Hyaluronic Acid 35 kDa Protects against a Hyperosmotic, Formula Feeding Model of Necrotizing Enterocolitis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Formula Preparation
2.2. Mouse Model
2.3. Histology and Injury Scoring
2.4. Cytokine Analysis
2.5. Microbiome Analysis via 16S Ribosomal RNA (rRNA) Sequencing
2.6. Ileal RNA-Seq Profiling and Enrichment Analysis
2.7. Statistical Analysis
3. Results
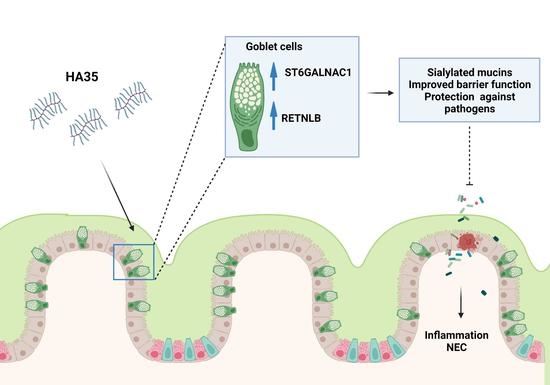
3.1. HA35 Protects against NEC Induced by Formula Feeding and Dithizone
3.2. Effects of Oral HA35 on Intestinal Microbiome Taxonomy and Functional Prediction
3.3. Effect of Oral HA35 on Ileal RNA-Seq Transcriptional Profiles
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Neu, J.; Walker, W.A. Necrotizing enterocolitis. N. Engl. J. Med. 2011, 364, 255–264. [Google Scholar] [CrossRef] [Green Version]
- Fredriksson, F.; Engstrand Lilja, H. Survival rates for surgically treated necrotising enterocolitis have improved over the last four decades. Acta Paediatr. 2019, 108, 1603–1608. [Google Scholar] [CrossRef] [PubMed]
- Neu, J.; Pammi, M. Necrotizing enterocolitis: The intestinal microbiome, metabolome and inflammatory mediators. Semin. Fetal Neonatal Med. 2018, 23, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.N.; Sangild, P.T. Pathogenesis and biomarkers for necrotizing enterocolitis: Getting any closer? EBioMedicine 2019, 45, 13–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bazacliu, C.; Neu, J. Pathophysiology of Necrotizing Enterocolitis: An Update. Curr. Pediatric Rev. 2019, 15, 68–87. [Google Scholar] [CrossRef]
- Sullivan, S.; Schanler, R.J.; Kim, J.H.; Patel, A.L.; Trawöger, R.; Kiechl-Kohlendorfer, U.; Chan, G.M.; Blanco, C.L.; Abrams, S.; Cotten, C.M.; et al. An exclusively human milk-based diet is associated with a lower rate of necrotizing enterocolitis than a diet of human milk and bovine milk-based products. J. Pediatrics 2010, 156, 562–567.e561. [Google Scholar] [CrossRef] [Green Version]
- Maffei, D.; Schanler, R.J. Human milk is the feeding strategy to prevent necrotizing enterocolitis! Semin. Perinatol. 2017, 41, 36–40. [Google Scholar] [CrossRef] [Green Version]
- Denning, T.L.; Bhatia, A.M.; Kane, A.F.; Patel, R.M.; Denning, P.W. Pathogenesis of NEC: Role of the innate and adaptive immune response. Semin. Perinatol. 2017, 41, 15–28. [Google Scholar] [CrossRef] [Green Version]
- Hill, D.R.; Rho, H.K.; Kessler, S.P.; Amin, R.; Homer, C.R.; McDonald, C.; Cowman, M.K.; de la Motte, C.A. Human milk hyaluronan enhances innate defense of the intestinal epithelium. J. Biol. Chem. 2013, 288, 29090–29104. [Google Scholar] [CrossRef] [Green Version]
- Hill, D.R.; Kessler, S.P.; Rho, H.K.; Cowman, M.K.; de la Motte, C.A. Specific-sized hyaluronan fragments promote expression of human beta-defensin 2 in intestinal epithelium. J. Biol. Chem. 2012, 287, 30610–30624. [Google Scholar] [CrossRef] [Green Version]
