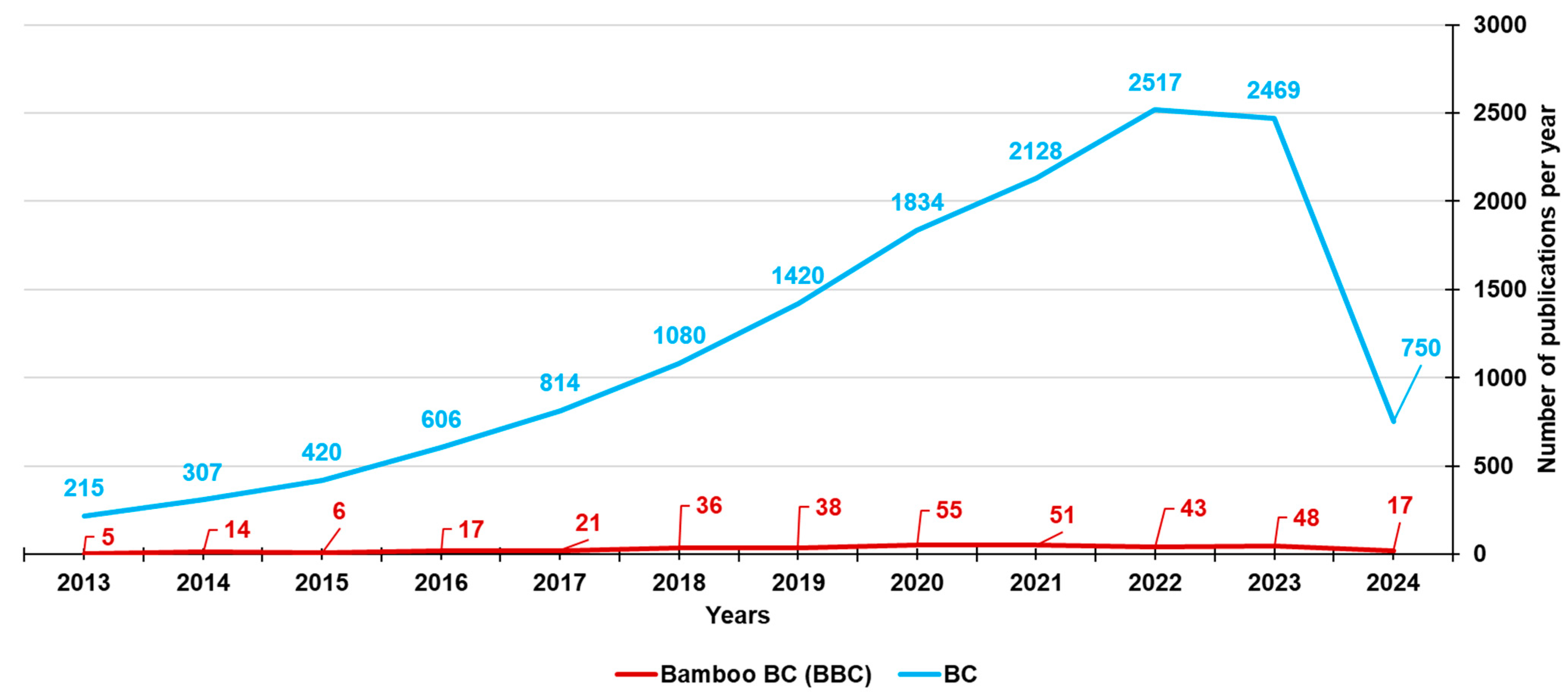
Bamboo-Based Biochar: A Still Too Little-Studied Black Gold and Its Current Applications
Abstract
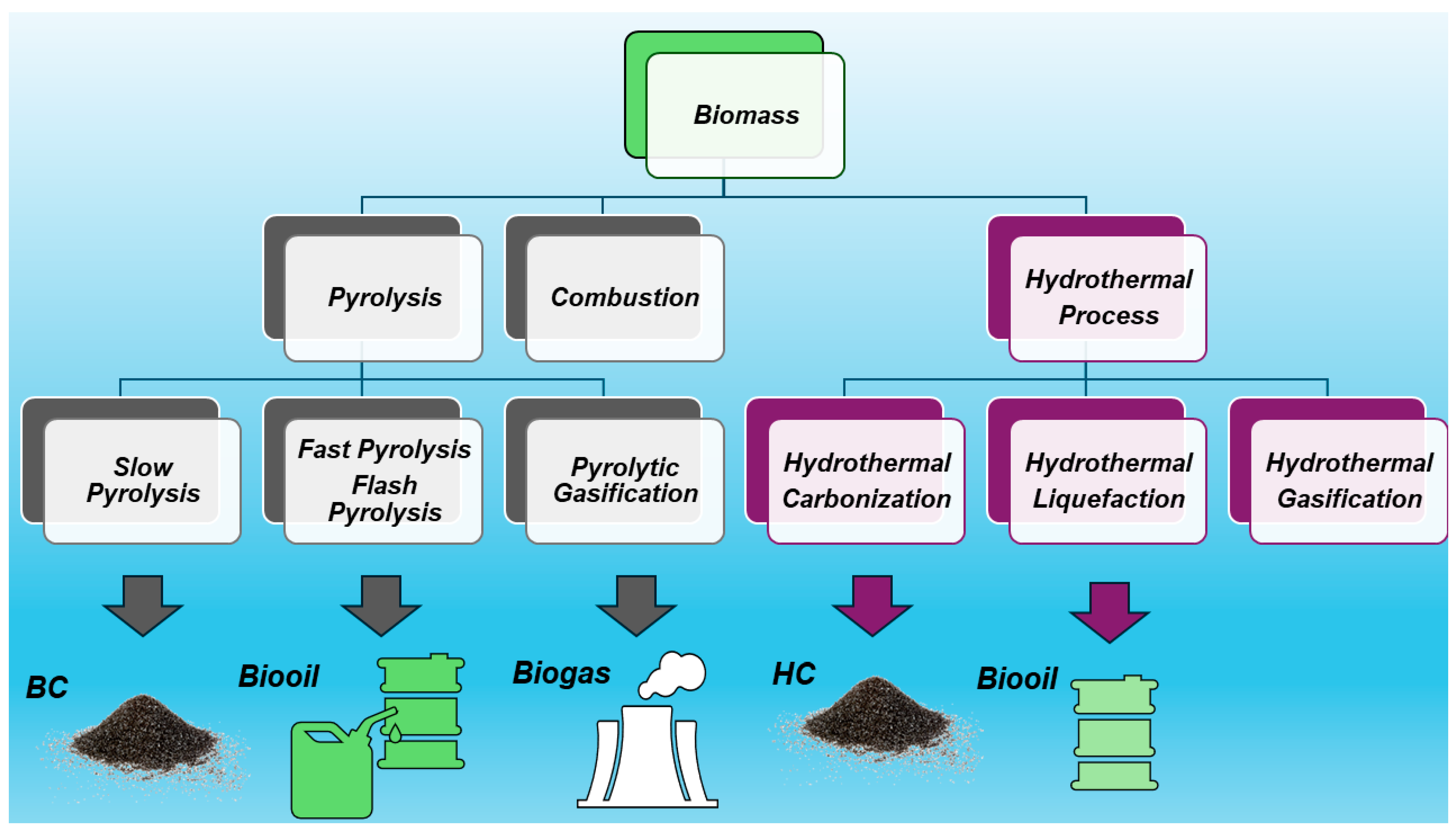
:1. Introduction: Biochar (BC)
2. What Can Biochar Do?
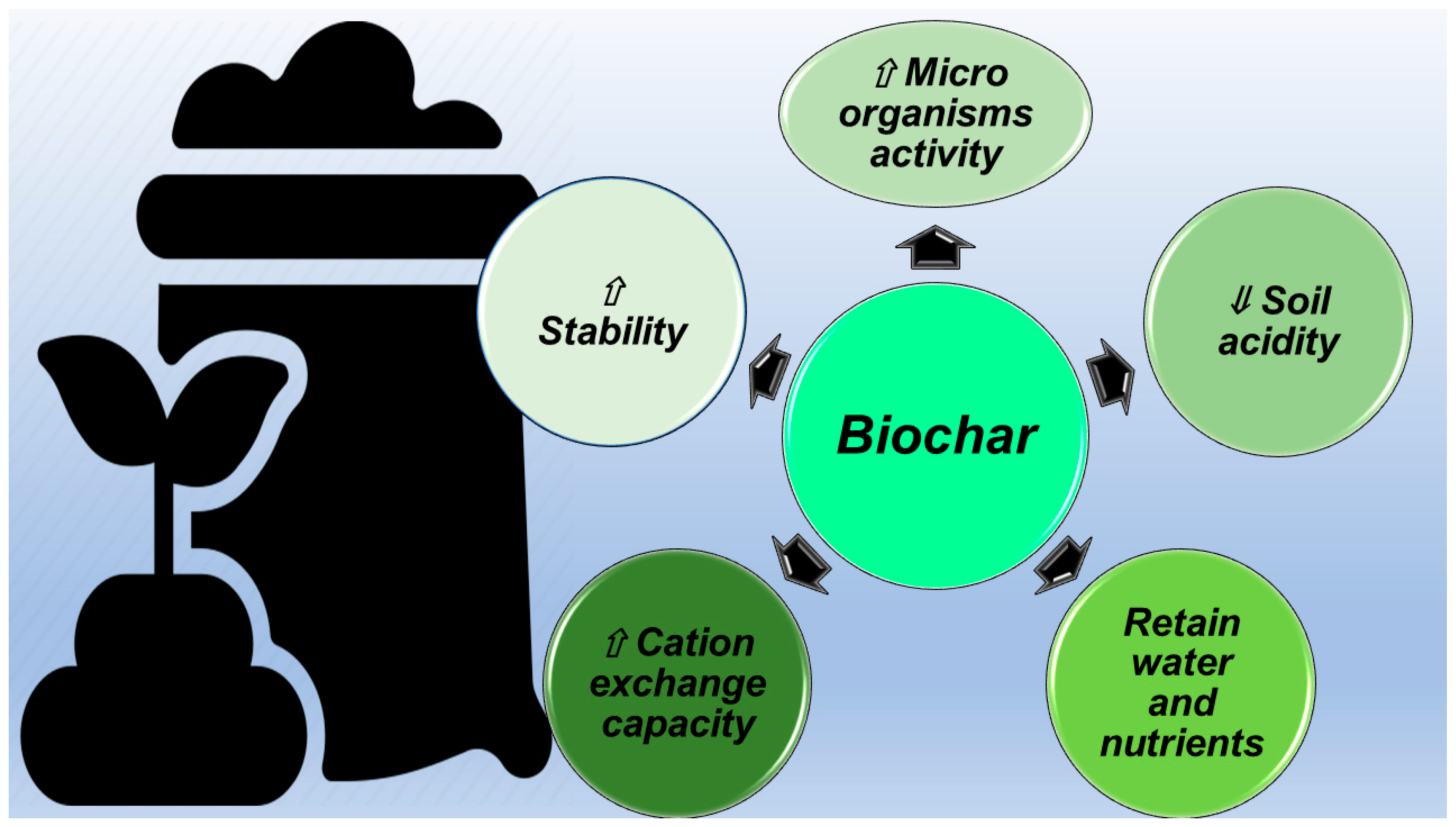
2.1. BC as Climate and Soil Improver
Critical Considerations
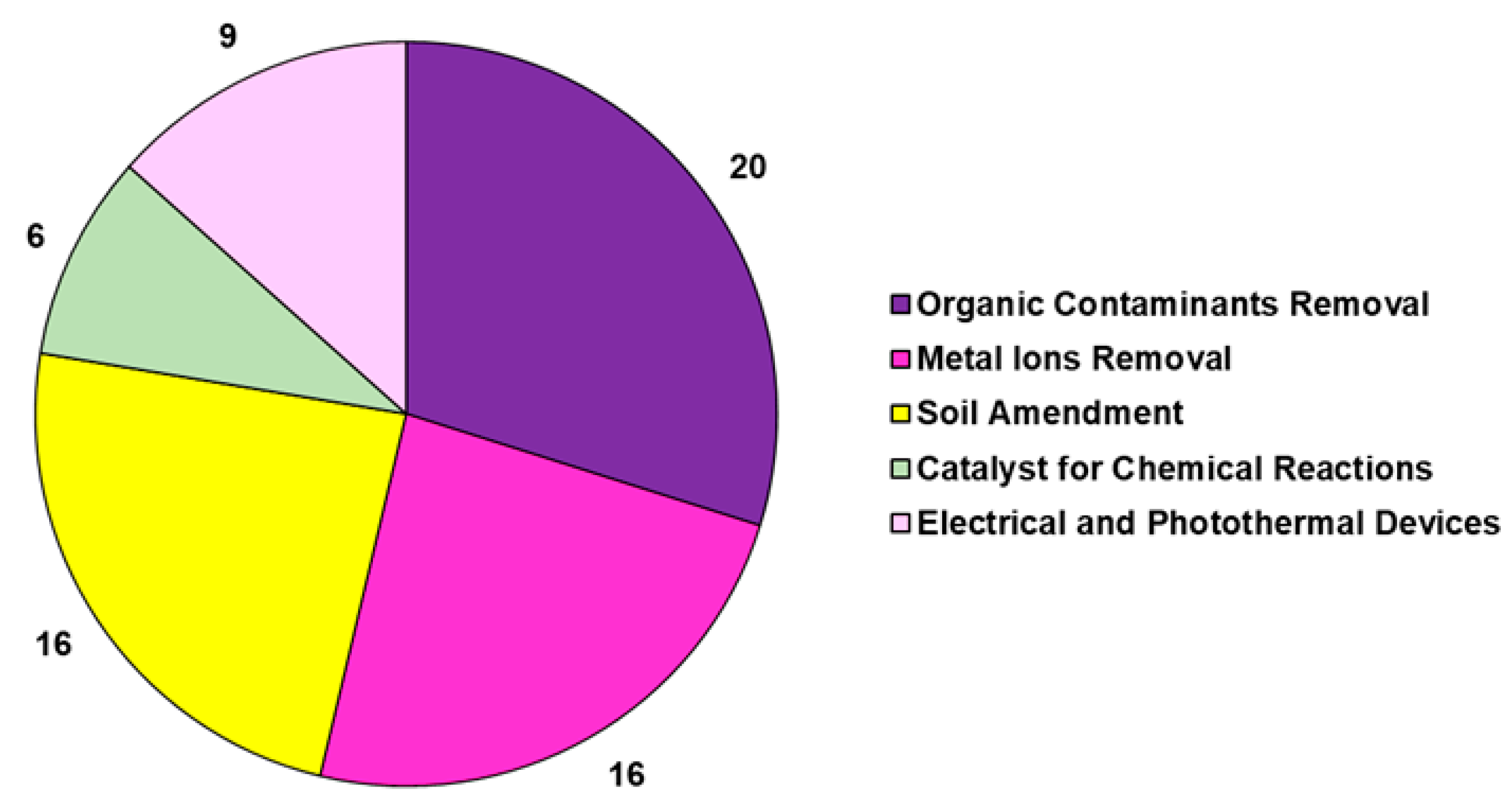
2.2. Xenobiotics Removal by Biochar (BC)
Xenobiotics Removal by Degradative Oxidation
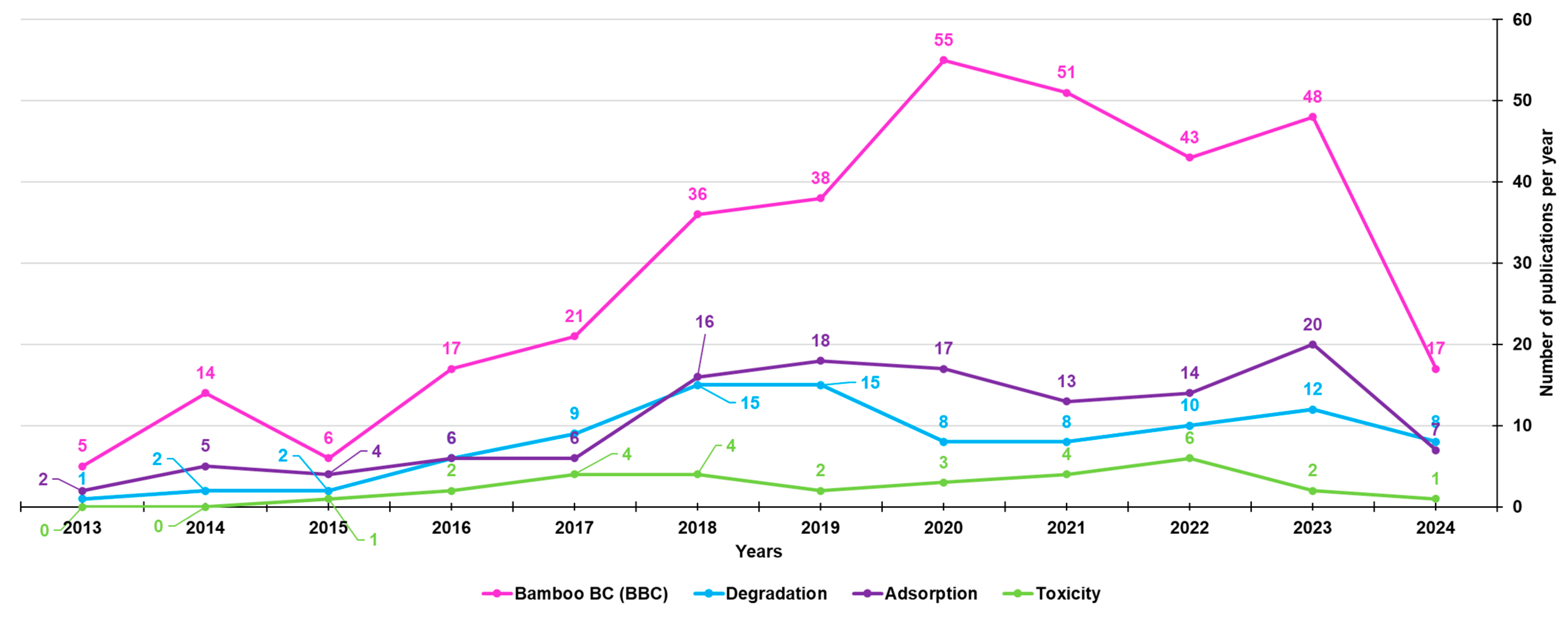
3. Bamboo-Derived Biochar (BBC)
