1. Introduction
Newcastle disease virus (NDV) is characterized by selective viral replication of tumor cells [
1]; this virus is known to be oncolytic i.e., it kills infected tumor cells and oncolytic therapy with NDV has been studied [
2]. Moreover, in addition to direct tumor cytotoxicity due to its oncolytic properties, NDV-infected tumor cells were reported to induce the maturation of dendritic cell by type I interferon (IFN) [
3,
4], activation of antigen specific T cells [
5], immunostimulatory effects such as the activation of NK cells by hemagglutinin-neuraminidase (HN) protein expressed on an infected cell surface [
6], and increase in MHC class I expression [
7]. Currently, an NDV-infected tumor vaccine (NDV-TV), using NDV as an adjuvant of tumor cell vaccine, has been studied and clinically tested in humans with mammary adenocarcinoma, ovarian cancer [
8], colon cancer [
9], squamous cell carcinoma of the head and neck [
9,
10], and glioblastoma multiforme [
11]. These reports indicated an effective anti-tumor response induced by NDV-TV.
Previously, we generated a green fluorescent protein (GFP)-expressing recombinant NDV (rNDV). The virus induced cell death only in infected cells without producing any infectious virus particles, because the viral F protein cleavage site amino acid sequence was transformed (G-R-Q-G/S-R; L) [
12]. Furthermore, we examined the induction of anti-tumor response induced by the rNDV infected tumor vaccine (rNDV-TV). rNDV-TV was prepared as tumor cells were infected with rNDV in vitro, then irradiated by UV for inactivation of both rNDV and tumor cells. The rNDV-TV was administered to mice as an immunogen (
Figure 1a). In murine melanoma, the mice inoculated with rNDV-TV had significantly suppression of tumor metastasis compared to the control. And, the rNDV-TV vaccinated group survived longer than the control group. [
13]. Also in murine fibrosarcoma, the mice inoculated with rNDV-TV induced an effective anti-tumor response and complete tumor exclusion [
14]. These experiments revealed the induction of an anti-tumor response by rNDV-TV. However, the results indicated that there were differences in the anti-tumor effect of rNDV-TV between used tumor cell lines.
Therefore, in this study we focused on the difference in the anti-tumor response of rNDV-TV and the rNDV infectivity between several murine tumor cell lines. Moreover, to improve the anti-tumor response of rNDV-TV, the possibility of a new therapeutic strategy was investigated.
2. Materials and Methods
2.1. Animals
Specific pathogen-free C57BL/6 and C3H/HeN murine strains were obtained from CLEA Japan, Inc. (Tokyo, Japan). Mice were maintained in a sterile isolator and treated according to the Laboratory Animal Control Guidelines of the National Guidelines for the Care and Use of Laboratory Animals (Rakuno Gakuen University approval number: VH14A4).
2.2. Cell Culture and Virus Production
The
murine fibrosarcoma cell line WEHI164 (laboratory stock cell line), mammary tumor cell line BALB-MC (derived from BALB/c mice, #JCRB0233.0, JCRB cell bank, Ibaraki, Japan), melanoma cell line B16 and Lewis lung carcinoma cell line 3LL (derived from C57BL/6, #JCRB0202 and # JCRB1348, JCRB cell bank, Ibaraki, Japan), squamous cell carcinoma SCC VII (provided from Dr. Inanami, Hokkaido University, Sapporo, Japan), and bladder carcinoma MBT-2 (derived from C3H/HeN, # IFO50041, JCRB cell bank, Ibaraki, Japan) were used. WEHI164 and 3LL were cultured in RPMI-1640 medium (Sigma-Aldrich, St. Louis, MO, USA), B16 was cultured in Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, St. Louis, MO, USA), and the others were cultured in Minimum Essential Medium (MEM; Gibco, Waltham, MA, USA). The cells were cultured in each medium supplemented with 5% fetal bovine serum (FBS; Gibco, Waltham, MA, USA), 200 U/mL penicillin (Meiji, Tokyo, Japan), and 200 μg/mL streptomycin (Meiji, Tokyo, Japan) at 37 °C in a humidified atmosphere with 5% CO
2. GFP-expressing rNDV was generated as previously described [
12] and proliferated in embryonated chicken eggs. rNDV was collected from the allantoic fluid, and the number of focus-forming units (FFUs) was determined.
