Arsenic Speciation and Extraction and the Significance of Biodegradable Acid on Arsenic Removal—An Approach for Remediation of Arsenic-Contaminated Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Soil Samples
2.2. Experimental Design
2.2.1. Sequential Extraction Protocol
2.2.2. Washing Experiments
2.3. Analytical Methods and Statistical Analysis
3. Results
3.1. Soil Characterizations
3.2. Sequential Extraction of Arsenic in Soil
3.3. Soil Washing Experiment Using Biodegradable Acids
3.3.1. Arsenic Extraction
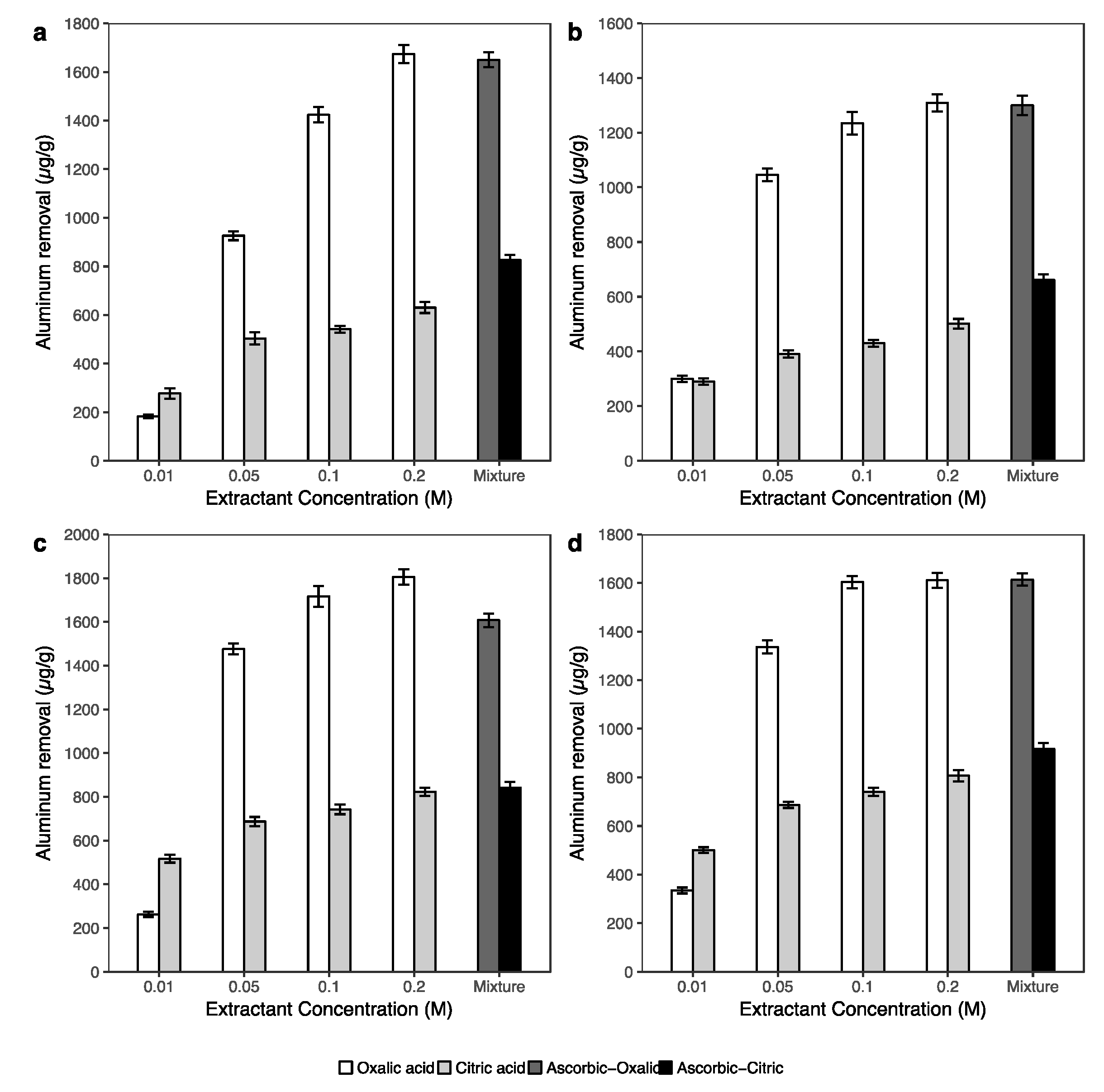
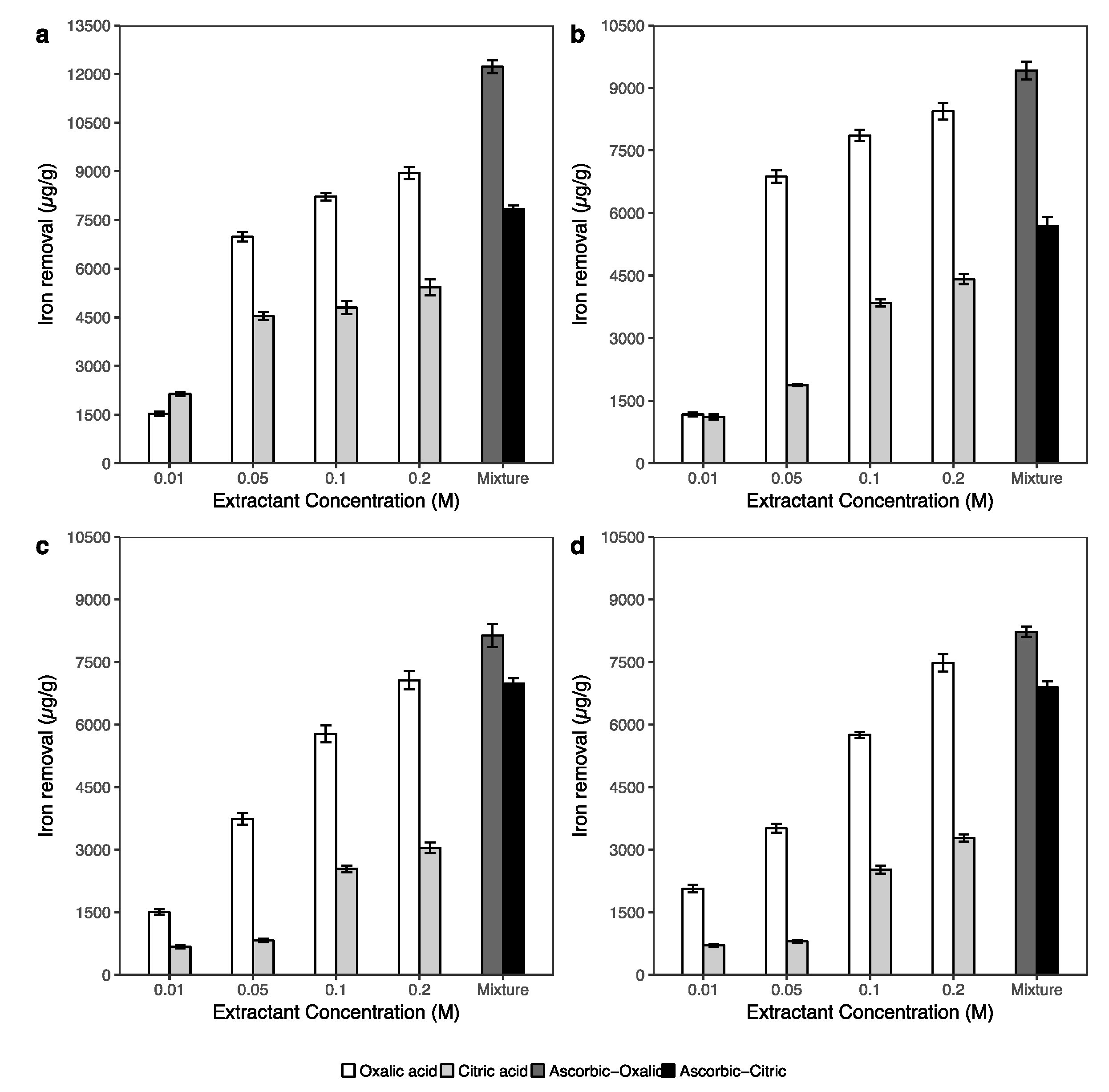
3.3.2. Iron, Aluminum and Manganese Extraction
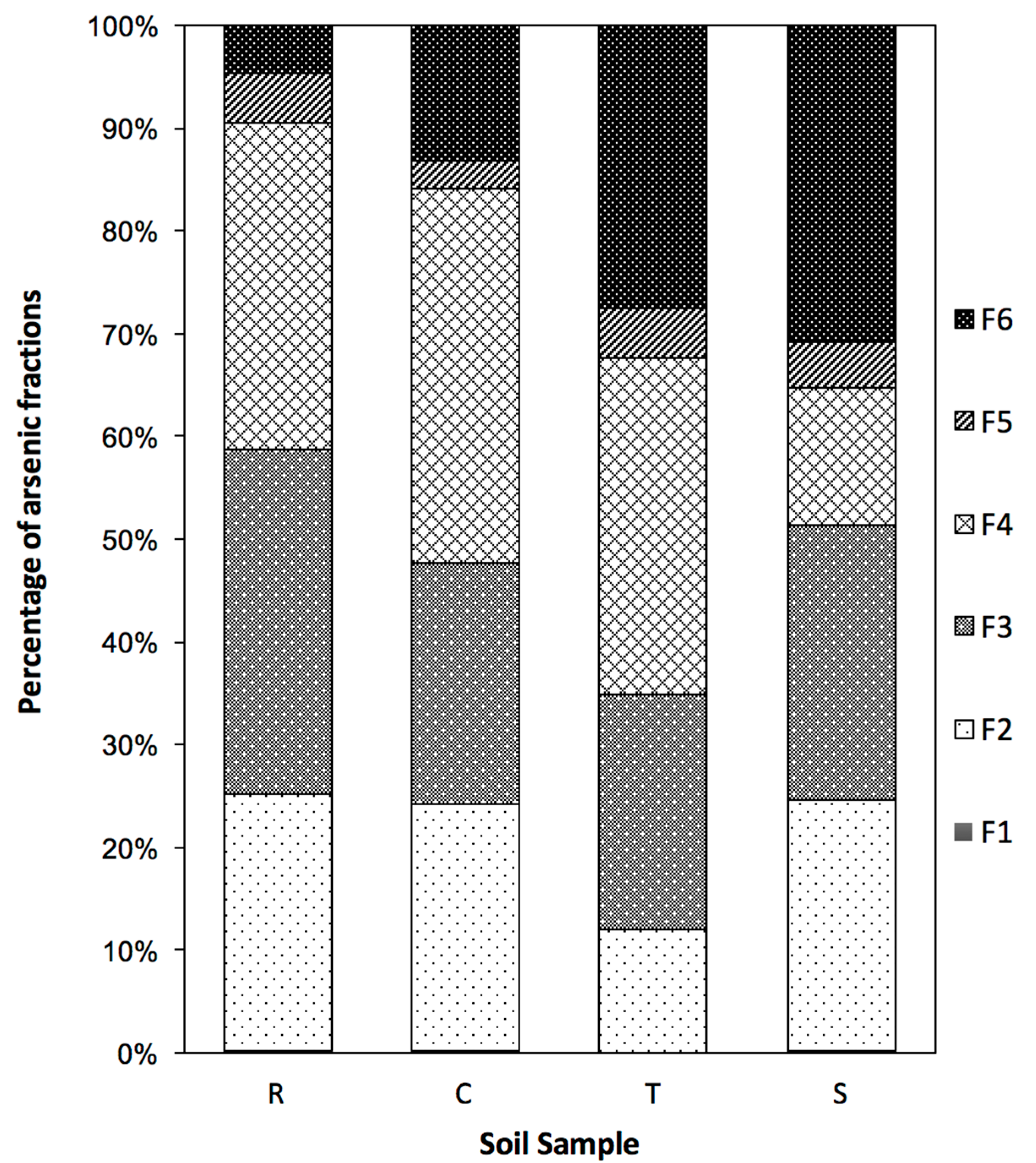
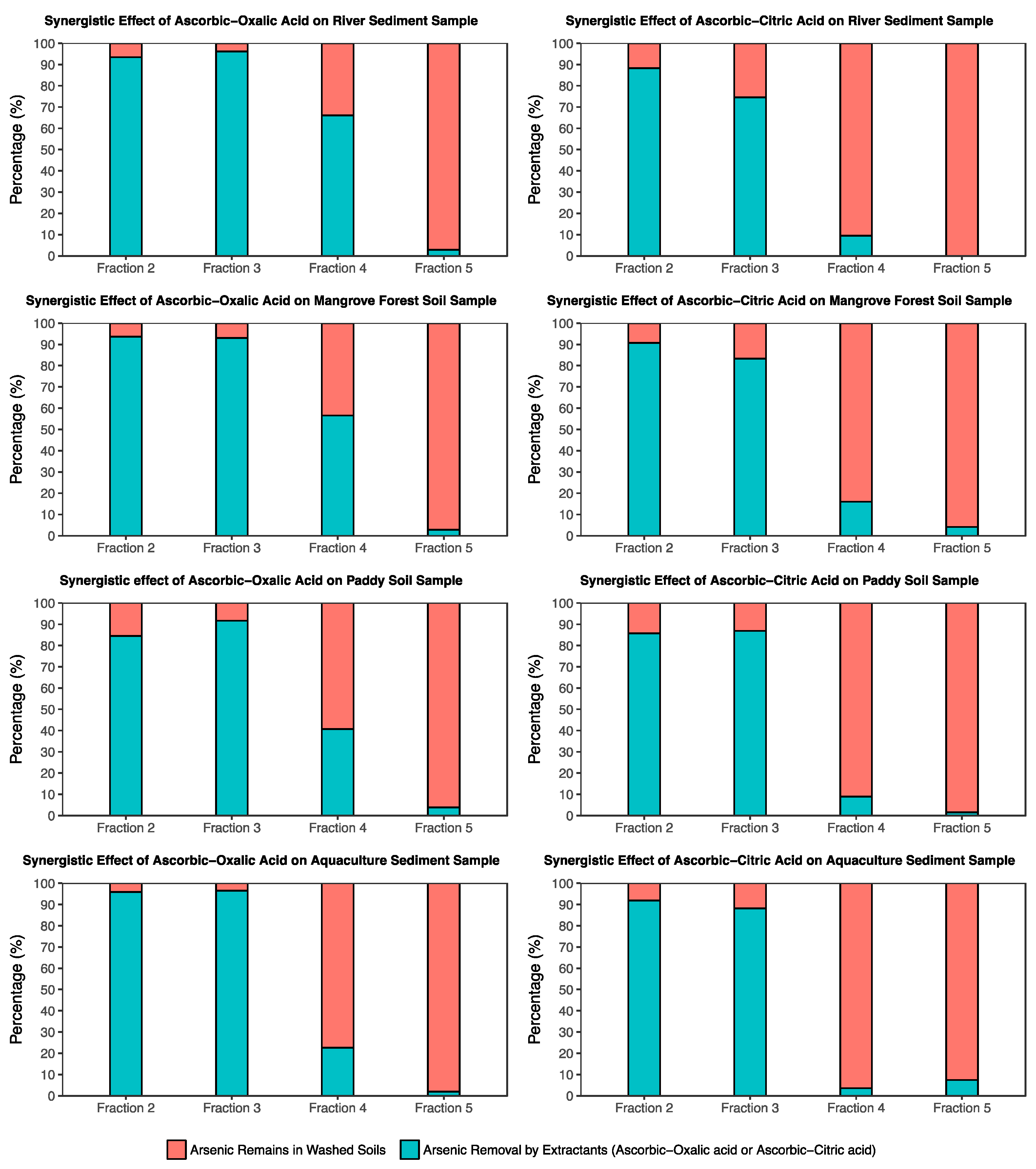
