Analysis of the Vigor of Pinus hartwegii Lindl. along an Altitudinal Gradient Using UAV Multispectral Images: Evidence of Forest Decline Possibly Associated with Climatic Change
Abstract
:1. Introduction
2. Materials and Methods
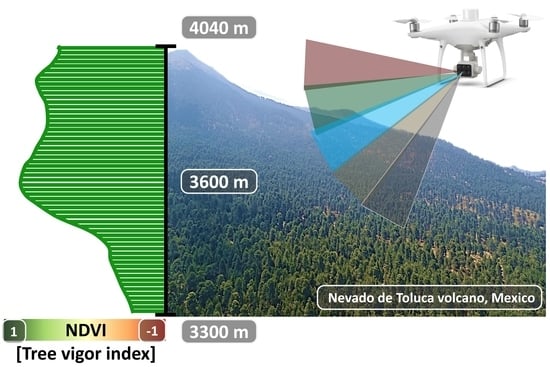
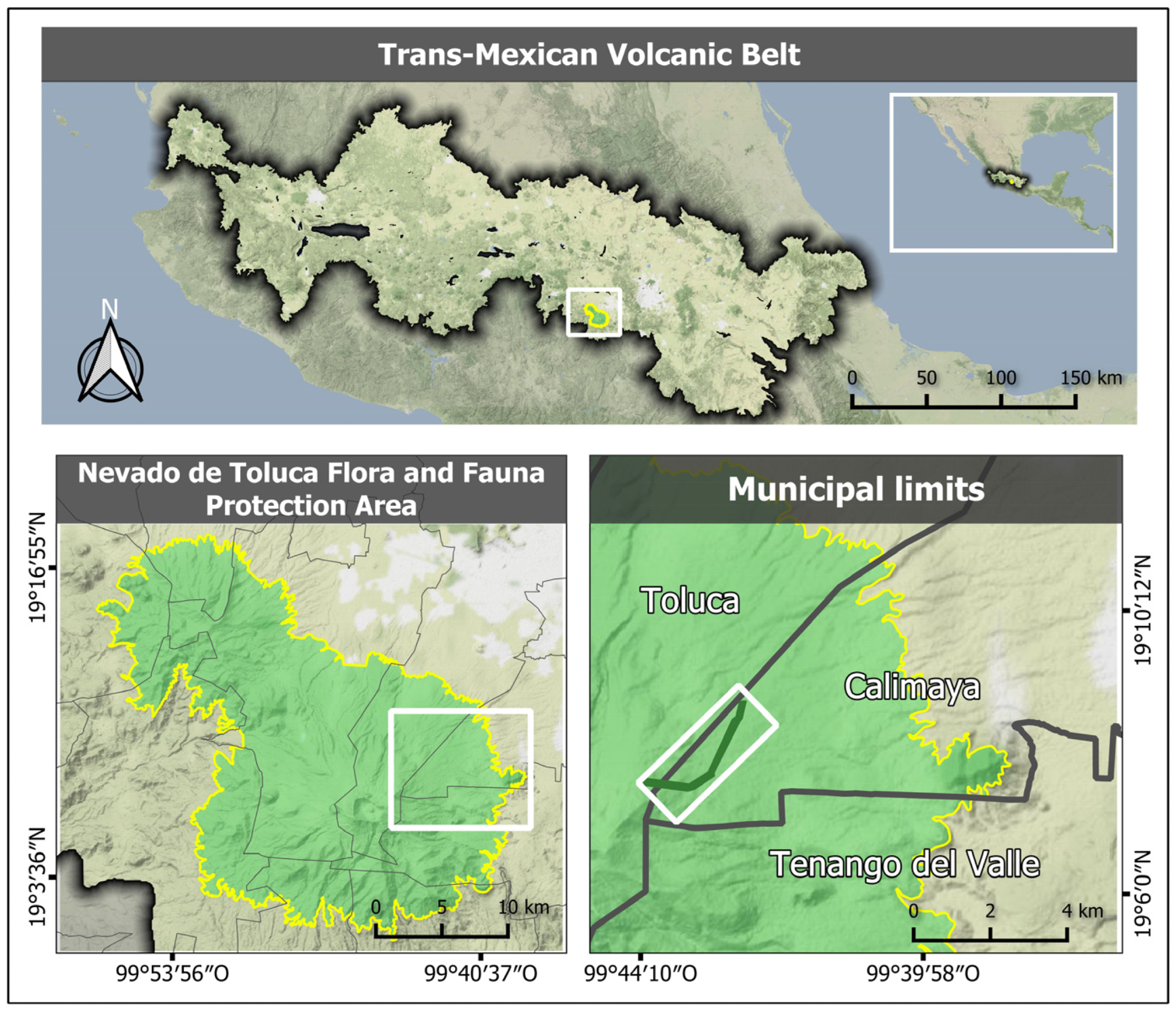
2.1. Study Area
2.2. UAV Platform Equipped with a Multispectral Camera
2.3. Planning and Execution of Flights along the Altitudinal Gradient
2.4. Photogrammetric Processing of UAV Images
2.5. Calculation of Vegetation Indices
2.6. Detection and Extraction of Information at Individual Tree Level
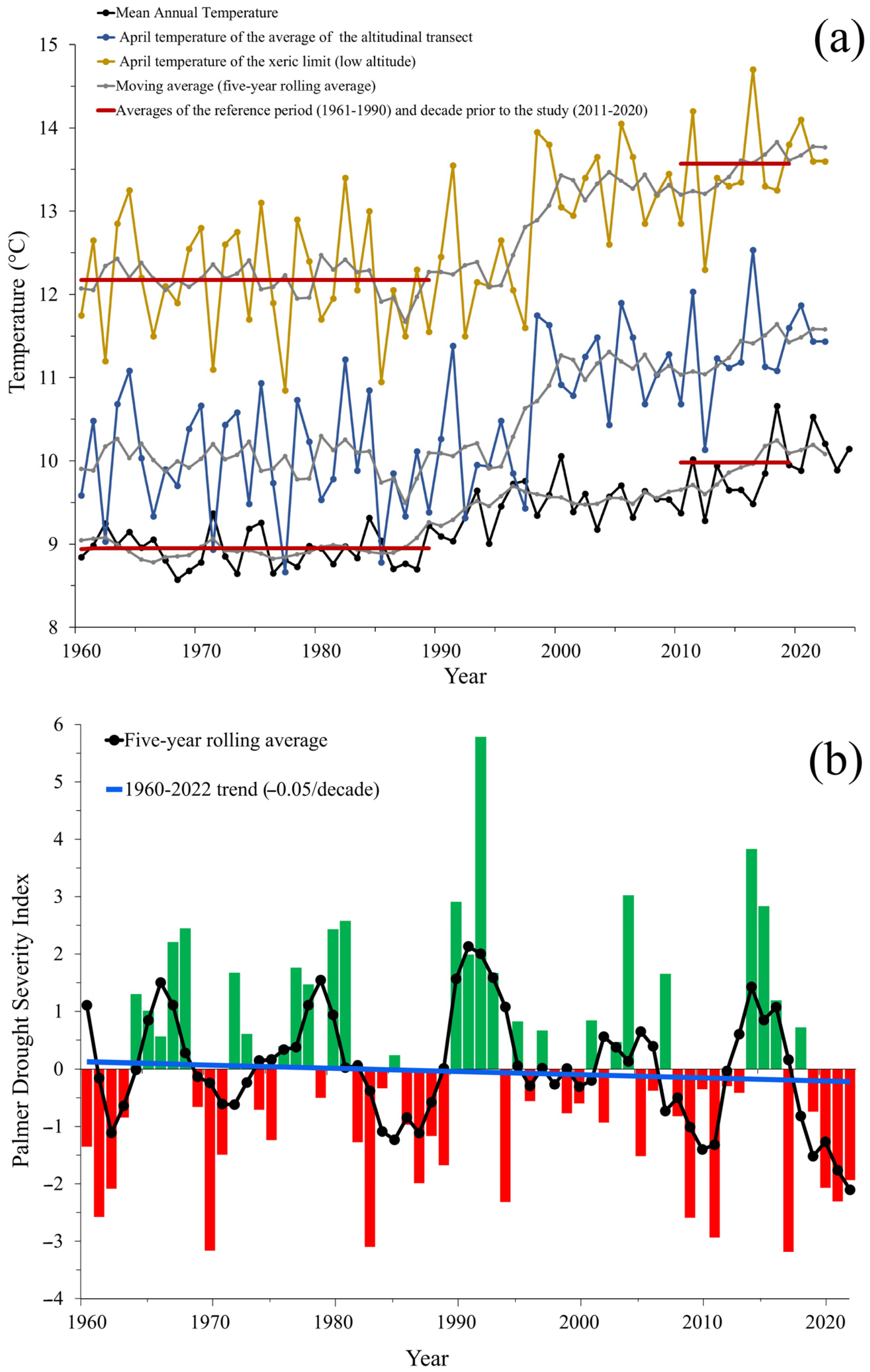
2.7. Climate Change Time-Series Analyses
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lauer, W. The Altitudinal Belts of the Vegetation in the Central Mexican Highlands and Their Climatic Conditions. Arct. Alp. Res. 1973, 5, A99–A113. [Google Scholar] [CrossRef]
- Lauer, W.; Klaus, D. Geoecological Investigations on the Timberline of Pico De Orizaba, Mexico. Arct. Alp. Res. 1975, 7, 315–330. [Google Scholar] [CrossRef]
- Viveros-Viveros, H.; Sáenz-Romero, C.; Vargas-Hernández, J.J.; López-Upton, J.; Ramírez-Valverde, G.; Santacruz-Varela, A. Altitudinal Genetic Variation in Pinus hartwegii Lindl. I: Height Growth, Shoot Phenology, and Frost Damage in Seedlings. For. Ecol. Manag. 2009, 257, 836–842. [Google Scholar] [CrossRef]
- Viveros-Viveros, H.; Sáenz-Romero, C.; López-Upton, J.; Vargas-Hernández, J.J. Growth and Frost Damage Variation among Pinus pseudostrobus, P. montezumae and P. hartwegii Tested in Michoacán, México. For. Ecol. Manag. 2007, 253, 81–88. [Google Scholar] [CrossRef]
- Iglesias, L.G.; Solís-Ramos, L.Y.; Viveros-Viveros, H. Variación Morfométrica En Dos Poblaciones Naturales de Pinus hartwegii Lindl. Del Estado de Veracruz. Phyton 2012, 81, 239–246. [Google Scholar]
- Alfaro-Ramírez, F.U.; Ramírez-Albores, J.E.; Vargas-Hernández, J.J.; Franco-Maass, S.; Pérez-Suárez, M. Potential Reduction of Hartweg´s Pine (Pinus hartwegii Lindl.) Geographic Distribution. PLoS ONE 2020, 15, e0229178. [Google Scholar] [CrossRef] [Green Version]
- Malhi, Y.; Franklin, J.; Seddon, N.; Solan, M.; Turner, M.G.; Field, C.B.; Knowlton, N. Climate Change and Ecosystems: Threats, Opportunities and Solutions. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190104. [Google Scholar] [CrossRef] [Green Version]
- Ripple, W.J.; Wolf, C.; Newsome, T.M.; Barnard, P.; Moomaw, W.R. World Scientists’ Warning of a Climate Emergency. Bioscience 2020, 70, 8–12. [Google Scholar] [CrossRef]
- Beniston, M. Climatic Change in Mountain Regions: A Review of Possible Impacts. Clim. Change 2003, 59, 5–31. [Google Scholar] [CrossRef]
- Fort, M. Impact of Climate Change on Mountain Environment Dynamics: An Introduction. J. Alp. Res. 2015, 103, 2–7. [Google Scholar] [CrossRef] [Green Version]
