New Deployable Expandable Electrodes in the Electroporation Treatment in a Pig Model: A Feasibility and Usability Preliminary Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Model
2.2. Electrodes Description
2.3. Operative Electroporation Procedure
2.3.1. Liver Intervention in Laparoscopy or Laparotomy Approach
2.3.2. Endoscopic Trans-Oral and Trans-Anal Approach
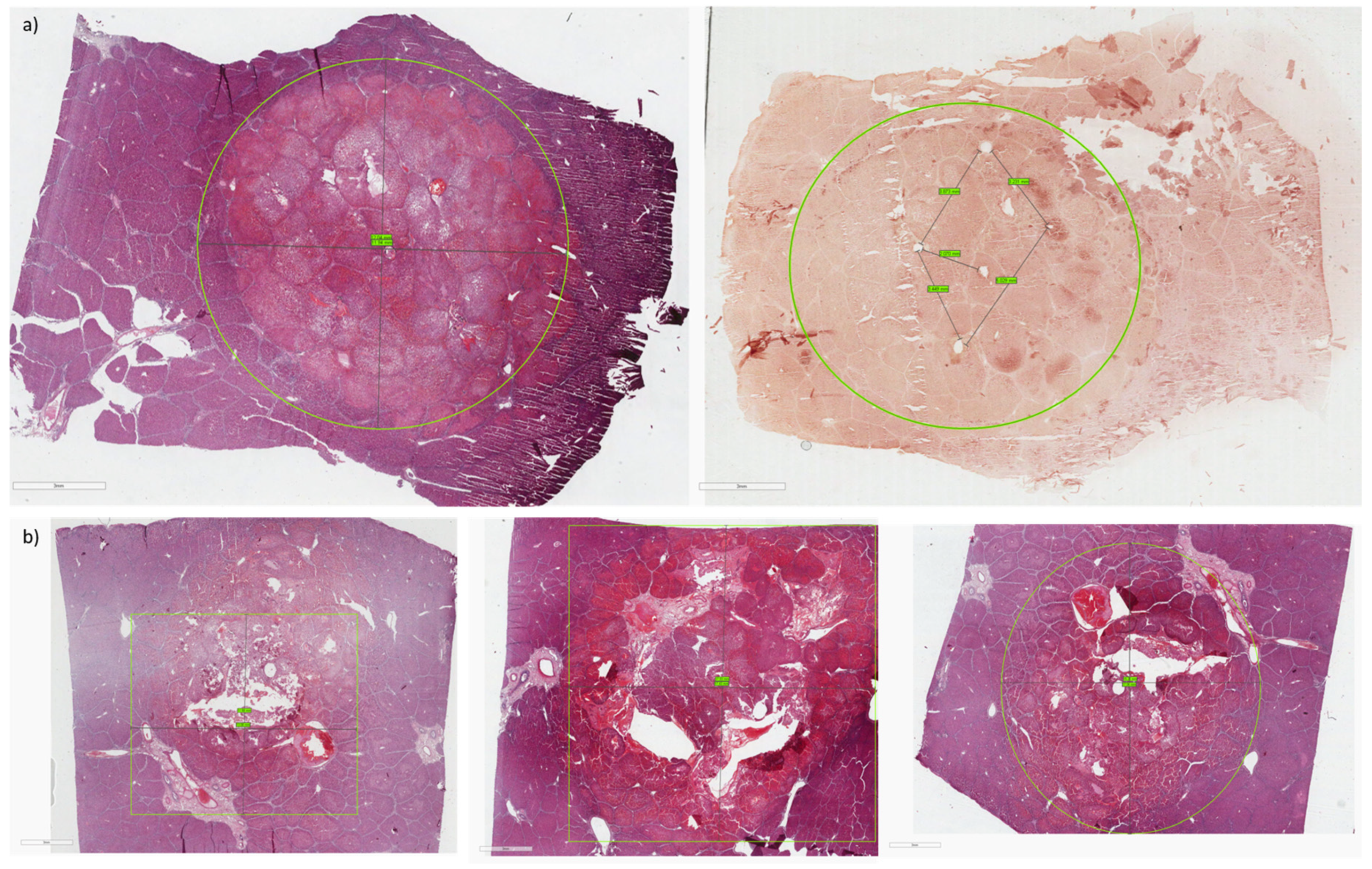
2.4. Imaging Data Analysis
- Calculation of the volume of the smallest ellipsoid containing the ablated area. The ellipsoid is generated by considering, as its axes, the maximum diameters of the area in the 3 orthogonal planes. The volume of the ellipsoid is given by:where a, b, and c are the semi-axes of the ellipsoid (or the radii of the area in the 3 planes).