3.4. Arsenic Fraction after the Washing Experiment
4. Discussion
4.1. Toxicity Potential of Arsenic in Soil Samples
4.2. Effectiveness of Biodegradable Organic Acid in Arsenic Removal
5. Conclusions
- (1)
- Most of the arsenic in the four investigated soils was bound with amorphous Fe, Mn (F3), and crystalline Al and iron (F4) and was in the exchangeable fraction (F2). The As content in the organic matter fraction (F5) ranged from 3–5% in all samples.
- (2)
- Biodegradable organic acids showed high potential for remediation the arsenic contaminated soil. However, the efficiency of As removal using the combination of 0.1 M ascorbic with 0.2 M oxalic and/or 0.2 M citric acids was better than the single acid (0.2 M oxalic and/or 0.2 M citric acid) extractions.
- (3)
- The results of extracted arsenic using the combination of 0.2 M oxalic acid and 0.1 M ascorbic acid solution suggested that this solution can effectively wash arsenic from soils associated with crystalline Fe oxides. In addition, the solution of 0.2 M citric and 0.1 M ascorbic acids can be applied to arsenic-contaminated soil, which has a low crystalline Fe oxide content in soil.
- (4)
- The amount of extracted arsenic under the effects of biodegradable acids was strongly correlated with the dissolution of iron and aluminum in soils.
- (5)
- The soils treated using biodegradable acids had low remaining concentrations of arsenic that are primarily contained in crystalline iron oxides and organic matter fractions.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tanaka, T. Distribution of arsenic in the natural environment with emphasis on rocks and soils. Appl. Organomet. Chem. 1988, 2, 283–295. [Google Scholar] [CrossRef]
- Frankerberger, W. Environmental Chemistry of Arsenic; Wiley: Hoboken, NJ, USA, 2002; ISBN 0824706765. [Google Scholar]
- Stoeppler, M. Arsenic. In Elements and Their Compounds in the Environment; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2004; pp. 1321–1364. ISBN 9783527619634. [Google Scholar]
- Health Canada. Guidelines for Canadian Drinking Water Quality—Summary Table. Water and Air; Health Canada: Ottawa, ON, Canada, 2014.