- Sáenz-Romero, C.; Mendoza-Maya, E.; Gómez-Pineda, E.; Blanco-García, A.; Endara-Agramont, A.R.; Lindig-Cisneros, R.; López-Upton, J.; Trejo-Ramírez, O.; Wehenkel, C.; Cibrián-Tovar, D.; et al. Recent Evidence of Mexican Temperate Forest Decline and the Need for Ex Situ Conservation, Assisted Migration, and Translocation of Species Ensembles as Adaptive Management to Face Projected Climatic Change Impacts in a Megadiverse Country. Can. J. For. Res. 2020, 50, 843–854. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Z.; Wang, Y.; Zhang, Y.; Shen, J.; Qin, D.; Li, S. Trade-off Analyses of Multiple Mountain Ecosystem Services along Elevation, Vegetation Cover and Precipitation Gradients: A Case Study in the Taihang Mountains. Ecol. Indic. 2019, 103, 94–104. [Google Scholar] [CrossRef]
- Sáenz-Romero, C.; Rehfeldt, G.E.; Crookston, N.L.; Duval, P.; St-Amant, R.; Beaulieu, J.; Richardson, B.A. Spline Models of Contemporary, 2030, 2060 and 2090 Climates for Mexico and Their Use in Understanding Climate-Change Impacts on the Vegetation. Clim. Change 2010, 102, 595–623. [Google Scholar] [CrossRef]
- Hammond, W.M.; Williams, A.P.; Abatzoglou, J.T.; Adams, H.D.; Klein, T.; López, R.; Sáenz-Romero, C.; Hartmann, H.; Breshears, D.D.; Allen, C.D. Global Field Observations of Tree Die-off Reveal Hotter-Drought Fingerprint for Earth’s Forests. Nat. Commun. 2022, 13, 1761. [Google Scholar] [CrossRef]
- Forzieri, G.; Dakos, V.; McDowell, N.G.; Ramdane, A.; Cescatti, A. Emerging Signals of Declining Forest Resilience under Climate Change. Nature 2022, 608, 534–539. [Google Scholar] [CrossRef]
- Stevens, M. 102 Million Dead California Trees ‘Unprecedented in Our Modern History’, Officials Say. Los Angeles Times. 2016. Available online: https://www.latimes.com/local/lanow/la-me-dead-trees-20161118-story.html (accessed on 3 April 2023).
- Jay, L.; Josue, M.-A.; John, D.; Kathleen, S. Lessons from California’s 2012–2016 Drought. J. Water Resour. Plan Manag. 2018, 144, 04018067. [Google Scholar] [CrossRef] [Green Version]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.T.; et al. A Global Overview of Drought and Heat-Induced Tree Mortality Reveals Emerging Climate Change Risks for Forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef] [Green Version]
- Mátyás, C. Forecasts Needed for Retreating Forests. Nature 2010, 464, 1271. [Google Scholar] [CrossRef]
- Czúcz, B.; Gálhidy, L.; Mátyás, C. Present and Forecasted Xeric Climatic Limits of Beech and Sessile Oak Distribution at Low Altitudes in Central Europe. Ann. For. Sci. 2011, 68, 99–108. [Google Scholar] [CrossRef] [Green Version]
- Stojanović, D.B.; Kržič, A.; Matović, B.; Orlović, S.; Duputie, A.; Djurdjević, V.; Galić, Z.; Stojnić, S. Prediction of the European Beech (Fagus Sylvatica L.) Xeric Limit Using a Regional Climate Model: An Example from Southeast Europe. Agric. For. Meteorol. 2013, 176, 94–103. [Google Scholar] [CrossRef]
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. On Underestimation of Global Vulnerability to Tree Mortality and Forest Die-off from Hotter Drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Huang, C.; Anderegg, W.R.L.; Asner, G.P. Remote Sensing of Forest Die-off in the Anthropocene: From Plant Ecophysiology to Canopy Structure. Remote Sens. Environ. 2019, 231, 111233. [Google Scholar] [CrossRef]