- Real calculation of the segmented area volume using the CT imaging parameters: total number of pixels contained in the segmented area (npixel), horizontal pixel spacing (PSo), vertical pixel spacing (PSv), space between slice (SBS), slice thickness (ST). By considering the presence of a possible gap (SBS - ST) between one slice and the next, the volume is given by:
2.5. Histological Analysis
2.6. Immunohistochemical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Ethical Approvals
Funding
Acknowledgments
Conflicts of Interest
References
- Cadossi, R.; Ronchetti, M.; Cadossi, M. Locally enhanced chemotherapy by electroporation: Clinical experiences and perspective of use of electrochemotherapy. Future Oncol. 2014, 10, 877–890. [Google Scholar] [CrossRef] [Green Version]
- Sersa, G.; Miklavcic, D.; Cemazar, M.; Rudolf, Z.; Pucihar, G.; Snoj, M. Electrochemotherapy in treatment of tumours. Eur. J. Surg. Oncol. EJSO 2008, 34, 232–240. [Google Scholar] [CrossRef] [Green Version]
- Spratt, D.E.; Gordon Spratt, E.A.; Wu, S.; DeRosa, A.; Lee, N.Y.; Lacouture, M.E.; Barker, C.A. Efficacy of skin-directed therapy for cutaneous metastases from advanced cancer: A meta-analysis. J. Clin. Oncol. 2014, 32, 3144–3155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mali, B.; Jarm, T.; Snoj, M.; Sersa, G.; Miklavcic, D. Antitumor effectiveness of electrochemotherapy: A systematic review and meta-analysis. Eur. J. Surg. Oncol. 2013, 39, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.A.; Davalos, R.V.; Miklavcic, D. A numerical investigation of the electric and thermal cell kill distributions in electroporation-based therapies in tissue. PLoS ONE 2014, 9, e103083. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, F.H.; Cindrič, H.; Kos, B.; Fujimori, M.; Petre, E.N.; Miklavčič, D.; Solomon, S.B.; Srimathveeravalli, G. Peri-tumoral Metallic Implants Reduce the Efficacy of Irreversible Electroporation for the Ablation of Colorectal Liver Metastases. Cardiovasc. Intervent. Radiol. 2019, 43, 3. [Google Scholar] [CrossRef] [PubMed]
- Izzo, F.; Granata, V.; Grassi, R.; Fusco, R.; Palaia, R.; Delrio, P.; Carrafiello, G.; Azoulay, D.; Petrillo, A.; Curley, S.A. Radiofrequency Ablation and Microwave Ablation in Liver Tumors: An Update. Oncologist 2019, 24, 2018–2033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.; Zheng, Y.; He, W.; Zou, R.; Qiu, J.; Shen, J.; Yang, Z.; Zhang, Y.; Wang, C.; Wang, Y.; et al. Microwave vs radiofrequency ablation for hepatocellular carcinoma within the Milan criteria: A propensity score analysis. Aliment. Pharmacol. Ther. 2018, 48, 671–681. [Google Scholar] [CrossRef]
- Edd, J.F.; Horowitz, L.; Davalos, R.V.; Mir, L.M.; Rubinsky, B. In vivo results of a new focal tissue ablation technique: Irreversible electroporation. IEEE Trans Biomed. Eng. 2006, 53, 1409–1415. [Google Scholar] [CrossRef]
- Mir, L.M.; Gehl, J.; Sersa, G.; Collins, G.C.; Garbay, J.R.; Billard, V.; Mir, L.G.; Garbaya, G.R.; Geertsend, P.F.; Gehl, G.; et al. Standard operating procedures of the electrochemotherapy: Instructions for the use of bleomycin or cisplatin administered either systemically or locally and electric pulses delivered by the CliniporatorTM by means of invasive or noninvasive electrodes. Eur. J. Cancer Suppl. 2006, 4, 14–25. [Google Scholar] [CrossRef]
