Effects of Natural Products on Fructose-Induced Nonalcoholic Fatty Liver Disease (NAFLD)
Abstract
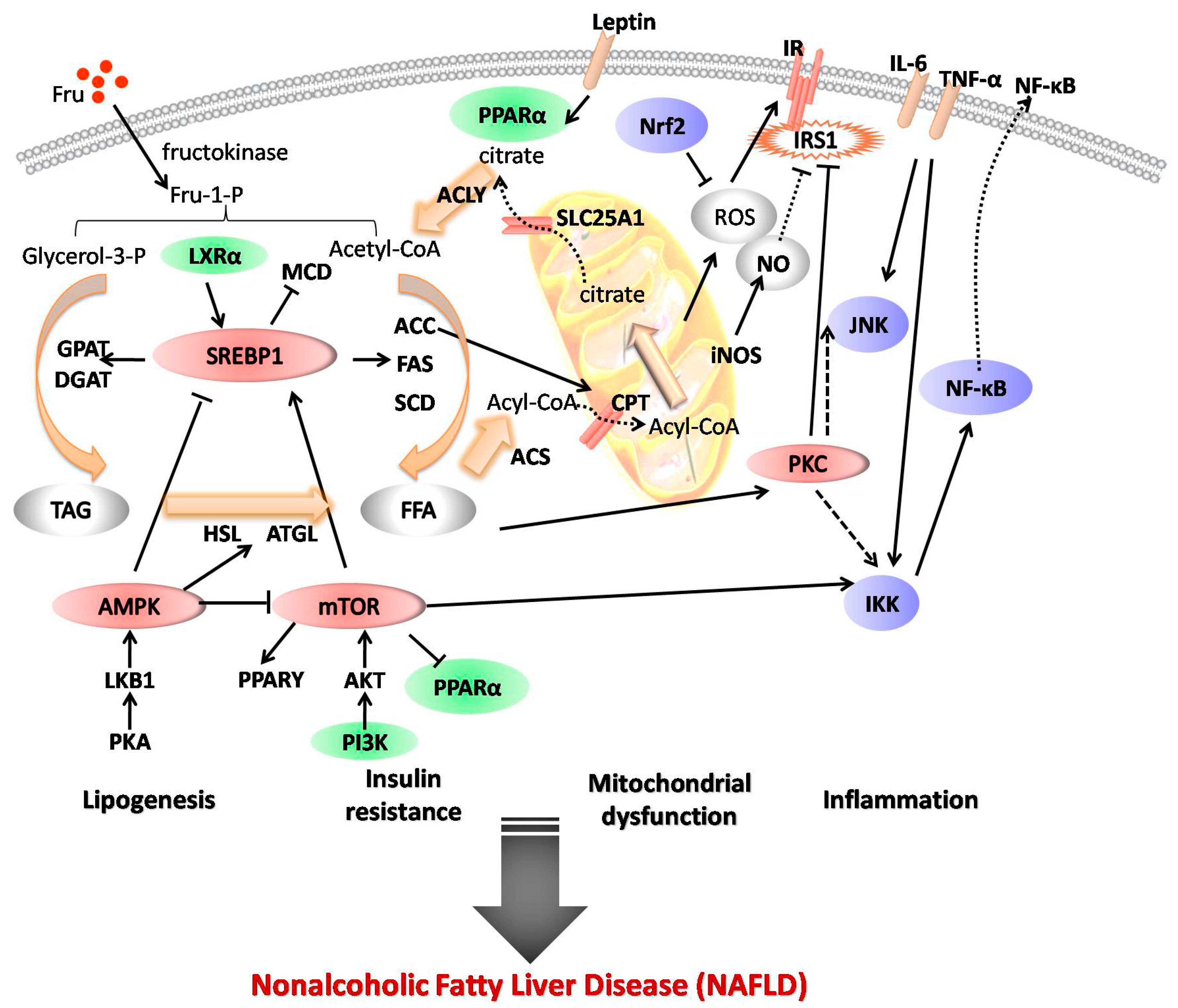
:1. Introduction
2. Mechanisms of Fructose Action in NAFLD
3. Effects of Natural Products on Fructose-Induced NAFLD
3.1. Regulation of Lipogenesis
3.2. Repair of Mitochondrial Dysfunction
3.3. Inhibition of Inflammatory Pathways
3.4. Improvement of Insulin Resistance
4. Conclusions
5. Prospect
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACC | Acetyl-CoA carboxylase |
| ACLY | ATP-citrate lyase |
| ACS | Acyl-CoA synthase |
| AMPK | Adenosine 5′-monophosphate (AMP)-activated protein kinase |
| ATGL | Adipose triglyceride lipase |
| CPT-1 | Carnitine palmitoyl transferase 1 |
| DGAT | Diacylglycerol acyltransferase |
| eNOS | Endothelial nitric oxide synthase |
| FAS | Fatty acid synthase |
| FFA | Free fatty acid |
| Fru | Fructose |
| Fru-1-P | Fructose-1-phophate |
| Glut5 | Glucose transporter 5 |
| GPAT | Glycerol-3-phosphate acyltransferase |
| HNF1α | Hepatocyte nuclear factor 1α |
| HSL | Hormone-sensitive lipase |
| IKK | Inhibitor of nuclear factor κB (IκB) kinase |
| IL-6 | Interleukin-6 |
| iNOS | Inducible nitric oxide synthase |
| IR | Insulin receptor |
| IRS-1 | Insulin receptor substrate-1 |
| JNK | c-Jun amino terminal kinase |
| LKB1 | Liver kinase B1 |
| LXRα | Liver X receptor α |
| MAPK | Mitogen-activated protein kinase |
| MCD | Malonyl-CoA decarboxylase |
| mTOR | Mammalian target of rapamycin |
| NAFLD | Nonalcoholic fatty liver disease |
| NF-κB | Nuclear factor kappa B |
| Nrf2 | Nuclear factor-erythroid 2-related factor 2 |
| PI3K | Phosphoinositide 3-kinase |
| PKA | Protein kinase A |
| PKC | Protein kinase C |
| PPARα | Peroxisome proliferator–activated receptor α |
| PPARγ | Peroxisome proliferator activated receptor γ |
| ROS | Reactive oxygen species |
| SCD | Stearoyl-CoA desaturase |
| SREBP-1c | Sterol regulatory element binding protein 1c |
| TAG | Triglyceride |
| TNF-α | Tumor necrosis factor α |
| VLDL | Very low density lipoprotein |
References
- Lim, J.S.; Mietus-Snyder, M.; Valente, A.; Schwarz, J.M.; Lustig, R.H. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Vos, M.B.; Lavine, J.E. Dietary fructose in nonalcoholic fatty liver disease. Hepatology 2013, 57, 2525–2531. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Popkin, B.M. Dietary sugar and body weight: Have we reached a crisis in the epidemic of obesity and diabetes?: Health be damned! Pour on the sugar. Diabetes Care 2014, 37, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, X.; Cirillo, P.; Sautin, Y.; McCall, S.; Bruchette, J.L.; Diehl, A.M.; Johnson, R.J.; Abdelmalek, M.F. Fructose consumption as a risk factor for non-alcoholic fatty liver disease. J. Hepatol. 2008, 48, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.A.; St-Onge, M.P.; Siebert, E.A.; Medici, V.; Stanhope, K.L.; Havel, P.J. Differential responses of plasma adropin concentrations to dietary glucose or fructose consumption in humans. Sci. Rep. 2015, 5, 14691. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulou, A.; Weiskirchen, S.; Weiskirchen, R. Lipocalin 2 (LCN2) expression in hepatic malfunction and therapy. Front. Physiol. 2016, 7, 430. [Google Scholar] [CrossRef] [PubMed]