- Gallardo-Salazar, J.L.; Sáenz-Romero, C.; Lindig-Cisneros, R.; López-Toledo, L.; Blanco-García, J.A.; Endara-Agramont, Á.R. Three Decades of Remote Sensing Analysis of Forest Decline Related to Climate Change: A Bibliometric Study. Cuad. Investig. Geográfica 2023. [Google Scholar] [CrossRef]
- Paneque-Gálvez, J.; McCall, M.; Napoletano, B.; Wich, S.; Koh, L. Small Drones for Community-Based Forest Monitoring: An Assessment of Their Feasibility and Potential in Tropical Areas. Forests 2014, 5, 1481–1507. [Google Scholar] [CrossRef] [Green Version]
- Torresan, C.; Berton, A.; Carotenuto, F.; Di Gennaro, S.F.; Gioli, B.; Matese, A.; Miglietta, F.; Vagnoli, C.; Zaldei, A.; Wallace, L. Forestry Applications of UAVs in Europe: A Review. Int. J. Remote Sens. 2017, 38, 2427–2447. [Google Scholar] [CrossRef]
- Gallardo-Salazar, J.L.; Carrillo-Aguilar, D.M.; Pompa-García, M.; Aguirre-Salado, C.A. Multispectral Indices and Individual-Tree Level Attributes Explain Forest Productivity in a Pine Clonal Orchard of Northern Mexico. Geocarto Int. 2021, 37, 4441–4453. [Google Scholar] [CrossRef]
- Dash, J.P.; Watt, M.S.; Pearse, G.D.; Heaphy, M.; Dungey, H.S. Assessing Very High Resolution UAV Imagery for Monitoring Forest Health during a Simulated Disease Outbreak. ISPRS J. Photogramm. Remote Sens. 2017, 131, 1–14. [Google Scholar] [CrossRef]
- Näsi, R.; Honkavaara, E.; Lyytikäinen-Saarenmaa, P.; Blomqvist, M.; Litkey, P.; Hakala, T.; Viljanen, N.; Kantola, T.; Tanhuanpää, T.; Holopainen, M. Using UAV-Based Photogrammetry and Hyperspectral Imaging for Mapping Bark Beetle Damage at Tree-Level. Remote Sens. 2015, 7, 15467–15493. [Google Scholar] [CrossRef] [Green Version]
- Brovkina, O.; Cienciala, E.; Surový, P.; Janata, P. Unmanned Aerial Vehicles (UAV) for Assessment of Qualitative Classification of Norway Spruce in Temperate Forest Stands. Geo-Spat. Inf. Sci. 2018, 21, 12–20. [Google Scholar] [CrossRef] [Green Version]
- Lin, Q.; Huang, H.; Wang, J.; Huang, K.; Liu, Y. Detection of Pine Shoot Beetle (PSB) Stress on Pine Forests at Individual Tree Level Using UAV-Based Hyperspectral Imagery and Lidar. Remote Sens. 2019, 11, 2540. [Google Scholar] [CrossRef] [Green Version]
- De Castro, A.I.; Shi, Y.; Maja, J.M.; Peña, J.M. UAVs for Vegetation Monitoring: Overview and Recent Scientific Contributions. Remote Sens. 2021, 13, 2139. [Google Scholar] [CrossRef]
- Kopačková-Strnadová, V.; Koucká, L.; Jelének, J.; Lhotáková, Z.; Oulehle, F. Canopy Top, Height and Photosynthetic Pigment Estimation Using Parrot Sequoia Multispectral Imagery and the Unmanned Aerial Vehicle (UAV). Remote Sens. 2021, 13, 705. [Google Scholar] [CrossRef]
- Ecke, S.; Dempewolf, J.; Frey, J.; Schwaller, A.; Endres, E.; Klemmt, H.-J.; Tiede, D.; Seifert, T. UAV-Based Forest Health Monitoring: A Systematic Review. Remote Sens. 2022, 14, 3205. [Google Scholar] [CrossRef]
