Potential Distribution Projections for Senegalia senegal (L.) Britton under Climate Change Scenarios
Abstract
:1. Introduction
2. Material and Methods
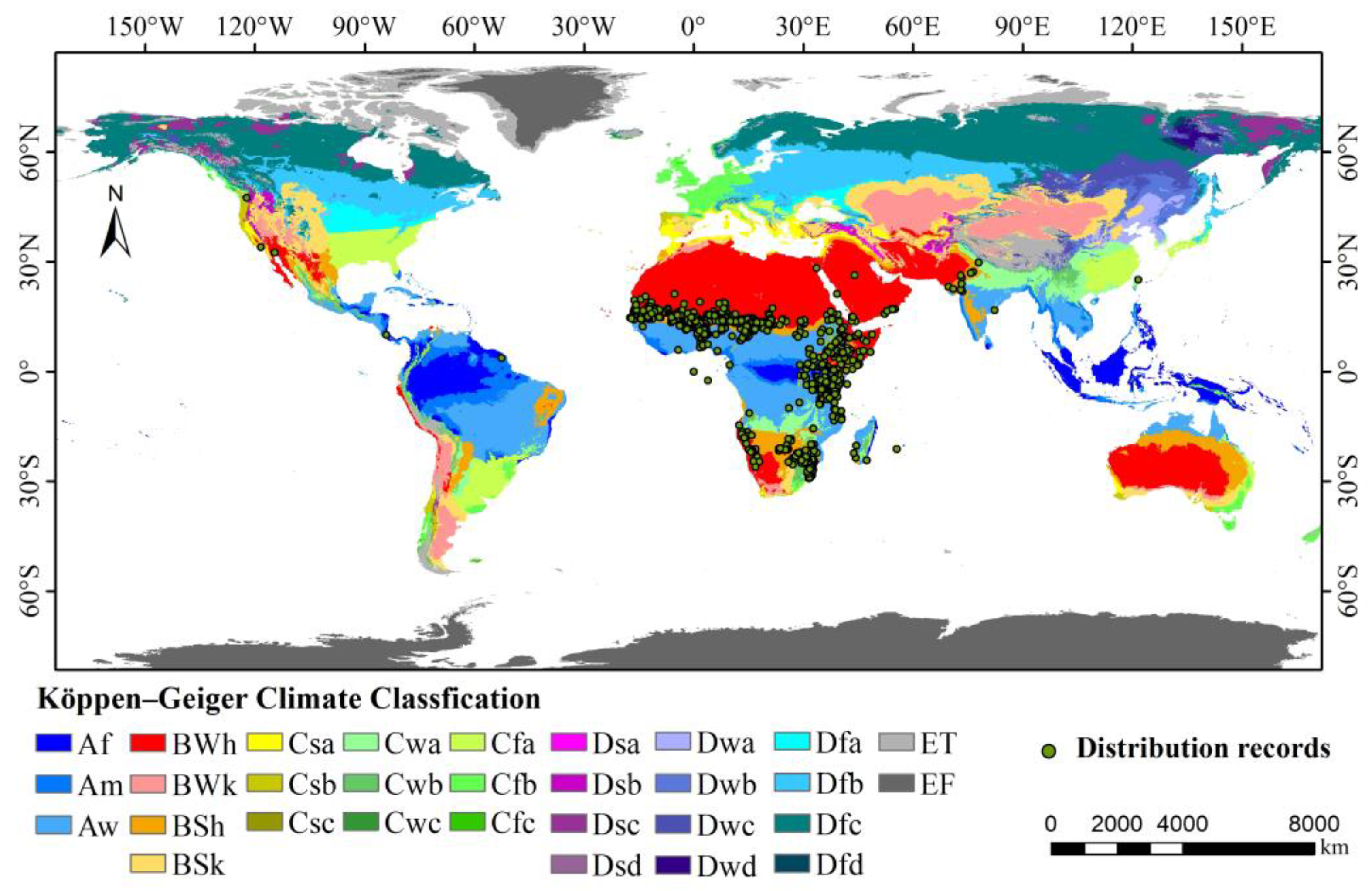
2.1. Occurrence Data
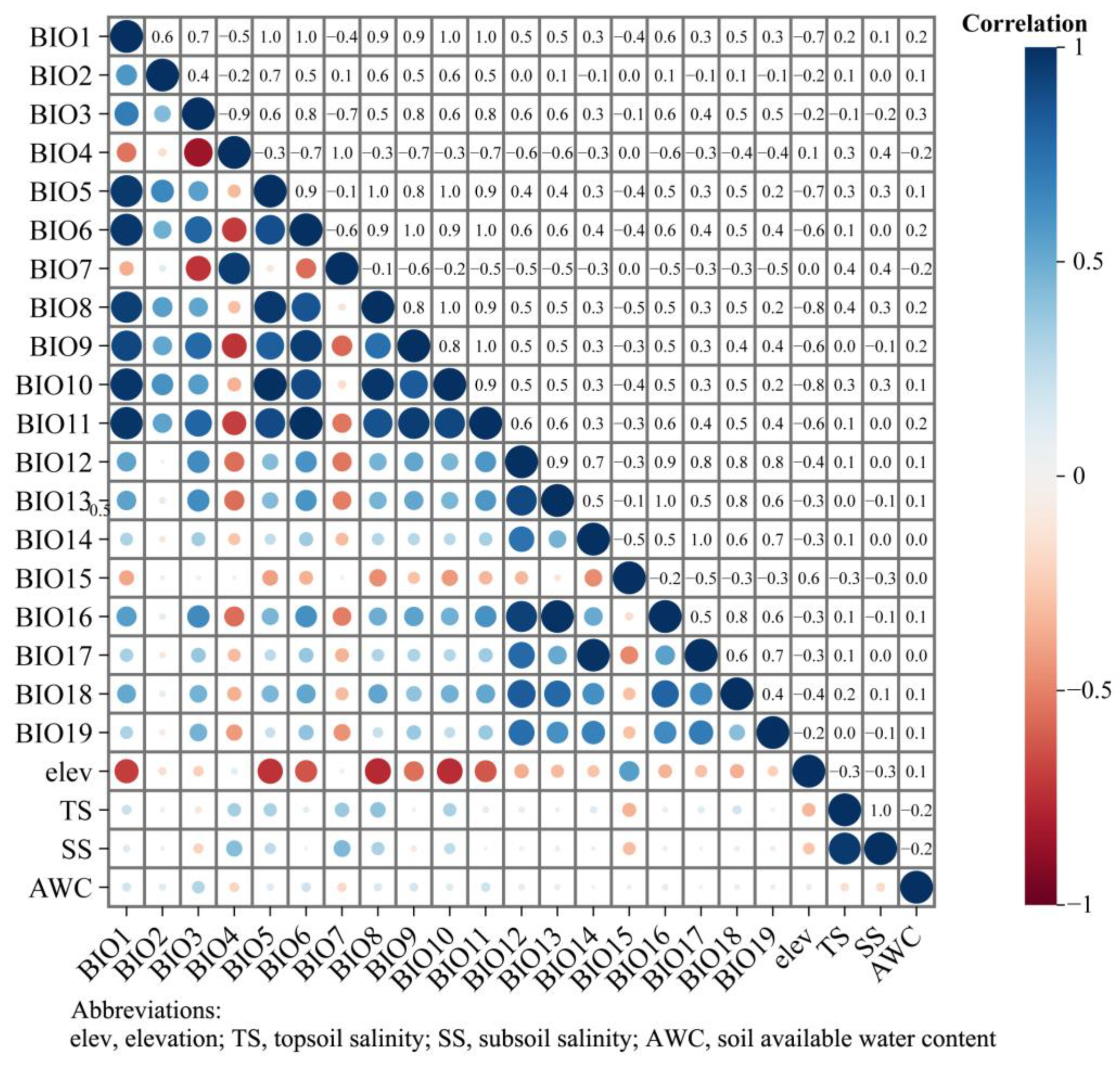
2.2. Environment Variables
2.3. Modeling Optimization
2.4. MaxEnt Modeling and Evaluation
2.5. Classification of Suitable Regions and Spatial Pattern Changes
3. Results
3.1. Assessment of Key Environment Variables
3.2. Response Curve Analysis of Key Environmental Variables
3.3. Potentially Suitable Habitats under Current Climatic Conditions
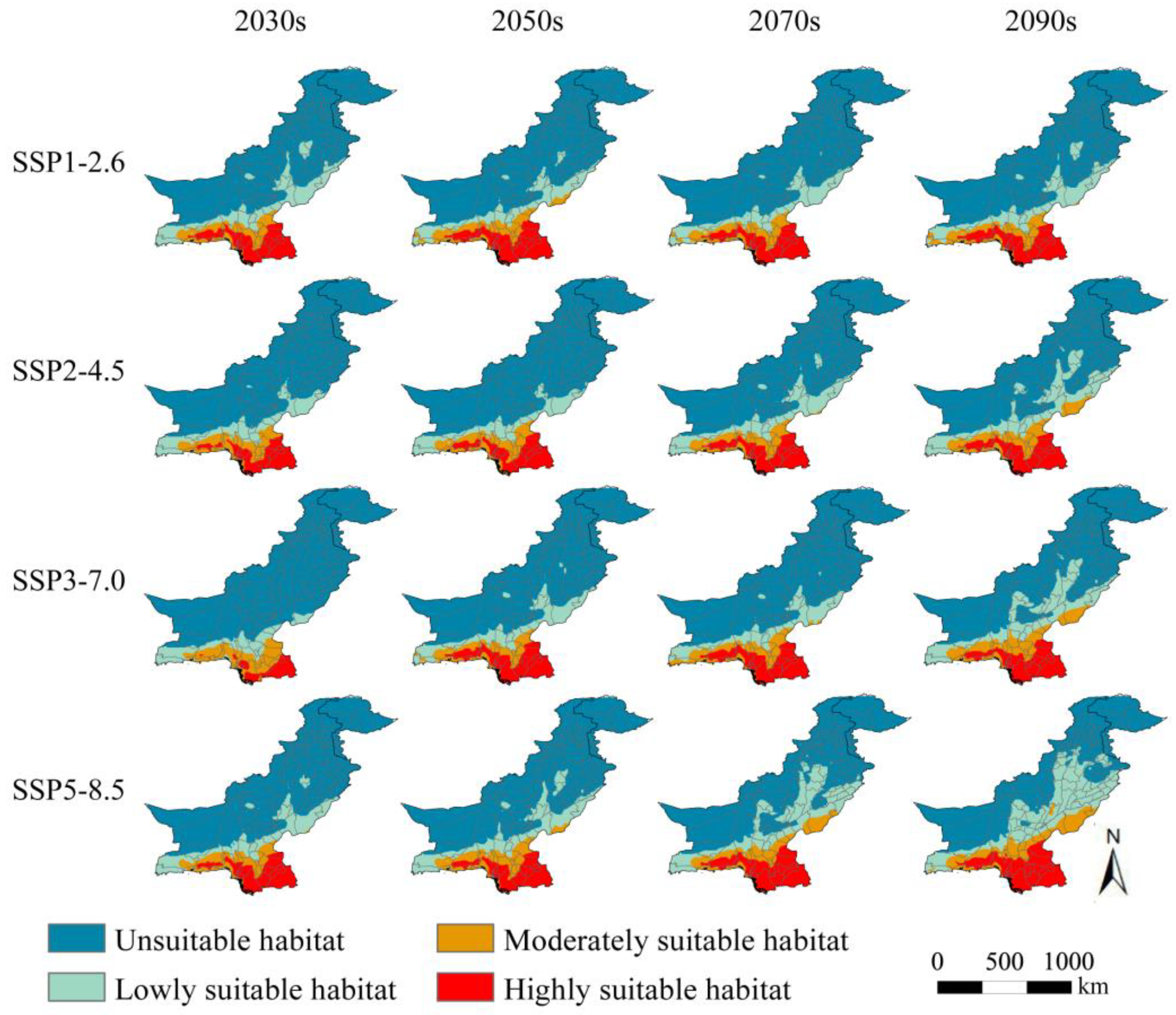
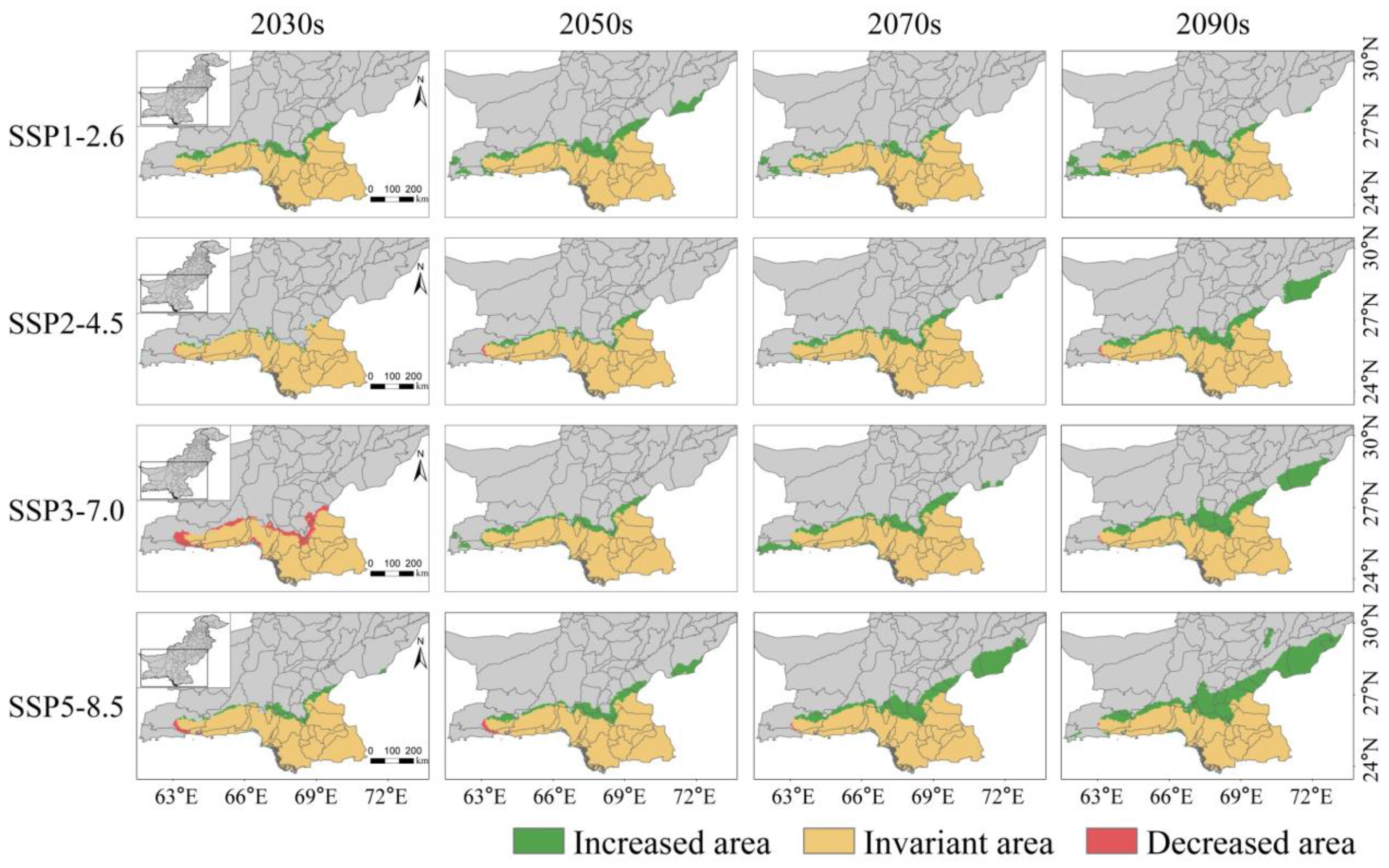
3.4. Potentially Suitable Habitats in Pakistan under Different Future Climate Scenarios
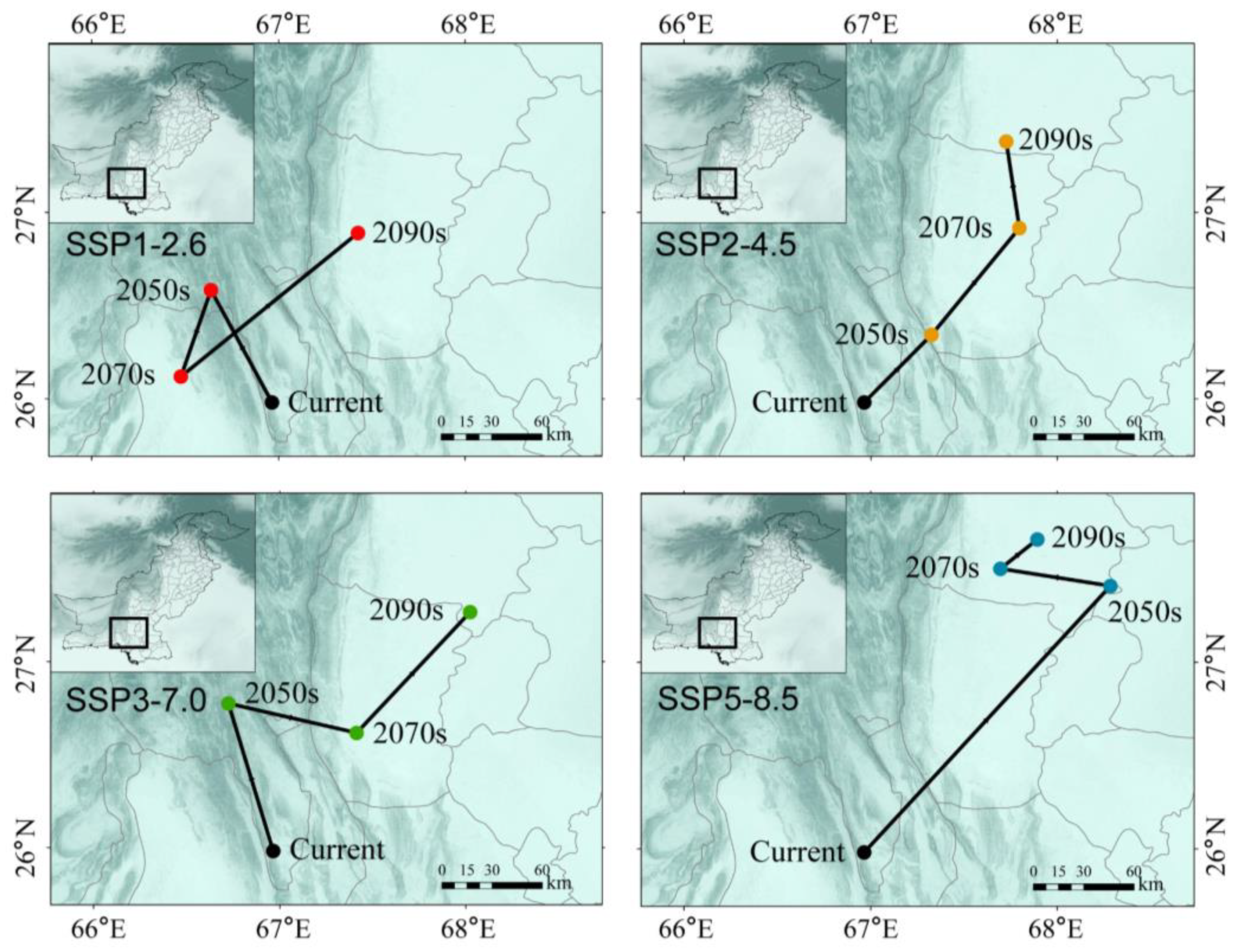
3.5. Spatial Pattern Changes of Potential Habitat Regions in Pakistan
4. Discussion
4.1. Effects of Environmental Variables on the Distribution of S. senegal
4.2. Changes in Suitable Habitat for S. senegal in Pakistan
4.3. Recommendations for the Introduction of S. senegal for Afforestation in Pakistan
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- D’Odorico, P.; Bhattachan, A.; Davis, K.F.; Ravi, S.; Runyan, C.W. Global desertification: Drivers and feedbacks. Adv. Water Resour. 2013, 51, 326–344. [Google Scholar] [CrossRef]
- Huang, J.; Yu, H.; Guan, X.; Wang, G.; Guo, R. Accelerated dryland expansion under climate change. Nat. Clim. Chang. 2016, 6, 166–171. [Google Scholar] [CrossRef]
- Qi, K.; Zhu, J.; Zheng, X.; Wang, G.G.; Li, M. Impacts of the world’s largest afforestation program (Three-North Afforestation Program) on desertification control in sandy land of China. GISci. Remote Sens. 2023, 60, 2167574. [Google Scholar] [CrossRef]
- Diatta, O.; Sarr, M.S.; Hansen, J.K.; Diallo, A.M.; Nielsen, L.R.; Raebild, A.; Kjaer, E.D. Survival and growth of Acacia senegal (L.) Wild. (Senegalia senegal (L.) Britton) provenances depend on the rainfall at the site of origin. Ann. For. Sci. 2021, 78, 82. [Google Scholar] [CrossRef]
- Bakhoum, N.; Fall, D.; Fall, F.; Diouf, F.; Hirsch, A.M.; Balachandar, D.; Diouf, D. Senegalia senegal (synonym: Acacia senegal), its importance to sub-Saharan Africa, and its relationship with a wide range of symbiotic soil microorganisms. S. Afr. J. Bot. 2018, 119, 362–368. [Google Scholar] [CrossRef]
- Sanchez, C.; Nigen, M.; Tamayo, V.M.; Doco, T.; Williams, P.; Amine, C.; Renard, D. Acacia gum: History of the future. Food Hydrocoll. 2018, 78, 140–160. [Google Scholar] [CrossRef]