- Dhar, R.K.; Zheng, Y.; Rubenstone, J.; van Geen, A. A rapid colorimetric method for measuring arsenic concentrations in groundwater. Anal. Chim. Acta 2004, 526, 203–209. [Google Scholar] [CrossRef]
- Nriagu, J.O.; Bhattacharya, P.; Mukherjee, A.B.; Bundschuh, J.; Zevenhoven, R.; Loeppert, R.H. Arsenic in soil and groundwater: An overview. In Arsenic in Soil and Groundwater Environment: Biogeochemical Interactions, Health Effects and Remediation; Bhattacharya, P., Mukherjee, A.B., Bundschuh, J., Zevenhoven, R., Loeppert, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 3–60. ISBN 1875-1121. [Google Scholar]
- Duruibe, J.O.; Ogwuegbu, M.O.C.; Egwurugwu, J.N. Heavy metal pollution and human biotoxic effects. Int. J. Phys. Sci. 2007, 2, 112–118. [Google Scholar] [CrossRef]
- Bosch, A.C.; O’Neill, B.; Sigge, G.O.; Kerwath, S.E.; Hoffman, L.C. Heavy metals in marine fish meat and consumer health: A review. J. Sci. Food Agric. 2016, 96, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Leung, H.M.; Leung, A.O.W.; Wang, H.S.; Ma, K.K.; Liang, Y.; Ho, K.C.; Cheung, K.C.; Tohidi, F.; Yung, K.K.L. Assessment of heavy metals/metalloid (As, Pb, Cd, Ni, Zn, Cr, Cu, Mn) concentrations in edible fish species tissue in the Pearl River Delta (PRD), China. Mar. Pollut. Bull. 2014, 78, 235–245. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, M.J.; Zarcinas, B.A.; Stevens, D.P.; Cook, N. Soil testing for heavy metals. Commun. Soil Sci. Plant Anal. 2000, 31, 1661–1700. [Google Scholar] [CrossRef]
- McBride, M.B. Environmental Chemistry of Soils; Oxford University Press, Inc.: New York, NY, USA, 1994; ISBN 0195070119. [Google Scholar]
- Nguyen Van, T.; Ozaki, A.; Nguyen Tho, H.; Nguyen Duc, A.; Tran Thi, Y.; Kurosawa, K. Arsenic and Heavy Metal Contamination in Soils under Different Land Use in an Estuary in Northern Vietnam. Int. J. Environ. Res. Public Health 2016, 13, 1091. [Google Scholar] [CrossRef] [PubMed]
- Adamo, P.; Arienzo, M.; Imperato, M.; Naimo, D.; Nardi, G.; Stanzione, D. Distribution and partition of heavy metals in surface and sub-surface sediments of Naples city port. Chemosphere 2005, 61, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yang, F.; Yan, X.; Huo, Z.; Zhang, G. Heavy Metal Concentrations in Surface Sediments and Manila Clams (Ruditapes philippinarum) from the Dalian Coast, China after the Dalian Port Oil Spill. Biol. Trace Elem. Res. 2012, 149, 241–247. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Li, R.; Chai, M.; Qiu, G. Threat of heavy metal contamination in eight mangrove plants from the Futian mangrove forest, China. Environ. Geochem. Health 2014, 36, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Suñer, M.A.; Devesa, V.; Muñoz, O.; López, F.; Montoro, R.; Arias, A.M.; Blasco, J. Total and inorganic arsenic in the fauna of the Guadalquivir estuary: Environmental and human health implications. Sci. Total Environ. 1999, 242, 261–270. [Google Scholar] [CrossRef]
- Kirby, J.; Maher, W.; Chariton, A.; Krikowa, F. Arsenic concentrations and speciation in a temperate mangrove ecosystem, NSW, Australia. Appl. Organomet. Chem. 2002, 16, 192–201. [Google Scholar] [CrossRef]
- Qiu, Y.W.; Yu, K.F.; Zhang, G.; Wang, W.X. Accumulation and partitioning of seven trace metals in mangroves and sediment cores from three estuarine wetlands of Hainan Island, China. J. Hazard. Mater. 2011, 190, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.R.; Hong, H.L.; Yan, C.L. Arsenic Accumulation and Translocation in Mangrove (Aegiceras corniculatum L.) Grown in Arsenic Contaminated Soils. Int. J. Environ. Res. Public Health 2015, 12, 7244–7253. [Google Scholar] [CrossRef] [PubMed]
- Peshut, P.J.; Morrison, R.J.; Brooks, B.A. Arsenic speciation in marine fish and shellfish from American Samoa. Chemosphere 2008, 71, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Raknuzzaman, M.; Ahmed, M.K.; Islam, M.S.; Habibullah-Al-Mamun, M.; Tokumura, M.; Sekine, M.; Masunaga, S. Trace metal contamination in commercial fish and crustaceans collected from coastal area of Bangladesh and health risk assessment. Environ. Sci. Pollut. Res. 2016, 23, 17298–17310. [Google Scholar] [CrossRef] [PubMed]