- Alwahsh, S.M.; Xu, M.; Seyhan, H.A.; Ahmad, S.; Mihm, S.; Ramadori, G.; Schultze, F.C. Diet high in fructose leads to an overexpression of lipocalin-2 in rat fatty liver. World J. Gastroenterol. 2014, 20, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.F.; Suzuki, A.; Guy, C.; Unalp-Arida, A.; Colvin, R.; Johnson, R.J.; Diehl, A.M. Increased fructose consumption is associated with fibrosis severity in patients with nonalcoholic fatty liver disease. Hepatology 2010, 51, 1961–1971. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Segal, M.S.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.H.; Gersch, M.S.; Benner, S.; Sánchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar] [PubMed]
- Mayes, P.A. Intermediary metabolism of fructose. Am. J. Clin. Nutr. 1993, 58, 754S–765S. [Google Scholar] [PubMed]
- DeBosch, B.J.; Chen, Z.; Saben, J.L.; Finck, B.N.; Moley, K.H. Glucose transporter 8 (glut8) mediates fructose-induced de novo lipogenesis and macrosteatosis. J. Biol. Chem. 2014, 289, 10989–10998. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.J.; Perez-Pozo, S.E.; Sautin, Y.Y.; Manitius, J.; Sanchez-Lozada, L.G.; Feig, D.I.; Shafiu, M.; Segal, M.; Glassock, R.J.; Shimada, M.; et al. Hypothesis: Could excessive fructose intake and uric acid cause type 2 diabetes? Endocr. Rev. 2009, 30, 96–116. [Google Scholar] [CrossRef] [PubMed]
- Basciano, H.; Federico, L.; Adeli, K. Fructose, insulin resistance, and metabolic dyslipidemia. Nutr. Metab. 2005, 2, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, S.Z.; Empie, M.W. Fructose metabolism in humans-what isotopic tracer studies tell us. Nutr. Metab. 2012, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.G.; Farrell, G.C. Epidemiology of non-alcoholic fatty liver disease in China. J. Hepatol. 2009, 50, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Tziomalos, K.; Athyros, V.G.; Paschos, P.; Karagiannis, A. Nonalcoholic fatty liver disease and statins. Meta. Clin. Exper. 2015, 64, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Calzadilla Bertot, L.; Adams, L.A. The natural course of non-alcoholic fatty liver disease. Int. J. Mol. Sci. 2016, 17, E774. [Google Scholar] [CrossRef] [PubMed]
- De la Iglesia, R.; Loria-Kohen, V.; Zulet, M.A.; Martinez, J.A.; Reglero, G.; Ramirez de Molina, A. Dietary strategies implicated in the prevention and treatment of metabolic syndrome. Int. J. Mol. Sci. 2016, 17, E1877. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.H.; Lee, B.H.; Pan, T.M. Leptin-induced mitochondrial fusion mediates hepatic lipid accumulation. Int. J. Obes. 2015, 39, 1750–1756. [Google Scholar] [CrossRef] [PubMed]
- Alwahsh, S.M.; Xu, M.; Schultze, F.C.; Wilting, J.; Mihm, S.; Raddatz, D.; Ramadori, G. Combination of alcohol and fructose exacerbates metabolic imbalance in terms of hepatic damage, dyslipidemia, and insulin resistance in rats. PLoS ONE 2014, 9, e104220. [Google Scholar] [CrossRef] [PubMed]