3.1. The Use of Bamboo Biomass to Prepare BC: A Plethora of Advantages over Wood
3.2. Composition of Bamboo
3.2.1. Chemical Composition of Bamboo
3.2.2. Elemental Composition of Bamboo
3.3. Bamboo-Derived Biochar (BBC)
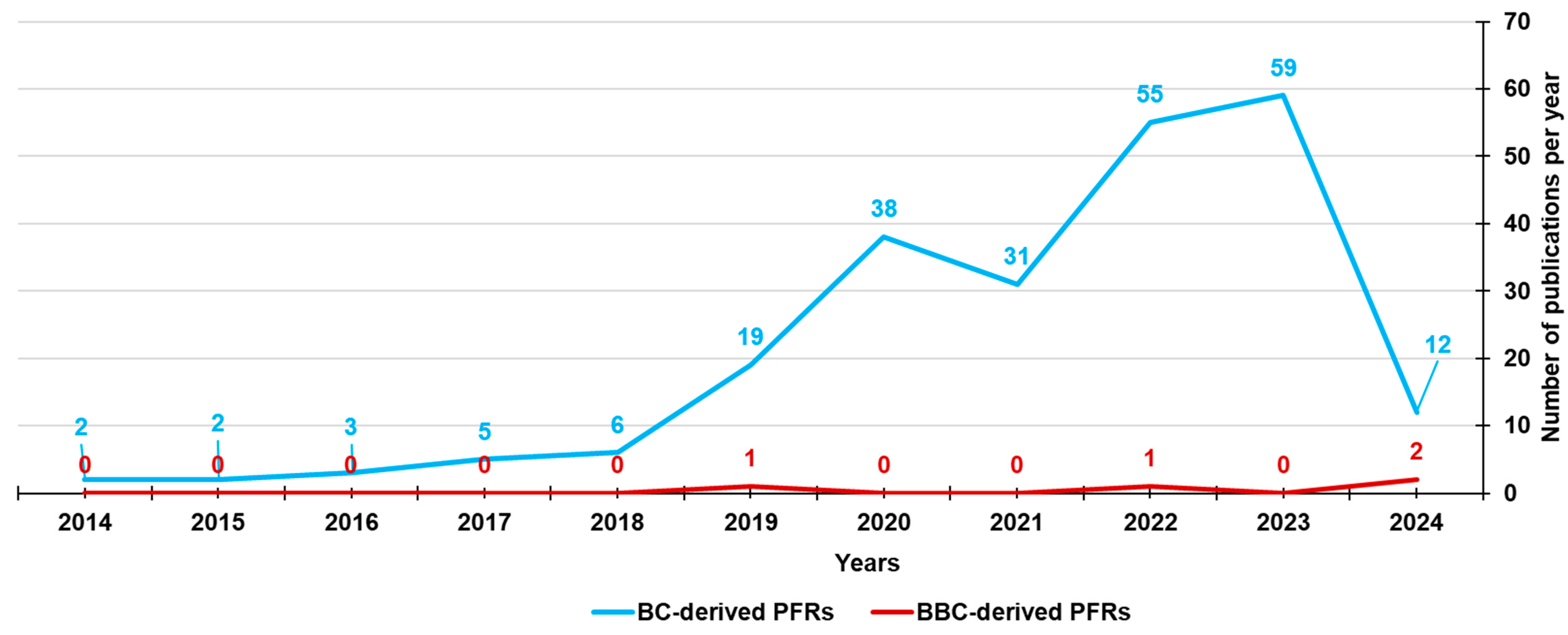
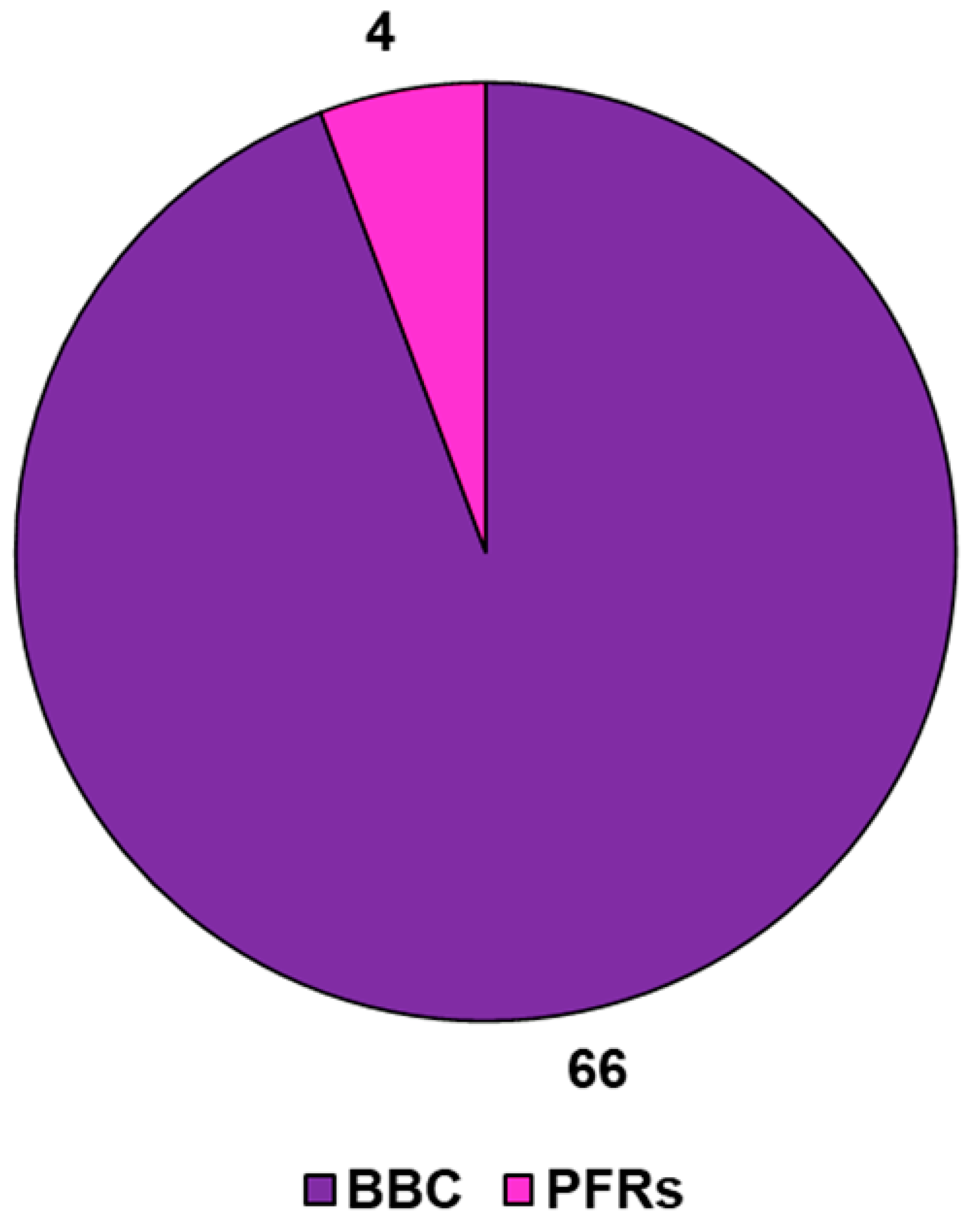
4. BBC-Derived Persistent Free Radicals
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Biochar Initiative. Available online: https://biochar-international.org/about-biochar/faqs/ (accessed on 11 January 2024).