- Odee, D.W.; Wilson, J.; Cavers, S. Prospects for Genetic Improvement of Acacia Senegal: Can Molecular Approaches Deliver Better Gum Yield and Quality?. In Gum Arabic; Kennedy, J.F., Phillips, G.O., Williams, P.A., Eds.; The Royal Society of Chemistry: London, UK, 2011. [Google Scholar]
- Raddad, E.Y.; Luukkanen, O. The influence of different Acacia senegal agroforestry systems on soil water and crop yields in clay soils of the Blue Nile region, Sudan. Agric. Water Manag. 2007, 87, 61–72. [Google Scholar] [CrossRef]
- Diallo, M.D.; Goalbaye, T.; Mahamat-Saleh, M.; Sarr, P.S.; Masse, D.; Wood, S.A.; Diop, L.; Dick, R.P.; Diop, A.; Guisse, A. Effects of major woody species of the Senegalese Great Green Wall on N mineralization and microbial biomass in soils. Bois For. Trop. 2017, 333, 43–54. [Google Scholar]
- Yonli, H.H.; Sene, G.; Sanon, K.B.; Dianda, M.; Khasa, D.P. Senegalia senegal (L.) Britton Response to Microbial and Manure Amendments for the Rehabilitation of Waste Rock Dumps in the Essakane Gold Mining Site, Burkina Faso. Front. Environ. Sci. 2022, 10, 803009. [Google Scholar] [CrossRef]
- Fall, D.; Bakhoum, N.; Sall, S.N.; Zoubeirou, A.M.; Sylla, S.N.; Diouf, D. Rhizobial Inoculation Increases Soil Microbial Functioning and Gum Arabic Production of 13-Year-Old Senegalia senegal (L.) Britton, Trees in the North Part of Senegal. Front. Plant Sci. 2016, 7, 1355. [Google Scholar] [CrossRef]
- Zhao, Y.; Deng, X.; Xiang, W.; Chen, L.; Ouyang, S. Predicting potential suitable habitats of Chinese fir under current and future climatic scenarios based on Maxent model. Ecol. Inform. 2021, 64, 101393. [Google Scholar] [CrossRef]
- Evans, J.M.; Fletcher, R.J., Jr.; Alavalapati, J. Using species distribution models to identify suitable areas for biofuel feedstock production. Glob. Chang. Biol. Bioenergy 2010, 2, 63–78. [Google Scholar] [CrossRef]
- Zhao, Z.; Xiao, N.; Shen, M.; Li, J. Comparison between optimized MaxEnt and random forest modeling in predicting potential distribution: A case study with Quasipaa boulengeri in China. Sci. Total Environ. 2022, 842, 156867. [Google Scholar] [CrossRef]
- Kang, J.; Liu, M.; Qu, M.; Guang, X.; Chen, J.; Zhao, Y.; Huang, B. Identifying the potential soil pollution areas derived from the metal mining industry in China using MaxEnt with mine reserve scales (MaxEnt_MRS). Environ. Pollut. 2023, 329, 121687. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Bao, R.; Li, X.; Zheng, J. Feature tuning improves MAXENT predictions of the potential distribution of Pedicularis longiflora Rudolph and its variant. PeerJ 2022, 10, e13337. [Google Scholar] [CrossRef]