- Kabata-pendias, A. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FL, USA, 2010; ISBN 9781420093681. [Google Scholar]
- Wenzel, W.W.; Kirchbaumer, N.; Prohaska, T.; Stingeder, G.; Lombi, E.; Adriano, D.C. Arsenic fractionation in soils using an improved sequential extraction procedure. Anal. Chim. Acta 2001, 436, 309–323. [Google Scholar] [CrossRef]
- Fedotov, P.S.; Fitz, W.J.; Wennrich, R.; Morgenstern, P.; Wenzel, W.W. Fractionation of arsenic in soil and sludge samples: Continuous-flow extraction using rotating coiled columns versus batch sequential extraction. Anal. Chim. Acta 2005, 538, 93–98. [Google Scholar] [CrossRef]
- Hong, K.J.; Tokunaga, S.; Kajiuchi, T. Extraction of heavy metals from MSW incinerator fly ashes by chelating agents. J. Hazard. Mater. 2000, 75, 57–73. [Google Scholar] [CrossRef]
- Im, J.; Yang, K.; Moon, S.; Kim, Y.J.; Nam, K. Role of phosphate and Fe-oxides on the acid-aided extraction efficiency and readsorption of As in field-aged soil. J. Hazard. Mater. 2015, 300, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Luna, J.M.; Rufino, R.D.; Sarubbo, L.A. Biosurfactant from Candida sphaerica UCP0995 exhibiting heavy metal remediation properties. Process Saf. Environ. Prot. 2016, 102, 558–566. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Mukherjee, S.; Hashim, M.A.; Sen Gupta, B. Remediation of Arsenic Contaminated Soil Using Phosphate and Colloidal Gas Aphron Suspensions Produced from Sapindus mukorossi. Bull. Environ. Contam. Toxicol. 2017, 98, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Carson, K.C.; Holliday, S.; Glenn, A.R.; Dilworth, M.J. Siderophore and organic acid production in root nodule bacteria. Arch. Microbiol. 1992, 157, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Reichard, P.U.; Kretzschmar, R.; Kraemer, S.M. Dissolution mechanisms of goethite in the presence of siderophores and organic acids. Geochim. Cosmochim. Acta 2007, 71, 5635–5650. [Google Scholar] [CrossRef]
- Onireti, O.O.; Lin, C.; Qin, J. Combined Effects of Low-molecular-weight Organic Acids on Mobilization of Arsenic and Lead from Multi-contaminated Soils. Chemosphere 2016, 170, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Vítková, M.; Komárek, M.; Tejnecky, V.; Hana, S. Interactions of nano-oxides with low-molecular-weight organic acids in a contaminated soil. J. Hazard. Mater. 2015, 293, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Bostick, B.C.; Mailloux, B.J.; Ross, J.M.; Chillrud, S.N. Effect of oxalic acid treatment on sediment arsenic concentrations and lability under reducing conditions. J. Hazard. Mater. 2016, 311, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.; Chen, J.; Wang, X. Removal of arsenic and cadmium with sequential soil washing techniques using Na2EDTA, oxalic and phosphoric acid: Optimization conditions, removal effectiveness and ecological risks. Chemosphere 2016, 156, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Qiu, R.; Zou, Z.; Zhao, Z.; Zhang, W.; Zhang, T.; Dong, H.; Wei, X. Removal of trace and major metals by soil washing with Na2EDTA and oxalate. J. Soils Sediments 2010, 10, 45–53. [Google Scholar] [CrossRef]
- Isosaari, P.; Sillanpää, M. Effects of oxalate and phosphate on electrokinetic removal of arsenic from mine tailings. Sep. Purif. Technol. 2012, 86, 26–34. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Ure, A.M.; Quevauviller, P.; Muntau, H.; Griepink, B. Speciation of Heavy Metals in Soils and Sediments. An Account of the Improvement and Harmonization of Extraction Techniques Undertaken under the Auspices of the BCR of the Commission of the European Communities. Int. J. Environ. Anal. Chem. 1993, 51, 135–151. [Google Scholar] [CrossRef]
- Sparks, D.L.; Page, A.L.; Helmke, P.A.; Loeppert, R.H. Methods of Soil Analysis Part 3—Chemical Methods; SSSA Book Ser. 5.3; American Society of Agronomy, Soil Science Society of America: Fitchburg, WI, USA, 1996. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Method 3050B: Acid Digenstion of Sediments, Sludges and Soils; USEPA: Washington, DC, USA, 1996.