- Eitel, J.U.H.; Vierling, L.A.; Litvak, M.E.; Long, D.S.; Schulthess, U.; Ager, A.A.; Krofcheck, D.J.; Stoscheck, L. Broadband, Red-Edge Information from Satellites Improves Early Stress Detection in a New Mexico Conifer Woodland. Remote Sens. Envrion. 2011, 115, 3640–3646. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Camino, C.; Beck, P.S.A.; Calderon, R.; Hornero, A.; Hernández-Clemente, R.; Kattenborn, T.; Montes-Borrego, M.; Susca, L.; Morelli, M.; et al. Previsual Symptoms of Xylella Fastidiosa Infection Revealed in Spectral Plant-Trait Alterations. Nat. Plants 2018, 4, 432–439. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.; Tang, L.; Hupy, J.P.; Wang, Y.; Shao, G. A Commentary Review on the Use of Normalized Difference Vegetation Index (NDVI) in the Era of Popular Remote Sensing. J. For. Res. 2021, 32, 1–6. [Google Scholar] [CrossRef]
- Datt, B. A New Reflectance Index for Remote Sensing of Chlorophyll Content in Higher Plants: Tests Using Eucalyptus Leaves. J. Plant Physiol. 1999, 154, 30–36. [Google Scholar] [CrossRef]
- Mejía Canales, A.; Franco-Maass, S.; Endara Agramont, A.R.; Ávila Akerberg, V. Caracterización Del Sotobosque En Bosques Densos de Pino y Oyamel En El Nevado de Toluca, México. Madera Bosques 2018, 24. [Google Scholar] [CrossRef]
- Chávez-Aguilar, G.; Campos-Ángeles, G.V.; Pérez-Suárez, M. Estructura y Composición Del Bosque de Pinus hartwegii Lindl. En Su Distribución Altitudinal En El Nevado de Toluca. Rev. Mex. Cienc. For. 2022, 13, 54–76. [Google Scholar] [CrossRef]
- Alfaro-Ramírez, F.U.; Arredondo-Moreno, J.T.; Pérez-Suárez, M.; Endara-Agramont, Á.R. Pinus hartwegii Lindl. Treeline Ecotone: Structure and Altitudinal Limits at Nevado de Toluca, Mexico. Rev. Chapingo Ser. Cienc. For. Ambiente 2017, 23, 261–273. [Google Scholar] [CrossRef]
- Pérez-Suárez, M.; Ramírez-Albores, J.E.; Vargas-Hernández, J.J.; Alfaro-Ramírez, F.U. A Review of the Knowledge of Hartwegʼs Pine (Pinus hartwegii Lindl.): Current Situation and the Need for Improved Future Projections. Trees 2022, 36, 25–37. [Google Scholar] [CrossRef]
- Gómez-Pineda, E.; Sáenz-Romero, C.; Ortega-Rodríguez, J.M.; Blanco-García, A.; Madrigal-Sánchez, X.; Lindig-Cisneros, R.; Lopez-Toledo, L.; Pedraza-Santos, M.E.; Rehfeldt, G.E. Suitable Climatic Habitat Changes for Mexican Conifers along Altitudinal Gradients under Climatic Change Scenarios. Ecol. Appl. 2020, 30, e02041. [Google Scholar] [CrossRef]
- Loya-Rebollar, E.; Sáenz-Romero, C.; Lindig-Cisneros, R.A.; Lobit, P.; Villegas-Moreno, J.A.; Sánchez-Vargas, N.M. Clinal Variation in Pinus hartwegii Populations and Its Application for Adaptation to Climate Change. Silvae Genet. 2013, 62, 86–95. [Google Scholar] [CrossRef] [Green Version]
- Gallardo-Salazar, J.L.; Pompa-García, M.; Aguirre-Salado, C.; López-Serrano, P.; Meléndez-Soto, A. Drones: Technology with a Promising Future in Forest Management. Rev. Mex. For. Cienc. 2020, 11. [Google Scholar] [CrossRef]
- CONANP Programa de Manejo Área de Protección de Flora y Fauna Nevado de Toluca. Available online: https://simec.conanp.gob.mx/pdf_libro_pm/104_libro_pm.pdf (accessed on 2 April 2022).
