1. Early History on Vitamin C and Infections
Vitamin C was identified in the early twentieth century in the search for a substance, the deficiency of which would cause scurvy [
1,
2]. Scurvy was associated with pneumonia in the early literature, which implies that the factor that cured scurvy might also have an effect on pneumonia.
Alfred Hess (1920) summarized a series of autopsy findings as follows: “pneumonia, lobular or lobar, is one of the most frequent complications (of scurvy) and causes of death” and “secondary pneumonias, usually broncho-pneumonic in type, are of common occurrence and in many (scurvy) epidemics constitute the prevailing cause of death” [
3]. He later commented that in “infantile scurvy ... a lack of the antiscorbutic factor (vitamin C) which leads to scurvy, at the same time predisposes to infections (particularly of the respiratory tract) ... Similar susceptibility to infections goes hand in hand with adult scurvy” [
4]. In the early 1900s, Casimir Funk, who coined the word “vitamin”, noted that an epidemic of pneumonia in the Sudan disappeared when antiscorbutic (vitamin C-containing) treatment was given to the numerous cases of scurvy that appeared at about the same time [
5].
The great majority of mammals synthesize vitamin C in their bodies, but primates and the guinea pig cannot. Therefore, the guinea pig is a useful animal model on which to study vitamin C deficiency. Bacteria were often found in histological sections of scorbutic guinea pigs, so much so that some early authors assumed that scurvy might be an infectious disease. However, Hess (1920) concluded that such results merely showed that the tissues of scorbutic animals frequently harbor bacteria, and “there is no doubt that the invasion of the blood-stream does occur readily in the course of scurvy, but this takes place generally after the disease has developed and must be regarded as a secondary phenomenon and therefore unessential from an etiological standpoint. Indeed one of the striking and important symptoms of scurvy is the marked susceptibility to infection” [
3]. When summarizing autopsy findings of experimental scurvy in the guinea pig, Hess also noted that “Pneumonia is met with very frequently and constitutes a common terminal infection”.
Vitamin C was considered as an explanation for scurvy, which was regarded as a disease of the connective tissues, since many of the symptoms such as poor wound healing implied crucial effects on the connective tissues. Therefore, the mainstream view in medicine regarded vitamin C as a vitamin that safeguards the integrity of connective tissues [
6]. The implications of the earlier research by Hess and others were superseded. This historical background might explain the current lack of interest in the effects of vitamin C on infections, even though firm evidence that vitamin C influences infections has been available for decades.
Early literature on vitamin C and infections was reviewed by Clausen (1934), Robertson (1934), and Perla and Marmorston (1937) [
5,
7,
8]. Those reviews are thorough descriptions of the large number of early studies on the topic of this review. Scanned versions of those reviews and English translations of many non-English papers cited in this review are available at the home page of this author [
9]. The book on scurvy by Hess (1920) is available in a digitized format [
3].
4. The Common Cold
The term “the common cold” does not refer to any precisely defined disease, yet the set of symptoms that is called “the common cold” is personally familiar to practically everybody [
61]. Typically the symptoms consist of nasal discharge, sore throat, cough, with or without fever. Young children typically have half a dozen colds per year, and the incidence decreases with age so that elderly people have colds about once per year [
62]. The common cold is the leading cause of acute morbidity and of visits to a physician in high-income countries, and a major cause of absenteeism from work and school. The economic burden of the common cold is comparable to that of hypertension or stroke [
63].
The most relevant definition of the common cold is based on the symptoms; thus the “common cold” does not always entail a viral etiology. Although the majority of common cold episodes are caused by respiratory viruses, similar symptoms are also caused by certain bacterial infections and by some non-infectious causes such as allergic and mechanical irritation. The cough and sore throat after running a marathon does not necessarily imply a viral etiology, although some researchers have assumed so. It is still reasonable to use the term the “common cold” in such a context on the grounds of the symptom-based definition.
4.1. Vitamin C and the Common Cold
Interest in the effects of vitamin C on the common cold originated soon after purified vitamin C became available. The first controlled trials on vitamin C were carried out as early as the 1940s. For example, in the 1950s, a British study examined the clinical effects of vitamin C deprivation, and reported that “the geometric mean duration of colds was 6.4 days in vitamin C-deprived subjects and 3.3 days in non-deprived subjects”, and the authors concluded that the absence of vitamin C tended to cause colds to last longer [
12].
Figure 1 shows the number of participants in placebo-controlled studies in which ≥1 g/day of vitamin C was administered. It also illustrates the main time points of the history of vitamin C and the common cold.
In 1970, Linus Pauling, a Nobel laureate in chemistry and also a Nobel Peace Prize winner, wrote a book on vitamin C and the common cold [
64]. He also published two meta-analyses, which were among the earliest meta-analyses in medicine [
65,
66]. Pauling identified four placebo-controlled studies from which he calculated that there was strong evidence that vitamin C decreased the “integrated morbidity” of colds (
p = 0.00002 [
65]). By integrated morbidity, Pauling meant the total burden of the common cold: the combination of the incidence and duration of colds. In his analysis, Pauling put the greatest weight on the study by Ritzel (1961), which was a randomized controlled trial (RCT) with double-blinded placebo control and the subjects were schoolchildren in a skiing camp in the Swiss Alps [
67]. Ritzel’s study was methodologically the best of the four and used the highest dose of vitamin C, 1 g/day, and therefore Pauling concluded that gram doses of vitamin C would be beneficial against colds [
64,
65,
66].