- Araujo, M.B.; Anderson, R.P.; Marcia Barbosa, A.; Beale, C.M.; Dormann, C.F.; Early, R.; Garcia, R.A.; Guisan, A.; Maiorano, L.; Naimi, B.; et al. Standards for distribution models in biodiversity assessments. Sci. Adv. 2019, 5, eaat4858. [Google Scholar] [CrossRef]
- Araújo, M.B.; Whittaker, R.J.; Ladle, R.J.; Erhard, M. Reducing uncertainty in projections of extinction risk from climate change. Glob. Ecol. Biogeogr. 2005, 14, 529–538. [Google Scholar] [CrossRef]
- Sillero, N.; Barbosa, A.M. Common mistakes in ecological niche models. Int. J. Geogr. Inf. Sci. 2021, 35, 213–226. [Google Scholar] [CrossRef]
- Min, Y.; Zhang, Y.; Kang, J.; Feng, K. Study on Spatial-temporal Dynamic Monitoring of Degree of Desertification in CPEC based on MODIS Image. Remote Sens. Technol. Appl. 2021, 36, 827–837. [Google Scholar]
- Chen, I.C.; Hill, J.K.; Ohlemueller, R.; Roy, D.B.; Thomas, C.D. Rapid Range Shifts of Species Associated with High Levels of Climate Warming. Science 2011, 333, 1024–1026. [Google Scholar] [CrossRef]
- Harrison, P.A.; Berry, P.M.; Butt, N.; New, M. Modelling climate change impacts on species’ distributions at the European scale: Implications for conservation policy. Environ. Sci. Policy 2006, 9, 116–128. [Google Scholar] [CrossRef]
- Xiao, J.; Eziz, A.; Zhang, H.; Wang, Z.; Tang, Z.; Fang, J. Responses of four dominant dryland plant species to climate change in the Junggar Basin, northwest China. Ecol. Evol. 2019, 9, 13596–13607. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef]
- Alabar, F.; Politi, N.; Nazaro, P.; Amoroso, M.; Rivera, L. Changes in the potential distribution of valuable tree species based on their regeneration in the Neotropical seasonal dry forest of north-western Argentina. Environ. Conserv. 2022, 49, 83–89. [Google Scholar] [CrossRef]
- Guo, Y.; Zhao, Z.; Zhu, F.; Li, X. Climate change may cause distribution area loss for tree species in southern China. For. Ecol. Manag. 2022, 511, 120134. [Google Scholar] [CrossRef]
- Vieira, T.L.; Barbosa-Silva, R.G.; Acosta, A.L.; van den Berg, C. Expanding the Distribution of Prosthechea jauana (Orchidaceae) in the Pantepui and Highlighting the Urgent Need for Conservation Strategies in the Region in Face of Climate Change. Plants 2024, 13, 222. [Google Scholar] [CrossRef]