- Marty, M.; Sersa, G.; Garbay, J.R.; Gehl, J.; Collins, G.C.; Snoj, M.; Billard, V.; Geertsen, P.F.J.; Larkin, O.; Miklavcic, D.; et al. Electrochemotherapy—An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. Eur. J. Cancer Suppl. 2006, 4, 3–13. [Google Scholar] [CrossRef]
- Gehl, J.; Sersa, G.; Matthiessen, L.W.; Muir, T.; Soden, D.; Occhini, A.; Quaglino, P.; Curatolo, P.; Campana, L.G.; Kunte, C.; et al. Updated standard operating procedures for electrochemotherapy of cutaneous tumours and skin metastases. Acta Oncol. 2018, 57, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Miklavcic, D.; Snoj, M.; Zupanic, A.; Kos, B.; Cemazar, M.; Kropivnik, M.; Bracko, M.; Pecnik, T.; Gadzijev, E.; Sersa, G. Towards treatment planning and treatment of deep-seated solid tumors by electrochemotherapy. Biomed. Eng. Online 2010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miklavčič, D.; Mali, B.; Kos, B.; Heller, R.; Serša, G. Electrochemotherapy: From the drawing board into medical practice. Biomed. Eng. Online 2014, 13, 29. [Google Scholar] [CrossRef] [Green Version]
- Miklavčič, D.; Serša, G.; Brecelj, E.; Gehl, J.; Soden, D.; Bianchi, G.; Ruggieri, P.; Rossi, C.R.; Campana, L.G.; Jarm, T. Electrochemotherapy: Technological advancements for efficient electroporation-based treatment of internal tumors. Med. Biol. Eng. Comput. 2012, 50, 1213–1225. [Google Scholar]
- Edhemovic, I.; Gadzijev, E.M.; Brecelj, E.; Miklavcic, D.; Kos, B.; Zupanic, A.; Mali, B.; Jarm, T.; Pavliha, D.; Marcan, M.; et al. Electrochemotherapy: A new technological approach in treatment of metastases in the liver. Technol. Cancer Res. Treat. 2011, 10, 475–485. [Google Scholar] [CrossRef] [Green Version]
- Tarantino, L.; Busto, G.; Nasto, A.; Fristachi, R.; Cacace, L.; Talamo, M.; Accardo, C.; Bortone, S.; Gallo, P.; Tarantino, P.; et al. Percutaneous electrochemotherapy in the treatment of portal vein tumor thrombosis at hepatic hilum in patients with hepatocellular carcinoma in cirrhosis: A feasibility study. World J. Gastroenterol. 2017, 23, 906–918. [Google Scholar] [CrossRef]
- Djokic, M.; Djokic, M.; Cemazar, M.; Popovi, P.; Kos, B.; Dezman, R.; Bosnjak, M.; Zakelj, M.N.; Miklavcic, D.; Potrc, S.; et al. Electrochemotherapy as treatment option for hepatocellular carcinoma, a prospective pilot study. Eur. J. Surg. Oncol. 2018, 44, 651–657. [Google Scholar] [CrossRef] [Green Version]
- Cornelis, F.H.; Korenbaum, C.; Ben Ammar, M.; Tavolaro, S.; Nouri-Neuville, M.; Lotz, J.P. Multimodal image-guided electrochemotherapy of unresectable liver metastasis from renal cell cancer. Diagn. Interv. Imaging 2019, 100, 309–311. [Google Scholar] [CrossRef]
- Scala, D.; Rega, D.; Ruffolo, F.; Pace, U.; Sassaroli, C.; Cardone, E.; Grimaldi, A.M.; Caraco, C.; Mozzillo, N.; Delrio, P. Electrochemotherapy for rectal cancer after neoadjuvant radiotherapy: A case report. Eur. J. Surg. Oncol. 2015, 41, S13–S14. [Google Scholar] [CrossRef]
- Bourke, M.; Salwa, S.; Forde, P.F.; Sadadcheam, M.; Karkin, J.O.; Collins, C.; Zeesham, S.; Winter, D.; O’Sullivan, G.C.; Doden, G.M.; et al. P80 Endoscopically targeted electrochemotherapy for the treatment of colorectal cancer. Eur. J.Surg. Oncol. 2012, 38, 1127–1128. [Google Scholar] [CrossRef]
- Endoscopic Assisted Electrochemotherapy in Addition to Neoadjuvant Treatment of Locally Advanced Rectal Cancer (nECT). Available online: https://clinicaltrials.gov/ct2/show/NCT03040180 (accessed on 21 January 2019).