The activity of Pauling, in turn, led to a great upsurge in interest in vitamin C among lay people and also in academic circles in the early 1970s. From 1972 to 1979, in that eight-year period, 29 placebo-controlled studies were published, which amounted to a total of 8409 participants (
Figure 1) [
68,
69]. Thus, the mean number of participants per study was 290.
In the interval from 1972 to 1975, five placebo-controlled trials were published that used ≥2 g/day of vitamin C. Those five studies were published after Pauling’s book and therefore they formally tested Pauling’s hypothesis. A meta-analysis by Hemilä (1996) showed that there was very strong evidence from the five studies that colds were shorter or less severe in the vitamin C groups (
p = 10
−5), and therefore those studies corroborated Pauling’s hypothesis that vitamin C was indeed effective against colds [
70].
After the mid-1970s, however, interest in the topic plummeted so much so that during the 30-year period from 1985 to 2014, only 11 placebo-controlled trials comprising just 538 participants in total were published, with a mean of 49 participants per study (
Figure 1). Thus, the number of studies published after 1985 is much lower than during the 1970s. In addition, the few recent studies are much smaller than the trials published in the 1970s. Therefore, the great majority of the data on vitamin C and the common cold that are currently available originated within the decade after the publication of Pauling’s book.
This sudden lack of interest after the middle of the 1970s can be explained by three papers published in the same year by Chalmers (1975), Karlowski et al. (1975), and Dykes and Meier (1975) [
71,
72,
73] (
Figure 1). Few trials were started after 1975, which indicates the great impact of these three papers. First, the findings of the placebo controlled studies will be summarized, and then difficulties in the interpretation of common cold studies will be considered, and finally problems in the three papers that were published in 1975 will be discussed.
4.2. Vitamin C Does Not Decrease the Average Incidence of Colds in the General Community
Table 4 summarizes the findings of the studies on vitamin C and the common cold in the Cochrane review by Hemilä and Chalker (2013) [
68,
69]. Regularly administered vitamin C has not decreased the average number of colds among the general population (
Table 4). Another meta-analysis combined the findings of the six largest trials that had used ≥1 g/day of vitamin C and calculated that there was no difference in the vitamin and placebo groups with RR = 0.99 (95% CI 0.93, 1.04) [
74,
75].
Thus, there is no justification for “ordinary people” to take vitamin C regularly in order to prevent colds. However, this conclusion does not mean that regular vitamin C supplementation is ineffective for all people. There is strong evidence that vitamin C decreases the incidence of colds under special conditions and/or among certain population groups.
4.3. Vitamin C May Decrease Common Cold Incidence in Special Conditions
Vitamin C halved the incidence of colds in five RCTs during which the participants were under heavy short-term physical activity (
Table 4) [
68,
76]. Three of the studies used marathon runners in South Africa as subjects, whereas one study used Canadian military personnel on winter exercise, and the fifth study was on schoolchildren in a skiing camp in the Swiss Alps, i.e., the Ritzel (1961) trial [
67]. Thus, three studies were conducted under conditions of a hot environment and profound physical stress and the other two were carried out under cold environments and physical stress (see
Section 2.4).
Another group in which vitamin C has prevented colds is British men [
74,
75,
77]. Four trials found that vitamin C decreased the incidence of colds by 30%, and in another set of four trials, the proportion of men who had recurrent common cold infections during the study decreased by a mean of 46%. All these studies were carried out in the 1970s or earlier, and according to surveys, the intake of vitamin C in the United Kingdom was low when the studies were carried out, 0.03 to 0.06 g/day, and three of the U.K. trials specifically estimated that the dietary vitamin C intake was between 0.015 to 0.05 g/day [
74]. In particular, Baird (1979) administered only 0.08 g/day of vitamin C yet they observed 37% lower incidence of colds in the vitamin C group, indicating that it was the “marginal deficiency” and not a high dose that explained the benefit [
77,
78].
In addition, the levels of vitamin C are usually lower in men than in women, which may explain the benefit for British males, in comparison to no apparent effect in British females. Evidently, the dietary vitamin C intake in the United Kingdom has increased since the 1970s, and therefore these studies do not indicate that vitamin C supplementation would necessarily influence colds in ordinary British men nowadays. However, if low dietary vitamin C intake increases the risk of respiratory infections, then that may be currently relevant in other contexts, since there are still many population groups that have low intakes of vitamin C. A recent small study in the USA by Johnston (2014) was restricted to 28 males with marginally low vitamin C levels, mean 30 µmol/L, and found a decrease in common cold incidence, RR = 0.55 (95% CI: 0.33–0.94;
p = 0.04) [
79], which may also be explained by the low vitamin C levels.
4.4. Vitamin C Might Protect against the Common Cold in a Restricted Subgroup of the General Community
Although vitamin C has not influenced the average common cold incidence in the general community trials (
Table 4), some of them found that there was a subgroup of people who had obtained benefits from vitamin C. In a Canadian trial, Anderson (1972) [
80] reported that in the vitamin C group there were 10 percentage points more participants with no “days confined to house” because of colds (57% vs. 47%;
p = 0.01, [
1] (p. 44)). Thus, one in 10 benefited from vitamin C in this outcome. In a trial with Navajo schoolchildren, Coulehan (1974) [
81] found that in the vitamin C group there were 16 percentage points more children who were “never ill on active surveillance by a medically trained clerk or the school nurse” (44% vs. 29%;
p < 0.001; [
1] (p. 44)). A more recent study in the UK by van Straten (2002) reported that vitamin C decreased the number of participants who had recurrent colds by 17 percentage points [
82] (19% vs. 2%;
p < 0.001, [
1] (p. 47)). Thus, the statistical evidence of benefit for a restricted subgroup in these three trials is strong.