- Panwar, N.L.; Pawar, A.; Salvi, B.L. Comprehensive Review on Production and Utilization of Biochar. SN Appl. Sci. 2019, 1, 168. [Google Scholar] [CrossRef]
- Danesh, P.; Niaparast, P.; Ghorbannezhad, P.; Ali, I. Biochar Production: Recent Developments, Applications, and Challenges. Fuel 2023, 337, 126889. [Google Scholar] [CrossRef]
- Alfei, S.; Pandoli, O.G. Biochar-Derived Persistent Free Radicals: A Plethora of Environmental Applications in a Lights and Shadows Scenario. Preprints 2024. [Google Scholar] [CrossRef]
- Pandey, D.; Daverey, A.; Arunachalam, K. Biochar: Production, Properties and Emerging Role as a Support for Enzyme Immobilization. J. Clean. Prod. 2020, 255, 120267. [Google Scholar] [CrossRef]
- Antal, M.J.; Grønli, M. The Art, Science, and Technology of Charcoal Production. Ind. Eng. Chem. Res. 2003, 42, 1619–1640. [Google Scholar] [CrossRef]
- Bridgwater, A.V. The Production of Biofuels and Renewable Chemicals by Fast Pyrolysis of Biomass. Int. J. Glob. Energy Issues 2007, 27, 160–203. [Google Scholar] [CrossRef]
- DeSisto, W.J.; Hill, N.; Beis, S.H.; Mukkamala, S.; Joseph, J.; Baker, C.; Ong, T.-H.; Stemmler, E.A.; Wheeler, M.C.; Frederick, B.G.; et al. Fast Pyrolysis of Pine Sawdust in a Fluidized-Bed Reactor. Energy Fuels 2010, 24, 2642–2651. [Google Scholar] [CrossRef]
- Al-Rumaihi, A.; Shahbaz, M.; Mckay, G.; Mackey, H.; Al-Ansari, T. A Review of Pyrolysis Technologies and Feedstock: A Blending Approach for Plastic and Biomass towards Optimum Biochar Yield. Renew. Sustain. Energy Rev. 2022, 167, 112715. [Google Scholar] [CrossRef]
- National Energy Technology Laboratory. Syngas Composition. Available online: https://www.netl.doe.gov/research/coal/energy-systems/gasification/gasifipedia/syngas-composition (accessed on 16 January 2024).
- Machado, H.; Cristino, A.F.; Orišková, S.; Galhano dos Santos, R. Bio-Oil: The Next-Generation Source of Chemicals. Reactions 2022, 3, 118–137. [Google Scholar] [CrossRef]
- Shan Ahamed, T.; Anto, S.; Mathimani, T.; Brindhadevi, K.; Pugazhendhi, A. Upgrading of Bio-Oil from Thermochemical Conversion of Various Biomass—Mechanism, Challenges and Opportunities. Fuel 2021, 287, 119329. [Google Scholar] [CrossRef]
- Hu, X.; Gholizadeh, M. Progress of the Applications of Bio-Oil. Renew. Sustain. Energy Rev. 2020, 134, 110124. [Google Scholar] [CrossRef]
- Masoumi, S.; Borugadda, V.B.; Nanda, S.; Dalai, A.K. Hydrochar: A Review on Its Production Technologies and Applications. Catalysts 2021, 11, 939. [Google Scholar] [CrossRef]
- Karagöz, S.; Bhaskar, T.; Muto, A.; Sakata, Y.; Oshiki, T.; Kishimoto, T. Low-Temperature Catalytic Hydrothermal Treatment of Wood Biomass: Analysis of Liquid Products. Chem. Eng. J. 2005, 108, 127–137. [Google Scholar] [CrossRef]
- Alok, J. ‘Biochar’ Goes Industrial with Giant Microwaves to Lock Carbon in Charcoal. The Guardian. 13 March 2009. Available online: https://www.theguardian.com/environment/2009/mar/13/charcoal-carbon (accessed on 15 January 2024).
- Ng, W.C.; You, S.; Ling, R.; Gin, K.Y.-H.; Dai, Y.; Wang, C.-H. Co-Gasification of Woody Biomass and Chicken Manure: Syngas Production, Biochar Reutilization, and Cost-Benefit Analysis. Energy 2017, 139, 732–742. [Google Scholar] [CrossRef]
- Cantrell, K.B.; Hunt, P.G.; Uchimiya, M.; Novak, J.M.; Ro, K.S. Impact of Pyrolysis Temperature and Manure Source on Physicochemical Characteristics of Biochar. Bioresour. Technol. 2012, 107, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Funke, A.; Ziegler, F. Hydrothermal Carbonization of Biomass: A Summary and Discussion of Chemical Mechanisms for Process Engineering. Biofuels Bioprod. Biorefin. 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Klinghoffer, N.B.; Castaldi, M.J.; Nzihou, A. Influence of Char Composition and Inorganics on Catalytic Activity of Char from Biomass Gasification. Fuel 2015, 157, 37–47. [Google Scholar] [CrossRef]
- Nunoura, T.; Wade, S.R.; Bourke, J.P.; Antal, M.J. Studies of the Flash Carbonization Process. 1. Propagation of the Flaming Pyrolysis Reaction and Performance of a Catalytic Afterburner. Ind. Eng. Chem. Res. 2006, 45, 585–599. [Google Scholar] [CrossRef]
- Chen, Q.; Zhou, J.; Liu, B.; Mei, Q.; Luo, Z. Influence of Torrefaction Pretreatment on Biomass Gasification Technology. Chin. Sci. Bull. 2011, 56, 1449–1456. [Google Scholar] [CrossRef]
- Smidth, A.N. A New Boost for Biochar as a Natural Climate Solution. 2023. Available online: https://blog.nature.org/science-brief/a-new-boost-for-biochar-as-a-natural-climate-solution/#:~:text=biochar%20is%20a%20type%20of%20charcoal%20made%20from,other%20biomass%20would%2c%20leading%20to%20more%20carbon%20sequestration (accessed on 27 December 2023).
- Luo, L.; Wang, J.; Lv, J.; Liu, Z.; Sun, T.; Yang, Y.; Zhu, Y.-G. Carbon Sequestration Strategies in Soil Using Biochar: Advances, Challenges, and Opportunities. Environ. Sci. Technol. 2023, 57, 11357–11372. [Google Scholar] [CrossRef]
- Brassard, P.; Godbout, S.; Lévesque, V.; Palacios, J.H.; Raghavan, V.; Ahmed, A.; Hogue, R.; Jeanne, T.; Verma, M. Biochar for Soil Amendment. In Char and Carbon Materials Derived from Biomass; Elsevier: Amsterdam, The Netherlands, 2019; pp. 109–146. [Google Scholar]
- Godlewska, P.; Schmidt, H.P.; Ok, Y.S.; Oleszczuk, P. Biochar for Composting Improvement and Contaminants Reduction. A Review. Bioresour. Technol. 2017, 246, 193–202. [Google Scholar] [CrossRef]
- Xiang, W.; Zhang, X.; Chen, J.; Zou, W.; He, F.; Hu, X.; Tsang, D.C.W.; Ok, Y.S.; Gao, B. Biochar Technology in Wastewater Treatment: A Critical Review. Chemosphere 2020, 252, 126539. [Google Scholar] [CrossRef]
- Gupta, S.; Kua, H.W.; Koh, H.J. Application of Biochar from Food and Wood Waste as Green Admixture for Cement Mortar. Sci. Total Environ. 2018, 619–620, 419–435. [Google Scholar] [CrossRef]
- Suarez-Riera, D.; Restuccia, L.; Ferro, G.A. The Use of Biochar to Reduce the Carbon Footprint of Cement-Based Materials. Procedia Struct. Integr. 2020, 26, 199–210. [Google Scholar] [CrossRef]
- Qiu, B.; Shao, Q.; Shi, J.; Yang, C.; Chu, H. Application of Biochar for the Adsorption of Organic Pollutants from Wastewater: Modification Strategies, Mechanisms and Challenges. Sep. Purif. Technol. 2022, 300, 121925. [Google Scholar] [CrossRef]
- Kalu, S.; Kulmala, L.; Zrim, J.; Peltokangas, K.; Tammeorg, P.; Rasa, K.; Kitzler, B.; Pihlatie, M.; Karhu, K. Potential of Biochar to Reduce Greenhouse Gas Emissions and Increase Nitrogen Use Efficiency in Boreal Arable Soils in the Long-Term. Front. Environ. Sci. 2022, 10, 914766. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, B.; Gao, B.; Cheng, N.; Feng, Q.; Chen, M.; Wang, S. Degradation of Organic Pollutants from Water by Biochar-Assisted Advanced Oxidation Processes: Mechanisms and Applications. J. Hazard. Mater. 2023, 442, 130075. [Google Scholar] [CrossRef]
- Lyu, H.; Zhang, Q.; Shen, B. Application of Biochar and Its Composites in Catalysis. Chemosphere 2020, 240, 124842. [Google Scholar] [CrossRef]
- Schmidt, H.-P.; Hagemann, N.; Draper, K.; Kammann, C. The Use of Biochar in Animal Feeding. PeerJ 2019, 7, e7373. [Google Scholar] [CrossRef]
- Yu, D.; Yu, Y.; Tang, J.; Li, X.; Ke, C.; Yao, Z. Application Fields of Kitchen Waste Biochar and Its Prospects as Catalytic Material: A Review. Sci. Total Environ. 2022, 810, 152171. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zhang, W.; Dong, X.; Wang, F.; Yang, W.; Liu, C.; Chen, D. A Review on Recent Advances of Biochar from Agricultural and Forestry Wastes: Preparation, Modification and Applications in Wastewater Treatment. J. Environ. Chem. Eng. 2024, 12, 111638. [Google Scholar] [CrossRef]
- Wijitkosum, S. Biochar Derived from Agricultural Wastes and Wood Residues for Sustainable Agricultural and Environmental Applications. Int. Soil Water Conserv. Res. 2022, 10, 335–341. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Biochar Derived from Anaerobically Digested Sugar Beet Tailings: Characterization and Phosphate Removal Potential. Bioresour. Technol. 2011, 102, 6273–6278. [Google Scholar] [CrossRef] [PubMed]
- Puettmann, M.; Sahoo, K.; Wilson, K.; Oneil, E. Life Cycle Assessment of Biochar Produced from Forest Residues Using Portable Systems. J. Clean. Prod. 2020, 250, 119564. [Google Scholar] [CrossRef]
- Papageorgiou, A.; Azzi, E.S.; Enell, A.; Sundberg, C. Biochar Produced from Wood Waste for Soil Remediation in Sweden: Carbon Sequestration and Other Environmental Impacts. Sci. Total Environ. 2021, 776, 145953. [Google Scholar] [CrossRef] [PubMed]
- Laird, D. Biochar Amendments Make the Harvesting of Crop Residue for Bioenergy Production Sustainable. Nutr. Cycl. Agroecosyst. 2023. [Google Scholar] [CrossRef]
- Li, N.; He, M.; Lu, X.; Yan, B.; Duan, X.; Chen, G.; Wang, S.; Hou, L. Municipal Solid Waste Derived Biochars for Wastewater Treatment: Production, Properties and Applications. Resour. Conserv. Recycl. 2022, 177, 106003. [Google Scholar] [CrossRef]
- Shi, W.; Wang, H.; Yan, J.; Shan, L.; Quan, G.; Pan, X.; Cui, L. Wheat Straw Derived Biochar with Hierarchically Porous Structure for Bisphenol A Removal: Preparation, Characterization, and Adsorption Properties. Sep. Purif. Technol. 2022, 289, 120796. [Google Scholar] [CrossRef]
- Foong, S.Y.; Chan, Y.H.; Chin, B.L.F.; Lock, S.S.M.; Yee, C.Y.; Yiin, C.L.; Peng, W.; Lam, S.S. Production of Biochar from Rice Straw and Its Application for Wastewater Remediation—An Overview. Bioresour. Technol. 2022, 360, 127588. [Google Scholar] [CrossRef]
- Gunamantha, I.M.; Widana, G.A.B. Characterization the Potential of Biochar from Cow and Pig Manure for Geoecology Application. IOP Conf. Ser. Earth Environ. Sci. 2018, 131, 012055. [Google Scholar] [CrossRef]
- Rathnayake, D.; Schmidt, H.; Leifeld, J.; Mayer, J.; Epper, C.A.; Bucheli, T.D.; Hagemann, N. Biochar from Animal Manure: A Critical Assessment on Technical Feasibility, Economic Viability, and Ecological Impact. GCB Bioenergy 2023, 15, 1078–1104. [Google Scholar] [CrossRef]
- Drané, M.; Zbair, M.; Hajjar-Garreau, S.; Josien, L.; Michelin, L.; Bennici, S.; Limousy, L. Unveiling the Potential of Corn Cob Biochar: Analysis of Microstructure and Composition with Emphasis on Interaction with NO2. Materials 2023, 17, 159. [Google Scholar] [CrossRef] [PubMed]
- Sudarsan, J.S.; Prasanna, K.; Shiam Babu, R.; Sai Krishna, V.M.V. Biochar: A Sustainable Solution for Organic Waste Management a Way Forward towards Circular Economy. In Recent Trends in Solid Waste Management; Elsevier: Amsterdam, The Netherlands, 2023; pp. 215–230. [Google Scholar]
- Rawat, S.; Wang, C.-T.; Lay, C.-H.; Hotha, S.; Bhaskar, T. Sustainable Biochar for Advanced Electrochemical/Energy Storage Applications. J. Energy Storage 2023, 63, 107115. [Google Scholar] [CrossRef]
- Daly, J. Poo-Eating Beetles and Charcoal Used by WA Farmer to Combat Climate Change. ABC RURAL. 18 October 2019. Available online: https://www.abc.net.au/news/rural/2019-10-18/wa-farmer-uses-beetles-and-charcoal-to-combat-climate-change/11613846 (accessed on 16 January 2024).