- Martin, R.C., 2nd; Kwon, D.; Chalikonda, S.; Sellers, M.; Kotz, E.; Scoggins, C.; McMasters, K.M.; Watkins, K. Treatment of 200 locally advanced (stage III) pancreatic adenocarcinoma patients with irreversible electroporation: Safety and efficacy. Ann. Surg. 2015, 262, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Bimonte, S.; Leongito, M.; Granata, V.; Barbieri, A.; Del Vecchio, V.; Falco, M.; Nasto, A.; Albino, V.; Piccirillo, M.; Palaia, R.; et al. Electrochemotherapy in pancreatic adenocarcinoma treatment: Pre-clinical and clinical studies. Radiol. Oncol. 2016, 50, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Tafuto, S.; von Arx, C.; De Divitiis, C.; Maura, C.T.; Palaia, R.; Albino, V.; Fusco, R.; Membrini, M.; Petrillo, A.; Granata, V.; et al. ENETS Center of Excellence Multidisciplinary Group for Neuroendocrine Tumors in Naples (Italy). Electrochemotherapy as a new approach on pancreatic cancer and on liver metastases. Int. J. Surg. Suppl. 2015, 1, S78–S82. [Google Scholar] [CrossRef]
- Girelli, R.; Prejanò, S.; Cataldo, I.; Corbo, V.; Martini, L.; Scarpa, A.; Claudio, B. Feasibility and safety of electrochemotherapy (ECT) in the pancreas: A pre-clinical investigation. Radiol. Oncol. 2015, 49, 147–154. [Google Scholar] [CrossRef] [Green Version]
- Granata, V.; Fusco, R.; Piccirillo, M.; Palaia, R.; Petrillo, A.; Lastoria, S.; Izzo, F. Electrochemotherapy in locally advanced pancreatic cancer: Preliminary results. Int. J. Surg. 2015, 18, 230–236. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Setola, S.V.; Piccirillo, M.; Leongito, M.; Palaia, R.; Granata, F.; Lastoria, S.; Izzo, F.; Petrillo, A. Early radiological assessment of locally advanced pancreatic cancer treated with electrochemotherapy. World J. Gastroenterol. 2017, 23, 4767–4778. [Google Scholar] [CrossRef]
- Bianchi, G.; Campanacci, L.; Ronchetti, M.; Donati, D. Electrochemotherapy in the Treatment of Bone Metastases: A Phase II Trial. World J. Surg. 2016, 40, 3088–3094. [Google Scholar] [CrossRef] [Green Version]
- Ritter, A.; Bruners, P.; Isfort, P.; Barabasch, A.; Pfeffer, J.; Schmitz, J.; Pedersoli, F.; Baumann, M. Electroporation of the Liver: More Than 2 Concurrently Active, Curved Electrodes Allow New Concepts for Irreversible Electroporation and Electrochemotherapy. Technol. Cancer Res. Treat. 2018. [Google Scholar] [CrossRef] [Green Version]
- Ueshima, E.; Schattner, M.; Mendelsohn, R.; Gerdes, H.; Monette, S.; Takaki, H.; Durack, J.C.; Solomon, S.B.; Srimathveeravalli, G. Transmural ablation of the normal porcine common bile duct with catheter-directed irreversible electroporation is feasible and does not affect duct patency. Gastrointest Endosc. 2018, 87, 300.e1–300.e6. [Google Scholar] [CrossRef]