4.5. Vitamin C Shortens and Alleviates the Common Cold
The effect of vitamin C on the duration and severity of the common cold has been studied in regular supplementation trials and in therapeutic trials. Regular supplementation means that vitamin C was administered each day over the whole study period, and the outcome is the duration and severity of colds that occurred during the study. Therapeutic vitamin C trial means that vitamin C administration was started only after the first common cold symptoms had occurred and the duration of colds were then recorded.
In regular supplementation studies, ≥0.2 g/day of vitamin C decreased the duration of colds by 9% (
Table 4). When the dosage was ≥1 g/day of vitamin C, the mean duration of colds was shortened by 8% in adults and by 18% in children. Vitamin C also significantly alleviated the severity of the colds.
Therapeutic studies have hitherto not shown consistent benefit from vitamin C. However, therapeutic trials are more complex to conduct and interpret than regular supplementation trials. If the timing of the initiation of supplementation or the duration of supplementation influences the extent of the benefit, false negative findings may result from inappropriate study protocols. For example, four therapeutic studies used only 2–3 days of 2–4 g/day vitamin C supplementation, whereas the mean duration of colds in these studies was about a week. None of these studies detected any benefit from vitamin C [
68,
83]. On the other hand, Anderson (1974) [
84] found that 8 g/day on the first day only reduced the duration of colds significantly (
Figure 2). In addition, in a five-day therapeutic trial, Anderson (1975) [
85] reported a 25% reduction in “days spent indoors per subject” because of illness (
p = 0.048) in the vitamin C group (1 to 1.5 g/day) [
1] (p. 48). Finally, none of the therapeutic studies investigated children, although the effect of regular vitamin C has been greater in children (
Table 4). Thus, although the regular supplementation trials unambiguously show that vitamin C shortens and alleviates the common cold, there is no consistent evidence that therapeutic supplementation is effective.
4.6. Possible Differences in the Effects of Vitamin C between Subgroups
The regular supplementation study by Anderson (1972) is one of the largest that has been carried out [
80]. They found that the proportion of participants who were not confined to the house decreased by 10 percentage points in the vitamin C group. In addition, they found that per episode the days confined to the house was 21% shorter in the vitamin C group. Together these combine to a 30% reduction in the days confined to the house per person (
p = 0.001). Such a large effect gives statistical power for subgroup comparisons.
Anderson (1972) reported that vitamin C decreased total days confined to house by 46% in participants who had contact with young children, but just by 17% in participants who did not have contact with young children (
Table 5). Anderson (1972) also reported that vitamin C decreased total days confined to house by 43% in participants who usually had two or more colds per winter, but just by 13% in participants who usually had zero to one cold per winter (
Table 5).
In a study with adolescent competitive swimmers, Constantini (2011) found a significant difference between males and females in the effect of vitamin C, whereby the vitamin halved the duration and severity of colds in males but had no effect on females [
86]. In a study with British students, Baird (1979) also found a significant difference between males and females, but the outcome was the incidence of colds (
Table 5).
Carr (1981) found that vitamin C had a beneficial effect on the duration of colds for twins living separately, but not for twins living together [
87]. This subgroup difference might be explained by swapping of tablets by twins living together, which was not possible for twins living separately.
The significant within-trial differences in the effect of vitamin C on the common cold indicate that there is no universal effect of vitamin C valid over the whole population. Instead, the size of the vitamin C effect seems to depend on various characteristics of people (see
Section 2.8).
4.7. Dose Dependency of Vitamin C Supplementation Effect
An earlier meta-analysis of dose-dependency calculated that on average 1 g/day of vitamin C shortened the duration of colds in adults on average by 6% and in children by 17%; and ≥2 g/day vitamin C shortened the duration of colds in adults by 21% and in children by 26% [
83]. Thus, higher doses were associated with greater effects. In addition, children weigh less than adults and the greater effects in children may be explained by a greater dose per weight. Nevertheless, such a comparison suffers from numerous simultaneous differences between the trials. The most valid examination of dose–response is within a single study so that the virus distribution is similar in each trial arm and the outcome definition is identical.
Coulehan (1974) [
81] administered 1 g/day to children and observed a 12% reduction in common cold duration, and in parallel they administered 2 g/day to other children and observed a 29% reduction in cold duration. Although the point estimates suggest a dose–response, the study was small and the 95% CIs overlap widely [
68,
83].
