Tree Growth Rings in Tropical Peat Swamp Forests of Kalimantan, Indonesia
Abstract
:1. Introduction
2. Sites, Material and Methods
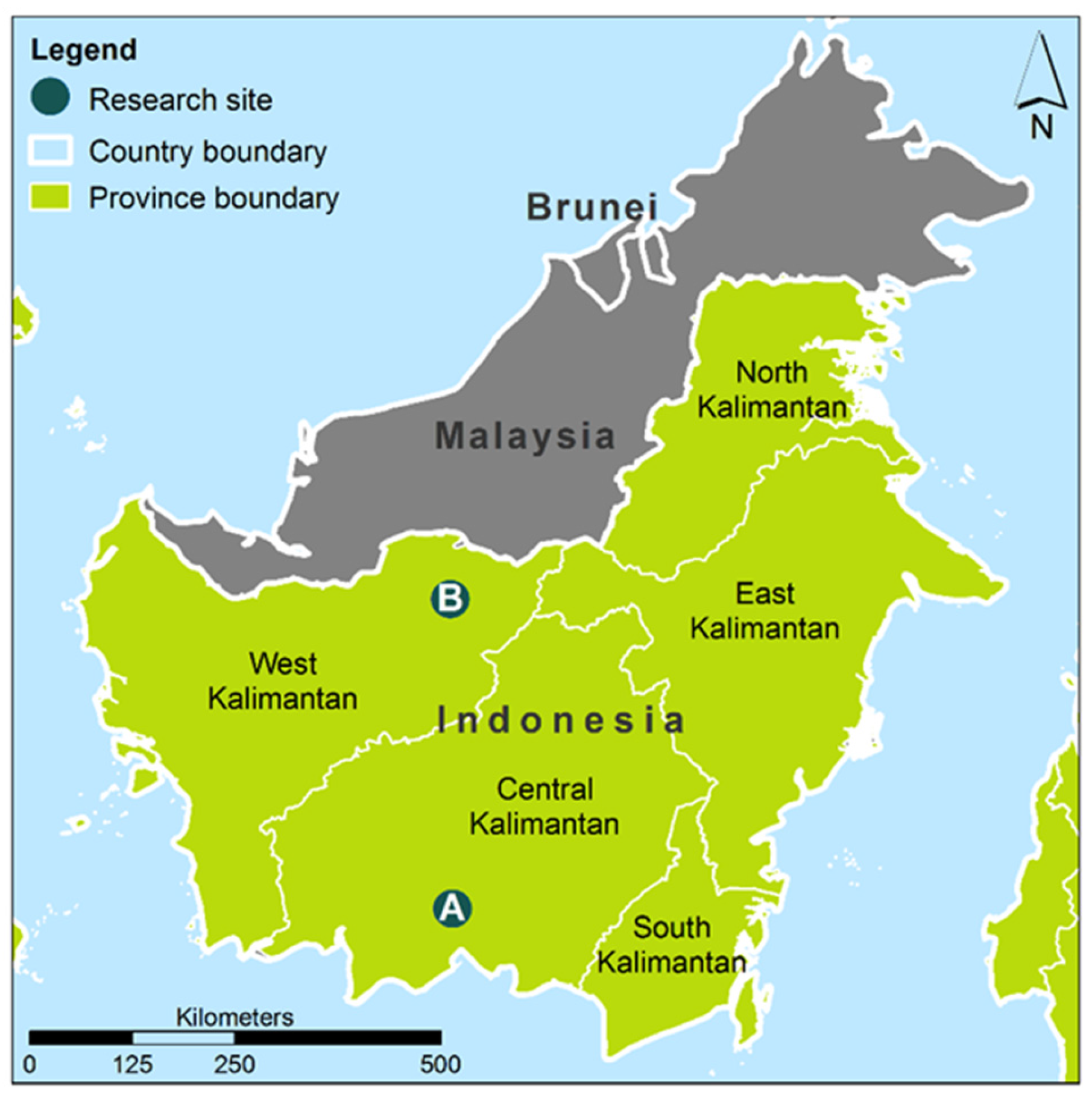
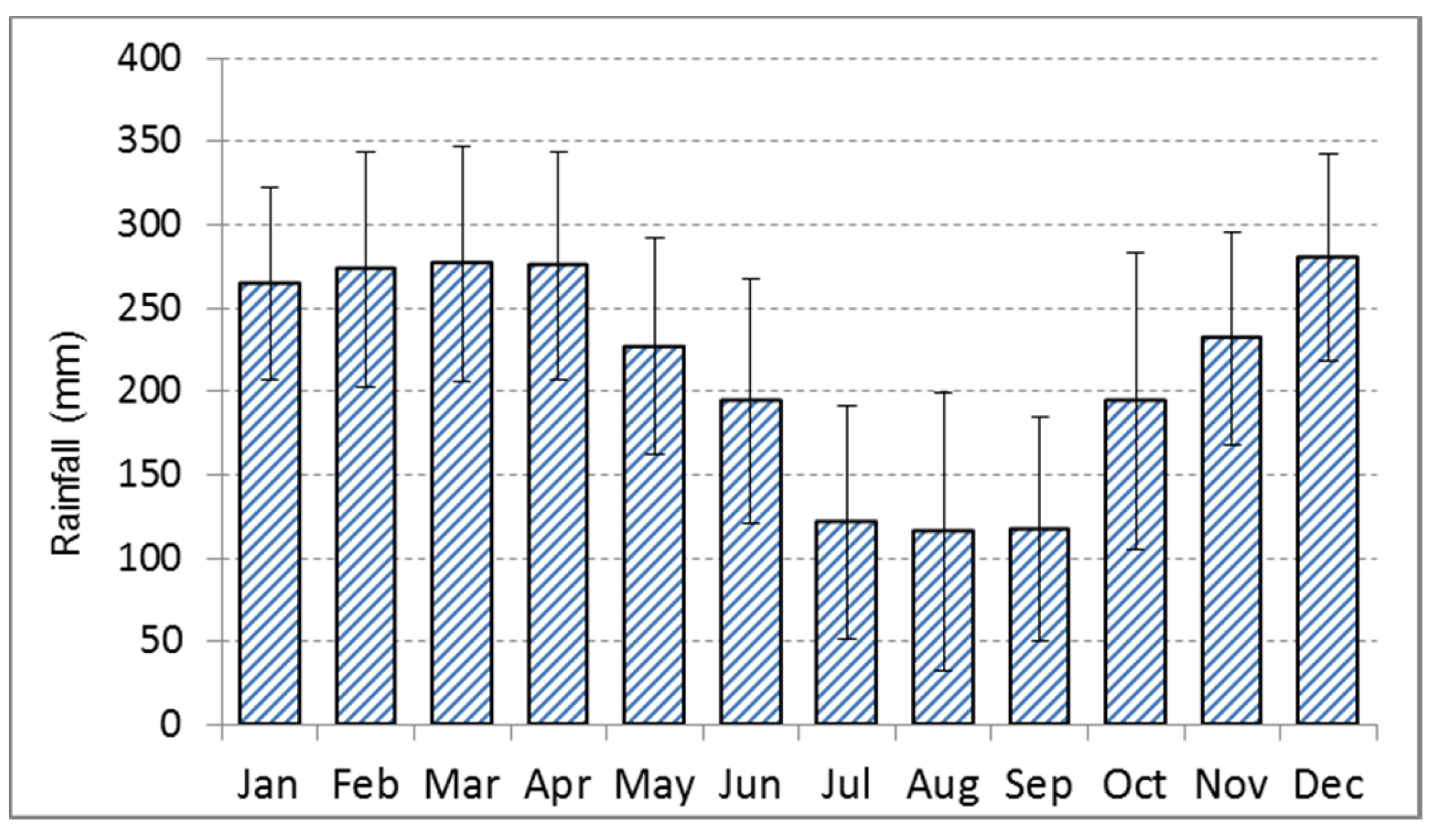
2.1. Study Sites, Climate and Sample Collection
2.2. Disc Treatment and Analysis
3. Results
3.1. Wood Anatomy
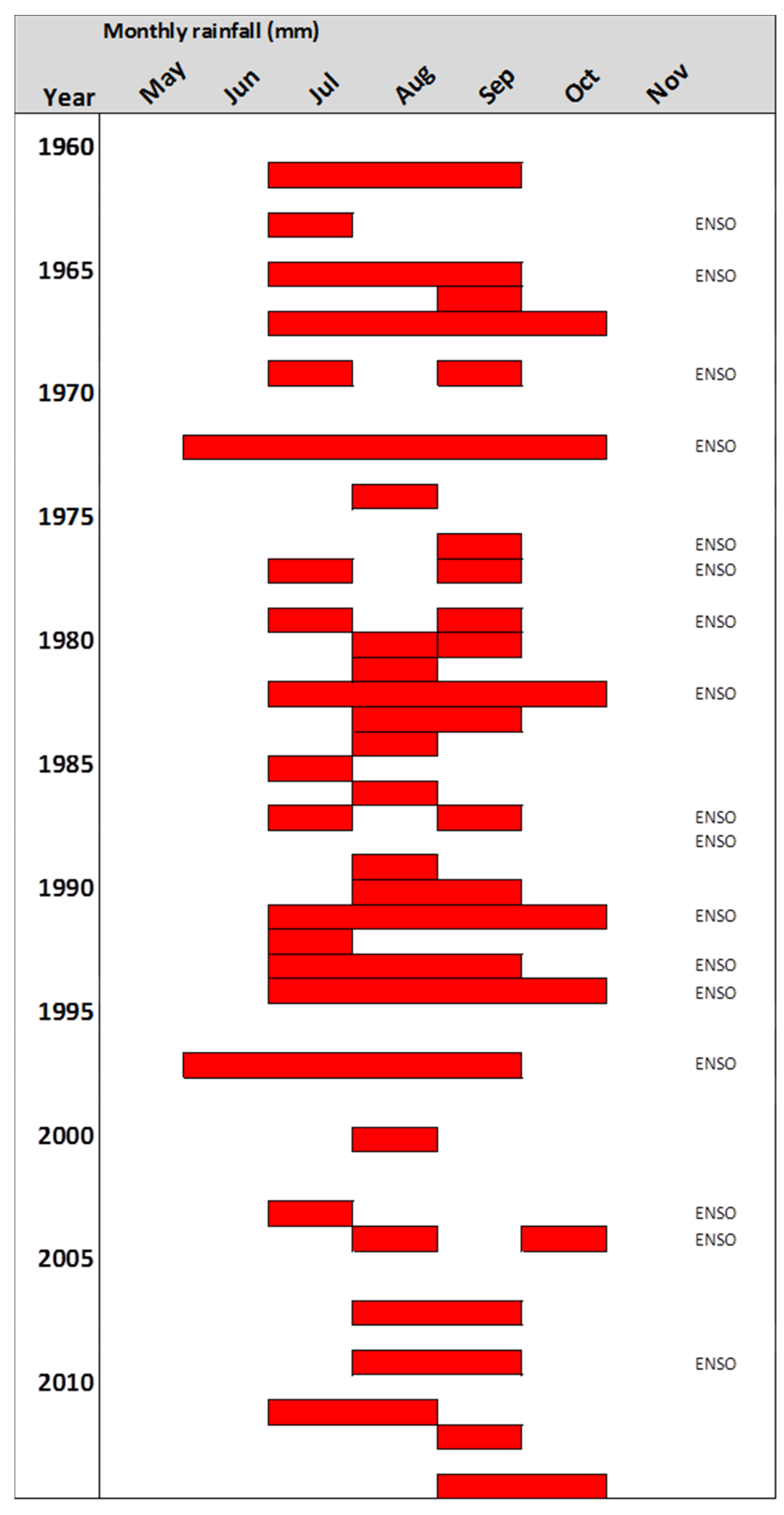
3.2. Periodicity of Ring Formation
3.3. Ring Curves and Growth Rates
4. Discussion
- (1)
- A periodically occurring stress factor induces cambial dormancy and consequently tree rings in the wood.
- (2)
- (3)
- In Diospyros, we found distinct growth zones (Figure 4b) based on density variations which obviously are not annual, but point to a stress factor with a non-annual periodicity.
- (4)
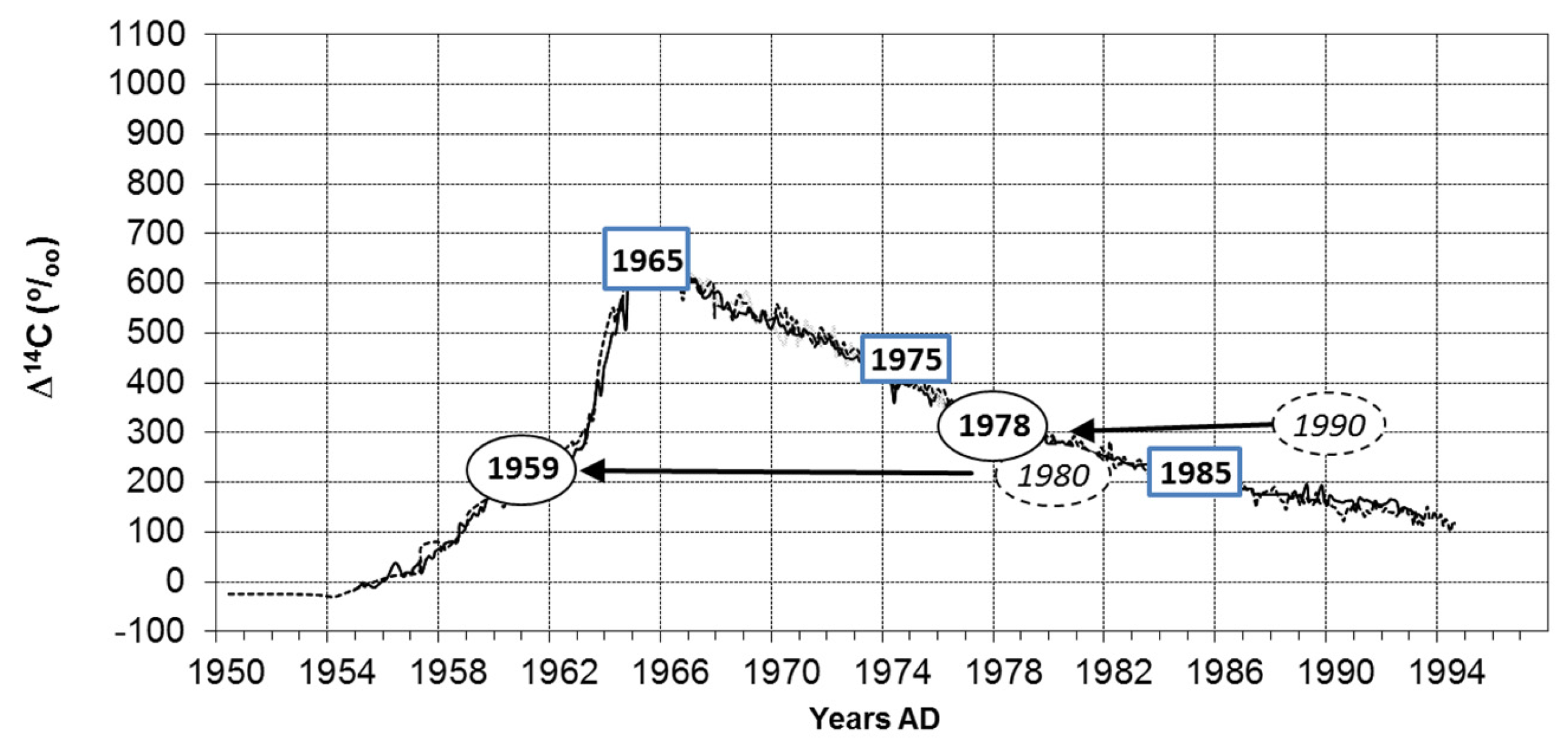
- For Horsfieldia, we demonstrated the existence of annual rings based on two independent methods. Successful radiocarbon based dating is supported by the similarity of time series from tree rings and precipitation (e.g., [49]). The anatomical feature of growth ring boundaries is the parenchyma bands as limits of growth zones overlaying less frequent patterns of density variations.
- (5)
- In a next step, we measured ring width curves from all species with marginal parenchyma bands as a growth zone delimiter. The successful cross dating of the tree ring sequences from Magnolia and Calophyllum with those from Horsfieldia indicates the annual nature of the growth rings in these species as well. The time series from Knema intermedia is too short for significant statistical comparisons but visually shows a good congruence with Horsfieldia and Calophyllum samples and the dry period rainfall curve. These findings indicate that all species with marginal parenchyma bands form annual rings.
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schweingruber, F.H. Tree Rings and Environment: Dendroecology; Paul Haupt: Berne, Switzerland, 1996; p. 609. [Google Scholar]
- Schimper, A.F.W. Pflanzen Geographie auf Physiologischer; Grundlage: Jena, Germany, 1898. [Google Scholar]
- Whitmore, T.C. Tropical Rain Forests of the Far East; Clarendon Press: Oxford, UK, 1984; p. 352. [Google Scholar]