- Walkley, A.; Black, I.A. An Examination of the Degtjareff Method for Determining Soil Organic Matter, and a Proposed Modification of the Chromic Acid Titration Method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Klute, A. Methods of Soil Analysis: Part 1—Physical and Mineralogical Methods; SSSA Book Ser. 5.1; American Society of Agronomy, Soil Science Society of America: Madison, WI, USA, 1986. [Google Scholar]
- Webster, R. Soil Sampling and Methods of Analysis. Eur. J. Soil Sci. 2008, 59, 3–14. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; The R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- RStudio Team. RStudio: Integrated Development Environment for R. Available online: https://www.rstudio.com/products/rstudio/ (accessed on 29 August 2017).
- Ministry of Natural Resources and Environment of The Socialist Republic of Vietnam (MONRE). National Technical Regulation on the Allowable Limits of Heavy Metals in the Soils; MONRE: Hanoi, Vietnam, 2008. [Google Scholar]
- Lee, J.-C.; Kim, E.J.; Baek, K. Synergistic effects of the combination of oxalate and ascorbate on arsenic extraction from contaminated soils. Chemosphere 2017, 168, 1439–1446. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Jeon, E.-K.; Baek, K. Role of reducing agent in extraction of arsenic and heavy metals from soils by use of EDTA. Chemosphere 2016, 152, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.; Víalakhi, G.; Venkateswaran, G.; Moorthy, P.N. Dissolution of Haematite in Citric Acid-EDTA-Ascorbic Acid Mixtures. J. Nucl. Sci. Technol. 1996, 33, 479–485. [Google Scholar] [CrossRef]
- Fitz, W.J.; Wenzel, W.W. Arsenic transformations in the soil–rhizosphere–plant system: Fundamentals and potential application to phytoremediation. J. Biotechnol. 2002, 99, 259–278. [Google Scholar] [CrossRef]
- Masscheleyn, P.H.; Delaune, R.D.; Patrick, W.H., Jr. Effect of Redox Potential and pH on Arsenic Speciation and Solubility in a Contaminated Soil. Environ. Sci. Technol. 1991, 25, 1414–1419. [Google Scholar] [CrossRef]
- Inskeep, W.P.; McDermott, T.R.; Fendorf, S. Arsenic (V)/(III) cycling in soil and natural waters: Chemical and microbiological processes. In Environmental Chemistry of Arsenic; Marcel Dekker: New York City, NY, USA, 2002; pp. 183–215. [Google Scholar]
- Takahashi, Y.; Minamikawa, R.; Hattori, K.H.; Kurishima, K.; Kihou, N.; Yuita, K. Arsenic Behavior in Paddy Fields during the Cycle of Flooded and Non-flooded Periods. Environ. Sci. Technol. 2004, 38, 1038–1044. [Google Scholar] [CrossRef] [PubMed]
- Roden, E.E.; Urrutia, M.M. Ferrous Iron Removal Promotes Microbial Reduction of Crystalline Iron(III) Oxides. Environ. Sci. Technol. 1999, 33, 1847–1853. [Google Scholar] [CrossRef]
- Mao, X.; Han, F.X.; Shao, X.; Arslan, Z.; McComb, J.; Chang, T.; Guo, K.; Celik, A. Remediation of lead-, arsenic-, and cesium-contaminated soil using consecutive washing enhanced with electro-kinetic field. J. Soils Sediments 2016, 16, 2344–2353. [Google Scholar] [CrossRef]
- Lee, S.O.; Tran, T.; Jung, B.H.; Kim, S.J.; Kim, M.J. Dissolution of iron oxide using oxalic acid. Hydrometallurgy 2007, 87, 91–99. [Google Scholar] [CrossRef]
- Fox, T.R.; Comerford, N.B. Low-Molecular-Weight Organic Acids in Selected Forest Soils of the Southeastern USA. Soil Sci. Soc. Am. J. 1990, 54, 1139–1144. [Google Scholar] [CrossRef]
- Hue, N.V.; Craddock, G.R.; Adams, F. Effect of Organic Acids on Aluminum Toxicity in Subsoils1. Soil Sci. Soc. Am. J. 1986, 50, 28–34. [Google Scholar] [CrossRef]
- Jones, D.L. Organic acids in the rhizosphere—A critical review. Plant Soil 1998, 205, 25–44. [Google Scholar] [CrossRef]
- Panias, D.; Taxiarchou, M.; Paspaliaris, I.; Kontopoulos, A. Mechanisms of dissolution of iron oxides in aqueous oxalic acid solutions. Hydrometallurgy 1996, 42, 257–265. [Google Scholar] [CrossRef]