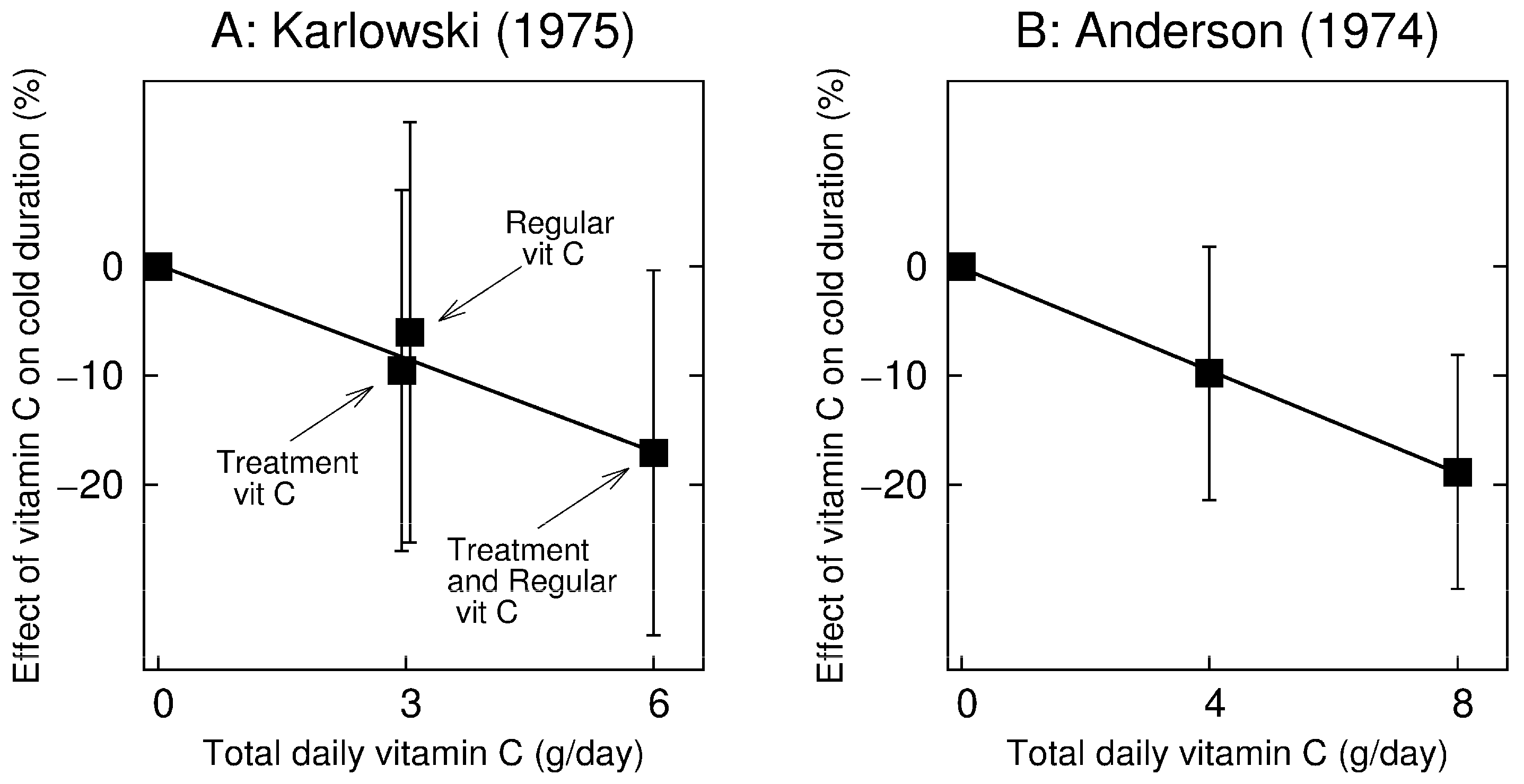
In a 2 × 2 design, Karlowski (1975) [
72] randomized participants to 3 g/day regular vitamin C and to 3 g/day vitamin C treatment for five days when the participant caught a cold. Thus, one study arm was administered placebo, the second was administered regular vitamin C, the third therapeutic, and the fourth arm was administered regular + therapeutic vitamin C (i.e., 6 g/day). The four arms of the Karlowski trial are shown in
Figure 2A. The 95% CIs show the comparisons with the placebo group. The test for trend for a linear regression model gives
p = 0.018.
Anderson (1974) [
84] randomized participants to a placebo and two vitamin C treatment arms which were administered vitamin C only on the first day of the cold. One treatment arm (arm #7) was given 4 g/day of, and another (arm #8) was given 8 g/day. These arms are compared with the placebo arm #4 in
Figure 2B. The 95% CIs show the comparisons with the placebo group. The test for trend in a linear regression model gives
p = 0.013.
Finally, some case reports have proposed that vitamin C doses should be over 15 g/day for the best treatment of colds [
88,
89]. Thus, it is possible that the doses used in most of the therapeutic studies, up to just 6–8 g/day, have not been sufficiently high to properly test the effects of vitamin C that might be achievable.
4.8. Vitamin C and Complications of the Common Cold
Given the strong evidence that regularly administered vitamin C shortens and alleviates common cold symptoms, it seems plausible that vitamin C might also alleviate complications of the common cold. One frequent complication is the exacerbation of asthma [
90].
A systematic review identified three studies that provided information on the potential pulmonary effects of vitamin C in sufferers of common cold–induced asthma [
91]. A trial conducted in Nigeria studied asthmatic patients whose asthma exacerbations resulted from respiratory infections. A vitamin C dose of 1 g/day decreased the occurrence of severe and moderate asthma attacks by 89% [
92]. Another study on patients who had infection-related asthma reported that 5 g/day vitamin C decreased the prevalence of bronchial hypersensitivity to histamine by 52 percentage points [
93]. A third study found that the administration of a single dose of 1 g vitamin C to non-asthmatic common cold patients decreased bronchial sensitivity in a histamine challenge test [
94].
