Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution
Abstract
:1. Introduction
2. Results and Discussion
2.1. Wear-Corrosion Tests
2.2. Macrophage Cell Response
3. Materials and Methods
3.1. Material
3.2. Wear-Corrosion Tests under Electrochemical Control
3.3. Isolation and Characterization of Particles
3.4. Macrophages Cell Cultures Assays
3.5. Mitochondrial Activity Measurement
3.6. Measurement of Lactate Dehydrogenase Activity
3.7. Statistical Analysis of Data
3.7.1. Wear Particles Analysis Data
3.7.2. Biocompatibility Analysis Data
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fujiwara, N.; Kobayashi, K. Macrophages in Inflammation in Current Drug Targets—Inflammation & Allergy; Zaenker, K.S., Ed.; Bentham Science Publisher: Berlin, Germany, 2005; pp. 281–286. [Google Scholar] [CrossRef]
- Poggio, C.E. Plasmacytoma of the mandible associated with a dental implant failure: A clinical report. Clin. Oral Implant. Res. 2007, 18, 540–543. [Google Scholar] [CrossRef] [PubMed]
- McGuff, H.S.; Heim-Hall, J.; Holsinger, F.C.; Jones, A.A.; O’Dell, D.S.; Hafemeister, A.C. Maxillary osteosarcoma associated with a dental implant: Report of a case and review of the literature regarding implant-related sarcomas. J. Am. Dent. Assoc. 2008, 139, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Ferrier, J.; Ross, S.M.; Kanehisa, J.; Aubin, J.E. Osteoclasts and osteoblasts migrate in opposite directions in response to a constant electrical-field. J. Cell. Physiol. 1986, 129, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.; Thorlin, T.; Robinson, K.R.; Nogi, T.; Mercola, M. Asymmetries in H+/K+-ATPase and cell membrane potentials comprise a very early step in left-right patterning. Cell 2002, 111, 77–89. [Google Scholar] [CrossRef]
- Becker, R.O.; Spadaro, J.A.; Marino, A.A. Clinical experiences with low intensity direct-current stimulation of bone-growth. Clin. Orthop. Relat. Res. 1977, 124, 75–83. [Google Scholar] [CrossRef]
- Levin, M. Large-scale biophysics: Ion flows and regeneration. Trends Cell Biol. 2007, 17, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Lokietek, W.; Pawluk, R.J.; Bassett, C.A. Muscle injury potentials source of voltage in undeformed rabbit tibia. J. Bone Jt. Surg. Br. 1974, 56, 361–369. [Google Scholar] [CrossRef]