- Tapia-Vázquez, I.; Sánchez-Cruz, R.; Arroyo-Domínguez, M.; Lira-Ruan, V.; Sánchez-Reyes, A.; del Rayo Sánchez-Carbente, M.; Padilla-Chacón, D.; Batista-García, R.A.; Folch-Mallol, J.L. Isolation and Characterization of Psychrophilic and Psychrotolerant Plant-Growth Promoting Microorganisms from a High-Altitude Volcano Crater in Mexico. Microbiol. Res. 2020, 232, 126394. [Google Scholar] [CrossRef]
- INEGI Cartografía Edafológica Temática, Escala 1:250,000. Available online: https://www.inegi.org.mx/temas/edafologia/ (accessed on 3 April 2023).
- Carlón Allende, T.; Villanueva Díaz, J.; Soto Castro, G.; Mendoza, M.E.; Macías, J.L. Tree Rings as Indicators of Climatic Variation in the Trans-Mexican Volcanic Belt, Central Mexico. Ecol. Indic. 2021, 120, 106920. [Google Scholar] [CrossRef]
- Bravo-Espinosa, M.; Mendoza, M.E.; Medina-Orozco, L.; Prat, C.; García-Oliva, F.; López-Granados, E. Runoff, Soil Loss, and Nutrient Depletion under Traditional and Alternative Cropping Systems in the Transmexican Volcanic Belt, Central Mexico. Land Degrad. Dev. 2009, 20, 640–653. [Google Scholar] [CrossRef]
- Cruzado-Vargas, A.L.; Blanco-García, A.; Lindig-Cisneros, R.; Gómez-Romero, M.; Lopez-Toledo, L.; de la Barrera, E.; Sáenz-Romero, C. Reciprocal Common Garden Altitudinal Transplants Reveal Potential Negative Impacts of Climate Change on Abies Religiosa Populations in the Monarch Butterfly Biosphere Reserve Overwintering Sites. Forests 2021, 12, 69. [Google Scholar] [CrossRef]
- Villers-Ruiz, L.; Castañeda-Aguado, D. Species and Plant Community Reorganization in the Trans-Mexican Volcanic Belt under Climate Change Conditions. J. Mt. Sci. 2013, 10, 923–931. [Google Scholar] [CrossRef]
- P4 Multispectral Specs. Available online: https://www.dji.com/mx/p4-multispectral/specs (accessed on 3 April 2023).
- Lu, H.; Fan, T.; Ghimire, P.; Deng, L. Experimental Evaluation and Consistency Comparison of UAV Multispectral Minisensors. Remote Sens. 2020, 12, 2542. [Google Scholar] [CrossRef]
- Syetiawan, A.; Gularso, H.; Kusnadi, G.I.; Pramudita, G.N. Precise Topographic Mapping Using Direct Georeferencing in UAV. IOP Conf. Ser. Earth Environ. Sci. 2020, 500, 12029. [Google Scholar] [CrossRef]
- De Jesús, A.; Breña-Naranjo, J.A.; Pedrozo-Acuña, A.; Alcocer Yamanaka, V.H. The Use of TRMM 3B42 Product for Drought Monitoring in Mexico. Water 2016, 8, 325. [Google Scholar] [CrossRef] [Green Version]
- DJI Ground Station Pro. Available online: https://www.dji.com/mx/ground-station-pro (accessed on 3 April 2023).
- OpenDroneMap Awesome. Drone. Software. Available online: www.opendronemap.org (accessed on 2 April 2023).