It has also been proposed that vitamin C might prevent sinusitis and otitis media [
95,
96], but to our knowledge there are no data from controlled studies.
A further complication of viral respiratory infections is pneumonia; this is discussed in the section on pneumonia.
6. Evaporation of Interest in Vitamin C and the Common Cold after 1975
Given the strong evidence from studies published before 1970 that vitamin C has beneficial effects against the common cold [
65], and from the ≥2 g/day vitamin C studies published between 1972 and 1975 [
70], it is puzzling that the interest in vitamin C and the common cold collapsed after 1975 so that few small trials on vitamin C and the common cold have been conducted thereafter (
Figure 1).
This sudden loss of interest can be explained by the publication of the three highly important papers in 1975 (
Figure 1). These papers are particularly influential because of their authors and the publication forums. Two of the papers were published in
JAMA [
72,
73], and the third paper was published in the
American Journal of Medicine [
71]. Both of these journals are highly influential medical journals with extensive circulations. Two of the papers were authored by Thomas Chalmers [
71,
72], who was a highly respected and influential pioneer of RCTs [
1,
102,
103], and the third paper was authored by Paul Meier [
73], who was a highly influential statistician, e.g., one of the authors of the widely used Kaplan–Meier method [
1,
104,
105].
Karlowski, Chalmers, et al. (1975) [
72] published the results of a RCT in
JAMA, in which 6 g/day of vitamin C significantly shortened the duration of colds (
Figure 2A). However, these authors claimed that the observed benefit was not caused by the physiological effects of vitamin C, but by the placebo effect. However, the “placebo-effect explanation” was shown afterwards to be erroneous. For example, Karlowski et al. had excluded 42% of common cold episodes from the subgroup analysis that was the basis for their conclusion, without giving any explanation of why so many participants were excluded. The numerous problems of the placebo explanation are detailed in a critique by Hemilä [
1,
106,
107]. Chalmers wrote a response [
108], but did not answer the specific issues raised [
109].
In the same year (1975), Chalmers published a review of the vitamin C and common cold studies. He pooled the results of seven studies and calculated that vitamin C would shorten colds only by 0.11 (SE 0.24) days [
71]. Such a small difference has no clinical importance and the SE indicates that it is simply explained by random variation. However, there were errors in the extraction of data, studies that used very low doses of vitamin C (down to 0.025 g/day) were included, and there were errors in the calculations [
1,
110]. Pauling had proposed that vitamin C doses should be ≥1 g/day. When Hemilä and Herman (1995) included only those studies that had used ≥1 g/day of vitamin C and extracted data correctly, they calculated that colds were 0.93 (SE 0.22) days shorter, which is over eight times that calculated by Chalmers, and highly significant (
p = 0.01) [
110].
The third paper was a review published in
JAMA by Michael Dykes and Paul Meier (1975). They analyzed selected studies and concluded that there was no convincing evidence that vitamin C has effects on colds [
73]. However, they did not calculate the estimates of the effect nor any
p-values, and many comments in their analysis were misleading. Pauling wrote a manuscript in which he commented upon the review by Dykes and Meier and submitted it to
JAMA. Pauling stated afterwards that his paper was rejected even after he twice made revisions to meet the suggestions of the referees and the manuscript was finally published in a minor journal [
111,
112]. The rejection of Pauling’s papers was strange since the readers of
JAMA were effectively prevented from seeing the other side of an important controversy. There were also other problems that were not pointed out by Pauling; see [
1,
70].
Although the three papers have serious biases, they have been used singly or in the combinations of two as references in nutritional recommendations, in medical textbooks, in texts on infectious diseases and on nutrition, when the authors claimed that vitamin C had been shown to be ineffective for colds [
1] (pp. 21–23, 36–38, 42–45). The American Medical Association, for example, officially stated that “One of the most widely misused vitamins is ascorbic acid. There is no reliable evidence that large doses of ascorbic acid prevent colds or shorten their duration” [
113], a statement that was based entirely on Chalmers’s 1975 review.
These three papers are the most manifest explanation for the collapse in the interest in vitamin C and the common cold after 1975, despite the strong evidence that had emerged by that time that ≥2 g/day vitamin C shortens and alleviates colds [
70].
7. Pneumonia
Pneumonia is the most common severe infection, which is usually caused by bacteria and viruses.
As recounted at the beginning of this review, the association between frank vitamin C deficiency and pneumonia was noted by Alfred Hess and other early authors, when the chemical identity of vitamin C was not yet known. Vitamin C was purified in the early 1930s and soon thereafter a few German and U.S. physicians proposed that vitamin C might be beneficial in the treatment of pneumonia. For example, Gander and Niederberger (1936) concluded from a series of 15 cases that “the general condition is always favorably influenced (by vitamin C) to a noticeable extent, as is the convalescence, which proceeds better and more quickly than in cases of pneumonia, which are not treated with vitamin C” [
114] and other German physicians also claimed benefits of vitamin C [
115,
116]. Translations of these papers are available [
9]. Case reports from the USA also suggested that vitamin C was beneficial against pneumonia [
117,
118,
119].
A Cochrane review on vitamin C and pneumonia identified three controlled trials that reported the number of pneumonia cases in participants who were administered vitamin C and two therapeutic trials in which pneumonia patients were given vitamin C [
27,
28].