- Dixit, S.; Hering, J.G. Comparison of Arsenic(V) and Arsenic(III) Sorption onto Iron Oxide Minerals: Implications for Arsenic Mobility. Environ. Sci. Technol. 2003, 37, 4182–4189. [Google Scholar] [CrossRef] [PubMed]
- Martell, A.E.; Smith, R.M. Carboxylic Acids. In Critical Stability Constants: First Supplement; Springer: Boston, MA, USA, 1982; pp. 284–332. ISBN 978-1-4615-6761-5. [Google Scholar]
- Martin, S. Precipitation and Dissolution of Iron and Manganese Oxides. In Environmental Catalysis; CRC Press: Boca Raton, FL, USA, 2005; pp. 61–82. ISBN 978-1-57444-462-9. [Google Scholar]
- Dos Santos Afonso, M.; Morando, P.J.; Blesa, M.A.; Banwart, S.; Stumm, W. The reductive dissolution of iron oxides by ascorbate. J. Colloid Interface Sci. 1990, 138, 74–82. [Google Scholar] [CrossRef]
- Banwart, S.; Davies, S.; Stumm, W. The role of oxalate in accelerating the reductive dissolution of hematite (α-Fe2O3) by ascorbate. Colloids Surf. 1989, 39, 303–309. [Google Scholar] [CrossRef]
- Suter, D.; Banwart, S.; Stumm, W. Dissolution of hydrous iron(III) oxides by reductive mechanisms. Langmuir 1991, 7, 809–813. [Google Scholar] [CrossRef]

| Fraction | Reagents | Extraction Condition | Arsenic Form | Reference |
|---|---|---|---|---|
| F1 | 0.05 M (NH4)2SO4 | 1:25 of soil to solution. 4-h shaking, 20 °C | Non-specifically bound | [23,24,26] |
| F2 | 0.05 M (NH4)H2PO4 | 1:25 of soil to solution. 16-h shaking, 20 °C | Specifically bound | [23,24,26] |
| F3 | 0.2 M NH4-Oxalate, pH = 3.25 | 1:25 of soil to solution. 6-h shaking in the dark, 20 °C | Amorphous iron, aluminum, manganese oxide bound | [23,24,26] |
| F4 | 0.2 M NH4-Oxalate + 0.1 M Ascorbic acid; pH = 3.25 | 1:25 of soil to solution. 30 min occasional agitation, 96 °C | Crystalline iron | [23,24,26] |
| F5 | H2O2 + HNO3 acid, pH = 2, 3.2 M NH4OAc in 20% HNO3 | 3-h occasional agitation at 85 °C | Organic matter and secondary sulfide bound | [37,38,39] |
| Total As | HNO3 acid, H2O2 | Hot-plate, 110 °C | Total concentration of arsenic | [40] |
| F6 | Total As—(F1 + F2 + F3 + F4 + F5) | Residual | ||
| Sample | Land Uses | Latitude | Longitude | pH | OC | CEC | Clay Content | Silt Content | Sand Content | Astot |
|---|---|---|---|---|---|---|---|---|---|---|
| (%) | (cmol kg-1) | (%) | (µg/g) | |||||||
| R | River bed | 20.34 | 106.39 | 6.92 | 1.03 | 13.2 | 5.5 | 49.3 | 45.2 | 31.0 |
| C | Mangrove forest Soil | 20.25 | 106.57 | 6.98 | 0.70 | 11.0 | 18.1 | 47.8 | 34.1 | 27.4 |
| T | Paddy soil | 20.27 | 106.45 | 6.88 | 1.50 | 11.4 | 25.3 | 58.1 | 16.6 | 16.8 |
| S | Aquaculture farm | 20.25 | 106.55 | 6.52 | 1.20 | 16.0 | 26.2 | 44.4 | 29.4 | 17.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen Van, T.; Osanai, Y.; Do Nguyen, H.; Kurosawa, K. Arsenic Speciation and Extraction and the Significance of Biodegradable Acid on Arsenic Removal—An Approach for Remediation of Arsenic-Contaminated Soil. Int. J. Environ. Res. Public Health 2017, 14, 990. https://doi.org/10.3390/ijerph14090990
Nguyen Van T, Osanai Y, Do Nguyen H, Kurosawa K. Arsenic Speciation and Extraction and the Significance of Biodegradable Acid on Arsenic Removal—An Approach for Remediation of Arsenic-Contaminated Soil. International Journal of Environmental Research and Public Health. 2017; 14(9):990. https://doi.org/10.3390/ijerph14090990
Chicago/Turabian StyleNguyen Van, Thinh, Yasuhito Osanai, Hai Do Nguyen, and Kiyoshi Kurosawa. 2017. "Arsenic Speciation and Extraction and the Significance of Biodegradable Acid on Arsenic Removal—An Approach for Remediation of Arsenic-Contaminated Soil" International Journal of Environmental Research and Public Health 14, no. 9: 990. https://doi.org/10.3390/ijerph14090990