- Cyphert, J.M.; Trempus, C.S.; Garantziotis, S. Size Matters: Molecular Weight Specificity of Hyaluronan Effects in Cell Biology. Int. J. Cell Biol. 2015, 2015, 563818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kessler, S.P.; Obery, D.R.; Nickerson, K.P.; Petrey, A.C.; McDonald, C.; de la Motte, C.A. Multifunctional Role of 35 Kilodalton Hyaluronan in Promoting Defense of the Intestinal Epithelium. J. Histochem. Cytochem. Off. J. Histochem. Soc. 2018, 66, 273–287. [Google Scholar] [CrossRef] [PubMed]
- Chaaban, H.; Burge, K.; Eckert, J.; Trammell, M.; Dyer, D.; Keshari, R.S.; Silasi, R.; Regmi, G.; Lupu, C.; Good, M.; et al. Acceleration of Small Intestine Development and Remodeling of the Microbiome Following Hyaluronan 35 kDa Treatment in Neonatal Mice. Nutrients 2021, 13, 2030. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Sherman, M.P.; Prince, L.S.; Bader, D.; Weitkamp, J.H.; Slaughter, J.C.; McElroy, S.J. Paneth cell ablation in the presence of Klebsiella pneumoniae induces necrotizing enterocolitis (NEC)-like injury in the small intestine of immature mice. Dis. Models Mech. 2012, 5, 522–532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunasekaran, A.; Eckert, J.; Burge, K.; Zheng, W.; Yu, Z.; Kessler, S.; de la Motte, C.; Chaaban, H. Hyaluronan 35 kDa enhances epithelial barrier function and protects against the development of murine necrotizing enterocolitis. Pediatric Res. 2019, 87, 1177–1184. [Google Scholar] [CrossRef]
- Lucas, A.; Cole, T.J. Breast milk and neonatal necrotising enterocolitis. Lancet 1990, 336, 1519–1523. [Google Scholar] [CrossRef]
- Elgin, T.G.; Kern, S.L.; McElroy, S.J. Development of the Neonatal Intestinal Microbiome and Its Association with Necrotizing Enterocolitis. Clin. Ther. 2016, 38, 706–715. [Google Scholar] [CrossRef] [Green Version]
- Lueschow, S.R.; Kern, S.L.; Gong, H.; Grobe, J.L.; Segar, J.L.; Carlson, S.J.; McElroy, S.J. Feeding Formula Eliminates the Necessity of Bacterial Dysbiosis and Induces Inflammation and Injury in the Paneth Cell Disruption Murine NEC Model in an Osmolality-Dependent Manner. Nutrients 2020, 12, 900. [Google Scholar] [CrossRef] [Green Version]
- Book, L.S.; Herbst, J.J.; Atherton, S.O.; Jung, A.L. Necrotizing enterocolitis in low-birth-weight infants fed an elemental formula. J. Pediatrics 1975, 87, 602–605. [Google Scholar] [CrossRef]
- Sántulli, T.V.; Schullinger, J.N.; Heird, W.C.; Gongaware, R.D.; Wigger, J.; Barlow, B.; Blanc, W.A.; Berdon, W.E. Acute necrotizing enterocolitis in infancy: A review of 64 cases. Pediatrics 1975, 55, 376–387. [Google Scholar] [CrossRef]
- Pearson, F.; Johnson, M.J.; Leaf, A.A. Milk osmolality: Does it matter? Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F166–F169. [Google Scholar] [CrossRef] [PubMed]
- Schanler, R.J.; Garza, C.; Nichols, B.L. Fortified mothers’ milk for very low birth weight infants: Results of growth and nutrient balance studies. J. Pediatrics 1985, 107, 437–445. [Google Scholar] [CrossRef]
- Ananthan, A.; Balasubramanian, H.; Rao, S.; Patole, S. Human Milk-Derived Fortifiers Compared with Bovine Milk-Derived Fortifiers in Preterm Infants: A Systematic Review and Meta-Analysis. Adv. Nutr. 2020, 11, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Koike, Y.; Chi, L.; Ahmed, A.; Miyake, H.; Li, B.; Lee, C.; Delgado-Olguín, P.; Pierro, A. Formula feeding and immature gut microcirculation promote intestinal hypoxia, leading to necrotizing enterocolitis. Dis. Models Mech. 2019, 12, dmm.040998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Chang, K.T.; Lian, D.W.; Lu, H.; Roy, S.; Laksmi, N.K.; Low, Y.; Krishnaswamy, G.; Pierro, A.; Ong, C.C. The role of ischemia in necrotizing enterocolitis. J. Pediatric Surg. 2016, 51, 1255–1261. [Google Scholar] [CrossRef]