- Rutledge, A.C.; Adeli, K. Fructose and the metabolic syndrome: Pathophysiology and molecular mechanisms. Nutr. Rev. 2007, 65, 13–23. [Google Scholar] [CrossRef]
- Dekker, M.J.; Su, Q.; Baker, C.; Rutledge, A.C.; Adeli, K. Fructose: A highly lipogenic nutrient implicated in insulin resistance, hepatic steatosis, and the metabolic syndrome. Am. J. Physiol. Endocr. Metab. 2010, 299, E685–E694. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Guo, J.; Su, Z. Advances in understanding the interrelations between leptin resistance and obesity. Physiol. Behav. 2014, 130, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Alwahsh, S.M.; Gebhardt, R. Dietary fructose as a risk factor for non-alcoholic fatty liver disease (NAFLD). Arch. Toxicol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Antu, K.A.; Riya, M.P.; Nair, A.; Mishra, A.; Srivastava, A.K.; Raghu, K.G. Symplocos cochinchinensis enhances insulin sensitivity via the down regulation of lipogenesis and insulin resistance in high energy diet rat model. J. Ethnopharmacol. 2016, 193, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, M.; Lin, X.; Li, Y.; Liu, C.; Yang, Y.; Yamahara, J.; Wang, J.; Li, Y. Modulation of hepatic sterol regulatory element-binding protein-1c-mediated gene expression contributes to Salacia oblonga root-elicited improvement of fructose-induced fatty liver in rats. J. Ethnopharmacol. 2013, 150, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Ehlers, S.J.; Lee, J.Y.; Fernandez, M.L.; Koo, S.I. Dietary green tea extract lowers plasma and hepatic triglycerides and decreases the expression of sterol regulatory element-binding protein-1c mRNA and its responsive genes in fructose-fed, ovariectomized rats. J. Nutr. 2009, 139, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Prakash, P.; Singh, V.; Jain, M.; Rana, M.; Khanna, V.; Barthwal, M.K.; Dikshit, M. Silymarin ameliorates fructose induced insulin resistance syndrome by reducing de novo hepatic lipogenesis in the rat. Eur. J. Pharmacol. 2014, 727, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Maithilikarpagaselvi, N.; Sridhar, M.G.; Swaminathan, R.P.; Sripradha, R.; Badhe, B. Curcumin inhibits hyperlipidemia and hepatic fat accumulation in high-fructose-fed male Wistar rats. Pharm. Biol. 2016, 54, 2857–2863. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, Y.; Zuo, G.; Xu, W.; Gao, H.; Yang, Y.; Yamahara, J.; Wang, J.; Li, Y. Oleanolic acid diminishes liquid fructose-induced fatty liver in rats: Role of modulation of hepatic sterol regulatory element-binding protein-1c-mediated expression of genes responsible for de novo fatty acid synthesis. Evid. Based Compl. Alt. 2013, 2013, 534084. [Google Scholar] [CrossRef] [PubMed]