2.3. Preparation of rNDV-TV, Ultraviolet Irradiated Tumor Vaccine (UV-TV) and Ruxolitinib-Treated rNDV-TV (rNDV-TV-Rux)
The tumor cells (105 cells/well) were cultured with each medium supplemented with 5% FBS in 6-well plates. For rNDV-TV-Rux preparation, the cells were cultured in the appropriate medium, containing 1 μg/mL of janus kinase (JAK) inhibitor, ruxolitinib (Rux; Cayman Chemical, Ann Arbor, MI, USA), for 20 h before rNDV infection. Then, the cells were infected with rNDV using a multiplicity of infection (MOI) of 2 and cultured at 37 °C for 24 h in a CO2 incubator. rNDV-infected tumor cells were harvested with ethylenediaminetetraacetic acid and phosphate-buffered saline (EDTA-PBS), then inactivated with UV irradiation (400 mJ/cm2). After washing with PBS, the cells were resuspended at 5 × 105 cells/mL in the correct medium. UV-TV was prepared by UV irradiating (400 mJ/cm2) each tumor cell, those without rNDV infection were also prepared.
2.4. Immunization Protocol
Each vaccine was administered intraperitoneally to mice at 105 cells/mouse four times at weekly intervals. For control, 0.2 mL of each cell culture medium was administered intraperitoneally to mice.
2.5. Cytotoxicity Assay
Spleen cells were collected from immunized mice, and splenic mononuclear cells (SMCs) were separated by density gradient centrifugation using Ficoll–Conray solution (d = 1.088). To induce the cytotoxic T lymphocytes (CTL), the SMCs were cultured with inactivated tumor cells for 5 days at an effector–target ratio (E:T) of 4:1. After a 5-day pre-stimulation with the tumor cells, effector SMCs were separated by density gradient centrifugation with Ficoll–Conray solution. SMCs were washed with PBS and co-cultured with each tumor cell at an E:T of 20:1 for 24 h. Cytotoxicity was determined by lactate dehydrogenase (LDH) release using a Cytotoxicity Detection KitPLUS (LDH) (Roche Diagnostics, Basel, Switzerland). The cytotoxicity rate was calculated as follows: Cytotoxicity (%) = (Experimental LDH − Effector spontaneous LDH − Target spontaneous LDH)/(Target maximum LDH − Target spontaneous LDH) × 100 (rNDV-TV and UV-TV group, each n = 3; control group, n = 2).
2.6. Viral Infection
The tumor cells (5 × 105 cells/well) were cultured with each cell adopted medium containing 5% FBS in 6-well plates. After washing with FBS-free medium, the cells were adsorbed with rNDV using a multiplicity of infection (MOI) of 2 for 1 h in a CO2 incubator. Then, the cells were cultured in medium with 5% FBS at 37 °C for 24 h in a CO2 incubator, and observed with a fluorescence microscope (Zeiss, Oberkochen, Germany). The infection rate was calculated as follow: Infection rate (%) = Tumor cells expressing GFP/Total tumor cells × 100.
2.7. RNA Extraction and cDNA Synthesis
Total RNA was extracted with the RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Sample were lysed with 350 μL of RLT buffer, and total RNA was eluted with 50 μL of RNase-free water through the RNeasy Mini spin column.
For cDNA synthesis, Transcriptor First Strand cDNA Synthesis Kit (Roche Diagnostics, Switzerland) was used. Briefly, 1 μg of total RNA was reverse-transcribed using oligo (dT) primers and incubated for 60 min at 50 °C after denaturation. The reverse transcriptase was then inactivated by 5 min heating at 85 °C.
2.8. Quantitative Polymerase Chain Reaction (qPCR)
qPCR was carried out with a Light Cycler 2.0 (Roche Diagnostics, Basel, Switzerland), QuantiTect SYBR Green Kit (Qiagen, Hilden, Germany) for the detection of genes. The target genes were normalized with glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The amplification conditions consisted of 45 cycles of 94 °C for 15 s, 60 °C for 30 s, and 72 °C for 15 s. The sequences of primers used in qPCR were shown in
supplementary Table S1.
2.9. JAK Inhibition
To confirm adequate quantity of Rux for suppression of JAK in tumor cells, tumor cells were incubated with various dose of Rux. Tumor cells (105 cells/well) were cultured with each cell adopted medium containing 5% FBS in 24-well plates for overnight. Then, the cells were washed with FBS-free medium once, and exchanged the medium containing from 0 to 1.0 μg/mL of ruxolitinib, and incubated for 20 h.