- Making Concrete Change: Innovation in Low-Carbon Cement and Concrete. Chatham House—International Affairs Think Tank. Available online: https://www.chathamhouse.org/2018/06/making-concrete-change-innovation-low-carbon-cement-and-concrete (accessed on 22 January 2024).
- Arvaniti, E.C.; Juenger, M.C.G.; Bernal, S.A.; Duchesne, J.; Courard, L.; Leroy, S.; Provis, J.L.; Klemm, A.; De Belie, N. Physical Characterization Methods for Supplementary Cementitious Materials. Mater. Struct. 2015, 48, 3675–3686. [Google Scholar] [CrossRef]
- Gupta, S.; Kua, H.W.; Pang, S.D. Effect of Biochar on Mechanical and Permeability Properties of Concrete Exposed to Elevated Temperature. Constr. Build. Mater. 2020, 234, 117338. [Google Scholar] [CrossRef]
- Varjani, S.; Kumar, G.; Rene, E.R. Developments in Biochar Application for Pesticide Remediation: Current Knowledge and Future Research Directions. J. Environ. Manag. 2019, 232, 505–513. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of Fast Pyrolysis of Biomass and Product Upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Premarathna, K.S.D.; Rajapaksha, A.U.; Sarkar, B.; Kwon, E.E.; Bhatnagar, A.; Ok, Y.S.; Vithanage, M. Biochar-Based Engineered Composites for Sorptive Decontamination of Water: A Review. Chem. Eng. J. 2019, 372, 536–550. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A Review of Biochars’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils. Environ. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A Quantitative Review of the Effects of Biochar Application to Soils on Crop Productivity Using Meta-Analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Kinney, T.J.; Masiello, C.A.; Dugan, B.; Hockaday, W.C.; Dean, M.R.; Zygourakis, K.; Barnes, R.T. Hydrologic Properties of Biochars Produced at Different Temperatures. Biomass Bioenergy 2012, 41, 34–43. [Google Scholar] [CrossRef]
- Qin, Y.; Li, G.; Gao, Y.; Zhang, L.; Ok, Y.S.; An, T. Persistent Free Radicals in Carbon-Based Materials on Transformation of Refractory Organic Contaminants (ROCs) in Water: A Critical Review. Water Res. 2018, 137, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Werner, C.; Schmidt, H.-P.; Gerten, D.; Lucht, W.; Kammann, C. Biogeochemical Potential of Biomass Pyrolysis Systems for Limiting Global Warming to 1.5 °C. Environ. Res. Lett. 2018, 13, 044036. [Google Scholar] [CrossRef]
- Yousaf, B.; Liu, G.; Wang, R.; Abbas, Q.; Imtiaz, M.; Liu, R. Investigating the Biochar Effects on C-mineralization and Sequestration of Carbon in Soil Compared with Conventional Amendments Using the Stable Isotope (δ13C) Approach. GCB Bioenergy 2017, 9, 1085–1099. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable Biochar to Mitigate Global Climate Change. Nat. Commun. 2010, 1, 56. [Google Scholar] [CrossRef] [PubMed]
- Cocora, L.; Brand, K.; Cocora, L.; Brand, K. United Nations University: Japan’s Charcoal Making Traditions Still Alive. Available online: https://ourworld.unu.edu/en/japans-charcoal-making-traditions-still-alive (accessed on 16 January 2024).
- Arshad, U.; Altaf, M.T.; Liaqat, W.; Ali, M.; Shah, M.N.; Jabran, M.; Ali, M.A. Biochar: Black Gold for Sustainable Agriculture and Fortification Against Plant Pathogens—A Review. Gesunde Pflanz. 2023. [Google Scholar] [CrossRef]
- Hamidzadeh, Z.; Ghorbannezhad, P.; Ketabchi, M.R.; Yeganeh, B. Biomass-Derived Biochar and Its Application in Agriculture. Fuel 2023, 341, 127701. [Google Scholar] [CrossRef]
- Nguyen, H.N.; Pignatello, J.J. Laboratory Tests of Biochars as Absorbents for Use in Recovery or Containment of Marine Crude Oil Spills. Environ. Eng. Sci. 2013, 30, 374–380. [Google Scholar] [CrossRef]
- Jothirani, R.; Kumar, P.S.; Saravanan, A.; Narayan, A.S.; Dutta, A. Ultrasonic Modified Corn Pith for the Sequestration of Dye from Aqueous Solution. J. Ind. Eng. Chem. 2016, 39, 162–175. [Google Scholar] [CrossRef]
- Suganya, S.; Senthil Kumar, P.; Saravanan, A.; Sundar Rajan, P.; Ravikumar, C. Computation of Adsorption Parameters for the Removal of Dye from Wastewater by Microwave Assisted Sawdust: Theoretical and Experimental Analysis. Environ. Toxicol. Pharmacol. 2017, 50, 45–57. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Renita, A.A. Hybrid Synthesis of Novel Material through Acid Modification Followed Ultrasonication to Improve Adsorption Capacity for Zinc Removal. J. Clean. Prod. 2018, 172, 92–105. [Google Scholar] [CrossRef]
- Luo, Z.; Yao, B.; Yang, X.; Wang, L.; Xu, Z.; Yan, X.; Tian, L.; Zhou, H.; Zhou, Y. Novel Insights into the Adsorption of Organic Contaminants by Biochar: A Review. Chemosphere 2022, 287, 132113. [Google Scholar] [CrossRef]
- Tao, W.; Zhang, P.; Li, H.; Yang, Q.; Oleszczuk, P.; Pan, B. Generation Mechanism of Persistent Free Radicals in Lignocellulose-Derived Biochar: Roles of Reducible Carbonyls. Environ. Sci. Technol. 2022, 56, 10638–10645. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Zhu, Y.; Zeng, G.; Zhang, Y.; Wang, Y.; Zhang, M.; Long, H.; Tang, W. Efficient Removal of Perfluorooctanoic Acid by Persulfate Advanced Oxidative Degradation: Inherent Roles of Iron-Porphyrin and Persistent Free Radicals. Chem. Eng. J. 2020, 392, 123640. [Google Scholar] [CrossRef]
- Cao, W.; Zeng, C.; Guo, X.; Liu, Q.; Zhang, X.; Mameda, N. Enhanced Electrochemical Degradation of 2,4-Dichlorophenol with the Assist of Hydrochar. Chemosphere 2020, 260, 127643. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zhang, Y.; Li, J.; Hao, Q.; Li, X.; Liu, F. Heterogeneous Activation of Peroxymonosulfate by a Biochar-Supported Co3O4 Composite for Efficient Degradation of Chloramphenicols. Environ. Pollut. 2020, 257, 113610. [Google Scholar] [CrossRef]
- Li, X.; Jia, Y.; Zhou, M.; Su, X.; Sun, J. High-Efficiency Degradation of Organic Pollutants with Fe, N Co-Doped Biochar Catalysts via Persulfate Activation. J. Hazard. Mater. 2020, 397, 122764. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Hu, W.; Wang, X.; Tong, W.; Li, P.; Zhou, H.; Wang, Y.; Zhang, Y. Molten Salt Induced Nitrogen-Doped Biochar Nanosheets as Highly Efficient Peroxymonosulfate Catalyst for Organic Pollutant Degradation. Environ. Pollut. 2020, 260, 114053. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Yang, L.; Qian, L.; Han, L.; Chen, M. Nano-Magnetite Supported by Biochar Pyrolyzed at Different Temperatures as Hydrogen Peroxide Activator: Synthesis Mechanism and the Effects on Ethylbenzene Removal. Environ. Pollut. 2020, 261, 114020. [Google Scholar] [CrossRef] [PubMed]
- Mian, M.M.; Liu, G.; Zhou, H. Preparation of N-Doped Biochar from Sewage Sludge and Melamine for Peroxymonosulfate Activation: N-Functionality and Catalytic Mechanisms. Sci. Total Environ. 2020, 744, 140862. [Google Scholar] [CrossRef]
- da Silva Veiga, P.A.; Schultz, J.; da Silva Matos, T.T.; Fornari, M.R.; Costa, T.G.; Meurer, L.; Mangrich, A.S. Production of High-Performance Biochar Using a Simple and Low-Cost Method: Optimization of Pyrolysis Parameters and Evaluation for Water Treatment. J. Anal. Appl. Pyrolysis 2020, 148, 104823. [Google Scholar] [CrossRef]
- Sun, P.; Zhang, K.-K.; Zhang, Y.; Zhang, Y.-R. Sunflower-Straw-Derived Biochar-Enhanced Fe(III)/S2O82− System for Degradation of Benzoic Acid. Huan Jing Ke Xue 2020, 41, 2301–2309. [Google Scholar] [CrossRef]
- Zeng, L.; Chen, Q.; Tan, Y.; Lan, P.; Zhou, D.; Wu, M.; Liang, N.; Pan, B.; Xing, B. Dual Roles of Biochar Redox Property in Mediating 2,4-Dichlorophenol Degradation in the Presence of Fe3+ and Persulfate. Chemosphere 2021, 279, 130456. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, Q.; Gao, Y.; Jian, H.; Wang, C.; Sun, H. Effects of Biochar-Dissolved Organic Matter on the Photodegradation of Sulfamethoxazole and Chloramphenicol in Biochar Solutions as Revealed by Oxygen Reduction Performances and Free Radicals. Sci. Total Environ. 2021, 781, 146807. [Google Scholar] [CrossRef]
- Zhong, D.; Jiang, Y.; Zhao, Z.; Wang, L.; Chen, J.; Ren, S.; Liu, Z.; Zhang, Y.; Tsang, D.C.W.; Crittenden, J.C. PH Dependence of Arsenic Oxidation by Rice-Husk-Derived Biochar: Roles of Redox-Active Moieties. Environ. Sci. Technol. 2019, 53, 9034–9044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Sun, P.; Zhang, Y. Decontamination of Cr(VI) Facilitated Formation of Persistent Free Radicals on Rice Husk Derived Biochar. Front. Environ. Sci. Eng. 2019, 13, 22. [Google Scholar] [CrossRef]
- Zhu, S.; Huang, X.; Yang, X.; Peng, P.; Li, Z.; Jin, C. Enhanced Transformation of Cr(VI) by Heterocyclic-N within Nitrogen-Doped Biochar: Impact of Surface Modulatory Persistent Free Radicals (PFRs). Environ. Sci. Technol. 2020, 54, 8123–8132. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, J.; Liu, J.; Liu, J.; Xia, F.; Wang, C.; Dahlgren, R.A.; Liu, W. Mechanism of Cr(VI) Removal by Magnetic Greigite/Biochar Composites. Sci. Total Environ. 2020, 700, 134414. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Kong, Y.; Zhao, S.; Jia, H.; Vione, D.; Kang, Y.; Gao, P. Enhancement of Cr(VI) Decontamination by Irradiated Sludge Biochar in Neutral Conditions: Evidence of a Possible Role of Persistent Free Radicals. Sep. Purif. Technol. 2021, 277, 119414. [Google Scholar] [CrossRef]
- Yu, J.; Zhu, Z.; Zhang, H.; Chen, T.; Qiu, Y.; Xu, Z.; Yin, D. Efficient Removal of Several Estrogens in Water by Fe-Hydrochar Composite and Related Interactive Effect Mechanism of H2O2 and Iron with Persistent Free Radicals from Hydrochar of Pinewood. Sci. Total Environ. 2019, 658, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.; Li, C.; Bu, L.; Tang, C.; Wang, S.; Duan, P.; Yao, L.; Tang, J.; Dionysiou, D.D.; Wu, Y. Bismuth Impregnated Biochar for Efficient Estrone Degradation: The Synergistic Effect between Biochar and Bi/Bi2O3 for a High Photocatalytic Performance. J. Hazard. Mater. 2020, 384, 121258. [Google Scholar] [CrossRef]
- Chen, Y.; Duan, X.; Zhang, C.; Wang, S.; Ren, N.; Ho, S.-H. Graphitic Biochar Catalysts from Anaerobic Digestion Sludge for Nonradical Degradation of Micropollutants and Disinfection. Chem. Eng. J. 2020, 384, 123244. [Google Scholar] [CrossRef]
- Xu, H.; Han, Y.; Wang, G.; Deng, P.; Feng, L. Walnut Shell Biochar Based Sorptive Remediation of Estrogens Polluted Simulated Wastewater: Characterization, Adsorption Mechanism and Degradation by Persistent Free Radicals. Environ. Technol. Innov. 2022, 28, 102870. [Google Scholar] [CrossRef]