- Groos, A.R.; Bertschinger, T.J.; Kummer, C.M.; Erlwein, S.; Munz, L.; Philipp, A. The Potential of Low-Cost UAVs and Open-Source Photogrammetry Software for High-Resolution Monitoring of Alpine Glaciers: A Case Study from the Kanderfirn (Swiss Alps). Geosciences 2019, 9, 356. [Google Scholar] [CrossRef] [Green Version]
- Deliry, S.I.; Avdan, U. Accuracy of Unmanned Aerial Systems Photogrammetry and Structure from Motion in Surveying and Mapping: A Review. J. Indian Soc. Remote Sens. 2021, 49, 1997–2017. [Google Scholar] [CrossRef]
- Iglhaut, J.; Cabo, C.; Puliti, S.; Piermattei, L.; O’Connor, J.; Rosette, J. Structure from Motion Photogrammetry in Forestry: A Review. Curr. For. Rep. 2019, 5, 155–168. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Qi, J.; Wan, P.; Wang, H.; Xie, D.; Wang, X.; Yan, G. An Easy-to-Use Airborne LiDAR Data Filtering Method Based on Cloth Simulation. Remote Sens. 2016, 8, 501. [Google Scholar] [CrossRef]
- Lee, S.; Yu, B.-H. Automatic Detection of Dead Tree from UAV Imagery. In Proceedings of the 39th Asian Conference on Remote Sensing, Kuala Lumpur, Malaysia, 15–19 October 2018. [Google Scholar]
- Rosas-Chavoya, M.; Gallardo-Salazar, J.L.; López-Serrano, P.M.; Alcántara-Concepción, P.C.; León-Miranda, A.K. QGIS a Constantly Growing Free and Open-Source Geospatial Software Contributing to Scientific Development. Cuad. Investig. Geográfica 2022, 48, 197–213. [Google Scholar] [CrossRef]
- Minařík, R.; Langhammer, J.; Lendzioch, T. Automatic Tree Crown Extraction from UAS Multispectral Imagery for the Detection of Bark Beetle Disturbance in Mixed Forests. Remote Sens. 2020, 12, 4081. [Google Scholar] [CrossRef]
- Jones, H.G.; Vaughan, R.A. Remote Sensing of Vegetation: Principles, Techniques, and Applications; Oxford University Press: Oxford, UK, 2010; ISBN 0199207798. [Google Scholar]
- Liu, S.; Zeng, W.; Wu, L.; Lei, G.; Chen, H.; Gaiser, T.; Srivastava, A.K. Simulating the Leaf Area Index of Rice from Multispectral Images. Remote Sens. 2021, 13, 3663. [Google Scholar] [CrossRef]
- Narmilan, A.; Gonzalez, F.; Salgadoe, A.S.A.; Kumarasiri, U.W.L.M.; Weerasinghe, H.A.S.; Kulasekara, B.R. Predicting Canopy Chlorophyll Content in Sugarcane Crops Using Machine Learning Algorithms and Spectral Vegetation Indices Derived from UAV Multispectral Imagery. Remote Sens. 2022, 14, 1140. [Google Scholar] [CrossRef]
- Nevalainen, O.; Honkavaara, E.; Tuominen, S.; Viljanen, N.; Hakala, T.; Yu, X.; Hyyppä, J.; Saari, H.; Pölönen, I.; Imai, N.N.; et al. Individual Tree Detection and Classification with UAV-Based Photogrammetric Point Clouds and Hyperspectral Imaging. Remote Sens. 2017, 9, 185. [Google Scholar] [CrossRef] [Green Version]
- Mohan, M.; Leite, R.V.; Broadbent, E.N.; Wan Mohd Jaafar, W.S.; Srinivasan, S.; Bajaj, S.; Dalla Corte, A.P.; do Amaral, C.H.; Gopan, G.; Saad, S.N.M.; et al. Individual Tree Detection Using UAV-Lidar and UAV-SfM Data: A Tutorial for Beginners. Open Geosci. 2021, 13, 1028–1039. [Google Scholar] [CrossRef]
- Gallardo-Salazar, J.L.; Pompa-García, M. Detecting Individual Tree Attributes and Multispectral Indices Using Unmanned Aerial Vehicles: Applications in a Pine Clonal Orchard. Remote Sens. 2020, 12, 4144. [Google Scholar] [CrossRef]
- Picos, J.; Bastos, G.; Míguez, D.; Alonso, L.; Armesto, J. Individual Tree Detection in a Eucalyptus Plantation Using Unmanned Aerial Vehicle (UAV)-LiDAR. Remote Sens. 2020, 12, 885. [Google Scholar] [CrossRef] [Green Version]
- Crabbé, A.; Cahy, T.; Somers, B.; Verbeke, L.P.; Van Coillie, F. Tree Density Calculator Software. Available online: https://bitbucket.org/kul-reseco/localmaxfilter (accessed on 2 April 2023).
- Ke, Y.; Quackenbush, L.J. A Review of Methods for Automatic Individual Tree-Crown Detection and Delineation from Passive Remote Sensing. Int. J. Remote Sens. 2011, 32, 4725–4747. [Google Scholar] [CrossRef]
- Li, W.; Fu, H.; Yu, L.; Cracknell, A. Deep Learning Based Oil Palm Tree Detection and Counting for High-Resolution Remote Sensing Images. Remote Sens. 2017, 9, 22. [Google Scholar] [CrossRef] [Green Version]
- Jiménez, J.; Aguirre, O.; Kramer, H. Tree Crown Structure Indicators in a Natural Uneven-Aged Mixed Coniferous Forest in Northeastern Mexico. In Proceedings of the Monitoring Science and Technology Symposium: Unifying Knowledge for Sustainability in the Western Hemisphere Proceedings RMRS-P-42CD; Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2006; pp. 649–654. [Google Scholar]
- Zhang, J.; You, S.; Gruenwald, L. Efficient Parallel Zonal Statistics on Large-Scale Global Biodiversity Data on GPUs. In Proceedings of the 4th International ACM SIGSPATIAL Workshop on Analytics for Big Geospatial Data, Bellevue, WA, USA, 3–6 November 2015; Association for Computing Machinery: New York, NY, USA, 2015; pp. 35–44. [Google Scholar]