2.10. Cytokine Expression
To monitor cytokine gene expression after tumor stimulation, the separated SMCs from immunized and non-immunized mice were used as effector cells and the tumor cells were used as target cells. Effector cells and target cells were co-cultured at an E:T of 20:1 for 6 h at 37° C. Then, SMCs were lysed with 350 μL of RLT buffer (Each group, n = 5). The samples were extract RNA for determine the gene expression.
2.11. Flow Cytometry
To monitor lymphocyte subsets in SMCs from each groups of mice, the cells were incubated with CD4-FITC mouse (Milteny Biotec, Bergisch Gladbach, Germany) and rat anti-mouse CD8a/Lyt-2-PE (Beckman Coulter, Brea, CA, USA), or FITC anti-mouse CD3 and PE anti-mouse CD19 (Bio Legend, Cromwell, CT, USA) for 30 min at room temperature. The cells were then washed with PBS twice, treated with 0.5% formalin-PBS, and used for flow cytometry analysis with a Coulter Epics XL (Beckman Coulter, Brea, CA, USA).
2.12. Statistical Analyses
Differences between two groups of data were calculated with the Student’s t test. Correlation analyses were performed using the Spearman’s correlation coefficient. Multiple comparison analyses were calculated with the Tukey–Kramer multiple comparison method. Values were regarded as significant at p < 0.05. The analyses were calculated with R version 1.0.143.
4. Discussion
In this study, we investigated the difference of rNDV infectivity in various tumor cell lines, and its influence on rNDV-TV. Then, we examined the anti-tumor response induced by rNDV-TV-Rux as a novel treatment for rNDV infectious tumor cells.
In B16-NDV immunized mice, the cytotoxicity in tumor cells was significantly higher than that in B16-UV immunized mice and the control group (
Figure 1b). In contrast, although there was no significant difference between the cytotoxicity in the SCC VII-NDV and SCC VII-UV immunized groups, the SCC VII-NDV immunized group significantly increased the cytotoxicity of tumor cells compared with the control group (
Figure 1c). Previously, we also confirmed the cytotoxicity of tumor cells with WEHI164-NDV. The WEHI164-NDV group indicated significantly higher cytotoxicity compared with the WEHI164-UV and control groups. Furthermore, the level of cytotoxicity induced by WEHI-NDV was higher than that of immunization with B16-NDV and SCC VII-NDV. These results indicated that rNDV-TV induced an effective anti-tumor response. However, the difference in anti-tumor response, induced by rNDV-TV, was observed between each tumor cell line. Furthermore, it was suggested that it may be difficult for some tumor cell lines to induce sufficient cytotoxicity by NDV, because there was no significant difference between the cytotoxicity induced by SCC VII-NDV and SCC VII-UV.
It has been reported that NDV is more readily infects tumor cells than normal cells [
1]. However, the difference in NDV infectivity between tumor cell lines has not been demonstrated until now. Then, to confirm the difference in susceptibility between tumor cell lines, the rNDV infection rate was examined in six kinds of tumor cell lines. The rNDV infection rate in each tumor cell line was as high as 80% in B16, WEHI 164, and 3LL. However, the infection rate was as low as 35% in SCC VII and MBT-2, and 13% in BALB-MC (
Figure 2). These results indicated the difference in rNDV infectivity between tumor cell lines and suggested that rNDV infectivity might be related to the efficacy of TV-NDV.
It has been reported that the difference in rNDV infectivity between tumor cells and normal cells is responsive to IFN-β [
15], the gene expression of RIG-I, IRF-3, IRF-7, and IFN-β before infection [
16] or the RIG-I gene and the receptor of type I IFN expression [
17]. Therefore, Type I IFN-related gene expression was examined as a factor affecting infectivity of rNDV. Type I IFN-related gene expression, before infection, showed a significant negative correlation with the rNDV infection rate compared with those after infection (
Table 1). Especially, OAS, OASL, Mx gene expression, and one of ISGs, which showed a significant negative correlation with rNDV infection. Consequently, it was suggested that ISG expression before infection affects rNDV susceptibility in tumor cells.