- Xing, X.; Li, D.; Chen, D.; Zhou, L.; Chonan, R.; Yamahara, J.; Wang, J.; Li, Y. Mangiferin treatment inhibits hepatic expression of acyl-coenzyme A: Diacylglycerol acyltransferase-2 in fructose-fed spontaneously hypertensive rats: A link to amelioration of fatty liver. Toxicol. Appl. Pharmacol. 2014, 280, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, Z.; Han, X.; Huang, D.; Lu, Y.; Yang, X. Enhancing the hepatic protective effect of genistein by oral administration with stachyose in mice with chronic high fructose diet consumption. Food Funct. 2016, 7, 2420–2430. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Kishimoto, K.; Miura, S.; Ezaki, O. Dietary β-conglycinin prevents fatty liver induced by a high-fat diet by a decrease in peroxisome proliferator-activated receptor γ2 protein. J. Nutr. Biochem. 2012, 23, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, T.O.; Hussaini, A.A.; Nafiu, B.Y.; Ibitoye, O.B. Aqueous seed extract of Hunteria umbellata (K. Schum.) Hallier f. (Apocynaceae) palliates hyperglycemia, insulin resistance, dyslipidemia, inflammation and oxidative stress in high-fructose diet-induced metabolic syndrome in rats. J. Ethnopharmacol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Li, W.; Huang, D.; Yang, X. Polyphenols from hawthorn peels and fleshes differently mitigate dyslipidemia, inflammation and oxidative stress in association with modulation of liver injury in high fructose diet-fed mice. Chem. Biol. Interact. 2016, 257, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Li, R.W.; Douglas, T.D.; Maiyoh, G.K.; Adeli, K.; Theriault, A.G. Green tea leaf extract improves lipid and glucose homeostasis in a fructose-fed insulin-resistant hamster model. J. Ethnopharmacol. 2006, 104, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.J.; Shi, L.; Song, G.Y.; Zhang, H.F.; Hu, Z.J.; Wang, C.; Zhang, D.H. Oxymatrine attenuates hepatic steatosis in non-alcoholic fatty liver disease rats fed with high fructose diet through inhibition of sterol regulatory element binding transcription factor 1 (Srebf1) and activation of peroxisome proliferator activated receptor α (PPARα). Eur. J. Pharmacol. 2013, 714, 89–95. [Google Scholar] [PubMed]
- Ge, C.X.; Yu, R.; Xu, M.X.; Li, P.Q.; Fan, C.Y.; Li, J.M.; Kong, L.D. Betaine prevented fructose-induced NAFLD by regulating LXRα/PPARα pathway and alleviating er stress in rats. Eur. J. Pharmacol. 2016, 770, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, P.; Reeta, K.H.; Maulik, S.K.; Dinda, A.K.; Gupta, Y.K. Protective effect of thymoquinone against high-fructose diet-induced metabolic syndrome in rats. Eur. J. Nutr. 2015, 54, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Yogalakshmi, B.; Sreeja, S.; Geetha, R.; Radika, M.K.; Anuradha, C.V. Grape seed proanthocyanidin rescues rats from steatosis: A comparative and combination study with metformin. J. Llipids 2013, 2013, 153897. [Google Scholar] [CrossRef] [PubMed]
- Kelany, M.E.; Hakami, T.M.; Omar, A.H. Curcumin improves the metabolic syndrome in high-fructose-diet-fed rats role of TNF-α, NF-κB, and oxidative stress. Can. J. Physiol. Pharmacol. 2016, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- De Castro, G.S.; Cardoso, J.F.; Calder, P.C.; Jordao, A.A.; Vannucchi, H. Fish oil decreases hepatic lipogenic genes in rats fasted and refed on a high fructose diet. Nutrients 2015, 7, 1644–1656. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Lanzi, C.; Perdicaro, D.J.; Antoniolli, A.; Fontana, A.R.; Miatello, R.M.; Bottini, R.; Vazquez Prieto, M.A. Grape pomace and grape pomace extract improve insulin signaling in high-fat-fructose fed rat-induced metabolic syndrome. Food Funct. 2016, 7, 1544–1553. [Google Scholar] [CrossRef] [PubMed]