- Wang, T.; Zheng, J.; Cai, J.; Liu, Q.; Zhang, X. Visible-Light-Driven Photocatalytic Degradation of Dye and Antibiotics by Activated Biochar Composited with K+ Doped g-C3N4: Effects, Mechanisms, Actual Wastewater Treatment and Disinfection. Sci. Total Environ. 2022, 839, 155955. [Google Scholar] [CrossRef] [PubMed]
- Odinga, E.S.; Waigi, M.G.; Gudda, F.O.; Wang, J.; Yang, B.; Hu, X.; Li, S.; Gao, Y. Occurrence, Formation, Environmental Fate and Risks of Environmentally Persistent Free Radicals in Biochars. Environ. Int. 2020, 134, 105172. [Google Scholar] [CrossRef] [PubMed]
- Fang, G.; Liu, C.; Gao, J.; Dionysiou, D.D.; Zhou, D. Manipulation of Persistent Free Radicals in Biochar To Activate Persulfate for Contaminant Degradation. Environ. Sci. Technol. 2015, 49, 5645–5653. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Upadhyay, A.; Ding, Y.; Emamverdian, A.; Shahzad, A. Bamboo: Origin, Habitat, Distributions and Global Prospective. In Biotechnological Advances in Bamboo; Springer: Singapore, 2021; pp. 1–31. [Google Scholar]
- Yadav, M.; Mathur, A. Bamboo as a Sustainable Material in the Construction Industry: An Overview. Mater. Today Proc. 2021, 43, 2872–2876. [Google Scholar] [CrossRef]
- Chaturvedi, K.; Singhwane, A.; Dhangar, M.; Mili, M.; Gorhae, N.; Naik, A.; Prashant, N.; Srivastava, A.K.; Verma, S. Bamboo for Producing Charcoal and Biochar for Versatile Applications. Biomass Convers. Biorefin. 2023. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, Q.; Sun, W.; Zhao, Y.; Wang, X.; Liu, X.; Li, Y. Bamboo Flattening Technique: A Literature and Patent Review. Eur. J. Wood Wood Prod. 2021, 79, 1035–1048. [Google Scholar] [CrossRef]
- Yang, L.; Lou, Z.; Han, X.; Liu, J.; Wang, Z.; Zhang, Y.; Wu, X.; Yuan, C.; Li, Y. Fabrication of a Novel Magnetic Reconstituted Bamboo with Mildew Resistance Properties. Mater. Today Commun. 2020, 23, 101086. [Google Scholar] [CrossRef]
- da Silveira, E.S.; Roel, A.R.; dos Santos Brito, V.H.; Pistori, H.; Cereda, M.P. Influência de Espécies de Bambu Como Alimento No Crescimento Populacional e Na Preferência Alimentar Do Caruncho-Do-Bambu. I. In Bambus no Brasil: Da Biologia à Tecnologia; Drumond, P.M., Wiedman, G., Eds.; Instituto Ciência Hoje—ICH: Rio de Janeiro, Brazil, 2017; pp. 130–144. [Google Scholar]
- Darabant, A.; Haruthaithanasan, M.; Atkla, W.; Phudphong, T.; Thanavat, E.; Haruthaithanasan, K. Bamboo Biomass Yield and Feedstock Characteristics of Energy Plantations in Thailand. Energy Procedia 2014, 59, 134–141. [Google Scholar] [CrossRef]
- Romero Millán, L.M.; Sierra Vargas, F.E.; Nzihou, A. Characterization of Steam Gasification Biochars from Lignocellulosic Agrowaste towards Soil Applications. Waste Biomass Valoriz. 2021, 12, 4141–4155. [Google Scholar] [CrossRef]
- Masís-Meléndez, F.; Segura-Chavarría, D.; García-González, C.A.; Quesada-Kimsey, J.; Villagra-Mendoza, K. Variability of Physical and Chemical Properties of TLUD Stove Derived Biochars. Appl. Sci. 2020, 10, 507. [Google Scholar] [CrossRef]
- Patel, B.; Patel, M.; Gami, B.; Patel, A. Cultivation of Bioenergy Crops in Gujarat State: A Consultative Survey Process to Understand the Current Practices of Landowners. Environ. Dev. Sustain. 2021, 23, 8991–9013. [Google Scholar] [CrossRef]
- Odega, C.A.; Ayodele, O.O.; Ogutuga, S.O.; Anguruwa, G.T.; Adekunle, A.E.; Fakorede, C.O. Potential Application and Regeneration of Bamboo Biochar for Wastewater Treatment: A Review. Adv. Bamboo Sci. 2023, 2, 100012. [Google Scholar] [CrossRef]
- Ginoble Pandoli, O.; Santos de Sá, D.; Nogueira Barbosa Junior, M.; Paciornik, S. Bamboo-Based Lignocellulose Biomass as Catalytic Support for Organic Synthesis and Water Treatments. In Bamboo Science and Technology; Springer: Singapore, 2023; pp. 297–327. [Google Scholar] [CrossRef]
- Kawakami, M.; Karato, T.; Takenaka, T.; Yokoyama, S. Structure Analysis of Coke, Wood Charcoal and Bamboo Charcoal by Raman Spectroscopy and Their Reaction Rate with CO2. ISIJ Int. 2005, 45, 1027–1034. [Google Scholar] [CrossRef]
- Liao, P.; Malik Ismael, Z.; Zhang, W.; Yuan, S.; Tong, M.; Wang, K.; Bao, J. Adsorption of Dyes from Aqueous Solutions by Microwave Modified Bamboo Charcoal. Chem. Eng. J. 2012, 195–196, 339–346. [Google Scholar] [CrossRef]
- Lehmann, J. Bio-Energy in the Black. Front. Ecol. Environ. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Wei, H.; Deng, S.; Hu, B.; Chen, Z.; Wang, B.; Huang, J.; Yu, G. Granular Bamboo-Derived Activated Carbon for High CO2 Adsorption: The Dominant Role of Narrow Micropores. ChemSusChem 2012, 5, 2354–2360. [Google Scholar] [CrossRef]
- Liu, D.; Song, J.; Anderson, D.P.; Chang, P.R.; Hua, Y. Bamboo Fiber and Its Reinforced Composites: Structure and Properties. Cellulose 2012, 19, 1449–1480. [Google Scholar] [CrossRef]
- Sahoo, S.S.; Vijay, V.K.; Chandra, R.; Kumar, H. Production and Characterization of Biochar Produced from Slow Pyrolysis of Pigeon Pea Stalk and Bamboo. Clean. Eng. Technol. 2021, 3, 100101. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, X.; Yuan, C.; Lou, Z.; Li, Y. Effect of Saturated Steam Heat Treatment on Physical and Chemical Properties of Bamboo. Molecules 2020, 25, 1999. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Yuan, T.; Wang, Q.; Wu, X.; Hu, S.; Hao, X.; Liu, X.; Li, Y. Fabrication of Crack-Free Flattened Bamboo and Its Macro-/MicroMorphological and Mechanical Properties. J. Renew. Mater. 2021, 9, 959–977. [Google Scholar] [CrossRef]
- Liese, W. The Anatomy of Bamboo Culms; INBAR: Beijing, China, 1998; 208p. [Google Scholar]
- Lee, C.-H.; Chung, M.-J.; Lin, C.-H.; Yang, T.-H. Effects of Layered Structure on the Physical and Mechanical Properties of Laminated Moso Bamboo (Phyllosachys edulis) Flooring. Constr. Build. Mater. 2012, 28, 31–35. [Google Scholar] [CrossRef]
- Wastowski, A.D. Química Da Madeira Editora Interciência, 1st ed.; Editora Interciência: Rio de Janeiro, Brazil, 2018; 556p. [Google Scholar]
- Yan, L.; Kasal, B.; Huang, L. A Review of Recent Research on the Use of Cellulosic Fibres, Their Fibre Fabric Reinforced Cementitious, Geo-Polymer and Polymer Composites in Civil Engineering. Compos. B Eng. 2016, 92, 94–132. [Google Scholar] [CrossRef]
- Liese, W.; Tang, T.K.H. Properties of the Bamboo Culm. In Bamboo; Springer: Cham, Switzerland, 2015; pp. 227–256. [Google Scholar] [CrossRef]
- Nayak, L.; Mishra, S.P. Prospect of Bamboo as a Renewable Textile Fiber, Historical Overview, Labeling, Controversies and Regulation. Fash. Text. 2016, 3, 2. [Google Scholar] [CrossRef]
- Wan, J.; Wang, Y.; Xiao, Q. Effects of Hemicellulose Removal on Cellulose Fiber Structure and Recycling Characteristics of Eucalyptus Pulp. Bioresour. Technol. 2010, 101, 4577–4583. [Google Scholar] [CrossRef]
- Mohamad Ibrahim, M.N.; Zakaria, N.; Sipaut, C.S.; Sulaiman, O.; Hashim, R. Chemical and Thermal Properties of Lignins from Oil Palm Biomass as a Substitute for Phenol in a Phenol Formaldehyde Resin Production. Carbohydr. Polym. 2011, 86, 112–119. [Google Scholar] [CrossRef]
- Nogueira, C.d.L. Painel de Bambu Laminado Colado Estrutural. Master’s Thesis, Universidade de São Paulo, Piracicaba, Brazil, 2008. [Google Scholar]
- Macedo, L.A.; Rousset, P.L.A.; Vale, A.T. Influência Da Composição Da Biomassa No Rendimento Em Condensáveis Da Torrefação de Resíduos Vegetais. Pesqui. Florest. Bras. 2014, 34, 417–424. [Google Scholar] [CrossRef]
- Rousset, P.; Aguiar, C.; Labbé, N.; Commandré, J.-M. Enhancing the Combustible Properties of Bamboo by Torrefaction. Bioresour. Technol. 2011, 102, 8225–8231. [Google Scholar] [CrossRef]
- Hernandez-Mena, L.E.; Pécoraa, A.A.B.; Beraldo, A.L. Slow Pyrolysis of Bamboo Biomass: Analysis of Biochar Properties. Chem. Eng. Trans. 2014, 37, 115–120. [Google Scholar] [CrossRef]
- Lin, L.-D.; Chang, F.-C.; Ko, C.-H.; Wang, C.-T. Bamboo-Derived Fuel from Dendrocalamus latiflorus, Phyllostachys makinoi, and Phyllostachys pubescens Waste. Bioresources 2016, 11, 8425–8434. [Google Scholar] [CrossRef]
- Neves, D.; Thunman, H.; Matos, A.; Tarelho, L.; Gómez-Barea, A. Characterization and Prediction of Biomass Pyrolysis Products. Prog. Energy Combust. Sci. 2011, 37, 611–630. [Google Scholar] [CrossRef]
- Gontijo, L.O.L.; Junior, M.N.B.; de Sá, D.S.; Letichevsky, S.; Pedrozo-Peñafiel, M.J.; Aucélio, R.Q.; Bott, I.S.; Diniz Lopes Alves, H.; Fragneaud, B.; Oliveira Maciel, I.; et al. 3D Conductive Monolithic Carbons from Pyrolyzed Bamboo for Microfluidic Self-Heating System. Carbon 2023, 213, 118214. [Google Scholar] [CrossRef]
- Lin, Q.; Gao, R.; Li, D.; Lu, Y.; Liu, S.; Yu, Y.; Huang, Y.; Yu, W. Bamboo-Inspired Cell-Scale Assembly for Energy Device Applications. NPJ Flex. Electron. 2022, 6, 13. [Google Scholar] [CrossRef]
- Deng, J.; Xiong, T.; Xu, F.; Li, M.; Han, C.; Gong, Y.; Wang, H.; Wang, Y. Inspired by Bread Leavening: One-Pot Synthesis of Hierarchically Porous Carbon for Supercapacitors. Green Chem. 2015, 17, 4053–4060. [Google Scholar] [CrossRef]
- Jiang, J.; Zhu, J.; Ai, W.; Fan, Z.; Shen, X.; Zou, C.; Liu, J.; Zhang, H.; Yu, T. Evolution of Disposable Bamboo Chopsticks into Uniform Carbon Fibers: A Smart Strategy to Fabricate Sustainable Anodes for Li-Ion Batteries. Energy Environ. Sci. 2014, 7, 2670–2679. [Google Scholar] [CrossRef]
- Yin, T.; Zhang, Z.; Xu, L.; Li, C.; Han, D. Preparation of Green High-performance Biomass-Derived Hard Carbon Materials from Bamboo Powder Waste. ChemistryOpen 2024. [Google Scholar] [CrossRef]
- Subyakto, S.; Budiman, I.; Pari, G. Effects of Temperature and Time of Carbonization on the Properties of Bamboo (Dendrocalamus asper) Carbon. Wood Res. J. 2017, 4, 68–73. [Google Scholar] [CrossRef]
- Yang, W.; Li, J.; Zhang, L.; Zhu, X.; Liao, Q. A Monolithic Air Cathode Derived from Bamboo for Microbial Fuel Cells. RSC Adv. 2017, 7, 28469–28475. [Google Scholar] [CrossRef]
- Bian, Y.; Du, Q.; Tang, K.; Shen, Y.; Hao, L.; Zhou, D.; Wang, X.; Xu, Z.; Zhang, H.; Zhao, L.; et al. Carbonized Bamboos as Excellent 3D Solar Vapor-Generation Devices. Adv. Mater. Technol. 2019, 4, 1800593. [Google Scholar] [CrossRef]