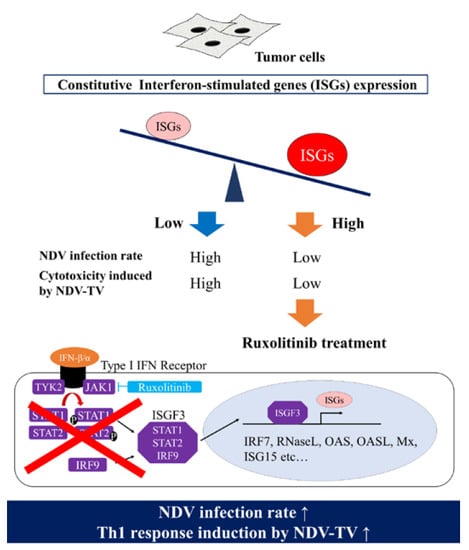
Furthermore, since the rNDV infectivity of the tumor cell lines showed a negative correlation with constitutive ISG expression, it was examined whether suppressing Janus kinases signal transducers, and activators of transcription (JAK-STAT) pathway, improves the rNDV infection rate in tumor cells. The rNDV infection rate was significantly improved in 6 tumor cell lines by Rux treatment suppressing JAK 1 and 2 (
Figure 3). ISG expression after Rux treatment was significantly reduced and showed a significant negative correlation with the rNDV infection rate (
Figure 4). These results revealed that the difference in rNDV infection rate in tumor cell lines was due to the degree of constitutive ISG expression, ISG expression decreased with JAK suppression, and the rNDV infection rate increased. Moreover, the finding that constitutive ISG expression, and the rNDV infection rate, show a negative correlation that might be useful as an indicator for application of viral therapy. This is a potential indicator because it is possible to predict the rNDV susceptibility in advance by confirmation of ISG expression in excised tumor tissue.
MBT-2 showed a low rNDV infection rate (
Figure 2), suppression of ISG expression by Rux treatment (
Figure 4), and an improved infection rate (
Figure 3). Therefore, the immune response induced by rNDV-TV-Rux was examined in MBT-2. In MBT-NDV-Rux immunized mice, the number of CD8
+ cells significantly increased in splenocytes seven days after immunization compared to the control and MBT-NDV groups (
Figure 5b). Previously, we showed that immunized CD8
+ cells are necessary for the anti-tumor response induced by rNDV-TV. The ratio of CD8
+ and CD4
+ cells were significantly increased in the mice transfused with natural killer T (NKT) cell, immunized CD8
+ and CD4
+ cells, and these mice survived compared with the mice transfused with NKT cell, non-immunized CD8
+ and CD4
+ cells [
14]. Therefore, it was suggested that the induction of CD8
+ cell growth by MBT-NDV-Rux might contribute to improvement of the anti-tumor response. Moreover, in IFN-γ gene expression in immunized splenocytes, the MBT-NDV-Rux immunized group significantly increased compared with the MBT-NDV immunized and control groups (
Figure 6). These results suggest that the induction of the Th1 immune response was increased by the improvement of the rNDV susceptibility in tumor cells with Rux treatment. Previous reports indicated that infectious NDV produces more type I IFNs than inactivated NDV [
18]. Type I IFN is considered an important factor in inducing tumor exclusion by rNDV-TV because type I IFN promotes maturation of dendritic cells and activates adaptive and innate immunity [
4,
5]. Virally derived HN protein from viruses expressed on the surface of NDV-infected cells induces antigen-specific cytotoxic activity by cytotoxic T cells (CTL) [
19], and activates NK cells to induce IFN-γ and TNF-α production [
6]. Furthermore, NH protein also induces IFN-α and TNF-α production from peripheral blood mononuclear cells [
20]. Therefore, tumor cell susceptibility to rNDV s is considered a factor that improves rNDV-TV induced immune response.
This study shows that rNDV susceptibility in tumor cells affects the effect of the tumor immunotherapy with rNDV-TV. Moreover, by improving the rNDV susceptibility of tumor cells using the JAK inhibitor ruxolitinib, it was shown that the anti-tumor response induced by rNDV-TV occurs even in tumor cells with low susceptibility. These results suggest that rNDV-TV-Rux can induce an effective antitumor response, even in rNDV low-infectious tumors, and rNDV-TV-Rux may be a new strategy for tumor immune viral therapy.