7.1. Vitamin C and the Incidence of Pneumonia
Table 7 shows the findings of the three vitamin C and pneumonia trials. Each of them found a ≥80% lower incidence of pneumonia for their vitamin C group [
27,
28,
120].
Glazebrook (1942) studied male students (15–20 years) in a boarding school in Scotland during World War II [
97]. No formal placebo was used; however, 0.05 to 0.3 g/day of vitamin C was added to the morning cocoa and to an evening glass of milk in the kitchen. Thus, the placebo effect does not seem to be a relevant concern in the dining hall. The ordinary diet of the schoolboys contained only 0.015 g/day vitamin C so that their intake was particularly low.
Kimbarowski (1967) studied the effect of 0.3 g/day of vitamin C on military recruits who had been hospitalized because of influenza type-A in the former Soviet Union [
121]. Thus, these pneumonia cases were complications of the viral respiratory infection. Vitamin C also shortened the mean stay in hospital for pneumonia treatment (9 vs. 12 days).
The latest of the three pneumonia prevention trials was carried out during a two-month recruit training period with U.S. Marine recruits by Pitt (1979) [
122]. The dose of vitamin C was 2 g/day. This was a randomized double-blind placebo-controlled trial, whereas the two earlier studies were not.
The findings of the three studies are consistent with the notion that the level of vitamin C intake may influence the risk of pneumonia. However, all the three studies were carried out using special participants under particular conditions, and their findings cannot be generalized to the ordinary current Western population. Dietary vitamin C intake was particularly low in the oldest study, and may also have been low in the second study. Thus, the benefit of vitamin C supplementation may be explained by the correction of marginal deficiency in these two older studies. However, in the study by Pitt (1979), the baseline plasma level of vitamin C, 57 µmol/L, corresponds to the dietary vitamin C intake of about 0.1 g/day [
11]. Furthermore, although the dose of 2 g/day was high, the plasma level of vitamin C increased only by 36% for the vitamin C group. This also indicates that the basal dietary intake vitamin C was high. Thus, treating marginal vitamin C deficiency is not a reasonable explanation for that latest study.
It is also worth noting that two of these trials used military recruits, and the third used young males who were accommodated in a boarding school [
123]. Therefore, the exposure to viruses and bacteria causing pneumonia may have been much higher compared to children and young adults living at home. In each of the three trials, the incidence of pneumonia in the control group was very high when compared with the incidence in the ordinary population [
124,
125]. A high incidence of pneumonia has been reported in military recruits [
126], but the incidence of pneumonia has been even higher in some child populations of the developing countries [
127] (
Table 7).
It seems reasonable to consider that these three studies observed a true effect of vitamin C against pneumonia in their specific circumstances. However, these findings should not be extrapolated to different circumstances. It would seem worthwhile to examine the effect of vitamin C in population groups that have a high incidence of pneumonia concomitantly with a low intake of vitamin C [
27,
41].
7.2. Vitamin C in the Treatment of Pneumonia
Two studies have reported on the therapeutic effect of vitamin C for pneumonia patients [
27,
28].
Hunt (1994) carried out a randomized, double-blind placebo controlled trial with elderly people in the UK (mean age 81 years), who were hospitalized because of acute bronchitis or pneumonia [
26]. The mean plasma vitamin C level at baseline was 23 µmol/L and one third of the patients had a vitamin C level of just ≤11 µmol/L. There was a significant difference in the effect of 0.2 g/day of vitamin C between patients who were more ill and those who were less ill when admitted to the hospital. Vitamin C reduced the respiratory symptom score in the more ill patients but not in their less ill counterparts. There were also six deaths during the study, all among the more ill participants: five in the placebo group, but only one in the vitamin C group.
Mochalkin (1970) examined the effect of vitamin C on pneumonia patients in the former Soviet Union [
25]. Although a placebo was not administered to the control group, two different doses of vitamin C were used and the observed difference between the low and high dosage cannot be explained by the placebo effect. The high-dose regime administered on average twice the quantity of vitamin C of the low dose, but both of them were related to the dosage of antibiotics so that the low dose vitamin C ranged from 0.25 to 0.8 g/day, and the high dose ranged from 0.5 to 1.6 g/day. The duration of hospital stay in the control group (no vitamin C supplementation) was 23.7 days. In the low dose vitamin C group the hospital stay was 19% shorter and in the high dose vitamin C group it was 36% shorter. A benefit was also reported on the normalization of chest X-ray, temperature, and erythrocyte sedimentation rate.
Although both of these therapeutic studies give support to the old case reports stating that vitamin C is beneficial for pneumonia patients, the findings cannot be directly generalized to typical pneumonia patients of Western countries.
8. Tetanus
Tetanus is a disease caused by the toxin of
Clostridium tetani, which may contaminate wounds. An early case report claimed that vitamin C was beneficial against tetanus in an unvaccinated six-year-old boy in the USA [
128]. A Cochrane review identified one controlled trial in which the effect of vitamin C on tetanus patients was examined [
129,
130].
Jahan (1984) studied the effect of 1 g/day of intravenous vitamin C on tetanus patients in Bangladesh [
131]. In children aged one to 12 years, there were no deaths in the vitamin C group, whereas there were 23 deaths in the control group (
p = 10
−9) [
1] (p. 17). In tetanus patients aged 13 to 30 years, there were 10 deaths in the vitamin C group compared with 19 deaths in the control group (
p = 0.03). The significant difference between the above-described age groups may be caused by the difference in the body weights of the patients. In the young children the same dose of vitamin C corresponds to a substantially higher dose per unit of weight. Although there were methodological weaknesses in the trial, they are unlikely explanations for the dramatic difference in the younger participants [
129].