- Walter, H.; Lieth, H. Klimadiagramm-Weltatlas; Gustav Fischer Verlag: Jena, Germany, 1967. [Google Scholar]
- Worbes, M. How to measure growth dynamics in tropical trees a review. IAWA J. 1995, 16, 337–351. [Google Scholar] [CrossRef]
- Klebs, G. Über die periodischen Erscheinungen tropischer Pflanzen. Biol. Cent. 1912, 32, 257–285. [Google Scholar]
- Geiger, F. Anatomische Untersuchungen über die Jahresringbildung von Tectona grandis. Jahrb. Wiss. Bot. 1915, 55, 521–607. [Google Scholar]
- Coster, C. Zur Anatomie und Physiologie der Zuwachszonen und Jahresringbildung in den Tropen. Ann. Jard. Bot. Buitenzorg 1927, 37, 49–161. [Google Scholar]
- Coster, C. Zur Anatomie und Physiologie der Zuwachszonen und Jahresringbildung in den Tropen. Ann. Jard. Bot. Buitenzorg 1928, 38, 1–114. [Google Scholar]
- Worbes, M. One Hundred Years of Tree ring Research in the Tropics—A Brief History and an Outlook to Future Challenges. Dendrochronologia 2002, 20, 217–231. [Google Scholar] [CrossRef]
- D’Arrigo, R.D.; Jacoby, G.C.; Krusic, P.J. Progress in dendroclimatic studies in Indonesia. TAO 1994, 5, 349–363. [Google Scholar]
- D’Arrigo, R.; Wilson, R.; Palmer, J.; Krusic, P.; Curtis, A.; Sakulich, J.; Bijaksana, S.; Zulaikah, S.; Ngkoimani, L.O.; Tudhope, A. The reconstructed Indonesian warm pool sea surface temperatures from tree rings and corals: Linkages to Asian monsoon drought and El Nino-Southern Oscillation. Paleoceanography 2006, 21. [Google Scholar] [CrossRef]
- Hennig, K.; Helle, G.; Heinrich, I.; Neuwirth, B.; Karyanto, O.; Winiger, M. Toward multi-parameter records (ring width, δ13C, δ18O) from tropical tree-rings—A case study on Tectona grandis from Java, Indonesia. In TRACE—Tree Rings in Archaeology, Climatology and Ecology; GFZ Potsdam, Scientific Technical Report STR 11/07; Maaten-Theunissen, M., Spiecker, H., Eds.; GFZ: Potsdam, Germany, 2011; Volume 9, pp. 158–165. [Google Scholar]
- Schollaen, K.; Karamperidou, C.; Krusic, P.; Cook, E.; Helle, G. Multiple tree-ring chronologies (ring width, δ13C and δ18O) reveal dry and rainy season signals of rainfall in Indonesia. Quat. Sci. Rev. 2013, 73, 170–181. [Google Scholar] [CrossRef]
- Worbes, M. Growth rings, increment and age of trees in inundation forests, savannas and a mountain forest in the Neotropics. IAWA Bull. 1989, 10, 109–122. [Google Scholar] [CrossRef]