- Pound, P.; Ritskes-Hoitinga, M. Is it possible to overcome issues of external validity in preclinical animal research? Why most animal models are bound to fail. J. Transl. Med. 2018, 16, 304. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Kessler, S.P.; Obery, D.R.; Homer, C.R.; McDonald, C.; de la Motte, C.A. Hyaluronan 35 kDa treatment protects mice from Citrobacter rodentium infection and induces epithelial tight junction protein ZO-1 in vivo. Matrix Biol. 2017, 62, 28–39. [Google Scholar] [CrossRef]
- Dvorak, B.; McWilliam, D.L.; Williams, C.S.; Dominguez, J.A.; Machen, N.W.; McCuskey, R.S.; Philipps, A.F. Artificial formula induces precocious maturation of the small intestine of artificially reared suckling rats. J. Pediatric Gastroenterol. Nutr. 2000, 31, 162–169. [Google Scholar] [CrossRef]
- Council, N.R. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; p. 246. [Google Scholar]
- McElroy, S.J.; Weitkamp, J.H. Innate Immunity in the Small Intestine of the Preterm Infant. Neoreviews 2011, 12, e517–e526. [Google Scholar] [CrossRef] [Green Version]
- Stanford, A.H.; Gong, H.; Noonan, M.; Lewis, A.N.; Gong, Q.; Lanik, W.E.; Hsieh, J.J.; Lueschow, S.R.; Frey, M.R.; Good, M.; et al. A direct comparison of mouse and human intestinal development using epithelial gene expression patterns. Pediatric Res. 2020, 88, 66–76. [Google Scholar] [CrossRef]
- Lueschow, S.R.; Stumphy, J.; Gong, H.; Kern, S.L.; Elgin, T.G.; Underwood, M.A.; Kalanetra, K.M.; Mills, D.A.; Wong, M.H.; Meyerholz, D.K.; et al. Loss of murine Paneth cell function alters the immature intestinal microbiome and mimics changes seen in neonatal necrotizing enterocolitis. PLoS ONE 2018, 13, e0204967. [Google Scholar] [CrossRef]
- Wynn, J.L.; Wilson, C.S.; Hawiger, J.; Scumpia, P.O.; Marshall, A.F.; Liu, J.H.; Zharkikh, I.; Wong, H.R.; Lahni, P.; Benjamin, J.T.; et al. Targeting IL-17A attenuates neonatal sepsis mortality induced by IL-18. Proc. Natl. Acad. Sci. USA 2016, 113, E2627–E2635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fricke, E.M.; Elgin, T.G.; Gong, H.; Reese, J.; Gibson-Corley, K.N.; Weiss, R.M.; Zimmerman, K.; Bowdler, N.C.; Kalantera, K.M.; Mills, D.A.; et al. Lipopolysaccharide-induced maternal inflammation induces direct placental injury without alteration in placental blood flow and induces a secondary fetal intestinal injury that persists into adulthood. Am. J. Reprod. Immunol. 2018, 79, e12816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, M.; Beiko, R.G. 16S rRNA Gene Analysis with QIIME2. Methods Mol. Biol. 2018, 1849, 113–129. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Baird, N.L.; Bowlin, J.L.; Cohrs, R.J.; Gilden, D.; Jones, K.L. Comparison of varicella-zoster virus RNA sequences in human neurons and fibroblasts. J. Virol. 2014, 88, 5877–5880. [Google Scholar] [CrossRef] [Green Version]