- McCaig, C.D.; Rajnicek, A.M.; Song, B.; Zhao, M. Controlling cell behavior electrically: Current views and future potential. Physiol. Rev. 2005, 85, 943–978. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Olivares-Navarrete, R.; Tannenbaum, R.; Boyan, B.D.; Schwartz, Z. Electrical implications of corrosion for osseointegration of titanium implants. J. Dent. Res. 2011, 90, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Faes, T.J.; van der Meij, H.A.; de Munck, J.C.; Heethaar, R.M. The electric resistivity of human tissues (100 Hz–10 MHz): A meta-analysis of review studies. Physiol. Meas. 1999, 20, R1–R10. [Google Scholar] [CrossRef] [PubMed]
- Borgens, R.B.; Jaffe, L.F.; Cohen, M.J. Large and persistent electrical currents enter the transected lamprey spinal-cord. Proc. Natl. Acad. Sci. USA 1980, 77, 1209–1213. [Google Scholar] [CrossRef] [PubMed]
- Fukada, E.; Yasuda, I. On the piezoelectric effect of bone. J. Phys. Soc. Jpn. 1957, 12, 1158–1162. [Google Scholar] [CrossRef]
- Guzelsu, N.; Demiray, H. Electro-mechanical properties and related models of bone tissues—Review. Int. J. Eng. Sci. 1979, 17, 813–851. [Google Scholar] [CrossRef]
- Rubinacci, A.; Black, J.; Brighton, C.T.; Friedenberg, Z.B. Changes in bioelectric potentials on bone associated with direct-current stimulation of osteogenesis. J. Orthop. Res. 1988, 6, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Díaz, I.; Martínez-Lerma, J.F.; Montoya, R.; Llorente, I.; Escudero, M.L.; García-Alonso, M.C. Study of overall and local electrochemical responses of oxide films grown on CoCr alloy under biological environments. Bioelectrochemistry 2017, 115, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hui, A.Y.; McCarty, W.J.; Masuda, K.; Firestein, G.S.; Sah, R.L. A systems biology approach to synovial joint lubrication in health, injury, and disease. Wiley Interdiscip. Rev. Syst. Biol. Med. 2012, 4, 15–37. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.W.E.; Kurz, S.; Virtanen, S.; Fervel, V.; Olsson, C.-O.A.; Mischler, S. Passive and transpassive behavior of CoCrMo in simulated biological solutions. Electrochim. Acta 2004, 49, 2167–2178. [Google Scholar] [CrossRef]
- Bitar, D.; Parvizi, J. Biological response to prosthetic debris. World J. Orthop. 2015, 6, 172–189. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.J.; Roebuck, K.A.; Archibeck, M.; Hallab, N.J.; Glant, T.T. Osteolysis: Basic science. Clin. Orthop. Relat. Res. 2001, 393, 71–77. [Google Scholar] [CrossRef]
- Dorr, L.D.; Bloebaum, R.; Emmanual, J.; Meldrum, R. Histologic, biochemical, and ion analysis of tissue and fluids retrieved during total hip-arthroplasty. Clin. Orthop. Relat. Res. 1990, 261, 82–95. [Google Scholar] [CrossRef]