- Schöngart, J.; Piedade, M.F.T.; Ludwigshausen, S.; Horna, V.; Worbes, M. Phenology and stem-growth periodicity of tree species in Amazonian floodplain forests. J. Trop. Ecol. 2002, 18, 581–597. [Google Scholar] [CrossRef]
- Huijnen, V.; Wooster, M.J.; Kaiser, J.W.; Gaveau, D.L.A.; Flemming, J.; Parrington, M.; Inness, A.; Murdiyarso, D.; Main, B.; van Weele, M. Fire carbon emissions over maritime southeast Asia in 2015 largest since 1997. Sci. Rep. 2016, 6, 26886. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.J.; Boesch, H.; Wooster, M.J.; Moore, D.P.; Webb, A.J.; Gaveau, D.; Murdiyarso, D. Atmospheric CH4 and CO2 enhancements and biomass burning emission ratios derived from satellite observations of the 2015 Indonesian fire plumes. Atmos. Chem. Phys. 2016, 16, 10111–10131. [Google Scholar] [CrossRef]
- Hirano, T.; Jauhiainen, J.; Inoue, T.; Takahashi, H. Controls on the Carbon Balance of Tropical Peatlands. Ecosystems 2009, 12, 873–887. [Google Scholar] [CrossRef]
- Dommain, R.; Couwenberg, J.; Joosten, H. Hydrological self-regulation of domed peatlands in south-east Asia and consequences for conservation and restoration. Mires Peat 2010, 6, 1–17. [Google Scholar]
- Junk, W.J.; Bayley, P.B.; Sparks, R.E. The flood pulse concept in river-floodplain-systems. In Canadian Special Publication of Fisheries and Aquatic Sciences 106, Proceedings of the International Large River Symposium, Honey Harbour, ON, Canada, 14–21 September 1989; Dodge, D.P., Ed.; Fisheries and Oceans: Ottawa, ON, Canada, 1989; pp. 110–127. [Google Scholar]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high-resolution grids of monthly climatic observations—The CRU TS3.10 Dataset. Int. J. Climatol. 2014, 34, 623–642. [Google Scholar] [CrossRef] [Green Version]
- Page, S.E.; Rieley, J.O.; Wust, R.A.J. Lowland tropical peatlands of Southeast Asia. In Peatlands: Basin Evolution and Depository of Records on Global Environmental and Climatic Changes; Developments in Earth Surface Processes Series; Martini, P., Martinez-Cortizas, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 145–172. [Google Scholar]