- Liu, J.; Yao, J.; Yuan, Y.; Liu, Q.; Zhang, W.; Zhang, X.; Gu, J. Surface-Carbonized Bamboos with Multilevel Functional Biostructures Deliver High Photothermal Water Evaporation Performance. Adv. Sustain. Syst. 2020, 4, 2000126. [Google Scholar] [CrossRef]
- Ye, L.; Zhang, J.; Zhao, J.; Luo, Z.; Tu, S.; Yin, Y. Properties of Biochar Obtained from Pyrolysis of Bamboo Shoot Shell. J. Anal. Appl. Pyrolysis 2015, 114, 172–178. [Google Scholar] [CrossRef]
- Aggangan, N.S.; Cortes, A.D.; Reaño, C.E. Growth Response of Cacao (Theobroma cacao L.) Plant as Affected by Bamboo Biochar and Arbuscular Mycorrhizal Fungi in Sterilized and Unsterilized Soil. Biocatal. Agric. Biotechnol. 2019, 22, 101347. [Google Scholar] [CrossRef]
- Chen, J.; Li, S.; Liang, C.; Xu, Q.; Li, Y.; Qin, H.; Fuhrmann, J.J. Response of Microbial Community Structure and Function to Short-Term Biochar Amendment in an Intensively Managed Bamboo (Phyllostachys praecox) Plantation Soil: Effect of Particle Size and Addition Rate. Sci. Total Environ. 2017, 574, 24–33. [Google Scholar] [CrossRef]
- Yin, Y.; Gu, M.; Zhang, W.; Yang, C.; Li, H.; Wang, X.; Chen, R. Relationships between Different Types of Biochar and N2O Emissions during Composting Based on Roles of NosZ-Carrying Denitrifying Bacterial Communities Enriched on Compost and Biochar Particles. Bioresour. Technol. 2024, 394, 130214. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Tao, X.; Du, Y.; Li, M.; Yang, S.; Zhang, W.; Yang, C.; Li, H.; Wang, X.; Chen, R. Biochar Improves the Humification Process during Pig Manure Composting: Insights into Roles of the Bacterial Community and Metabolic Functions. J. Environ. Manag. 2024, 355, 120463. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Méndez, A.S.; Ortega-Ramírez, E.; Lucho-Constantino, C.A.; Arce-Cervantes, O.; Vázquez-Rodríguez, G.A.; Coronel-Olivares, C.; Beltrán-Hernández, R.I. Bamboo Biochar and a Nopal-Based Biofertilizer as Improvers of Alkaline Soils with Low Buffer Capacity. Appl. Sci. 2021, 11, 6502. [Google Scholar] [CrossRef]
- Situmeang, Y.P.; Sudewa, K.A.; Holo, P.P. Utilization Biochar of Bamboo and Compost in Improving Yield of Pakchoy Plant. J. Biol. Chem. Res. 2017, 34, 713–722. [Google Scholar]
- Suthar, R.; Wang, C.; Nunes, M.; Chen, J.; Sargent, S.; Bucklin, R.; Gao, B. Bamboo Biochar Pyrolyzed at Low Temperature Improves Tomato Plant Growth and Fruit Quality. Agriculture 2018, 8, 153. [Google Scholar] [CrossRef]
- Villagra-Mendoza, K.; Masís-Meléndez, F.; Quesada-Kimsey, J.; García-González, C.A.; Horn, R. Physicochemical Changes in Loam Soils Amended with Bamboo Biochar and Their Influence in Tomato Production Yield. Agronomy 2021, 11, 2052. [Google Scholar] [CrossRef]
- Wang, Y.; Zhong, B.; Shafi, M.; Ma, J.; Guo, J.; Wu, J.; Ye, Z.; Liu, D.; Jin, H. Effects of Biochar on Growth, and Heavy Metals Accumulation of Moso Bamboo (Phyllostachy pubescens), Soil Physical Properties, and Heavy Metals Solubility in Soil. Chemosphere 2019, 219, 510–516. [Google Scholar] [CrossRef]
- Tarin, M.W.K.; Fan, L.; Xie, D.; Tayyab, M.; Rong, J.; Chen, L.; Muneer, M.A.; Zheng, Y. Response of Soil Fungal Diversity and Community Composition to Varying Levels of Bamboo Biochar in Red Soils. Microorganisms 2021, 9, 1385. [Google Scholar] [CrossRef]
- Wang, C.; Alidoust, D.; Yang, X.; Isoda, A. Effects of Bamboo Biochar on Soybean Root Nodulation in Multi-Elements Contaminated Soils. Ecotoxicol. Environ. Saf. 2018, 150, 62–69. [Google Scholar] [CrossRef]
- Tang, L.; Xiong, L.; Zhang, H.; Joseph, A.; Wang, Y.; Li, J.; Yuan, X.; Rene, E.R.; Zhu, N. Reduced Arsenic Availability in Paddy Soil through Fe-Organic Ligand Complexation Mediated by Bamboo Biochar. Chemosphere 2024, 349, 140790. [Google Scholar] [CrossRef]
- Hua, L.; Wu, W.; Liu, Y.; McBride, M.B.; Chen, Y. Reduction of Nitrogen Loss and Cu and Zn Mobility during Sludge Composting with Bamboo Charcoal Amendment. Environ. Sci. Pollut. Res. 2009, 16, 1–9. [Google Scholar] [CrossRef]
- Ji, Y.; Zhang, C.; Zhang, X.J.; Xie, P.F.; Wu, C.; Jiang, L. A High Adsorption Capacity Bamboo Biochar for CO2 Capture for Low Temperature Heat Utilization. Sep. Purif. Technol. 2022, 293, 121131. [Google Scholar] [CrossRef]
- Liu, Y.; Gan, L.; Chen, Z.; Megharaj, M.; Naidu, R. Removal of Nitrate Using Paracoccus sp. YF1 Immobilized on Bamboo Carbon. J. Hazard. Mater. 2012, 229–230, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Viglašová, E.; Galamboš, M.; Danková, Z.; Krivosudský, L.; Lengauer, C.L.; Hood-Nowotny, R.; Soja, G.; Rompel, A.; Matík, M.; Briančin, J. Production, Characterization and Adsorption Studies of Bamboo-Based Biochar/Montmorillonite Composite for Nitrate Removal. Waste Manag. 2018, 79, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, K.; Matsumoto, T.; Hatate, Y.; Nishihara, K.; Nakanishi, T. Removal of Nitrate-Nitrogen from Drinking Water Using Bamboo Powder Charcoal. Bioresour. Technol. 2004, 95, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Asada, T.; Ohkubo, T.; Kawata, K.; Oikawa, K. Ammonia Adsorption on Bamboo Charcoal with Acid Treatment. J. Health Sci. 2006, 52, 585–589. [Google Scholar] [CrossRef]
- Deng, P.; Wan, W.; Azeem, M.; Riaz, L.; Zhang, W.; Yang, Y.; Li, C.; Yuan, W. Characterization of Biochar Derived from Bamboo and Its Application to Modulate the Toxic Effects of Chromium on Wheat Plant. Biomass Convers. Biorefin. 2024, 14, 7643–7658. [Google Scholar] [CrossRef]
- Huang, A.; Bai, W.; Yang, S.; Wang, Z.; Wu, N.; Zhang, Y.; Ji, N.; Li, D. Adsorption Characteristics of Chitosan-Modified Bamboo Biochar in Cd(II) Contaminated Water. J. Chem. 2022, 2022, 6303252. [Google Scholar] [CrossRef]
- Mohamed, I.; Zhang, G.; Li, Z.; Liu, Y.; Chen, F.; Dai, K. Ecological Restoration of an Acidic Cd Contaminated Soil Using Bamboo Biochar Application. Ecol. Eng. 2015, 84, 67–76. [Google Scholar] [CrossRef]
- Ma, W.; Han, R.; Zhang, W.; Zhang, H.; Chen, L.; Zhu, L. Magnetic Biochar Enhanced Copper Immobilization in Agricultural Lands: Insights from Adsorption Precipitation and Redox. J. Environ. Manag. 2024, 352, 120058. [Google Scholar] [CrossRef]
- Zhang, X.; Xue, J.; Han, H.; Wang, Y. Study on Improvement of Copper Sulfide Acid Soil Properties and Mechanism of Metal Ion Fixation Based on Fe-Biochar Composite. Sci. Rep. 2024, 14, 247. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Long, Q.; Zhu, Y.; Lin, C.; Xu, X.; Pan, B.; Shi, W.; Guo, Y.; Deng, J.; Yao, Q.; et al. Multifunctional Self-Assembled Adsorption Microspheres Based on Waste Bamboo Shoot Shells for Multi-Pollutant Water Purification. Environ. Res. 2024, 249, 118452. [Google Scholar] [CrossRef]
- Tan, Z.; Qiu, J.; Zeng, H.; Liu, H.; Xiang, J. Removal of Elemental Mercury by Bamboo Charcoal Impregnated with H2O2. Fuel 2011, 90, 1471–1475. [Google Scholar] [CrossRef]
- Wang, F.Y.; Wang, H.; Ma, J.W. Adsorption of Cadmium (II) Ions from Aqueous Solution by a New Low-Cost Adsorbent—Bamboo Charcoal. J. Hazard. Mater. 2010, 177, 300–306. [Google Scholar] [CrossRef]
- Wang, S.Y.; Tsai, M.H.; Lo, S.F.; Tsai, M.J. Effects of Manufacturing Conditions on the Adsorption Capacity of Heavy Metal Ions by Makino Bamboo Charcoal. Bioresour. Technol. 2008, 99, 7027–7033. [Google Scholar] [CrossRef]
- Lo, S.-F.; Wang, S.-Y.; Tsai, M.-J.; Lin, L.-D. Adsorption Capacity and Removal Efficiency of Heavy Metal Ions by Moso and Ma Bamboo Activated Carbons. Chem. Eng. Res. Des. 2012, 90, 1397–1406. [Google Scholar] [CrossRef]
- González, P.G.; Pliego-Cuervo, Y.B. Adsorption of Cd(II), Hg(II) and Zn(II) from Aqueous Solution Using Mesoporous Activated Carbon Produced from Bambusa vulgaris Striata. Chem. Eng. Res. Des. 2014, 92, 2715–2724. [Google Scholar] [CrossRef]
- Lalhruaitluanga, H.; Prasad, M.N.V.; Radha, K. Potential of Chemically Activated and Raw Charcoals of Melocanna baccifera for Removal of Ni(II) and Zn(II) from Aqueous Solutions. Desalination 2011, 271, 301–308. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, B.; Yuan, S.; Wu, X.; Chen, J.; Wang, L. Adsorptive Removal of Chloramphenicol from Wastewater by NaOH Modified Bamboo Charcoal. Bioresour. Technol. 2010, 101, 7661–7664. [Google Scholar] [CrossRef]
- Li, Y.; Shao, J.; Wang, X.; Deng, Y.; Yang, H.; Chen, H. Characterization of Modified Biochars Derived from Bamboo Pyrolysis and Their Utilization for Target Component (Furfural) Adsorption. Energy Fuels 2014, 28, 5119–5127. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Fang, J.; Zhang, M.; Chen, H.; Zhou, Y.; Creamer, A.E.; Sun, Y.; Yang, L. Characterization and Environmental Applications of Clay–Biochar Composites. Chem. Eng. J. 2014, 242, 136–143. [Google Scholar] [CrossRef]
- Wang, T.; Liu, X.; Ma, C.; Liu, Y.; Dong, H.; Ma, W.; Liu, Z.; Wei, M.; Li, C.; Yan, Y. A Two Step Hydrothermal Process to Prepare Carbon Spheres from Bamboo for Construction of Core–Shell Non-Metallic Photocatalysts. New J. Chem. 2018, 42, 6515–6524. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, J.; Wu, J.; Liu, Q.; Zhang, H.; Jin, S. Adsorptive Removal of Fluoroquinolone Antibiotics Using Bamboo Biochar. Sustainability 2015, 7, 12947–12957. [Google Scholar] [CrossRef]
- Xu, T.; Lou, L.; Luo, L.; Cao, R.; Duan, D.; Chen, Y. Effect of Bamboo Biochar on Pentachlorophenol Leachability and Bioavailability in Agricultural Soil. Sci. Total Environ. 2012, 414, 727–731. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lin, X.; Wei, B.; Zhao, Y.; Wang, J. Evaluation of Adsorption Potential of Bamboo Biochar for Metal-Complex Dye: Equilibrium, Kinetics and Artificial Neural Network Modeling. Int. J. Environ. Sci. Technol. 2014, 11, 1093–1100. [Google Scholar] [CrossRef]
- Zhang, X.; Sarmah, A.K.; Bolan, N.S.; He, L.; Lin, X.; Che, L.; Tang, C.; Wang, H. Effect of Aging Process on Adsorption of Diethyl Phthalate in Soils Amended with Bamboo Biochar. Chemosphere 2016, 142, 28–34. [Google Scholar] [CrossRef]
- Liao, P.; Yuan, S.; Xie, W.; Zhang, W.; Tong, M.; Wang, K. Adsorption of Nitrogen-Heterocyclic Compounds on Bamboo Charcoal: Kinetics, Thermodynamics, and Microwave Regeneration. J. Colloid Interface Sci. 2013, 390, 189–195. [Google Scholar] [CrossRef]
- Liu, X.-M.; Huan, W.-W.; Kang, Y.; Guo, J.-Z.; Wang, Y.-X.; Li, F.-H.; Li, B. Effects of Cation Types in Persulfate on Physicochemical and Adsorptive Properties of Biochar Prepared from Persulfate-Pretreated Bamboo. Bioresour. Technol. 2024, 393, 130140. [Google Scholar] [CrossRef]