- Beck, H.E.; McVicar, T.R.; Vergopolan, N.; Berg, A.; Lutsko, N.J.; Dufour, A.; Zeng, Z.; Jiang, X.; van Dijk, A.I.J.M.; Miralles, D.G. High-resolution (1 km) Köppen-Geiger maps for 1901–2099 based on constrained CMIP6 projections. Sci. Data 2023, 10, 724. [Google Scholar] [CrossRef]
- Shao, M.; Fan, J.; Ma, J.; Wang, L. Identifying the natural reserve area of Cistanche salsa under the effects of multiple host plants and climate change conditions using a maximum entropy model in Xinjiang, China. Front. Plant Sci. 2022, 13, 934959. [Google Scholar] [CrossRef]
- Warren, D.L.; Wright, A.N.; Seifert, S.N.; Shaffer, H.B. Incorporating model complexity and spatial sampling bias into ecological niche models of climate change risks faced by 90 California vertebrate species of concern. Divers. Distrib. 2014, 20, 334–343. [Google Scholar] [CrossRef]
- Muscarella, R.; Galante, P.J.; Soley-Guardia, M.; Boria, R.A.; Kass, J.M.; Uriarte, M.; Anderson, R.P. ENMeval: An R package for conducting spatially independent evaluations and estimating optimal model complexity for MAXENT ecological niche models. Methods Ecol. Evol. 2014, 5, 1198–1205. [Google Scholar] [CrossRef]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudik, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Tarnian, F.; Kumar, S.; Azarnivand, H.; Chahouki, M.A.Z.; Mossivand, A.M. Assessing the effects of climate change on the distribution of Daphne mucronata in Iran. Environ. Monit. Assess. 2021, 193, 562. [Google Scholar] [CrossRef]
- Shao, M.; Wang, L.; Li, B.; Li, S.; Fan, J.; Li, C. Maxent Modeling for Identifying the Nature Reserve of Cistanche deserticola Ma under Effects of the Host (Haloxylon Bunge) Forest and Climate Changes in Xinjiang, China. Forests 2022, 13, 189. [Google Scholar] [CrossRef]
- Dillon, M.E.; Wang, G.; Huey, R.B. Global metabolic impacts of recent climate warming. Nature 2010, 467, U704–U788. [Google Scholar] [CrossRef]
- Sun, S.; Zhang, Y.; Huang, D.; Wang, H.; Cao, Q.; Fan, P.; Yang, N.; Zheng, P.; Wang, R. The effect of climate change on the richness distribution pattern of oaks (Quercus L.) in China. Sci. Total Environ. 2020, 744, 140786. [Google Scholar] [CrossRef]
- Waheed, M.; Haq, S.M.; Arshad, F.; Jameel, M.A.; Siddiqui, M.H.H.; Bussmann, R.W.W.; Manshoor, N.; Alamri, S. Where Will Threatened Aegle marmelos L., a Tree of the Semi-Arid Region, Go under Climate Change? Implications for the Reintroduction of the Species. Land 2023, 12, 1433. [Google Scholar] [CrossRef]