- Breckle, S.W. Walter’s Vegetation of the Earth; Springer: New York, NY, USA, 2002; p. 527. [Google Scholar]
- Gonzalez de Tanago, J.; Lau, A.; Bartholomeus, H.; Herold, M.; Avitabile, V.; Raumonen, P.; Martius, C.; Goodman, R.C.; Disney, M.; Manuri, S.; et al. Estimation of above-ground biomass of large tropical trees with Terrestrial LiDAR. Methods Ecol. Evol. 2017. accepted. [Google Scholar]
- Tarelkin, Y.; Delvaux, C.; De Ridder, M.; El Berkani, T.; De Cannière, C.; Beeckman, H. Growth-ring distinctness and boundary anatomy variability in tropical trees. IAWA J. 2016, 37, 275–294. [Google Scholar] [CrossRef]
- Worbes, M.; Junk, W.J. Dating tropical trees by means of 14C from bomb tests. Ecology 1989, 70, 503–507. [Google Scholar] [CrossRef]
- Schweingruber, F.H. Tree Rings—Basics and Applications; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1988; p. 276. [Google Scholar]
- Roig, F.A.; Jimenez Osornia, J.J.; Villanueva Diaz, J.; Luckman, B.; Tiessen, H.; Medina, A.; Noellemeyer, E.J. Anatomy of growth rings at the Yucatán Peninsula. Dendrochronologia 2005, 22, 187–193. [Google Scholar] [CrossRef]
- Worbes, M.; Raschke, N. Carbon allocation in a Costa Rican dry forest derived from tree ring analysis. Dendrochronologia 2012, 30, 231–238. [Google Scholar] [CrossRef]
- Nydal, R.; Lövseth, K. Tracing Bomb 14C in the Atmosphere 1962–1980. J. Geophys. Res. 1983, 88, 3621–3642. [Google Scholar] [CrossRef]
- Hua, Q.; Barbetti, M.; Worbes, M.; Head, J.; Levchenko, V.A. Review of Radiocarbon Data from Atmospheric and Tree Ring Samples for the Period 1950–1977 AD. IAWA J. 1999, 20, 261–284. [Google Scholar] [CrossRef]
- Brienen, R.J.W.; Schöngart, J.; Zuidema, P.A. Tree rings in the tropics: Insights into the ecology and climate sensitivity of tropical trees. Trop. Tree Physiol. 2016, 6, 439–461. [Google Scholar]
- Jacoby, G.C. Overview of tree-ring analysis in tropical regions. IAWA Bull. 1989, 10, 99–108. [Google Scholar] [CrossRef]
- Gourlay, I.D. The definition of seasonal growth zones in some African Acacia species—A review. IAWA J. 1995, 16, 353–359. [Google Scholar] [CrossRef]
- Dezzeo, N.; Worbes, M.; Ishil, I.; Herrera, R. Growth rings analysis of four tropical tree species in seasonally flooded forest of the Mapire River, a tributary of the lower Orinoco River, Venezuela. Plant Ecol. 2003, 168, 165–175. [Google Scholar] [CrossRef]