- Hameed, B.H.; Din, A.T.M.; Ahmad, A.L. Adsorption of Methylene Blue onto Bamboo-Based Activated Carbon: Kinetics and Equilibrium Studies. J. Hazard. Mater. 2007, 141, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Zhan, Z.; Dai, J.; Wu, X.; Zhang, W.; Wang, K.; Yuan, S. Adsorption of Tetracycline and Chloramphenicol in Aqueous Solutions by Bamboo Charcoal: A Batch and Fixed-Bed Column Study. Chem. Eng. J. 2013, 228, 496–505. [Google Scholar] [CrossRef]
- Men, Q.; Wang, T.; Ma, C.; Yang, L.; Liu, Y.; Huo, P.; Yan, Y. In-Suit Preparation of CdSe Quantum Dots/Porous Channel Biochar for Improving Photocatalytic Activity for Degradation of Tetracycline. J. Taiwan Inst. Chem. Eng. 2019, 99, 180–192. [Google Scholar] [CrossRef]
- Zhao, D.; Zhang, J.; Duan, E.; Wang, J. Adsorption Equilibrium and Kinetics of Dibenzothiophene from N-Octane on Bamboo Charcoal. Appl. Surf. Sci. 2008, 254, 3242–3247. [Google Scholar] [CrossRef]
- Li, S.; Umereweneza, D. Adsorption of N-Vinylpyrrolidone from Polyvinylpyrrolidone Solution onto Bamboo-Based Activated Carbon. Sep. Sci. Technol. 2012, 47, 104–111. [Google Scholar] [CrossRef]
- Lin, J.; Xu, Z.; Zhang, Q.; Cao, Y.; Mašek, O.; Lei, H.; Tsang, D.C.W. Enhanced Adsorption of Aromatic VOCs on Hydrophobic Porous Biochar Produced via Microwave Rapid Pyrolysis. Bioresour. Technol. 2024, 393, 130085. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Xie, T.; Liu, S.; Bai, L. Fabrication of a Biochar-Doped Monolithic Adsorbent and Its Application for the Extraction and Determination of Coumarins from Angelicae pubescentis Radix. J. Chromatogr. A 2024, 1714, 464564. [Google Scholar] [CrossRef] [PubMed]
- Jagnade, P.; Panwar, N.L. Experiment Investigation on the Performance of Activated Bamboo Biochar for the Adsorption of CO2 and PM2.5. Emergent Mater. 2024. [Google Scholar] [CrossRef]
- Wei, Y.; Shen, C.; Xie, J.; Bu, Q. Study on Reaction Mechanism of Superior Bamboo Biochar Catalyst Production by Molten Alkali Carbonates Pyrolysis and Its Application for Cellulose Hydrolysis. Sci. Total Environ. 2020, 712, 136435. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, Q.; Xiao, Y.; Zhang, C.; Fu, Z.; Liu, Y.; Yi, X.; Zheng, A.; Li, C.; Yin, D. Acid–Base Synergistic Catalysis of Biochar Sulfonic Acid Bearing Polyamide for Microwave-Assisted Hydrolysis of Cellulose in Water. Cellulose 2019, 26, 751–762. [Google Scholar] [CrossRef]
- Chen, W.; Fang, Y.; Li, K.; Chen, Z.; Xia, M.; Gong, M.; Chen, Y.; Yang, H.; Tu, X.; Chen, H. Bamboo Wastes Catalytic Pyrolysis with N-Doped Biochar Catalyst for Phenols Products. Appl. Energy 2020, 260, 114242. [Google Scholar] [CrossRef]
- Chen, W.; Li, K.; Xia, M.; Yang, H.; Chen, Y.; Chen, X.; Che, Q.; Chen, H. Catalytic Deoxygenation Co-Pyrolysis of Bamboo Wastes and Microalgae with Biochar Catalyst. Energy 2018, 157, 472–482. [Google Scholar] [CrossRef]
- Yang, H.; Chen, Z.; Chen, W.; Chen, Y.; Wang, X.; Chen, H. Role of Porous Structure and Active O-Containing Groups of Activated Biochar Catalyst during Biomass Catalytic Pyrolysis. Energy 2020, 210, 118646. [Google Scholar] [CrossRef]
- Niu, S.; Ning, Y.; Lu, C.; Han, K.; Yu, H.; Zhou, Y. Esterification of Oleic Acid to Produce Biodiesel Catalyzed by Sulfonated Activated Carbon from Bamboo. Energy Convers. Manag. 2018, 163, 59–65. [Google Scholar] [CrossRef]
- Situmeang, Y.P. The Use of Bamboo Biochar as a Soil Improver on the Growth and Yield of Mustard Plants. Agriwar J. 2022, 2, 14–18. [Google Scholar] [CrossRef]
- Zhang, S.-Z.; Cui, Z.-S.; Zhang, M.; Zhang, Z.-H. Biochar-Based Functional Materials as Heterogeneous Catalysts for Organic Reactions. Curr. Opin. Green Sustain. Chem. 2022, 38, 100713. [Google Scholar] [CrossRef]
- Huang, D.; Luo, H.; Zhang, C.; Zeng, G.; Lai, C.; Cheng, M.; Wang, R.; Deng, R.; Xue, W.; Gong, X.; et al. Nonnegligible Role of Biomass Types and Its Compositions on the Formation of Persistent Free Radicals in Biochar: Insight into the Influences on Fenton-like Process. Chem. Eng. J. 2019, 361, 353–363. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, M.; He, R.; Zhao, J.; Kang, W.; Lv, J. Effect of Pyrolysis Temperature on the Activated Permonosulfate Degradation of Antibiotics in Nitrogen and Sulfur-Doping Biochar: Key Role of Environmentally Persistent Free Radicals. Chemosphere 2022, 294, 133737. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, X.; Yang, K.; Lin, D. Differential Photodegradation Processes of Adsorbed Polychlorinated Biphenyls on Biochar Colloids with Various Pyrolysis Temperatures. Water Res. 2024, 251, 121174. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Ge, Q.; Wan, J.; Wang, Y.; Zhu, C. Insights into the Influence and Mechanism of Biomass Substrate and Thermal Conversion Conditions on Fe N Doped Biochar as a Persulfate Activator for Sulfamethoxazole Removal. Sci. Total Environ. 2024, 907, 168101. [Google Scholar] [CrossRef] [PubMed]

| Source Biomass | Ref. | Application | Refs. |
|---|---|---|---|
| Crop residue | [23] | Carbon sequestration Soil amendment Composting Wastewater treatment Concrete additive Adsorbing xenobiotics Reducing greenhouse gas emissions Pollutant degradation Catalysis Stock fodder | [24] [25] [26] [27] [28,29] [30] [31] [32] [33] [34] |
| Kitchen waste | [35] | ||
| Forestry | [36] | ||
| Agricultural waste | [37] | ||
| Sugar beet tailings | [38] | ||
| Forest residues | [39] | ||
| Waste wood | [40] | ||
| Bioenergy crops | [41] | ||
| Municipal solid waste | [42] | ||
| Wheat straw | [43] | ||
| Rice straw | [44] | ||
| Food manure | [45] | ||
| Animal manure | [46] | ||
| Corn cob | [47] |
| Application | Mechanisms | Refs. |
|---|---|---|
| Climate change mitigation | Sequestering carbon in soil, ⇓ CO2 emissions ⇓ NO2 emissions, ⇓ CH4 emissions Tackling 12% of current anthropogenic carbon emissions | [31] |
| Soil improvement | ⇑ Physicochemical and biological properties ⇑ Water retention capacity, ⇓ nutrient leaching ⇓ Acids, ⇑ microbial population and microbial activity Positive impacts on earthworm population Preventing desiccation | [25] |
| Waste management | Simply by pyrolyzing waste biomass * | [48] |
| Energy production | By conversion of waste biomass to BC **, providing liquid fuel (bio-oil) | [49] |
| Capturing contaminants | By adsorption of both organic xenobiotics and metal ions present in soil and water | [30,32] |
| Composting | ⇑ Physicochemical properties of composting ⇑ Enhance composting microbial activities ⇑ Organic matter decomposition | [26,32] |
| Catching Mechanism | Influencing Factors #, Details °, Examples § | Ref. |
|---|---|---|
| Adsorption * | ⇑ Surface area # Microporosity of BC # pH # | [71] |
| Hydrogen bond formation ** | For polar compounds °,** | |
| Electrostatic attraction/repulsion | For cationic compounds ° Interaction between positively charged cationic organic contaminants and negatively charged BC surfaces °,** | |
| Electrostatic outer sphere complexation | Due to metallic exchange with K+ and Na+ available in BC °,** | |
| Hydrophobic interactions *** | For non-polar compounds ° | |
| Diffusion | Non-ionic compounds can diffuse into the non-carbonized and carbonized fractions of BC ° | |
| Formation of surface complexes ** | pH # Ionic radius # Between metal cations and -OH, -COOH on BCs ° | |
| Precipitation | Lead precipitates as lead-phosphate-silicate in BC § Co-precipitates and inner-sphere complexes can form between metals and organic matter/mineral oxides of BC § |
| Components | Chemical Structure/Class of Substances | Description | (%) | Refs. |
|---|---|---|---|---|
| Cellulose | 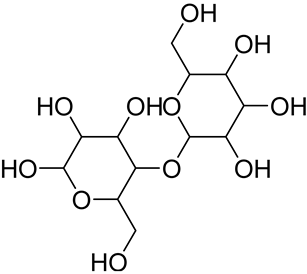 Portion of cellulose | Main material responsible for fibers’ stability and mechanical strength, allowing the formation of compact fibers Higher thermal stability and resistance to mechanical stresses in comparison to other non-cellulosic plant fiber components Exhibit areas with a flexible structure (amorphous cellulose) and areas with an ordinated, rigid, and non-flexible structure (crystalline cellulose) | 40–60 * | [118,119,120,121] |
| Hemicellulose (xylan) ** | 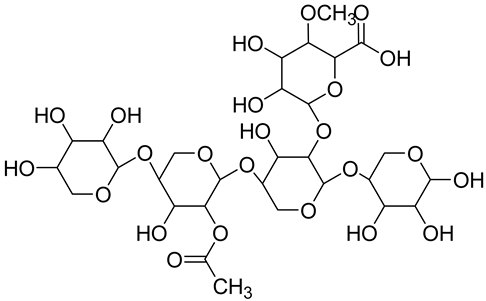 The most present hemicellulose in bamboo | Consists of a heterogeneous group of polysaccharides not forming a well-arranged fibrous network (amorphous structure) Low polymerization degree Easily absorbs water | 25 § | [116,122] |
| Lignin | 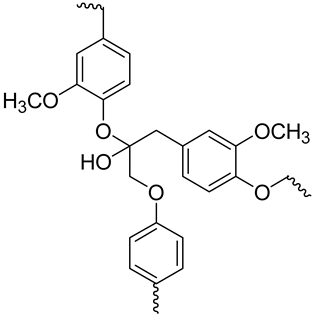 Portion of lignin | Amorphous phenolic macromolecule composed of phenyl propane units (C6-C3) No crystalline structure High resistance Cellulose and hemicellulose cells constitute the wall matrix Prevents cellulose and hemicellulose degradation, providing strength and rigidity to plant tissues Energy storage Polymerization degree (PD) > 15,000 High molecular weight | 20–30 | [116,120,121,123] |
| Extractives | N.R. | Aromatic organic compounds, including fatty acids, terpenes, flavonoids, and steroids | N.R. | [120,122,124] |
| Ash | N.R. | Mainly in the interior of the stem | 1–5 | N.R. |
| Starch | 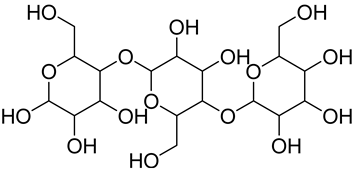 Portion of starch | Production by the cellular activity of chlorophyllized vegetables Attractive for xylophagous organisms, especially for Dinoderus minutus | 2–5 | [124] |
| Moisture | N.R. | N.R. | 6.1 | N.R. |
| Proteins | N.R. | N.R. | 1.5–6 | [124] |
| Glucose | N.R. | N.R. | 2 | [124] |
| Waxes, Resins | N.R. | N.R. | 2–3.5 | [124] |
| Silica | N.R. | Mainly in the epidermis, increasing from bottom to top Nonexistent in the internode tissues | 1–6 | N.R. |
| Biomass | C | H | N | O | H/C | O/C | Sources |
|---|---|---|---|---|---|---|---|
| Bambusa vulgaris | 49.60 | 6.10 | 0.40 | 44.00 | 0.12 | 0.89 | [125] |
| Bambusa vulgaris | 46.80 | 6.38 | 0.22 | 46.60 | 0.14 | 0.99 | [126] |
| Dendrocalamus giganteus | 44.26 | 5.48 | 0.46 | 42.66 | 0.12 | 0.96 | [127] |
| Dendrocalamus latiflorus | 44.22 | 6.10 | 0.07 | 45.63 | 0.14 | 1.03 | [128] |
| Phyllostachys makinoi | 43.90 | 6.06 | 0.06 | 41.47 | 0.14 | 0.94 | [128] |
| Phyllostachys pubescens | 45.25 | 5.71 | 0.08 | 43.89 | 0.13 | 0.97 | [128] |
| Wood * | 45.68 | 6.30 | 0.30 | 47.42 | 0.14 | 1.04 | [129] |
| Forest residue | 51.40 | 6.00 | 0.50 | 40.00 | 0.12 | 0.78 | [129] |