- Noshahr, Z.S.; Shahraki, M.R.; Ahmadvand, H.; Nourabadi, D.; Nakhaei, A. Protective effects of Withania somnifera root on inflammatory markers and insulin resistance in fructose-fed rats. Rep. Biochem. Mol. Biol. 2015, 3, 62–67. [Google Scholar]
- Yuan, L.; Han, X.; Li, W.; Ren, D.; Yang, X. Isoorientin prevents hyperlipidemia and liver injury by regulating lipid metabolism, antioxidant capability, and inflammatory cytokine release in high-fructose-fed mice. J. Agric. Food Chem. 2016, 64, 2682–2689. [Google Scholar] [CrossRef] [PubMed]
- Maithilikarpagaselvi, N.; Sridhar, M.G.; Swaminathan, R.P.; Zachariah, B. Curcumin prevents inflammatory response, oxidative stress and insulin resistance in high fructose fed male wistar rats: Potential role of serine kinases. Chem. Biol. Interact. 2016, 244, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Bettaieb, A.; Vazquez Prieto, M.A.; Rodriguez Lanzi, C.; Miatello, R.M.; Haj, F.G.; Fraga, C.G.; Oteiza, P.I. (−)-Epicatechin mitigates high-fructose-associated insulin resistance by modulating redox signaling and endoplasmic reticulum stress. Free Radic. Biol. Med. 2014, 72, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.M.; El-Denshary, E.S.; Abdallah, D.M. Geraniol, alone and in combination with pioglitazone, ameliorates fructose-induced metabolic syndrome in rats via the modulation of both inflammatory and oxidative stress status. PLoS ONE 2015, 10, e0117516. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneswari, S.; Yogalakshmi, B.; Sreeja, S.; Anuradha, C.V. Astaxanthin reduces hepatic endoplasmic reticulum stress and nuclear factor-kappab-mediated inflammation in high fructose and high fat diet-fed mice. Cell Stress Chaperon. 2014, 19, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Hu, L.; Zhang, Y.; Wang, Y.I. Hypolipidemic effects of flavonoids extracted from lomatogonium rotatum. Exp. Ther. Med. 2016, 11, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Zhao, Y.; Nie, Y.; Lu, X.; Sun, Y.; Yang, X. Chemical composition of Pleurotus eryngii polysaccharides and their inhibitory effects on high-fructose diet-induced insulin resistance and oxidative stress in mice. Food Funct. 2014, 5, 2609–2620. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Kim, J.E.; Nam, S.H.; Goo, J.S.; Choi, S.I.; Choi, Y.H.; Bae, C.J.; Woo, J.M.; Cho, J.S.; Hwang, D.Y. Differential regulation of the biosynthesis of glucose transporters by the PI3-K and MAPK pathways of insulin signaling by treatment with novel compounds from liriope platyphylla. Int. J. Mol. Med. 2011, 27, 319–327. [Google Scholar] [PubMed]