- Kodama, H.; Vroomen, L.G.; Ueshima, E.; Reilly, J.; Brandt, W.; Paluch, L.R.; Monette, S.; Jones, D.; Solomon, S.B.; Srimathveeravalli, G. Catheter-based endobronchial electroporation is feasible for the focal treatment of peribronchial tumors. J. Thorac. Cardiovasc. Surg. 2018, 155, 2150–2159.e3. [Google Scholar] [CrossRef] [PubMed]
- Agerholm-Larsen, B.; Iversen, H.K.; Ibsen, P.; Moller, J.M.; Mahmood, F.; Jensen, K.S.; Gehl, J. Preclinical validation of electrochemotherapy as an effective treatment for brain tumors. Cancer Res. 2011, 71, 3753–3762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahmood, F.; Gehl, J. Optimizing clinical performance and geometrical robustness of a new electrode device for intracranial tumor electroporation. Bioelectrochemistry 2011, 81, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Linnert, M.; Iversen, H.K.; Gehl, J. Multiple brain metastases—Current management and perspectives for treatment with electrochemotherapy. Radiol. Oncol. 2012, 46, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Soden, D.; Forde, P.; Sadadcharam, M.; Bourke, M.; O’Sullivan, G.C. Endoscopic Electroporation For Colorectal Cancer: Enhancing Drug Absorption. Available online: https://www.sages.org/meetings/annual-meeting/abstracts-archive/endoscopic-electroporation-for-colorectal-cancer-enhancing-drug-absorption/ (accessed on 22 June 2019).
- Miklavčič, D.; Corovic, S.; Pucihar, G.; Pavselj, N. Importance of tumour coverage by sufficiently high local electric field for effective electrochemotherapy. Eur. J. Cancer Suppl. 2006, 4, 45–51. [Google Scholar] [CrossRef]
- Haemmerich, D.; Schutt, D.; Wright, A.; Webster, J.; Mahvi, D. Electrical conductivity measurement of excised human metastatic liver tumours before and after thermal ablation. Physiol. Meas. 2009, 30, 459–466. [Google Scholar] [CrossRef] [Green Version]
- Peyman, A.; Kos, B.; Djokić, M.; Trotovšek, B.; Limbaeck-Stokin, C.; Serša, G.; Miklavčič, D. Variation in dielectric properties due to pathological changes in human liver. Bioelectromagnetics 2015, 36, 603–612. [Google Scholar] [CrossRef]
- Muftuler, L.T.; Hamamura, M.; Birgul, O.; Nalcioglu, O. Resolution and Contrast in Magnetic Resonance Electrical Impedance Tomography (MREIT) and its Application to Cancer Imaging. Technol. Cancer Res. Treat. 2004, 3, 599–609. [Google Scholar] [CrossRef] [Green Version]