- Wu, T.D.; Nacu, S. Fast and SNP-tolerant detection of complex variants and splicing in short reads. Bioinformatics 2010, 26, 873–881. [Google Scholar] [CrossRef] [Green Version]
- Trapnell, C.; Williams, B.A.; Pertea, G.; Mortazavi, A.; Kwan, G.; van Baren, M.J.; Salzberg, S.L.; Wold, B.J.; Pachter, L. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 2010, 28, 511–515. [Google Scholar] [CrossRef] [Green Version]
- Maheshwari, A.; Schelonka, R.L.; Dimmitt, R.A.; Carlo, W.A.; Munoz-Hernandez, B.; Das, A.; McDonald, S.A.; Thorsen, P.; Skogstrand, K.; Hougaard, D.M.; et al. Cytokines associated with necrotizing enterocolitis in extremely-low-birth-weight infants. Pediatric Res. 2014, 76, 100–108. [Google Scholar] [CrossRef]
- Papathanassiu, A.E.; Ko, J.H.; Imprialou, M.; Bagnati, M.; Srivastava, P.K.; Vu, H.A.; Cucchi, D.; McAdoo, S.P.; Ananieva, E.A.; Mauro, C.; et al. BCAT1 controls metabolic reprogramming in activated human macrophages and is associated with inflammatory diseases. Nat. Commun. 2017, 8, 16040. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Zhang, J.; Shi, Y.; Li, W.; Shi, H.; Ji, R.; Mao, F.; Qian, H.; Xu, W.; Zhang, X. CXCL5 promotes gastric cancer metastasis by inducing epithelial-mesenchymal transition and activating neutrophils. Oncogenesis 2020, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Eyking, A.; Ferber, F.; Köhler, S.; Reis, H.; Cario, E. TRIM58 Restrains Intestinal Mucosal Inflammation by Negatively Regulating TLR2 in Myeloid Cells. J. Immunol. 2019, 203, 1636–1649. [Google Scholar] [CrossRef] [PubMed]
- Morampudi, V.; Dalwadi, U.; Bhinder, G.; Sham, H.P.; Gill, S.K.; Chan, J.; Bergstrom, K.S.; Huang, T.; Ma, C.; Jacobson, K.; et al. The goblet cell-derived mediator RELM-β drives spontaneous colitis in Muc2-deficient mice by promoting commensal microbial dysbiosis. Mucosal Immunol. 2016, 9, 1218–1233. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.E.; Snook, A.E.; Li, P.; Stoecker, B.A.; Kim, G.W.; Magee, M.S.; Garcia, A.V.; Valentino, M.A.; Hyslop, T.; Schulz, S.; et al. GUCY2C opposes systemic genotoxic tumorigenesis by regulating AKT-dependent intestinal barrier integrity. PLoS ONE 2012, 7, e31686. [Google Scholar] [CrossRef] [Green Version]
- Song, D.; Cheng, Y.; Li, X.; Wang, F.; Lu, Z.; Xiao, X.; Wang, Y. Biogenic Nanoselenium Particles Effectively Attenuate Oxidative Stress-Induced Intestinal Epithelial Barrier Injury by Activating the Nrf2 Antioxidant Pathway. ACS Appl. Mater. Interfaces 2017, 9, 14724–14740. [Google Scholar] [CrossRef]
- Good, M.; Sodhi, C.P.; Yamaguchi, Y.; Jia, H.; Lu, P.; Fulton, W.B.; Martin, L.Y.; Prindle, T.; Nino, D.F.; Zhou, Q.; et al. The human milk oligosaccharide 2’-fucosyllactose attenuates the severity of experimental necrotising enterocolitis by enhancing mesenteric perfusion in the neonatal intestine. Br. J. Nutr. 2016, 116, 1175–1187. [Google Scholar] [CrossRef] [Green Version]
- Andersson, E.L.; Hernell, O.; Bläckberg, L.; Fält, H.; Lindquist, S. BSSL and PLRP2: Key enzymes for lipid digestion in the newborn examined using the Caco-2 cell line. J. Lipid Res. 2011, 52, 1949–1956. [Google Scholar] [CrossRef] [Green Version]