- Jacobs, J.J.; Urban, R.M.; Hallab, N.J.; Skipor, A.K.; Fischer, A.; Wimmer, M.A. Metal-on-metal bearing surfaces. J. Am. Acad. Orthop. Surg. 2009, 17, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Ponthiaux, P.; Wenger, F.; Drees, D.; Celis, J.P. Electrochemical techniques for studying tribocorrosion processes. Wear 2004, 256, 459–468. [Google Scholar] [CrossRef]
- Yan, Y.; Yang, H.; Su, Y.; Qiao, L. Study of the tribocorrosion behaviors of albumin on a cobalt-based alloy using scanning Kelvin probe force microscopy and atomic force microscopy. Electrochem. Commun. 2016, 64, 61–64. [Google Scholar] [CrossRef]
- Uesaka, S.; Miyazaki, K.; Ito, H. Age-related changes and sex differences in chondroitin sulfate isomers and hyaluronic acid in normal synovial fluid. Mod. Rheumatol. 2004, 14, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Igual-Muñoz, A.; Mischler, S. Effect of the environment on wear ranking and corrosion of biomedical CoCrMo alloys. J. Mater. Sci. Mater. Med. 2011, 22, 437–450. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.C.; Kilburn, M.R.; Heard, P.J.; Scott, T.B.; Hallam, K.R.; Allen, G.C.; Learmonth, I.D. The Entrapment of Corrosion Products from CoCr Implant Alloys in the Deposits of Calcium Phosphate: A Comparison of Serum, Synovial Fluid, Albumin, EDTA, and Water. J. Orthop. Res. 2006, 24, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Man, K.; Jiang, L.-H.; Foster, R.; Yang, X.B. Immunological response to total hip arthroplasty. J. Funct. Biomater. 2017, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R.M.; Pérez-Maceda, B.T.; Carboneras, M.; Onofre-Bustamante, E.; García-Alonso, M.C.; Escudero, M.L. Response of MC3T3-E1 osteoblasts, L929 fibroblasts and J774 macrophages to fluoride surface-modified AZ31 magnesium alloy. J. Biomed. Mater. Res. Part A 2013, 101, 2753–2762. [Google Scholar] [CrossRef] [PubMed]
- VanOs, R.; Lildhar, L.L.; Lehoux, E.A.; Beaulé, P.E.; Catelas, I. In vitro macrophage response to nanometer-size chromium oxide particles. J. Biomed. Mater. Res. Part B 2014, 102B, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Billi, F.; Benya, P.; Kavanaugh, A.; Adams, J.; McKellop, H.; Ebramzadeh, E. The John Charnley Award: An accurate and extremely sensitive method to separate, display, and characterize wear debris: Part 2: Metal and ceramic particles. Clin. Orthop. Relat. Res. 2012, 470, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Kavanaugh, A.E.; Benya, P.; Billi, F. A Method to Isolate and Characterize Wear Debris from Synovial Fluid and Tissues. In Metal-On-Metal Total Hip Replacement Devices; ASTM International: West Conshohocken, PA, USA, 2013. [Google Scholar] [CrossRef]