- Worbes, M. The Forest Ecosystems of the Floodplains. In The Amazonian Floodplains; Junk, W.J., Ed.; Ecology of a Pulsing System; Ecological Studies 126; Springer: New York, NY, USA, 1997; pp. 223–265. [Google Scholar]
- Pumijumnong, N. Dendrochronology in Southeast Asia. Trees 2013, 27, 343–358. [Google Scholar] [CrossRef]
- Sass, U.; Killmann, W.; Eckstein, D. Wood Formation in Two Species of Dipterocarpaceae in Peninsular Malaysia. IAWA J. 1995, 16, 371–384. [Google Scholar] [CrossRef]
- Robertson, I.; Froyd, C.A.; Walsh, R.P.D.; Newbery, D.M.; Woodborne, S.; Ong, R.C. The dating of dipterocarp tree rings: Establishing a record of carbon cycling and climatic change in the tropics. J. Quat. Sci. 2004, 19, 657–664. [Google Scholar] [CrossRef]
- Azim, A.A.A.; Okada, N. Occurence and anatomical feature of growth rings in tropical rainforest trees in Peninsular Malaysia: A preliminary study. Tropics 2014, 23, 15–31. [Google Scholar] [CrossRef]
- Nath, C.D.; Munoz, F.; Pélissier, R.; Burslem, D.F.R.P.; Muthusankar, G. Growth rings in tropical trees: Role of functional traits, environment, and phylogeny. Trees 2016, 30, 2153–2175. [Google Scholar] [CrossRef]
- Ogata, Y.; Nobuchi, T.; Fujita, M.; Sahri, M.H. Growth rings and tree growth in young para rubber trees from Peninsular Malaysia. IAWA J. 2001, 22, 43–56. [Google Scholar] [CrossRef]
- Newbery, D.M.; van der Burgt, X.M.; Worbes, M.; Chuyong, G.B. Transient dominance in a central African rain forest. Ecol. Monogr. 2013, 83, 339–382. [Google Scholar] [CrossRef]
- Hazlett, D. Seasonal cambial activity for Pentaclethra, Goelthalsia, and Carapa trees in a Costa Rica lowland forest. Biotropica 1987, 19, 357–360. [Google Scholar] [CrossRef]
- Fichtler, E.; Clark, D.A.; Worbes, M. Age and Long-Term Growth of Trees in an Old-Growth Tropical Rain Forest, Based on Analyses of Tree Rings and 14C1. Biotropica 2003, 35, 306–317. [Google Scholar] [CrossRef]
- Sundari, S.; Hirano, T.; Yamada, H.; Kusin, K.; Limin, S. Effect of groundwater level on soil respiration in tropical peat swamp forests. J. Agric. Meteorol. 2012, 68, 121–134. [Google Scholar] [CrossRef]