- Commentary on breast-feeding and infant formulas, including proposed standards for formulas. Pediatrics 1976, 57, 278–285. [CrossRef]
- Chandran, S.; Chua, M.C.; Lin, W.; Min Wong, J.; Saffari, S.E.; Rajadurai, V.S. Medications That Increase Osmolality and Compromise the Safety of Enteral Feeding in Preterm Infants. Neonatology 2017, 111, 309–316. [Google Scholar] [CrossRef]
- Buchanan, D.E.; Holzman, I.R. Hyperosmolal feedings and gastrointestinal blood flow and oxygenation in newborn lambs. J. Pediatric Gastroenterol. Nutr. 1987, 6, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Goldblum, O.M.; Holzman, I.R.; Fisher, S.E. Intragastric feeding in the neonatal dog. Its effect on intestinal osmolality. Am. J. Dis. Child. 1981, 135, 631–633. [Google Scholar] [CrossRef]
- Katz, L.; Hamilton, J.R. Response of the young pig to intrajejunal perfusion with a hypertonic nutrient solution. J. Pediatrics 1975, 87, 606–608. [Google Scholar] [CrossRef]
- Norris, H.T. Response of the small intestine to the application of a hypertonic solution. Am. J. Pathol. 1973, 73, 747–764. [Google Scholar] [PubMed]
- Miyake, H.; Chen, Y.; Koike, Y.; Hock, A.; Li, B.; Lee, C.; Zani, A.; Pierro, A. Osmolality of enteral formula and severity of experimental necrotizing enterocolitis. Pediatric Surg. Int. 2016, 32, 1153–1156. [Google Scholar] [CrossRef]
- Ossa, J.C.; Yáñez, D.; Valenzuela, R.; Gallardo, P.; Lucero, Y.; Farfán, M.J. Intestinal Inflammation in Chilean Infants Fed with Bovine Formula vs. Breast Milk and Its Association with Their Gut Microbiota. Front. Cell. Infect. Microbiol. 2018, 8, 190. [Google Scholar] [CrossRef]
- Burg, M.B.; Kwon, E.D.; Kültz, D. Regulation of gene expression by hypertonicity. Annu. Rev. Physiol. 1997, 59, 437–455. [Google Scholar] [CrossRef]
- Ahmed, H.M.; Kamel, N.M. The relation between serum levels of epidermal growth factor and necrotizing enterocolitis in preterm neonates. Korean J. Pediatric 2019, 62, 307–311. [Google Scholar] [CrossRef]
- Halpern, M.D.; Dominguez, J.A.; Dvorakova, K.; Holubec, H.; Williams, C.S.; Meza, Y.G.; Ruth, M.C.; Dvorak, B. Ileal cytokine dysregulation in experimental necrotizing enterocolitis is reduced by epidermal growth factor. J. Pediatric Gastroenterol. Nutr. 2003, 36, 126–133. [Google Scholar] [CrossRef]
- Torres-Castro, P.; Abril-Gil, M.; Rodríguez-Lagunas, M.J.; Castell, M.; Pérez-Cano, F.J.; Franch, À. TGF-β2, EGF, and FGF21 Growth Factors Present in Breast Milk Promote Mesenteric Lymph Node Lymphocytes Maturation in Suckling Rats. Nutrients 2018, 10, 1171. [Google Scholar] [CrossRef] [Green Version]
- Jain, S.K.; Baggerman, E.W.; Mohankumar, K.; Namachivayam, K.; Jagadeeswaran, R.; Reyes, V.E.; Maheshwari, A. Amniotic fluid-borne hepatocyte growth factor protects rat pups against experimental necrotizing enterocolitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, G361–G369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Strohmeier, V.; He, Z.; Deshpande, M.; Catalan-Dibene, J.; Durum, S.K.; Moran, T.M.; Kraus, T.; Xiong, H.; Faith, J.J.; et al. Interleukin 22 disrupts pancreatic function in newborn mice expressing IL-23. Nat. Commun. 2019, 10, 4517. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.X.; Rudloff, I.; Lao, J.C.; Pang, M.A.; Goldberg, R.; Bui, C.B.; McLean, C.A.; Stock, M.; Klassert, T.E.; Slevogt, H.; et al. Characterization of the pathoimmunology of necrotizing enterocolitis reveals novel therapeutic opportunities. Nat. Commun. 2020, 11, 5794. [Google Scholar] [CrossRef] [PubMed]