- Khan, A.M.; Li, Q.; Saqib, Z.; Khan, N.; Habib, T.; Khalid, N.; Majeed, M.; Tariq, A. MaxEnt Modelling and Impact of Climate Change on Habitat Suitability Variations of Economically Important Chilgoza Pine (Pinus gerardiana Wall.) in South Asia. Forests 2022, 13, 715. [Google Scholar] [CrossRef]
- Bhandari, M.S.; Meena, R.K.; Shankhwar, R.; Shekhar, C.; Saxena, J.; Kant, R.; Pandey, V.V.; Barthwal, S.; Pandey, S.; Chandra, G.; et al. Prediction Mapping Through Maxent Modeling Paves the Way for the Conservation of Rhododendron arboreum in Uttarakhand Himalayas. J. Indian Soc. Remote Sens. 2020, 48, 411–422. [Google Scholar] [CrossRef]
- Kogo, B.K.; Kumar, L.; Koech, R.; Kariyawasam, C.S. Modelling Climate Suitability for Rainfed Maize Cultivation in Kenya Using a Maximum Entropy (MaxENT) Approach. Agronomy 2019, 9, 727. [Google Scholar] [CrossRef]
- Ying, B.; Tian, K.; Guo, H.; Yang, X.; Li, W.; Li, Q.; Luo, Y.; Zhang, X. Prediction potential suitable habitats of Kandelia obvata in China under future climate scenarios based on MaxEnt model. Acta Ecol. Sin. 2024, 44, 224–234. [Google Scholar] [CrossRef]
- Thornton, P.K.; Jones, P.G.; Alagarswamy, G.; Andresen, J. Spatial variation of crop yield response to climate change in East Africa. Glob. Environ. Chang.-Hum. Policy Dimens. 2009, 19, 54–65. [Google Scholar] [CrossRef]
- He, Q.; Zhou, G. Climate-associated distribution of summer maize in China from 1961 to 2010. Agric. Ecosyst. Environ. 2016, 232, 326–335. [Google Scholar] [CrossRef]
- Raddad, E.Y.; Luukkanen, O. Adaptive genetic variation in water-use efficiency and gum yield in Acacia senegal provenances grown on clay soil in the Blue Nile region, Sudan. For. Ecol. Manag. 2006, 226, 219–229. [Google Scholar] [CrossRef]
- Benning, J.W.; Moeller, D.A. Plant-soil interactions limit lifetime fitness outside a native plant’s geographic range margin. Ecology 2021, 102, e03254. [Google Scholar] [CrossRef]
- Garcia, K.; Lasco, R.; Ines, A.; Lyon, B.; Pulhin, F. Predicting geographic distribution and habitat suitability due to climate change of selected threatened forest tree species in the. Philippines. Appl. Geogr. 2013, 44, 12–22. [Google Scholar] [CrossRef]
- Parmesan, C.; Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 2003, 421, 37–42. [Google Scholar] [CrossRef]
- Li, J.; Chang, H.; Liu, T.; Zhang, C. The potential geographical distribution of Haloxylon across Central Asia under climate change in the 21st century. Agric. For. Meteorol. 2019, 275, 243–254. [Google Scholar] [CrossRef]
