.3390/md7020153
Zhang X, Bommareddy A, Chen W, Hildreth MB, Kaushik RS, Zeman D, Khalifa S, Fahmy H, Dwivedi C. Chemopreventive Effects of Sarcophine-diol on Ultraviolet B-induced Skin Tumor Development in SKH-1 Hairless Mice. Marine Drugs. 2009; 7(2):153-165. https://doi.org/10.3390/md7020153
Chicago/Turabian StyleZhang, Xiaoying, Ajay Bommareddy, Wei Chen, Michael B. Hildreth, Radhey S. Kaushik, David Zeman, Sherief Khalifa, Hesham Fahmy, and Chandradhar Dwivedi. 2009. "Chemopreventive Effects of Sarcophine-diol on Ultraviolet B-induced Skin Tumor Development in SKH-1 Hairless Mice" Marine Drugs 7, no. 2: 153-165. https://doi.org/10.3390/md7020153