- Heida, F.H.; Beyduz, G.; Bulthuis, M.L.; Kooi, E.M.; Bos, A.F.; Timmer, A.; Hulscher, J.B. Paneth cells in the developing gut: When do they arise and when are they immune competent? Pediatric Res. 2016, 80, 306–310. [Google Scholar] [CrossRef] [Green Version]
- McElroy, S.J.; Prince, L.S.; Weitkamp, J.H.; Reese, J.; Slaughter, J.C.; Polk, D.B. Tumor necrosis factor receptor 1-dependent depletion of mucus in immature small intestine: A potential role in neonatal necrotizing enterocolitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G656–G666. [Google Scholar] [CrossRef] [Green Version]
- Markasz, L.; Wanders, A.; Szekely, L.; Lilja, H.E. Diminished DEFA6 Expression in Paneth Cells Is Associated with Necrotizing Enterocolitis. Gastroenterol. Res. Pract. 2018, 2018, 7345426. [Google Scholar] [CrossRef]
- Hodzic, Z.; Bolock, A.M.; Good, M. The Role of Mucosal Immunity in the Pathogenesis of Necrotizing Enterocolitis. Front. Pediatrics 2017, 5, 40. [Google Scholar] [CrossRef] [Green Version]
- Sherman, M.P.; Bennett, S.H.; Hwang, F.F.; Sherman, J.; Bevins, C.L. Paneth cells and antibacterial host defense in neonatal small intestine. Infect. Immun. 2005, 73, 6143–6146. [Google Scholar] [CrossRef] [Green Version]
- Maares, M.; Keil, C.; Straubing, S.; Robbe-Masselot, C.; Haase, H. Zinc Deficiency Disturbs Mucin Expression, O-Glycosylation and Secretion by Intestinal Goblet Cells. Int. J. Mol. Sci. 2020, 21, 6149. [Google Scholar] [CrossRef]
- Sicard, J.F.; Le Bihan, G.; Vogeleer, P.; Jacques, M.; Harel, J. Interactions of Intestinal Bacteria with Components of the Intestinal Mucus. Front. Cell. Infect. Microbiol. 2017, 7, 387. [Google Scholar] [CrossRef]
- Schwartz, J.L. Fcgbp—A Potential Viral Trap in RV144. Open AIDS J. 2014, 8, 21–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Post, S.; Jabbar, K.S.; Birchenough, G.; Arike, L.; Akhtar, N.; Sjovall, H.; Johansson, M.E.V.; Hansson, G.C. Structural weakening of the colonic mucus barrier is an early event in ulcerative colitis pathogenesis. Gut 2019, 68, 2142–2151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, W.; Shi, J.; Zhou, Y.; Liu, T.; Zhan, F.; Zhang, K.; Liu, N. Integrating proteomics and transcriptomics for the identification of potential targets in early colorectal cancer. Int. J. Oncol. 2019, 55, 439–450. [Google Scholar] [CrossRef] [Green Version]
- Munkley, J. The Role of Sialyl-Tn in Cancer. Int. J. Mol. Sci. 2016, 17, 275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.Y.; Cao, Y.X.; Zhou, X.; Wei, B.; Zhan, L.; Sun, S.Y. Stimulative role of ST6GALNAC1 in proliferation, migration and invasion of ovarian cancer stem cells via the Akt signaling pathway. Cancer Cell Int. 2019, 19, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, H.; Ohi, H.; Terao, T. Analysis of monoclonal antibodies reactive with meconium- and amniotic fluid-derived mucin. J. Clin. Lab. Anal. 1994, 8, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, E.; Macauley, M.S. Hypersialylation in Cancer: Modulation of Inflammation and Therapeutic Opportunities. Cancers 2018, 10, 207. [Google Scholar] [CrossRef] [Green Version]