- .Probst, U.; Fuhrmann, I.; Beyer, L.; Wiggermann, P. Electrochemotherapy as a New Modality in Interventional Oncology: A Review. Technol. Cancer Res. Treat. 2018. [Google Scholar] [CrossRef] [Green Version]
- Laudicella, M.; Walsh, B.; Munasinghe, A.; Faiz, O. Impact of laparoscopic versus open surgery on hospital costs for colon cancer: A population-based retrospective cohort study. BMJ Open 2016, 6, e012977. [Google Scholar] [CrossRef]
- Buia, A.; Stockhausen, F.; Hanisch, E. Laparoscopic surgery: A qualified systematic review. World J. Methodol. 2015, 5, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Dapri, G. 10-Year Experience with 1700 Single-Incision Laparoscopies. Surg. Technol. Int. 2019, 35, 71–83. [Google Scholar] [PubMed]
- Zhang, H.; Feng, Y.; Zhao, J.; Chen, R.; Chen, X.; Yin, X.; Cheng, W.; Li, D.; Li, J.; Huang, X.; et al. Total laparoscopic pancreaticoduodenectomy versus open pancreaticoduodenectomy (TJDBPS01): Study protocol for a multicentre, randomised controlled clinical trial. BMJ Open 2020, 10, e033490. [Google Scholar] [CrossRef] [PubMed]
- Pichi, B.; Pellini, R.; De Virgilio, A.; Spriano, G. Electrochemotherapy: A well accepted palliative treatment by patients with head and neck tumours. Acta Otorhinolaryngol. Italica 2018, 38, 181–187. [Google Scholar]
- Jarm, T.; Cemazar, M.; Miklavcic, D.; Sersa, G. Antivascular effects of electrochemotherapy: Implications in treatment of bleeding metastases. Expert. Rev. Anticancer Ther. 2010, 10, 729–746. [Google Scholar] [CrossRef]
- Kanas, G.P.; Taylor, A.; Primrose, J.N.; Langeberg, W.J.; Kelsh, M.A.; Mowat, F.S.; Alexander, D.D.; Choti, M.A.; Poston, G. Survival after liver resection in metastatic colorectal cancer: Review and meta-analysis of prognostic factors. Clin. Epidemiol. 2012, 4, 283–301. [Google Scholar]
- Wong, H.H.; Lemoine, N.R. Novel therapies for pancreatic cancer: Setbacks and progress. Future Oncol. 2010, 6, 1061–1064. [Google Scholar] [CrossRef] [Green Version]
- Wandel, A.; Ben-David, E.; Ulusoy, B.S.; Neal, R.; Faruja, M.; Nissenbaum, I.; Gourovich, S.; Goldberg, S.N. Optimizing Irreversible Electroporation Ablation with a Bipolar Electrode. J. Vasc. Interv. Radiol. 2016, 27, 1441–1450.e2. [Google Scholar] [CrossRef]

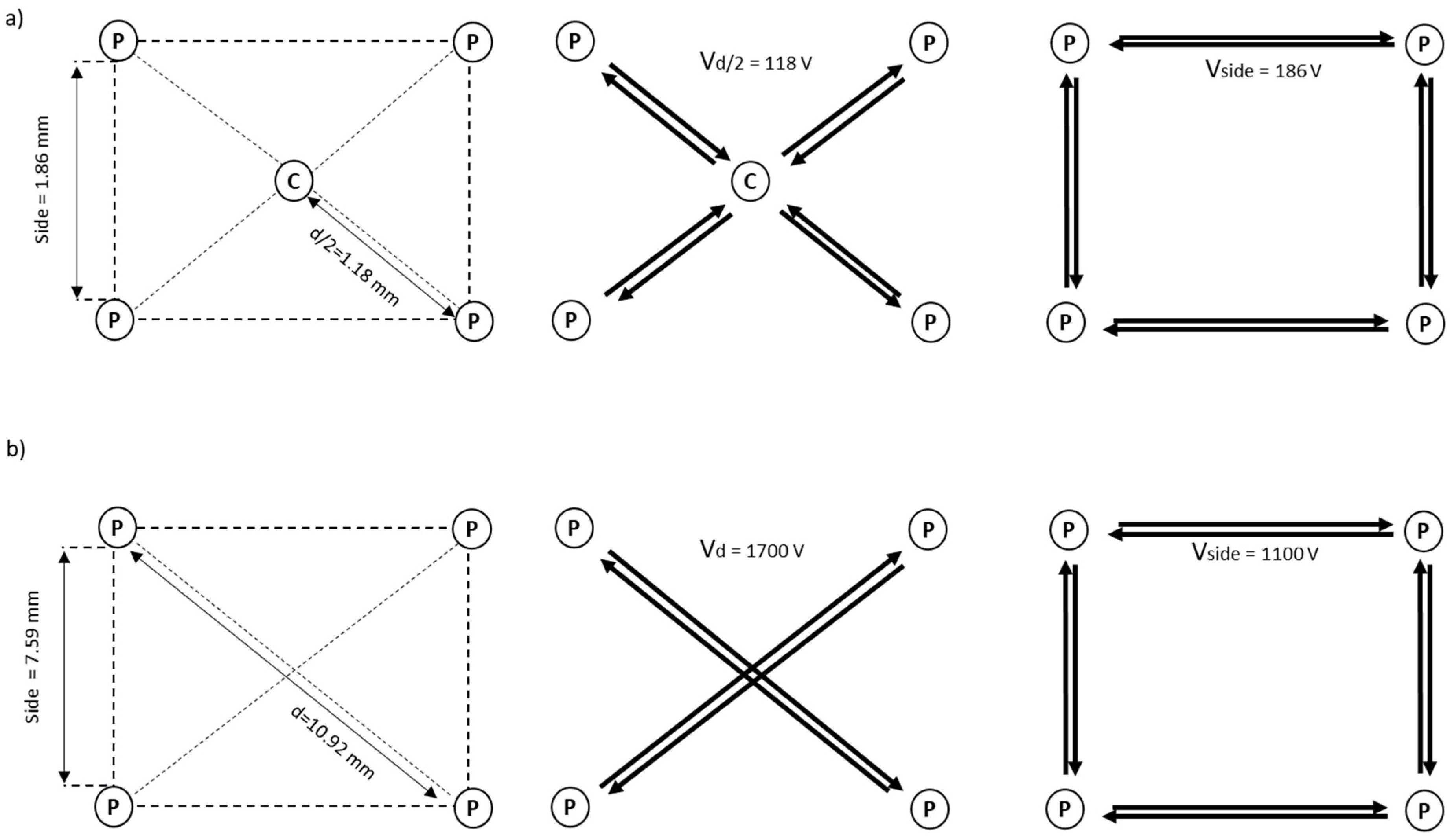






| Electrode Model | Geometry Configuration | Electric Parameters | |||
|---|---|---|---|---|---|
| Expandable electrode prototype with 5 needles with zero divergence | |||||
| Ø shaft [mm] | 5 | Side [mm] | 1.86 | Vside [V] | 186 |
| Shaft Length [cm] | 50 | Semi-diagonal d/2 [mm] | 1.18 | Vd/2 [V] | 118 |
| Divergence [°] | 0 | Pulses Number | 80 | ||
| Maximum Deployment [cm] | 4 | ||||