- Buras, A.; Schunk, C.; Zeiträg, C.; Herrmann, C.; Kaiser, L.; Lemme, H.; Straub, C.; Taeger, S.; Gößwein, S.; Klemmt, H.-J.; et al. Are Scots Pine Forest Edges Particularly Prone to Drought-Induced Mortality? Environ. Res. Lett. 2018, 13, 025001. [Google Scholar] [CrossRef]
- Ye, H.; Huang, W.; Huang, S.; Cui, B.; Dong, Y.; Guo, A.; Ren, Y.; Jin, Y. Recognition of Banana Fusarium Wilt Based on UAV Remote Sensing. Remote Sens. 2020, 12, 938. [Google Scholar] [CrossRef] [Green Version]
- Alley, W.M. The Palmer Drought Severity Index: Limitations and Assumptions. J. Clim. Appl. Meteorol. 1984, 23, 1100–1109. [Google Scholar] [CrossRef]
- Gómez-Pineda, E.; Hammond, W.M.; Trejo-Ramirez, O.; Gil-Fernández, M.; Allen, C.D.; Blanco-García, A.; Sáenz-Romero, C. Drought Years Promote Bark Beetle Outbreaks in Mexican Forests of Abies Religiosa and Pinus Pseudostrobus. For. Ecol. Manag. 2022, 505, 119944. [Google Scholar] [CrossRef]
- Abatzoglou, J.T.; Dobrowski, S.Z.; Parks, S.A.; Hegewisch, K.C. TerraClimate, a High-Resolution Global Dataset of Monthly Climate and Climatic Water Balance from 1958–2015. Sci. Data 2018, 5, 170191. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023. [Google Scholar]
- He, J.; Li, Y.; Zhang, K. Research of UAV Flight Planning Parameters. Positioning 2012, 3, 43–45. [Google Scholar] [CrossRef] [Green Version]
- Carrillo-Arizmendi, L.; Pérez-Suárez, M.; Vargas-Hernández, J.J.; Rozenberg, P.; Martínez-Campos, A.R. Warming Effects on Tree-Ring Variables in P. hartwegii Lindl. at the Extremes of Its Natural Elevational Distribution in Central Mexico. Agric. For. Meteorol. 2022, 324, 109109. [Google Scholar] [CrossRef]
- Sexton, J.P.; McIntyre, P.J.; Angert, A.L.; Rice, K.J. Evolution and Ecology of Species Range Limits. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 415–436. [Google Scholar] [CrossRef] [Green Version]
- Mátyás, C.; Berki, I.; Bidló, A.; Csóka, G.; Czimber, K.; Führer, E.; Gálos, B.; Gribovszki, Z.; Illés, G.; Hirka, A.; et al. Sustainability of Forest Cover under Climate Change on the Temperate-Continental Xeric Limits. Forests 2018, 9, 489. [Google Scholar] [CrossRef] [Green Version]
- Mátyás, C.; BERKI, I.; Czúcz, B.; Gálos, B.; Móricz, N.; Rasztovits, E. Future of Beech in Southeast Europe from the Perspective of Evolutionary Ecology. Acta Silv. Lignaria Hung. 2010, 6, 91–110. [Google Scholar]
- Ricker, M.; Gutiérrez-García, G.; Daly, D.C. Modeling Long-Term Tree Growth Curves in Response to Warming Climate: Test Cases from a Subtropical Mountain Forest and a Tropical Rainforest in Mexico. Can. J. For. Res. 2007, 37, 977–989. [Google Scholar] [CrossRef]
- Choat, B.; Brodribb, T.J.; Brodersen, C.R.; Duursma, R.A.; López, R.; Medlyn, B.E. Triggers of Tree Mortality under Drought. Nature 2018, 558, 531–539. [Google Scholar] [CrossRef]
- Cambrón-Sandoval, V.H.; Méndez-González, J.; Ledesma-Colunga, M.C.; Luna-Soria, H.; García-Aranda, M.A.; Cerano-Paredes, J.; Obregón-Zúñiga, J.A.; Vergara-Pineda, S. Variabilidad Climática e Interacción con Otros Factores que Afectan la Dinámica Poblacional de Descortezadores en Bosques Amenazados de México; CONAFOR-CONACYT: Querétaro, Mexico, 2018; ISBN 978-607-513-336-2.
- Tejeda-Landero, V.M.; Sánchez-Velásquez, L.R.; Viveros-Viveros, H.; Aparicio-Rentería, A.; Flores-Peredo, R. Seed Bank Formation and Removal of Pinus hartwegii (Pinaceae) Seeds along an Altitudinal Gradient in the Cofre de Perote National Park, Veracruz, Mexico. Bot. Sci. 2019, 97, 623–629. [Google Scholar] [CrossRef] [Green Version]
- Alba-Landa, J.; Aparicio-Rentería, A.; Márquez-Ramírez, J. Potencial y eficiencia de producción de semillas de Pinus hartwegii lindl. de dos poblaciones de México. For. Veracruzana 2003, 5, 25–28. [Google Scholar]
- Astudillo-Sánchez, C.C.; Fowler, M.S.; Villanueva-Díaz, J.; Endara-Agramont, A.R.; Soria-Díaz, L. Recruitment and Facilitation in Pinus hartwegii, a Mexican Alpine Treeline Ecotone, with Potential Responses to Climate Warming. Trees 2019, 33, 1087–1100. [Google Scholar] [CrossRef] [Green Version]
- Castellanos-Acuña, D.; Vance-Borland, K.W.; St. Clair, J.B.; Hamann, A.; López-Upton, J.; Gómez-Pineda, E.; Ortega-Rodríguez, J.M.; Sáenz-Romero, C. Climate-Based Seed Zones for Mexico: Guiding Reforestation under Observed and Projected Climate Change. New For. 2018, 49, 297–309. [Google Scholar] [CrossRef]
- Manzanilla-Quiñones, U.; Aguirre-Calderón, Ó.A.; Jiménez-Pérez, J.; Treviño-Garza, E.J.; Yerena-Yamallel, J.I. Distribución Actual y Futura Del Bosque Subalpino de Pinus hartwegii Lindl En El Eje Neovolcánico Transversal. Madera Bosques 2019, 25. [Google Scholar] [CrossRef]