- Propheter, D.C.; Chara, A.L.; Harris, T.A.; Ruhn, K.A.; Hooper, L.V. Resistin-like molecule β is a bactericidal protein that promotes spatial segregation of the microbiota and the colonic epithelium. Proc. Natl. Acad. Sci. USA 2017, 114, 11027–11033. [Google Scholar] [CrossRef] [Green Version]
- Bergstrom, K.S.; Morampudi, V.; Chan, J.M.; Bhinder, G.; Lau, J.; Yang, H.; Ma, C.; Huang, T.; Ryz, N.; Sham, H.P.; et al. Goblet Cell Derived RELM-β Recruits CD4+ T Cells during Infectious Colitis to Promote Protective Intestinal Epithelial Cell Proliferation. PLoS Pathog. 2015, 11, e1005108. [Google Scholar] [CrossRef]
- Riehl, T.E.; Ee, X.; Stenson, W.F. Hyaluronic acid regulates normal intestinal and colonic growth in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G377–G388. [Google Scholar] [CrossRef] [Green Version]
- Gorkiewicz, G.; Thallinger, G.G.; Trajanoski, S.; Lackner, S.; Stocker, G.; Hinterleitner, T.; Gülly, C.; Högenauer, C. Alterations in the colonic microbiota in response to osmotic diarrhea. PLoS ONE 2013, 8, e55817. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Formula Component | Amount (g/kg Body Weight) |
|---|---|
| Evaporated milk | 615 |
| Bovine serum albumin | 36 |
| Intralipid 20% intravenous fat emulsion | 292 |
| Sunflower oil | 10 |
| Teklad vitamin mix | 4 |
| Supplemental vitamin mixture 1 | 0.56 |
| Salt mixture solution 2 | 10 |
| Non-calcium mineral mixture 3 | 6 |
| Calcium lactate solution (40 g/L) | 10 |
| CaCO3 solution (40 g/L) | 10 |
| CuSO4 (30 g/L) | 0.57 |
| ZnSO4 (380 g/L) | 0.07 |
| Double-distilled water (ddH2O) | 6 |
| Treatment Group | Feeding Duration/Type | P14 Dithizone (Y/N) |
|---|---|---|
| Sham | P7–P13: Water + Dam P14: Saline | N |
| HA | P7–P13: HA + Dam P14: Saline | N |
| Formula | P7–P13: Water + Dam P14: RMS | N |
| Formula + HA | P7–P13: HA + Dam P14: RMS | N |
| NEC | P7–P13: Water + Dam P14: RMS | Y |
| NEC + HA | P7–P13: HA + Dam P14: RMS | Y |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burge, K.; Eckert, J.; Wilson, A.; Trammell, M.; Lueschow, S.R.; McElroy, S.J.; Dyer, D.; Chaaban, H. Hyaluronic Acid 35 kDa Protects against a Hyperosmotic, Formula Feeding Model of Necrotizing Enterocolitis. Nutrients 2022, 14, 1779. https://doi.org/10.3390/nu14091779
Burge K, Eckert J, Wilson A, Trammell M, Lueschow SR, McElroy SJ, Dyer D, Chaaban H. Hyaluronic Acid 35 kDa Protects against a Hyperosmotic, Formula Feeding Model of Necrotizing Enterocolitis. Nutrients. 2022; 14(9):1779. https://doi.org/10.3390/nu14091779
Chicago/Turabian StyleBurge, Kathryn, Jeffrey Eckert, Adam Wilson, MaJoi Trammell, Shiloh R. Lueschow, Steven J. McElroy, David Dyer, and Hala Chaaban. 2022. "Hyaluronic Acid 35 kDa Protects against a Hyperosmotic, Formula Feeding Model of Necrotizing Enterocolitis" Nutrients 14, no. 9: 1779. https://doi.org/10.3390/nu14091779