- Bertrand, R.; Lenoir, J.; Piedallu, C.; Riofrio-Dillon, G.; de Ruffray, P.; Vidal, C.; Pierrat, J.-C.; Gegout, J.-C. Changes in plant community composition lag behind climate warming in lowland forests. Nature 2011, 479, 517–520. [Google Scholar] [CrossRef]
- Tang, M.; Chen, J.; Yang, L.; Jia, X.; Liu, J.; Duan, J. Distribution and change of major biofuel tree species in China under climate change. Acta Ecol. Sin. 2023, 43, 10156–10170. [Google Scholar] [CrossRef]
- Ashraf, U.; Ali, H.; Chaudry, M.N.; Ashraf, I.; Batool, A.; Saqib, Z. Predicting the Potential Distribution of Olea ferruginea in Pakistan incorporating Climate Change by Using Maxent Model. Sustainability 2016, 8, 722. [Google Scholar] [CrossRef]
- Ali, K.; Ahmad, H.; Khan, N.; Jury, S. Future of Abies pindrow in Swat district, northern Pakistan. J. For. Res. 2014, 25, 211–214. [Google Scholar] [CrossRef]
- Abdelaal, M.; Fois, M.; Fenu, G.; Bacchetta, G. Using MaxEnt modeling to predict the potential distribution of the endemic plant Rosa arabica Crep. in Egypt. Ecol. Inform. 2019, 50, 68–75. [Google Scholar] [CrossRef]
- Gebrewahid, Y.; Abrehe, S.; Meresa, E.; Eyasu, G.; Abay, K.; Gebreab, G.; Kidanemariam, K.; Adissu, G.; Abreha, G.; Darcha, G. Current and future predicting potential areas of Oxytenanthera abyssinica (A. Richard) using MaxEnt model under climate change in Northern Ethiopia. Ecol. Process. 2020, 9, 6. [Google Scholar] [CrossRef]
- Puchalka, R.; Dyderski, M.K.; Vitkova, M.; Sadlo, J.; Klisz, M.; Netsvetov, M.; Prokopuk, Y.; Matisons, R.; Mionskowski, M.; Wojda, T.; et al. Black locust (Robinia pseudoacacia L.) range contraction and expansion in Europe under changing climate. Glob. Chang. Biol. 2021, 27, 1587–1600. [Google Scholar] [CrossRef]
- Bradley, B.A.; Wilcove, D.S.; Oppenheimer, M. Climate change increases risk of plant invasion in the Eastern United States. Biol. Invasions 2010, 12, 1855–1872. [Google Scholar] [CrossRef]
- Arshad, F.; Waheed, M.; Fatima, K.; Harun, N.; Iqbal, M.; Fatima, K.; Umbreen, S. Predicting the Suitable Current and Future Potential Distribution of the Native Endangered Tree Tecomella undulata (Sm.) Seem. in Pakistan. Sustainability 2022, 14, 7215. [Google Scholar] [CrossRef]
- Qazi, A.W.; Saqib, Z.; Zaman-ul-Haq, M.; Gardezi, S.M.H.; Khan, A.M.; Khan, I.; Munir, A.; Ahmed, I. Modelling Impacts of Climate Change on Habitat Suitability of Three Endemic Plant Species in Pakistan. Pol. J. Environ. Stud. 2023, 32, 3281–3290. [Google Scholar] [CrossRef]