- Wang, J.; Witte, F.; Xi, T.; Zheng, Y.; Yang, K.; Yang, Y.; Zhao, D.; Meng, J.; Li, Y.; Li, W.; et al. Recommendation for modifying current cytotoxicity testing standards for biodegradable magnesium-based materials. Acta Biomater. 2015, 21, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Kruskal, W.H.; Wallis, W.A. Use of Ranks in One-Criterion Variance Analysis. J. Am. Stat. Assoc. 1952, 47, 583–621. [Google Scholar] [CrossRef]
- Mann, H.B.; Whitney, D.R. On a Test of Whether one of Two Random Variables is Stochastically Larger than the Other. Ann. Math. Stat. 1947, 18, 50–60. [Google Scholar] [CrossRef]
- Minitab Inc. Minitab Statistical Software. (n.d.). Available online: www.minitab.com/en-us/ (accessed on 16 April 2018).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 16 April 2018).







| Condition Number | n | Mean | SD | Minimum | Maximum | C25 | Median | C75 | p * | |
|---|---|---|---|---|---|---|---|---|---|---|
| Cr | 1 | 7 | 33.28 | 6.92 | 21.61 | 43.13 | 29.33 | 34.38 | 38.45 | |
| 2 | 24 | 28.53 | 8.73 | 14.06 | 48.82 | 21.28 | 27.52 | 33.66 | 0.034 | |
| 3 | 7 | 39.09 | 11.02 | 22.34 | 57.01 | 33.65 | 37.60 | 47.67 | ||
| Co | 1 | 7 | 4.31 | 6.02 | 0.00 | 13.81 | 0.00 | 0.00 | 11.54 | |
| 2 | 24 | 7.73 | 16.94 | 0.00 | 54.07 | 0.00 | 0.00 | 5.99 | 0.001 | |
| 3 | 7 | 36.08 | 17.59 | 15.14 | 58.64 | 17.57 | 32.12 | 51.48 | ||
| Mo | 1 | 7 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |
| 2 | 24 | 0.45 | 1.54 | 0.00 | 5.63 | 0.00 | 0.00 | 0.00 | 0.006 | |
| 3 | 7 | 2.71 | 2.60 | 0.00 | 5.48 | 0.00 | 4.00 | 5.45 | ||
| P | 1 | 7 | 18.59 | 5.02 | 11.46 | 26.36 | 15.76 | 16.87 | 23.16 | |
| 2 | 24 | 14.11 | 5.90 | 0.96 | 27.48 | 11.64 | 13.24 | 17.83 | 0.051 | |
| 3 | 7 | 9.40 | 7.30 | 1.30 | 19.57 | 1.89 | 8.79 | 16.10 | ||
| Al | 1 | 7 | 1.52 | 4.01 | 0.00 | 10.62 | 0.00 | 0.00 | 0.00 | |
| 2 | 24 | 0.18 | 0.68 | 0.00 | 3.17 | 0.00 | 0.00 | 0.00 | 0.123 | |
| 3 | 7 | 0.76 | 1.06 | 0.00 | 2.57 | 0.00 | 0.00 | 1.85 | ||
| O | 1 | 7 | 40.10 | 10.67 | 23.79 | 54.36 | 33.72 | 41.31 | 51.57 | |
| 2 | 24 | 41.82 | 17.72 | 3.52 | 72.84 | 34.64 | 49.54 | 51.69 | 0.002 | |
| 3 | 7 | 10.91 | 7.03 | 3.76 | 23.11 | 4.83 | 8.28 | 16.05 | ||
| C | 1 | 7 | 2.19 | 5.80 | 0.00 | 15.35 | 0.00 | 0.00 | 0.00 | |
| 2 | 24 | 6.98 | 7.49 | 0.00 | 19.94 | 0.00 | 6.39 | 14.16 | 0.022 | |
| 3 | 7 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Comparison between Pairwise | |||