| Active Part [cm] | 2 | ||||
| Expandable electrode prototype with 5 needles with non-zero divergence | |||||
| Ø shaft [mm] | 5 | Deployments | S2, S3 * | Vside (S2) [V] | 1200 |
| Shaft Length [cm] | 20 | Side–d/2 (S2) [mm] | 13.69–9.55 | Vd/2 (S2) [V] | 900 |
| Divergence [°] | 20 | Side–d/2 (S3) [mm] | 18.84–13.19 | Vside (S3)[V] | 1700 |
| Deployment [cm] | 4 | Vd/2 (S3) [V] | 1100 | ||
| Active Part [cm] | 2 | Pulses Number | 120 | ||
| Expandable electrode prototype with 4 needles with non-zero divergence | |||||
| Ø shaft [mm] | 5 | Deployments | S2, S3, max * | Vside (S2) [V] | 1100 |
| Shaft Length [cm] | 20 | Side–d (S2) [mm] | 7.59–10.92 | Vd (S2) [V] | 1700 |
| Divergence [°] | 10 | Side–d (S3) [mm] | 10.08–14.44 | Vside (S3)[V] | 1500 |
| Deployment [cm] | 4 | Side–d (max) [mm] | 12.58–17.98 | Vd (S3) [V] | 2200 |
| Active Part [cm] | 2 | Vside (max) [V] | 1900 | ||
| Vd (max) [V] | 2700 | ||||
| Pulses Number | 80 | ||||
| 2a [cm] | 2b [cm] | 2c [cm] | Vellipsoid [cm3] | npixel | VCT [cm3] | |
|---|---|---|---|---|---|---|
| Expandable electrode prototype with 5 needles and zero divergence | 2.05 | 1.53 | 1.77 | 2.91 | 990 | 2.38 |
| Expandable electrode prototype with 4 needles and a 10° divergence | 2.6 | 2.14 | 2.14 | 6.23 | 2911 | 6.99 |
| Expandable electrode prototype with 5 needles and a 20° divergence | 3.04 | 2.49 | 2.57 | 10.19 | 4218 | 10.13 |
| Liver District | Oral Cavity | Anal Cavity | |
|---|---|---|---|
| Usability | Suitable | Suitable | Suitable |
| Appropriateness of the handle | Suggested a greater tactile feedback to better monitor the needle advancement even by counting the deployments. | Suggested modification of the positioning of the graduated scale relative to the movement of central needle cursor (left cursor). It would be preferable to have the zero position at the upper end of the cursor (i.e., the entire blue element). Suggested a greater tactile feedback to better monitor the needle advancement even by counting the deployments. | Proper handle. It is possible to exploit the shape of the lower half-shell to prepare the electrode (post-insertion) and maneuver it with greater precision. |
| Adequacy of the handling system (cursors) | Excessive sliding of central needle cursor. Suggested greater friction, in order to increase the precision during the movement of the central needle. | Excessive sliding of central needle cursor. Suggested greater friction, in order to increase the precision during the movement of the central needle. | Excessive sliding of central needle cursor. Suggested greater friction, in order to increase the precision during the movement of the central needle. |