- Ong, C.S.P.; Juan, J.C.; Yule, C.M. Litterfall production and chemistry of Koompassia malaccensis and Shorea uliginosa in a tropical peat swamp forest: Plant nutrient regulation and climate relationship. Trees 2015, 29, 527–537. [Google Scholar] [CrossRef]
- Schöngart, J.; Gribel, R.; Ferreira da Fonseca-Junior, S.; Haugaasen, T. Age and growth patterns of Brazil Nut trees (Bertholletia excelsa Bonpl.) in Amazonia, Brazil. Biotropica 2015, 47, 550–558. [Google Scholar]
- Worbes, M.; Blanchart, S.; Fichtler, E. Relations between water balance, wood traits and phenological behavior of tree species from a tropical dry forest in Costa Rica—A multifactorial study. Tree Physiol. 2013, 33, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Trouet, V.; Mukelabai, M.; Verheyden, A.; Beeckman, H. Cambial growth season of brevi-deciduous Brachystegia spiciformis trees from South Central Africa restricted to four months. PLoS ONE 2012, 7, e47364. [Google Scholar] [CrossRef] [PubMed]
- Szejner, P. Tropical Dendrochronology: Exploring Tree Rings of Pinus oocarpa in Eastern Guatemala. Master’s Thesis, University of Goettingen, Goettingen, Germany, 2011. [Google Scholar]
- Baker, P.J.; Bunyavejchewin, S.; Oliver, C.D.; Ashton, P.S. Disturbance history and historical stand dynamics of a seasonal tropical forest in western Thailand. Ecol. Monogr. 2005, 75, 317–343. [Google Scholar] [CrossRef]
- Groenendijk, P.; Sass-Klaassen, U.; Bongers, F.; Zuidema, P.A. Potential of tree-ring analysis in a wet tropical forest: A case study on 22 commercial tree species in Central Africa. For. Ecol. Manag. 2014, 323, 65–78. [Google Scholar] [CrossRef]
- Worbes, M.; Staschel, R.; Roloff, A.; Junk, W.J. Tree Ring Analysis Reveals Age Structure, Dynamics and Wood Production of a Natural Forest Stand in Cameroon. For. Ecol. Manag. 2003, 173, 105–123. [Google Scholar] [CrossRef]
- Daryono, H. Potensi, permasalahan dan kebijakan yang diperlukan dalam pengelolaan hutan dan lahan rawa gambut secara lestari. J. Anal. Kebijak. Kehutan. 2009, 6, 71–101. [Google Scholar]


| No. | Family | Species | Author | DBH (cm) | Wood Density g/cm3 | Growth Zone Types | Distinctiveness | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Density Variation | Marginal Parenchyma | Pattern of Fiber/Parenchyma | Vessel Distribution | |||||||