- Narasimhan, A.; Chinnaiyan, M.; Karundevi, B. Ferulic acid regulates hepatic Glut2 gene expression in high fat and fructose-induced type-2 diabetic adult male rat. Eur. J. Pharmacol. 2015, 761, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Sadi, G.; Ergin, V.; Yilmaz, G.; Pektas, M.B.; Yildirim, O.G.; Menevse, A.; Akar, F. High-fructose corn syrup-induced hepatic dysfunction in rats: Improving effect of resveratrol. Eur. J. Nutr. 2015, 54, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Pektas, M.B.; Koca, H.B.; Sadi, G.; Akar, F. Dietary fructose activates insulin signaling and inflammation in adipose tissue: Modulatory role of resveratrol. Biomed. Res. Int. 2016, 2016, 8014252. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, E.; Karthik, D.; Anuradha, C.V. Genistein sensitizes hepatic insulin signaling and modulates lipid regulatory genes through p70 ribosomal S6 kinase-1 inhibition in high-fat-high-fructose diet-fed mice. Pharm. Biol. 2013, 51, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneswari, S.; Anuradha, C.V. Astaxanthin prevents loss of insulin signaling and improves glucose metabolism in liver of insulin resistant mice. Can. J. Physiol. Pharmacol. 2012, 90, 1544–1552. [Google Scholar] [CrossRef] [PubMed]
- Alwahsh, S.M.; Ramadori, G. How does bariatric surgery improve type II diabetes? The neglected importance of the liver in clearing glucose and insulin from the portal blood. J. Obes. Weight Loss Ther. 2015, 5, 1000280. [Google Scholar] [CrossRef]
- Gupta, A.; Miegueu, P.; Lapointe, M.; Poirier, P.; Martin, J.; Bastien, M.; Tiwari, S.; Cianflone, K. Acute post-bariatric surgery increase in orexin levels associates with preferential lipid profile improvement. PLoS ONE 2014, 9, e84803. [Google Scholar] [CrossRef] [PubMed]
- Dib, N.; Kiciak, A.; Pietrzak, P.; Ferenc, K.; Jaworski, P.; Kapica, M.; Tarnowski, W.; Zabielski, R. Early-effect of bariatric surgery (scopinaro method) on intestinal hormones and adipokines in insulin resistant wistar rat. J. Physiol. Pharmacol. 2013, 64, 571–577. [Google Scholar] [PubMed]
- Barrière, G.; Tartary, M.; Rigaud, M. Metformin: A rising star to fight the epithelial mesenchymal transition in oncology. Anticancer Agents Med. Chem. 2013, 13, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Diehl, A.M.; Wiseman, M.; Farr, G.H., Jr.; Perrillo, R.P. Metformin in the treatment of non-alcoholic steatohepatitis: A pilot open label trial. Aliment. Pharmacol. Ther. 2004, 20, 23–28. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Q.; Wang, T.; Li, J.; Wang, S.; Qiu, F.; Yu, H.; Zhang, Y.; Wang, T. Effects of Natural Products on Fructose-Induced Nonalcoholic Fatty Liver Disease (NAFLD). Nutrients 2017, 9, 96. https://doi.org/10.3390/nu9020096
Chen Q, Wang T, Li J, Wang S, Qiu F, Yu H, Zhang Y, Wang T. Effects of Natural Products on Fructose-Induced Nonalcoholic Fatty Liver Disease (NAFLD). Nutrients. 2017; 9(2):96. https://doi.org/10.3390/nu9020096
Chicago/Turabian StyleChen, Qian, Tingting Wang, Jian Li, Sijian Wang, Feng Qiu, Haiyang Yu, Yi Zhang, and Tao Wang. 2017. "Effects of Natural Products on Fructose-Induced Nonalcoholic Fatty Liver Disease (NAFLD)" Nutrients 9, no. 2: 96. https://doi.org/10.3390/nu9020096