| Flexibility and shape of the shaft | Adapted. | Adapted to the anatomy of the oral cavity. The not excessive flexibility of the cannula allows a better holding of the shape given to the shaft. | Adapted to the anatomy of anal cavity. |
| Needles penetration | No critical issues emerged. | No critical issues emerged. | No critical issues emerged. |
| Electrode compatibility with laparoscopic trocar | The electrode is compatible with the 5 mm laparoscopic trocar. Not recommended the use of the electrode with laparoscopic trocar with larger diameter. The shaft would have excessive freedom of movement, thus reducing precision during use. | Not evaluated. | The electrode is compatible with the 5 mm single-port trocar. Not recommended the use of the electrode with trocar with larger diameter. The shaft would have excessive freedom of movement, thus reducing precision during use. |
| Electrode compatibility with clamps | The laparoscopic forceps used showed that it is possible to grasp the shaft and move it with adequate stability. Compatible with surgical instruments to imprint an angle to the shaft. | Compatible with surgical instruments to imprint an angle to the shaft. | The laparoscopic forceps used showed that it is possible to grasp the shaft and move it with adequate stability. To shape the shaft using pliers it is necessary that these have a curved shape, to prepare the electrode along the orthogonal direction. |
| Criticalities found | None. | None. | None. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izzo, F.; Ionna, F.; Granata, V.; Albino, V.; Patrone, R.; Longo, F.; Guida, A.; Delrio, P.; Rega, D.; Scala, D.; et al. New Deployable Expandable Electrodes in the Electroporation Treatment in a Pig Model: A Feasibility and Usability Preliminary Study. Cancers 2020, 12, 515. https://doi.org/10.3390/cancers12020515
Izzo F, Ionna F, Granata V, Albino V, Patrone R, Longo F, Guida A, Delrio P, Rega D, Scala D, et al. New Deployable Expandable Electrodes in the Electroporation Treatment in a Pig Model: A Feasibility and Usability Preliminary Study. Cancers. 2020; 12(2):515. https://doi.org/10.3390/cancers12020515
Chicago/Turabian StyleIzzo, Francesco, Franco Ionna, Vincenza Granata, Vittorio Albino, Renato Patrone, Francesco Longo, Agostino Guida, Paolo Delrio, Daniela Rega, Dario Scala, and et al. 2020. "New Deployable Expandable Electrodes in the Electroporation Treatment in a Pig Model: A Feasibility and Usability Preliminary Study" Cancers 12, no. 2: 515. https://doi.org/10.3390/cancers12020515