9. Other Infections
The effect of vitamin C supplementation on the common cold has been most extensively studied. One important reason for extensive research on vitamin C and the common cold seems to be the wide publicity given to it by Pauling [
1,
132]. Probably some researchers wanted to show that Pauling was either right or wrong, whereas others just wanted to study a topic about which a Nobel Prize winner had put his credibility on the line. Another reason for the large number of studies on the common cold is that it is a non-severe ubiquitous infection, and it is very easy to find common cold patients in schools and work places. It is much more difficult to study more serious infections.
The three infections discussed above, the common cold, pneumonia, and tetanus, were selected on the basis that the effects of vitamin C have been evaluated in Cochrane reviews, which entails a thorough literature search and a careful analysis of the identified trials. However, the selection of these three infections does not imply that the effects of vitamin C are limited to them.
Table 2 indicates that vitamin C may have effects on various infections caused by viruses, bacteria,
Candida albicans and protozoa. Vitamin C might have similar effects in humans. However, it also seems evident that the role of additional vitamin C depends on various factors such as the initial dietary intake level, other nutritional status, the exposure level to pathogens, the level of exercise and temperature stress, etc.
Three extensive searches of the older literature on vitamin C and infections have been published, and they give an extensive list of references, but none of these publications gave a balanced discussion of the findings [
133,
134,
135]. A few studies on the possible effects of vitamin C on other infections are outlined below, but this selection is not systematic.
Terezhalmy (1978) [
136] used a double-blind placebo-controlled RCT and found that the duration of pain caused by herpes labialis was shortened by 51%, from 3.5 to 1.3 days (
p = 10
−8), when patients were administered 1 g/day of vitamin C together with bioflavonoids [
1] (pp. 15–17). Furthermore, when vitamin C treatment was initiated within 24 hours of the onset of the symptoms, only six out of 26 patients (23%) developed herpes vesicles, whereas with later initiation of vitamin C, eight out of 12 patients (67%) developed vesicles (
p = 0.003 in the test of interaction). Vitamin C was administered with bioflavonoids, so the study was not specific to vitamin C, but there is no compelling evidence to indicate that bioflavonoids affect infections.
Herpes zoster (reactivation of varicella zoster virus) can cause long lasting post-herpetic neuralgia (PHN). Chen (2009) found that patients with PHN had significantly lower plasma vitamin C plasma than healthy volunteers, and their RCT showed that vitamin C administration significantly decreased the pain level of PHN [
137]. A number of other reports have also suggested that vitamin C may be effective against the pain caused by herpes zoster [
138,
139,
140,
141,
142].
Patrone (1982) and Levy (1996) reported that vitamin C administration was beneficial to patients who had recurrent infections, mainly of the skin [
143,
144]. Many of the patients had impaired neutrophil functions and therefore the findings cannot be generalized to the ordinary population.
Galley (1997) reported that vitamin C increased the cardiac index in patients with septic shock [
145]. Pleiner (2002) reported that intravenous vitamin C administration preserved vascular reactivity to acetylcholine in study participants who had been experimentally administered
Escherichia coli endotoxin [
146].
It seems unlikely that the effects of vitamin C on herpetic pain, cardiac index and the vascular system are mediated through effects on the immune system. Such effects are probably caused by other mechanisms instead. The question of the possible benefits of vitamin C against infections is therefore not just a question about the immune system effects of the vitamin, as was discussed earlier in this review (see
Section 2.7).
Some physicians used vitamin C for a large set of infectious disease patients and described their experiences in case reports that are worth reading [
89,
147].
10. Observational Studies on Vitamin C and Infections
Cohort studies on vitamins are often unreliable because diet is strongly associated with numerous lifestyle factors that cannot be fully adjusted for in statistical models. Therefore, there may always remain an unknown level of residual confounding [
148]. The main source of vitamin C in the diet is fruit, and high dietary vitamin C intake essentially always means a high fruit intake [
149]. Thus, any substantial correlations between vitamin C intake and infections could also reflect some other substances in fruit. Only two observational studies are commented upon in this section.
Merchant (2004) studied men whose ages ranged from 40 to 75 years in the USA and found no association between their vitamin C intake and community-acquired pneumonia [
124]. These males were U.S. health professionals; thus they were of a population that has a great interest in factors that affect health. The incidence of pneumonia was only three cases per 1000 person-years (
Table 7). The median vitamin C intake of the lowest quintile was 0.095 g/day and of the highest quintile it was 1.1 g/day. In contrast, the overall median of the adult U.S. population is about 0.1 g/day, and 10% of the U.S. population has an intake level of less than 0.04 g/day [
14]. Thus, Merchant and colleagues’ cohort study indicates that increasing the vitamin C intake upwards from the median level in the USA will not lead to any further decline in the already low pneumonia incidence among male health professionals. However, the study is uninformative about whether decreasing vitamin C level downwards from 0.1 g/day might increase pneumonia risk, or about whether vitamin C might have effects in populations that have particularly high incidences of pneumonia (
Table 7). Even though we must be cautious about interpreting observational studies, it seems that biological differences, rather than methodological differences, are most reasonable explanations for the divergence between the findings in the Merchant et al. cohort study and the three controlled trials shown in
Table 7.