|---|---|---|---|
| p ** 1 vs. 2 | p ** 1 vs. 3 | p ** 2 vs. 3 | |
| Cr | 0.119 | 0.277 | 0.021 |
| Co | 0.556 | 0.002 | 0.001 |
| Mo | 0.438 | 0.025 | 0.008 |
| P | - | - | - |
| Al | - | - | - |
| O | 0.508 | 0.002 | 0.002 |
| C | 0.098 | 0.317 | 0.017 |
| (a) | ||
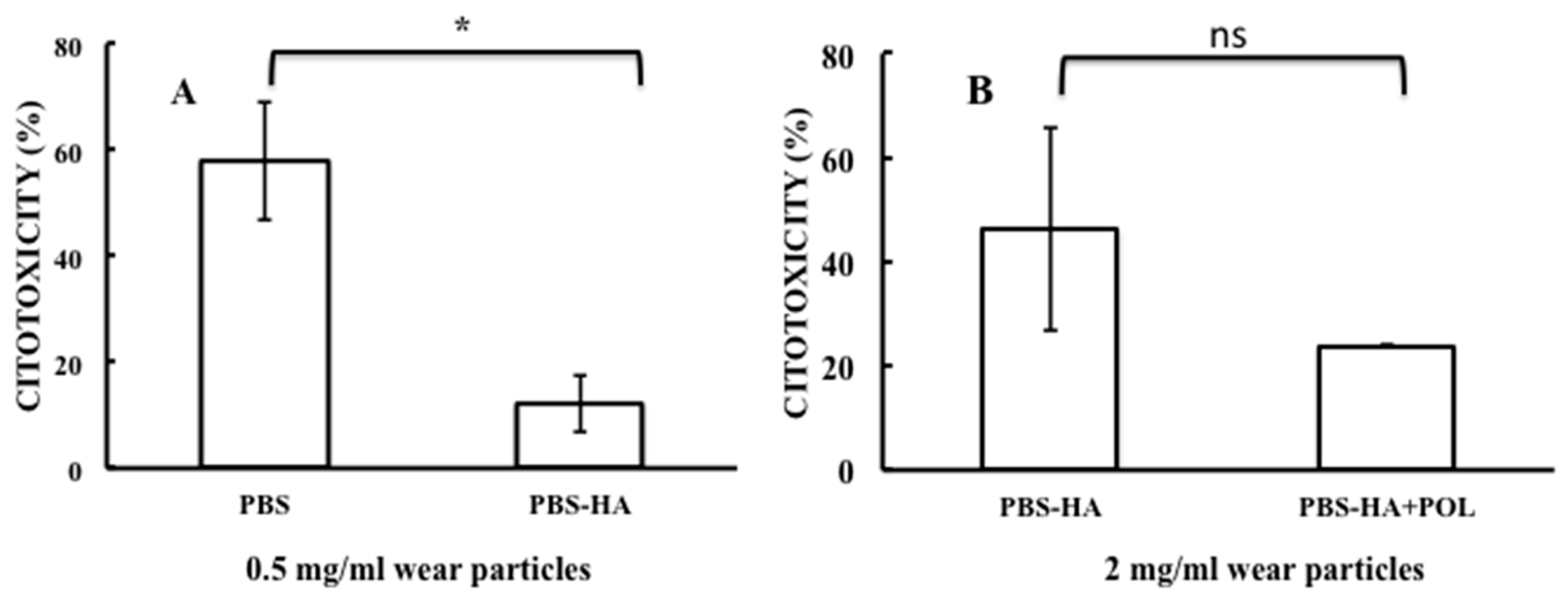
| P2 (0.5 mg/mL) vs. P3 (0.5 mg/mL) | ||
| Mean of P2 | Mean of P3 | P Value |
| 57.84 | 12.24 | 0.015 |
| (b) | ||
| P3 (2 mg/mL) vs. P6 (2 mg/mL) | ||
| Mean of P3 (2 mg/mL) | Mean of P6 (2 mg/mL) | P Value |
| 46.18 | 23.9 | 0.248 |
| ANOVA | ||||
|---|---|---|---|---|
| Sum Sq | Df | F Value | Pr (>F) | |
| P | 5745.2 | 1 | 266.75 | 4.64 × 10−9 |
| conc | 2191.39 | 2 | 50.87 | 2.76 × 10−6 |
| P × conc | 2648.33 | 2 | 61.48 | 1.07 × 10−6 |
| Residuals | 236.91 | 11 | - | - |
| P | Conc | Mean | Lower.CL | Upper.CL | Group |
|---|---|---|---|---|---|
| P3 | 2 | 25.7 | 17.1 | 34.3 | a |
| P3 | 1 | 60.9 | 52.3 | 69.4 | b |
| P3 | 0.5 | 87.5 | 77 | 98 | c |
| P6 | 1 | 89.8 | 81.3 | 98.4 | c |
| P6 | 2 | 94.3 | 85.8 | 102.9 | c |
| P6 | 0.5 | 95 | 86.4 | 103.5 | c |
| C | Co | Cr | Mo | Ni | S | P | Al | W | Mn | Fe | Si | N | Ti | Cu | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HC | 0.22 | 62 | 29.4 | 6.4 | 0.1 | 0.004 | 0.001 | 0.01 | 0.03 | 0.7 | 0.16 | 0.7 | 0.16 | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perez-Maceda, B.T.; López-Fernández, M.E.; Díaz, I.; Kavanaugh, A.; Billi, F.; Escudero, M.L.; García-Alonso, M.C.; Lozano, R.M. Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution. Materials 2018, 11, 756. https://doi.org/10.3390/ma11050756
Perez-Maceda BT, López-Fernández ME, Díaz I, Kavanaugh A, Billi F, Escudero ML, García-Alonso MC, Lozano RM. Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution. Materials. 2018; 11(5):756. https://doi.org/10.3390/ma11050756
Chicago/Turabian StylePerez-Maceda, Blanca Teresa, María Encarnación López-Fernández, Iván Díaz, Aaron Kavanaugh, Fabrizio Billi, María Lorenza Escudero, María Cristina García-Alonso, and Rosa María Lozano. 2018. "Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution" Materials 11, no. 5: 756. https://doi.org/10.3390/ma11050756
APA StylePerez-Maceda, B. T., López-Fernández, M. E., Díaz, I., Kavanaugh, A., Billi, F., Escudero, M. L., García-Alonso, M. C., & Lozano, R. M. (2018). Macrophage Biocompatibility of CoCr Wear Particles Produced under Polarization in Hyaluronic Acid Aqueous Solution. Materials, 11(5), 756. https://doi.org/10.3390/ma11050756