- Millar, C.I.; Stephenson, N.L. Temperate Forest Health in an Era of Emerging Megadisturbance. Science 2015, 349, 823–826. [Google Scholar] [CrossRef]
- Hill, A.P.; Nolan, C.J.; Hemes, K.S.; Cambron, T.W.; Field, C.B. Low-Elevation Conifers in California’s Sierra Nevada Are out of Equilibrium with Climate. PNAS Nexus 2023, 2, pgad004. [Google Scholar] [CrossRef]
- Minařík, R.; Langhammer, J. Use of a Multispectral UAV Photogrammetry for Detection and Tracking of Forest Disturbance Dynamics. Int. Arch. Photogramm. Remote Sens. Spat. Inf. Sci. 2016, XLI-B8, 711–718. [Google Scholar] [CrossRef] [Green Version]
- Cristiano, P.; Madanes, N.; Campanello, P.; di Francescantonio, D.; Rodríguez, S.; Zhang, Y.-J.; Carrasco, L.; Goldstein, G. High NDVI and Potential Canopy Photosynthesis of South American Subtropical Forests despite Seasonal Changes in Leaf Area Index and Air Temperature. Forests 2014, 5, 287–308. [Google Scholar] [CrossRef] [Green Version]
- Gamon, J.A.; Kovalchuck, O.; Wong, C.Y.S.; Harris, A.; Garrity, S.R. Monitoring Seasonal and Diurnal Changes in Photosynthetic Pigments with Automated PRI and NDVI Sensors. Biogeosciences 2015, 12, 4149–4159. [Google Scholar] [CrossRef] [Green Version]
- Rullan-Silva, C.D.; Olthoff, A.E.; Delgado de la Mata, J.A.; Pajares-Alonso, J.A. Remote Monitoring of Forest Insect Defoliation—A Review. For. Syst 2013, 22, 377. [Google Scholar] [CrossRef] [Green Version]
- Di Bella, C.M.; Paruelo, J.M.; Becerra, J.E.; Bacour, C.; Baret, F. Effect of Senescent Leaves on NDVI-Based Estimates of FAPAR: Experimental and Modelling Evidences. Int. J. Remote Sens. 2004, 25, 5415–5427. [Google Scholar] [CrossRef]
- Chavana-Bryant, C.; Malhi, Y.; Wu, J.; Asner, G.P.; Anastasiou, A.; Enquist, B.J.; Cosio Caravasi, E.G.; Doughty, C.E.; Saleska, S.R.; Martin, R.E.; et al. Leaf Aging of Amazonian Canopy Trees as Revealed by Spectral and Physiochemical Measurements. New Phytol. 2017, 214, 1049–1063. [Google Scholar] [CrossRef] [Green Version]
- Yao, H.; Qin, R.; Chen, X. Unmanned Aerial Vehicle for Remote Sensing Applications—A Review. Remote Sens. 2019, 11, 1443. [Google Scholar] [CrossRef] [Green Version]
- Puliti, S.; Breidenbach, J.; Astrup, R. Estimation of Forest Growing Stock Volume with UAV Laser Scanning Data: Can It Be Done without Field Data? Remote Sens. 2020, 12, 1245. [Google Scholar] [CrossRef] [Green Version]
- Gonroudobou, O.B.H.; Silvestre, L.H.; Diez, Y.; Nguyen, H.T.; Caceres, M.L.L. Treetop Detection in Mountainous Forests Using UAV Terrain Awareness Function. Computation 2022, 10, 90. [Google Scholar] [CrossRef]
- LaRue, E.A.; Fahey, R.T.; Alveshere, B.C.; Atkins, J.W.; Bhatt, P.; Buma, B.; Chen, A.; Cousins, S.; Elliott, J.M.; Elmore, A.J.; et al. A Theoretical Framework for the Ecological Role of Three-dimensional Structural Diversity. Front. Ecol. Environ. 2023, 21, 4–13. [Google Scholar] [CrossRef]








| Descriptive Measure | NDVI | LCI |
|---|---|---|
| Minimum | −0.45 | −0.24 |
| First quartile | 0.11 | 0.01 |
| Median | 0.28 | 0.03 |
| Mean | 0.26 | 0.04 |
| Third quartile | 0.40 | 0.07 |
| Maximum | 0.70 | 0.31 |
| Standard deviation | 0.18 | 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallardo-Salazar, J.L.; Lindig-Cisneros, R.A.; Lopez-Toledo, L.; Endara-Agramont, A.R.; Blanco-García, A.; Sáenz-Romero, C. Analysis of the Vigor of Pinus hartwegii Lindl. along an Altitudinal Gradient Using UAV Multispectral Images: Evidence of Forest Decline Possibly Associated with Climatic Change. Forests 2023, 14, 1176. https://doi.org/10.3390/f14061176
Gallardo-Salazar JL, Lindig-Cisneros RA, Lopez-Toledo L, Endara-Agramont AR, Blanco-García A, Sáenz-Romero C. Analysis of the Vigor of Pinus hartwegii Lindl. along an Altitudinal Gradient Using UAV Multispectral Images: Evidence of Forest Decline Possibly Associated with Climatic Change. Forests. 2023; 14(6):1176. https://doi.org/10.3390/f14061176
Chicago/Turabian StyleGallardo-Salazar, José Luis, Roberto A. Lindig-Cisneros, Leonel Lopez-Toledo, Angel R. Endara-Agramont, Arnulfo Blanco-García, and Cuauhtémoc Sáenz-Romero. 2023. "Analysis of the Vigor of Pinus hartwegii Lindl. along an Altitudinal Gradient Using UAV Multispectral Images: Evidence of Forest Decline Possibly Associated with Climatic Change" Forests 14, no. 6: 1176. https://doi.org/10.3390/f14061176
APA StyleGallardo-Salazar, J. L., Lindig-Cisneros, R. A., Lopez-Toledo, L., Endara-Agramont, A. R., Blanco-García, A., & Sáenz-Romero, C. (2023). Analysis of the Vigor of Pinus hartwegii Lindl. along an Altitudinal Gradient Using UAV Multispectral Images: Evidence of Forest Decline Possibly Associated with Climatic Change. Forests, 14(6), 1176. https://doi.org/10.3390/f14061176