| Pine | 47.79 | 5.80 | 0.10 | 45.31 | 0.12 | 0.95 | [129] |
| Rice husk | 47.30 | 6.10 | 0.90 | 45.70 | 0.13 | 0.97 | [125] |
| Sugar cane bagasse | 48.10 | 5.90 | 0.50 | 45.50 | 0.12 | 0.95 | [125] |
| Jatropha bark | 50.80 | 6.50 | 1.50 | 41.30 | 0.13 | 0.81 | [125] |
| Elephant grass | 49.20 | 6.10 | 1.10 | 43.60 | 0.12 | 0.89 | [125] |
| Bamboo Biomass | Pyrolysis Conditions | Reactor | Char (%) Other (%) | Characteristics Applications | Refs. |
|---|---|---|---|---|---|
| Dendrocalamus giganteus | 300 °C Slow pyrolysis | Fixed bad type | BC 80% ## Oil 35% $ Gas 40% @ | ⇑ Porosity ⇑ Carbon concentration As AC after chemical/physical modification Similar to wood biochar Energy source Soil ameliorant | [127] |
| 200–1000 °C | Tube furnace (MTI) | N.R. | 700 °C ⇑ Resistivity ⇑ Thermal conductivity ⇑ Thermal heating rate As a 3D microfluidic heater | [130] | |
| 1000 °C ⇑ Electric conductivity As working electrode | |||||
| Phyllostachy edulis | N.R. | N.R. | N.R. | Ag-carbon electrodes for energy device applications | [131] |
| Bamboo waste | KHCO3 400 °C/3 h | Muffle furnace | N.R. | Excellent electrochemical performance as supercapacitor electrode materials | [132] |
| Bamboo chopsticks | 800 °C/2 h (alkali) | N.R. | N.R. | Sustainable anodes for Li-ion batteries | [133] |
| Bamboo powder waste (alkali-activated) | 1000 °C/15 min | Tube furnace | N.R. | Sustainable anodes for Na-ion batteries | [134] |
| Dendrocalamus asper | 400 700 800 900 °C | N.R. | N.R. | N.R. | [135] |
| Phyllostachys pubescens Mazel | 900 °C | Tube furnace | N.R. | BCT-derived air cathode for microbial fuel cells | [136] |
| Phyllostachys edulis | 350 °C/60 min 500 °C/40 min 900 °C/240 min | N.R. | N.R. | 3D solar vapor-generation device for water desalination | [137] |
| Local defoliated bamboo | Surface-carbonized | N.R. | N.R. | Efficient photothermal-conversion devices | [138] |
| Agricultural by-product (BSS) d (D. latiforus Munro) | 300 °C to 500 °C | Tubular furnace | 48% | As AC when chemically/physically modified soil ameliorant ⇑ Porosity ⇑ Carbon concentration | [139] |
| Dry bamboo stalks | 400 °C to 600 °C Slow pyrolysis | Muffle furnace | 32% to 27% | In place of industrially produced AC ⇑ Porosity ⇑ Carbon concentration | [113] |
| Bamboo waste | 500 °C | Fabricated close tank | N.R. | ⇑ Soil fertility and crop growth | [140] |
| Bamboo tick (P. praecox) | 700 °C/4 h | Closed container | N.R. | ⇑ Soil acidification ⇑ Soil C and nutrient retention ⇑ Microbial community abundance ⇓ CO2 | [141] |
| Bamboo | 500 °C | N.R. | N.R. | ⇓ NO2 emissions in thermophilic phase of composting ⇑ nosZ-carrying denitrifying bacteria | [142] |
| Commercial BBC | N.R. | N.R. | N.R. | ⇑ Humidification during pig manure composting ⇑ Humic acid (HA) ⇑ HA/Fulvic acid (FA) ratio ⇑ Bacteria transforming organic matter | [143] |
| Residual bamboo biomass | 450–550 °C | N.R. | N.R. | Soil amendment | [144] |
| Bamboo waste | N.R. | N.R. | N.R. | Improving yield of pakchoy plant | [145] |
| Bamboo feedstock | 300–600 °C | Furnace apparatus | N.R. | ⇑ Tomato plant growth ⇑ Fruit quality | [146] |
| Bamboo stems (culms) a | 400 °C/30 min | Sealed metallic kiln | N.R. | ⇑ Physicochemical properties of SL, SiL ⇑ Tomato productivity | [147] |
| N.R. | 500 °C/2 h | N.R. | N.R. | ⇓ Cu uptake in roots ⇓ Solubility of soil heavy metals | [148] |
| N.R. | N.R. | N.R. | N.R. | ⇑ pH in red soil ⇑ Soil nutrients ⇑ Abundance of Basidiomycota Mucoromycota, Chytridiomycota | [149] |
| N.R. | <500 °C | N.R. | N.R. | ⇓ Mobile Cd, Cu, Mn, Ni, Zn ⇓ Pb, Mn, Cd, Zn, Cu, Ni uptake in soybean shoots ⇑ Root nodulation ⇑ Soybean growth ⇑ Plant K and Mo uptake ⇑ Soybean physiological performance | [150] |
| Bamboo chips | 300 °C/1 h 450 °C/1 h 600 °C/1 h | Muffle furnace | N.R. | Remediation of As-contaminated paddy soil via iron–organic ligand complexation | [151] |
| Bamboo charcoal particles b | 600 °C | N.R. | N.R. | As stabilizer for heavy metals Nitrogen retention in sludge composting | [152] |
| Bamboo sawdust | 1000 °C | N.R. | N.R. | For CO2 capturing ⇓ Regeneration temperature Excellent adsorption capacity | [153] |
| Bamboo carbon | 600 °C/2 h | Muffle furnace | N.R. | BBC §-immobilized Paracoccus sp. YF1 for nitrates remediation | [154] |
| P. virdiglaucesons | 460 °C Slow pyrolysis | Rotary furnace | 50% | Nitrate absorption from wastewater or industrial effluents * | [155] |
| Residual of Moso bamboo manufacturing | 900 °C/1 h | Electric furnace | N.R. | Nitrate-nitrogen adsorption | [156] |
| Giant timber bamboo (P. bambusoides) | 400, 700, 1000 °C/1 h | Charcoal kiln | N.R. | Ammonia absorption | [157] |
| Healthy dried stems without leaves of bamboo | 500 °C/20 min | Muffle furnace | N.R. | Modulate the toxic effects of chromium | [158] |
| Offcuts of bamboo c | 900 °C/4 h | Vacuum annealing furnace | N.R. | Remediation of Cd (II) in water | [159] |
| Bamboo residues | 400 °C | N.R. | N.R. | Restoration of acidic Cd-contaminated soil | [160] |
| Local bamboo | 600 °C/4 h | N.R. | N.R. | Cu adsorption from soil ⇓ Cu accumulation in lettuce | [161] |
| Bamboo pieces | 600 °C/5 h | Tube furnace | N.R. | Cu absorption from soil ⇓ Soil acidity ⇓ Zn and Pb | [162] |
| Bamboo shoot shells | 500 °C/3 h | Muffle furnace | N.R. | Removal of Ag (I) and Pd (II) Removal of TC and MB | [163] |
| N.R. | N.R. | N.R. | N.R. | Removal of elemental mercury | [164] |
| N.R. °°° | N.R. | N.R. | N.R. | Removal of Cd (II) ions from water | [165] |
| Makino bamboo (P. makinoi Hayata) d | 800–900 °C/2 h | Furnace | N.R. | Removal of heavy metal ions from water | [166] |
| Moso (P. pubescens) and Ma (D. latiflorus) bamboo slices e | 800 °C/1 h | Furnace | N.R. | Removal of heavy metal ions from water | [167] |
| B. vulgaris striata f | 650 °C/2 h | N.R. | N.R. | Adsorption of Cd (II), Hg (II), and Zn (II) from aqueous solution | [168] |
| Bundles of bamboo culms (Melocanna baccifera) g | N.R./2–4 h | Kiln | N.R. | Removal of Ni (II) and Zn (II) from aqueous solutions | [169] |
| Commercial bamboo charcoal | >450 °C | N.R. | N.R. | Activated by NaOH treatment ⇑ Percentage of surface graphitic carbon ⇑ Oxygen-containing groups ⇑ π–π interactions Adsorptive removal of chloramphenicol | [170] |
| Bamboo pieces | 550 °C | Fluidized bed reactor | N.R. | 100% Furfural removal | [171] |
| Bamboo | 600 °C/1 h | Tube furnace | N.R. | Removal of MB by electrostatic interactions | [172] |
| Bamboo from authors’ campus (Jiangsu University, China) | 200 °C/6 h 180 °C/3 h | Teflon-lined stainless steel autoclave | N.R. | As core–shell non-metallic photocatalysts for the photocatalytic decomposition of tetracyclines | [173] |
| Bamboo sawdust | 500 °C | N.R. | N.R. | Removal of fluoroquinolone antibiotics | [174] |
| Bamboo waste | 600 °C | N.R. | N.R. | In situ remediation of PCP | [175] |
| Bamboo waste | 1000 °C | N.R. | N.R. | Removal of MCAB-172 | [176] |
| Bamboo waste | 820 °C | Stainless steel vessel | N.R. | ⇓ Bioavailability of DEP | [177] |
| Moso (P. pubescens) bamboo | 800 °C Fast pyrolysis | N.R. | N.R. | Removal of pyridine, indole, quinoline | [178] |
| Bundles of bamboo culms (Melocanna baccifera) h | 800 °C Fast pyrolysis | N.R. | N.R. | Removal of MB and AO7 | [109] |
| Bamboo dust i | 240 °C/2 h | Tube furnace | N.R. | Removal of MB | [179] |
| Bamboo g | 700 °C/1 h (1st pyrolysis) 850 °C/2 h (activation) | Tube furnace | N.R. | Removal of MB | [180] |
| Moso (P. pubescens) bamboo sections | 700 °C Fast pyrolysis | N.R. | N.R. | Removal of CAF and TC | [181] |
| Bamboo waste (China) | 650 °C/1 h | Muffle furnace | N.R. | CdSe quantum dots/porous channel BBC for improved photocatalytic degradation of TC | [182] |
| Purchased BBC | N.R. | N.R. | N.R. | Removal of DBT | [183] |
| Bamboo sawdust g | 873.15 K/1 h (carbonization) 1073 K/0.5 h (activation) | Tube furnace | N.R. | Removal of NVP | [184] |
| Bamboo waste | 25 °C up to 850 °C | Microwave (2450 MHz) | N.R. | Absorption of toluene and benzene ⇑ Humidity resistance | [185] |
| Bamboo | N.R. | N.R. | N.R. | Extraction and determination of coumarins from Angelicae pubescentis Radix | [186] |
| Bamboo wood (KOH-activated) | 500–700 °C/60–120 min | Vacuum pyrolysis machine | 29–34% char 37–39% oil 26–33% gas | Adsorption of CO2 and PM2.5 | [187] |
| Bamboo power * | 450–600 °C | 2 L Cylindrical reactor | 9–29.82% ** 0.91–2.41% *** | As BC-supported sulfonic acid catalyst for cellulose hydrolysis | [188] |
| Phyllostachys edulis | 353 K/3 h | N.R. | N.R. | BBC sulfonic acid bearing polyamide for microwave-assisted hydrolysis of cellulose | [189] |
| Bamboo waste | 600 °C/30 min Fast pyrolysis | Fixed-bed system | 84.7 wt% °° | To prepare phenols | [190] |
| Bamboo waste # | 600 °C/30 min | Fixed-bed reactor | Char 19% Oil 42% H2O 18% Gas 18% | Formation of aromatics and phenols | [191] |
| Bamboo waste ° | 600 °C/30 min | Fixed-bed reactor | N.R. | Formation of phenols (67%) | [192] |
| Bamboo activated carbon (BAC) | N.R. | N.R. | N.R. | Sulfonated BAC-based catalyst for oleic acid esterification | [193] |
| Source of BBC | Application | Process | Radicals Active Site | Radicals | Refs. | |
|---|---|---|---|---|---|---|
| Bamboo | Tetracycline degradation | Fenton-like | PFRs | •OH | [196] | |
| Moso bamboo ** | PFX, OTC, CTC degradation | Oxidation PDS activation | OCFRs CCFRs-O CCFRs | g2 > 2.0040 2.0030 < g3 < 2.0040 g1 < 2.0030 | •OH SO4•− | [197] |
| Bamboo chips | PCB28 degradation | Electron transfer * Oxidation * | PFRs * | •OH * | [198] | |
| Bamboo | SMX, TOC degradation | Electron transfer Oxidation PDS activation | PFRs | •OH, SO4•− •O2 | [199] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alfei, S.; Pandoli, O.G. Bamboo-Based Biochar: A Still Too Little-Studied Black Gold and Its Current Applications. J. Xenobiot. 2024, 14, 416-451. https://doi.org/10.3390/jox14010026
Alfei S, Pandoli OG. Bamboo-Based Biochar: A Still Too Little-Studied Black Gold and Its Current Applications. Journal of Xenobiotics. 2024; 14(1):416-451. https://doi.org/10.3390/jox14010026
Chicago/Turabian StyleAlfei, Silvana, and Omar Ginoble Pandoli. 2024. "Bamboo-Based Biochar: A Still Too Little-Studied Black Gold and Its Current Applications" Journal of Xenobiotics 14, no. 1: 416-451. https://doi.org/10.3390/jox14010026