A cohort analysis of Finnish male smokers that is part of the Alpha-Tocopherol Beta-Carotene Cancer prevention (ATBC) Study found a significant inverse association between dietary vitamin C intake and tuberculosis risk in participants who were not administered vitamin E supplements [
150,
151]. The highest quartile had the median dietary vitamin C intake level of 0.15 g/day, whereas the lowest quartile had an intake level of only 0.052 g/day. The adjusted risk of tuberculosis in the lowest vitamin C intake quartile was 150% higher than that of the highest intake quartile. This is consistent with the animal studies that found that low vitamin C intake increases the susceptibility to, and severity of, tuberculosis (
Table 1,
Table 2 and
Table 3).
11. Potentially Harmful Interactions between Vitamins C and E
Vitamin C and vitamin E are both antioxidants and they protect against ROS. Therefore, these substances are of parallel interest as water-soluble vitamin C regenerates the lipid-soluble vitamin E in vitro [
152]. Dietary vitamin C intake modified the effect of vitamin E on mortality in the ATBC Study, which indicates that these substances may also have clinically important interactions [
153]. However, the major sources of the vitamin C in this subgroup were fruit, vegetables and berries and other substances in these foods might also have explained the modification of the vitamin E effect. Such a possibility was refuted by calculating the residual intake of fruit, vegetables and berries, and showing that the residual did not modify the effect of vitamin E. Vitamin C was thus indicated as the specific modifying factor. A similar approach was used to show that vitamin C specifically modified the effect of vitamin E on pneumonia [
154].
Two subgroups of the ATBC Study were identified in which the combination of high dietary vitamin C intake and vitamin E supplementation increased the risk of pneumonia by 248% and 1350% when compared with high vitamin C intake without vitamin E (
Table 8). In the former subgroup, one extra case of pneumonia was caused for every 13 participants and in the latter subgroup, for every 28 participants. In both subgroups, the residual intake of fruit, vegetables and berries did not modify the effect of vitamin E, indicating specificity of vitamin C. The total number of participants in the ATBC Study was 29,133 and in that respect the identified subgroups were relatively small and at 1081 individuals only amounted to 4% of all the ATBC participants. However, in these two subgroups the harm arising from the combination of vitamins C and E was substantial [
154].
Another subgroup analysis of the ATBC Study found that the combination of high vitamin C intake together with vitamin E supplementation increased the risk of tuberculosis in heavy smokers by 125% compared with high vitamin C alone subgroup (
Table 8). Thus, one extra case of tuberculosis arose in every 240 participants who had high intakes of vitamins C and E [
150,
151].
ROS have been implicated in the pathogenesis of diverse diseases, including infections. Antioxidants have been assumed to be beneficial since they react with ROS. However, given the suggestions that people should take vitamins C and E to improve their immune system, the subgroup findings in
Table 8 are somewhat alarming. Nevertheless, the harm in the three subgroups is limited to the combination of vitamins C and E. This author does not know of any findings that indicate that similar doses of vitamin C alone might cause harm.
12. Misconceptions and Prejudices about Vitamin C and Infections
In the first half of the 20th century, a large number of papers were published in the medical literature on vitamin C and infections and several physicians were enthusiastic about vitamin C. The topic was not dismissed because of large-scale controlled trials showing that vitamin C was ineffective. Instead, many rather large trials found benefits of vitamin C. There seem to be four particular reasons why the interest in vitamin C and infections disappeared.
First, antibiotics were introduced in the mid-20th century. They have specific and sometimes very dramatic effects on bacterial infections and therefore are much more rational first line drugs for patients with serious infections than vitamin C. Secondly, vitamin C was identified as the explanation for scurvy, which was considered a disease of the connective tissues. Evidently it seemed irrational to consider that a substance that “only” participates in collagen metabolism might also have effects on infections. However, the biochemistry and actions of vitamin C are complex and not limited to collagen metabolism. Thirdly, the three papers published in 1975 appeared to herald the loss of interest in vitamin C and the common cold (
Figure 1) and it seems likely that they increased the negative attitude towards vitamin C for other infections as well. Fourthly, “if a treatment bypasses the medical establishment and is sold directly to the public ... the temptation in the medical community is to accept uncritically the first bad news that comes along” [
155].
The belief that vitamin C is “ineffective” has been widely spread. For example, a survey of general practitioners in the Netherlands revealed that 47% of respondents considered that homeopathy is efficacious for the treatment of the common cold, whereas only 20% of those respondents considered that vitamin C was [
156]. Prejudices against vitamin C are not limited to the common cold. Richards compared the attitudes and arguments of physicians to three putative cancer medicines, 5-fluorouracil, interferon and vitamin C, and documented unambiguous bias against vitamin C [
157,
158,
159]. Goodwin and Tangum gave several examples to support the conclusion that there has been a systematic bias against the concept that vitamins may yield benefits in levels higher than the minimum needed to avoid the classic deficiency diseases [
160].
The use of vitamin C for preventing and treating colds falls into the category of alternative medicine under the classifications used by the National Institutes of Health in the USA and of the Cochrane collaboration. However, such categorization does not reflect the level of evidence for vitamin C, but reflects the low level of acceptance amongst the medical community, and may further amplify the inertia and prejudices against vitamin C [
161].