| 1 | Anacardiaceae | Campnosperma coriaceum | (Jack) Hallier f. | 31.6 | 0.43 ± 0.06 | X | X | +/− | ||
| 2 | Annonaceae | Xylopia fusca | Maingayi ex Hook. f. and Thomson | 28.0 | 0.34 ± 0.00 | X | − | |||
| 3 | Annonaceae | Mezzettia umbellata | Becc. | 15.8 | 0.65 ± 0.1 * | X | +/− | |||
| 4 | Annonaceae | Mezzettia parviflora | Becc. | 17.5 | 0.65 ± 0.1 * | X | +/− | |||
| 5 | Apocynaceae | Alstonia pneumatophora | Baker ex Den Berger | 27.3 | 0.33 ± 0.05 | X | − | |||
| 6 | Chrysobalanaceae | Parastemon urophyllus | (Wall. ex A.DC.)A. DC. | 38.8 | 0.96 ± 0.1 | X | X | +/− | ||
| 7 | Clusiaceae | Calophyllum hosei | Ridl. | 21.9 | 0.61 ± 0.10 * | X | + | |||
| 8 | Dipterocarpaceae | Shorea parvifolia | Dyer | 24.4 | 0.63 ± 0.14 * | X | X | + | ||
| 9 | Dipterocarpaceae | Shorea sp | 98.2 | 0.63 ± 0.14 * | X | X | + | |||
| 10 | Dipterocarpaceae | Shorea teysmanniana | Dyer ex. Brandis | 52.7 | 0.63 ± 0.14 * | X | X | + | ||
| 11 | Dipterocarpaceae | Dipterocarpus sp | 19.3 | 0.72 ± 0.1 * | X | X | − | |||
| 12 | Ebenaceae | Diospyros evena | Bakh. | 63.2 | 0.65 ± 0.70 | X | + | |||
| 13 | Euphorbiaceae | Hevea brasiliensis | (Willd. ex A.Juss.) Müll.Arg. | 19.0 | 0.48 ± 0.09 | X | + | |||
| 14 | Hypericaceae | Cratoxylum glaucum | Korth. | 33.9 | 0.54 ± 0.06 | X | X | + | ||
| 15 | Lauraceae | Dehaasia caesia | Blume | 21.6 | 0.77 ± 0.08 | X | − | |||
| 16 | Magnoliaceae | Magnolia bintuluensis | (A. Agostini) Noot. | 21.4 | 0.56 ± 0.02 | X | +/− | |||
| 17 | Meliaceae | Aglaia rubiginosa | (Hiern.) Pannell | 63.3 | 0.76 ± 0.11 * | X | + | |||
| 18 | Myristicaceae | Horsfieldia crassifolia | (Hook.f. and Thomson) Warb. | 32.1 | 0.48 ± 0.00 | X | + | |||
| 19 | Myristicaceae | Knema intermedia | Warb | 24.0 | 0.58 ± 0.06 * | X | + | |||
| 20 | Myrtaceae | Syzygium chloranthum | (Duthie) Merr. and L.M. Perry | 21.1 | 0.71 ± 0.10 * | X | X | − | ||
| 21 | Myrtaceae | Syzygium glaucum | (King) Chantaran. and J.Parn | 21.0 | 0.71 ± 0.10 | X | X | − | ||
| 22 | Myrtaceae | Syzygium syzygioides | (Miq.) Merr. and L.M.Perry | 27.3 | 0.85 ± 0.08 | X | X | +/− | ||
| 23 | Myrtaceae | Tristaniopsis merguensis | (Griff.) Peter G.Wilson and J.T. Waterh. | 12.6 | 1.05 ± 0.12 | ? | ? | ? | ? | − |