- Gilani, H.; Goheer, M.A.; Ahmad, H.; Hussain, K. Under predicted climate change: Distribution and ecological niche modelling of six native tree species in Gilgit-Baltistan, Pakistan. Ecol. Indic. 2020, 111, 106049. [Google Scholar] [CrossRef]
- Gaafar, A.M.; Salih, A.A.; Luukkanen, O.; El Fadl, M.A.; Kaarakka, V. Improving the traditional Acacia senegal-crop system in sudan: The effect of tree density on water use, gum production and crop yields. Agrofor. Syst. 2006, 66, 1–11. [Google Scholar] [CrossRef]
- Githae, E.W.; Gachene, C.K.K.; Njoka, J.T.; Omondi, S.F. Nitrogen Fixation by Natural Populations of Acacia Senegal in the Drylands of Kenya Using 15N Natural Abundance. Arid Land Res. Manag. 2013, 27, 327–336. [Google Scholar] [CrossRef]
- Diallo, A.M.; Nielsen, L.R.; Kjaer, E.D.; Petersen, K.K.; Raebild, A. Polyploidy can Confer Superiority to West African Acacia senegal (L.) Willd. Trees. Front. Plant Sci. 2016, 7, 821. [Google Scholar] [CrossRef] [PubMed]
- Akram, M.; Kahlown, M.A.; Soomro, Z.A. Desertification Control for Sustainable Land Use in the Cholistan Desert, Pakistan. In Proceedings of the International Scientific Conference on Desertification and Drylands Research, Tunis, Tunisia, 19–21 June 2006; Springer: Dordrecht, The Netherlands, 2008; pp. 483–492. [Google Scholar]
- Khan, A.N.; Ali, A. Desertification Risk Reduction Approaches in Pakistan. In Disaster Risk Reduction Approaches in Pakistan; AttaUrRahman, F.R.S., Khan, A.N., Shaw, R., Eds.; Disaster Risk Reduction; Springer: Tokyo, Janpan, 2015; pp. 161–173. [Google Scholar]
- Anjum, S.A.; Wang, L.-C.; Xue, L.-L.; Saleem, M.F.; Wang, G.-X.; Zou, C.-M. Desertification in Pakistan: Causes, impacts and management. J. Food Agric. Environ. 2010, 8, 1203–1208. [Google Scholar]
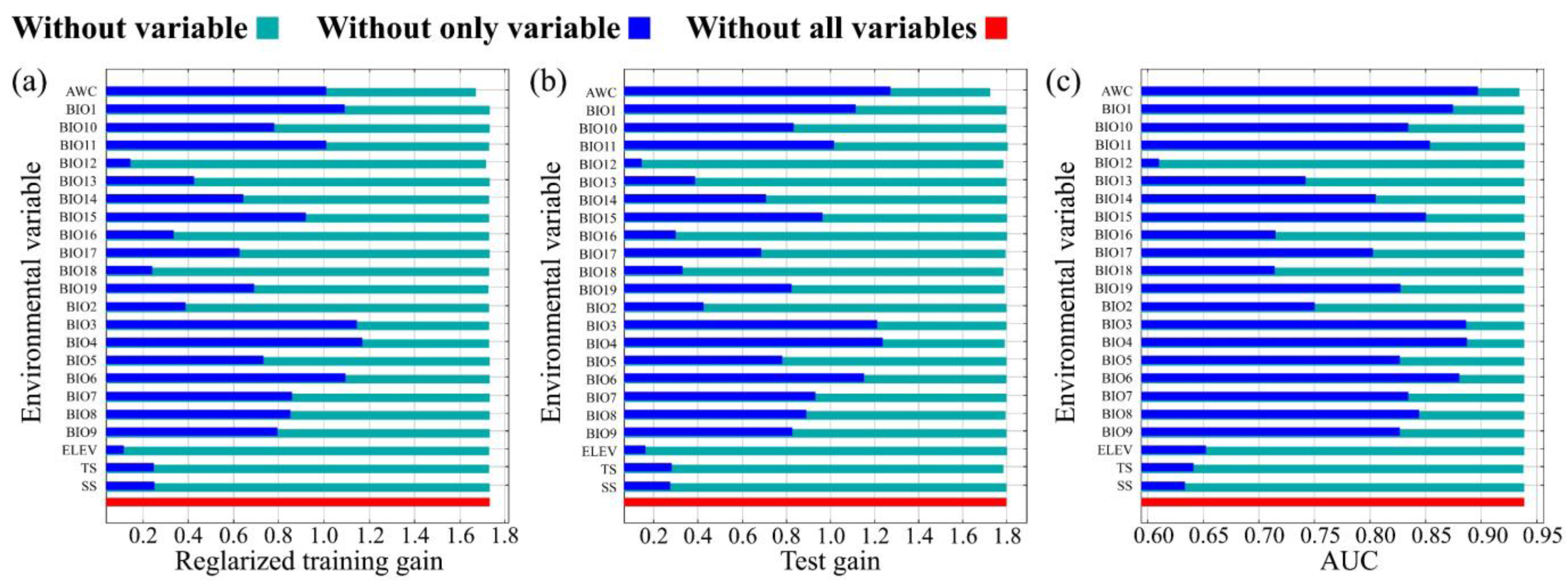


| Number | Variable | Percent Contribution | Permutation Importance |
|---|---|---|---|
| 1 | Isothermality (BIO3) | 30.5 | 8.1 |
| 2 | Precipitation of driest month (BIO14) | 12.7 | 0.3 |
| 3 | Mean temperature of coldest quarter (BIO11) | 10 | 33 |
| 4 | Precipitation of coldest quarter (BIO19) | 5.9 | 1.4 |
| 5 | Annual mean temperature (BIO1) | 5.5 | 0 |
| 6 | Annual precipitation (BIO12) | 5.3 | 22.4 |
| 7 | Precipitation seasonality (BIO15) | 4.6 | 3.3 |
| 8 | Precipitation of wettest month (BIO13) | 3.7 | 0 |