| 24 | Primulaceae | Rapanea borneensis | (Scheff.) Mez. | 20.7 | 0.72 ± 0.09 * | X | +/− | |||
| 25 | Sapotaceae | Palaquium ridleyi | King and Gamble | 17.1 | 0.39 ± 0.1 | X | X | +/− | ||
| 26 | Sapotaceae | Madhuca motleyana | (de Vriese) J.F.Macbr. | 29.8 | 0.53 ± 0.06 | X | X | − | ||
| 27 | Tetrameristaceae | Tetramerista glabra | Miq. | 57.6 | 0.73 ± 0.13 * | X | X | + | ||
| Growth Zone Type | Floodplain (Amazonia, 76 spp) | Dry Forest (Mexico, 52 spp) | Swamp Forest (Borneo, 26 spp) |
|---|---|---|---|
| Distinct (+) | 38 | 35 | 42 |
| More-or-less-distinct (+/−) | 51 | 52 | 27 |
| Indistinct (−) | 11 | 12 | 31 |
| Magnolia | Calophyllum | Chrono Horsfieldia | |
|---|---|---|---|
| Horsfieldia 3 | n.s. | 63 ** | 77 *** |
| Horsfieldia 4 | n.s. | n.s. | n.s. |
| Horsfieldia 15 | 67 ** | 60 * | 70 *** |
| Horsfieldia 40 | 53 (n.s.) | n.s. | 96 *** |
| Annual | n.s. | n.s. | 62 ** |
| Dry season | n.s. | n.s. | 61 ** |
| Rainy Season | n.s. | 59 * | n.s. |
| Period | Mean | Year <2000 | Year >2000 |
|---|---|---|---|
| Horsfieldia 3 | 0.21 ± 0.23 | 0.23 ± 0.16 | 0.64 ± 0.23 |
| Horsfieldia 4 | 0.34 ± 0.27 | 0.35 ± 0.29 | 0.34 ± 0.13 |
| Horsfieldia 15 | 0.29 ± 0.29 | 0.19 ± 0.14 | 0.75 ± 0.37 |
| Horsfieldia 40 | 0.30 ± 0.24 | 0.26 ± 0.19 | 0.58 ± 0.32 |
| Magnolia | 0.14 ± 0.09 | 0.15 ± 0.09 | 0.12 ± 0.05 |
| Calophyllum | 0.24 ± 0.09 | 0.23 ± 0.07 | 0.31 ± 0.13 |
| Chrono Horsfieldia | 0.27 ± 0.18 | 0.23 ± 0.15 | 0.45 ± 0.20 |
| Knema | 0.46 ± 0.20 | ||
| Diospyros | 0.26 ± 0.01 | ||
| Hevea (from terra firme rainforest) | 1.10 ± 0.10 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Worbes, M.; Herawati, H.; Martius, C. Tree Growth Rings in Tropical Peat Swamp Forests of Kalimantan, Indonesia. Forests 2017, 8, 336. https://doi.org/10.3390/f8090336
Worbes M, Herawati H, Martius C. Tree Growth Rings in Tropical Peat Swamp Forests of Kalimantan, Indonesia. Forests. 2017; 8(9):336. https://doi.org/10.3390/f8090336
Chicago/Turabian StyleWorbes, Martin, Hety Herawati, and Christopher Martius. 2017. "Tree Growth Rings in Tropical Peat Swamp Forests of Kalimantan, Indonesia" Forests 8, no. 9: 336. https://doi.org/10.3390/f8090336