| 9 | Available Water Content (AWC) | 3.6 | 5.7 |
| 10 | Min temperature of coldest month (BIO6) | 3.2 | 10.5 |
| 11 | Max temperature of warmest month (BIO5) | 2.6 | 0 |
| 12 | Mean temperature of warmest quarter (BIO10) | 2.3 | 0.6 |
| 13 | Mean temperature of wettest quarter (BIO8) | 2 | 0.1 |
| 14 | Mean diurnal range (BIO2) | 1.9 | 0.1 |
| 15 | Precipitation of wettest quarter (BIO16) | 1.3 | 2.6 |
| 16 | Temperature seasonality (BIO4) | 1.1 | 6 |
| 17 | Precipitation of warmest quarter (BIO18) | 0.9 | 1.9 |
| 18 | Mean temperature of driest quarter (BIO9) | 0.7 | 0 |
| 19 | Elevation (Elev) | 0.6 | 1.1 |
| 20 | Temperature annual range (BIO7) | 0.5 | 1 |
| 21 | Topsoil salinity (TS) | 0.5 | 1.2 |
| 22 | Precipitation of driest quarter (BIO17) | 0.2 | 0.7 |
| 23 | Subsoil salinity (SS) | 0.2 | 0 |
| Environmental Variables | Climate Scenario | Current | 2050s | 2070s | 2090s |
|---|---|---|---|---|---|
| BIO3 | SSP1-2.6 | 48.65 | 45.14 | 50.44 | 44.51 |
| SSP2-4.5 | 48.65 | 43.19 | 42.65 | 39.62 | |
| SSP3-7.0 | 48.65 | 43.48 | 43.59 | 39.29 | |
| SSP5-8.5 | 48.65 | 41.43 | 38.48 | 37.08 | |
| BIO4 | SSP1-2.6 | 550.62 | 626.35 | 563.49 | 744.97 |
| SSP2-4.5 | 550.62 | 653.49 | 744.72 | 768.59 | |
| SSP3-7.0 | 550.62 | 642.31 | 704.32 | 737.19 | |
| SSP5-8.5 | 550.62 | 743.82 | 753.74 | 756.59 | |
| BIO11/℃ | SSP1-2.6 | 16.81 | 15.46 | 21.31 | 19.30 |
| SSP2-4.5 | 16.81 | 17.98 | 19.79 | 20.07 | |
| SSP3-7.0 | 16.81 | 15.52 | 21.24 | 21.33 | |
| SSP5-8.5 | 16.81 | 19.55 | 21.60 | 22.83 | |
| BIO12/mm | SSP1-2.6 | 424.00 | 507.50 | 245.25 | 145.25 |
| SSP2-4.5 | 424.00 | 285.75 | 145.25 | 129.00 | |
| SSP3-7.0 | 424.00 | 454.75 | 165.25 | 129.00 | |
| SSP5-8.5 | 424.00 | 121.75 | 132.75 | 122.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, J.; Shi, J.; Zhang, P.; Shao, M.; Zhou, N.; Wang, Y.; Xu, X. Potential Distribution Projections for Senegalia senegal (L.) Britton under Climate Change Scenarios. Forests 2024, 15, 379. https://doi.org/10.3390/f15020379
Fang J, Shi J, Zhang P, Shao M, Zhou N, Wang Y, Xu X. Potential Distribution Projections for Senegalia senegal (L.) Britton under Climate Change Scenarios. Forests. 2024; 15(2):379. https://doi.org/10.3390/f15020379
Chicago/Turabian StyleFang, Jiaqi, Jianfei Shi, Ping Zhang, Minghao Shao, Na Zhou, Yongdong Wang, and Xinwen Xu. 2024. "Potential Distribution Projections for Senegalia senegal (L.) Britton under Climate Change Scenarios" Forests 15, no. 2: 379. https://doi.org/10.3390/f15020379







