Photogenerated Carrier-Assisted Electrocatalysts for Efficient Water Splitting
Abstract
:1. Introduction
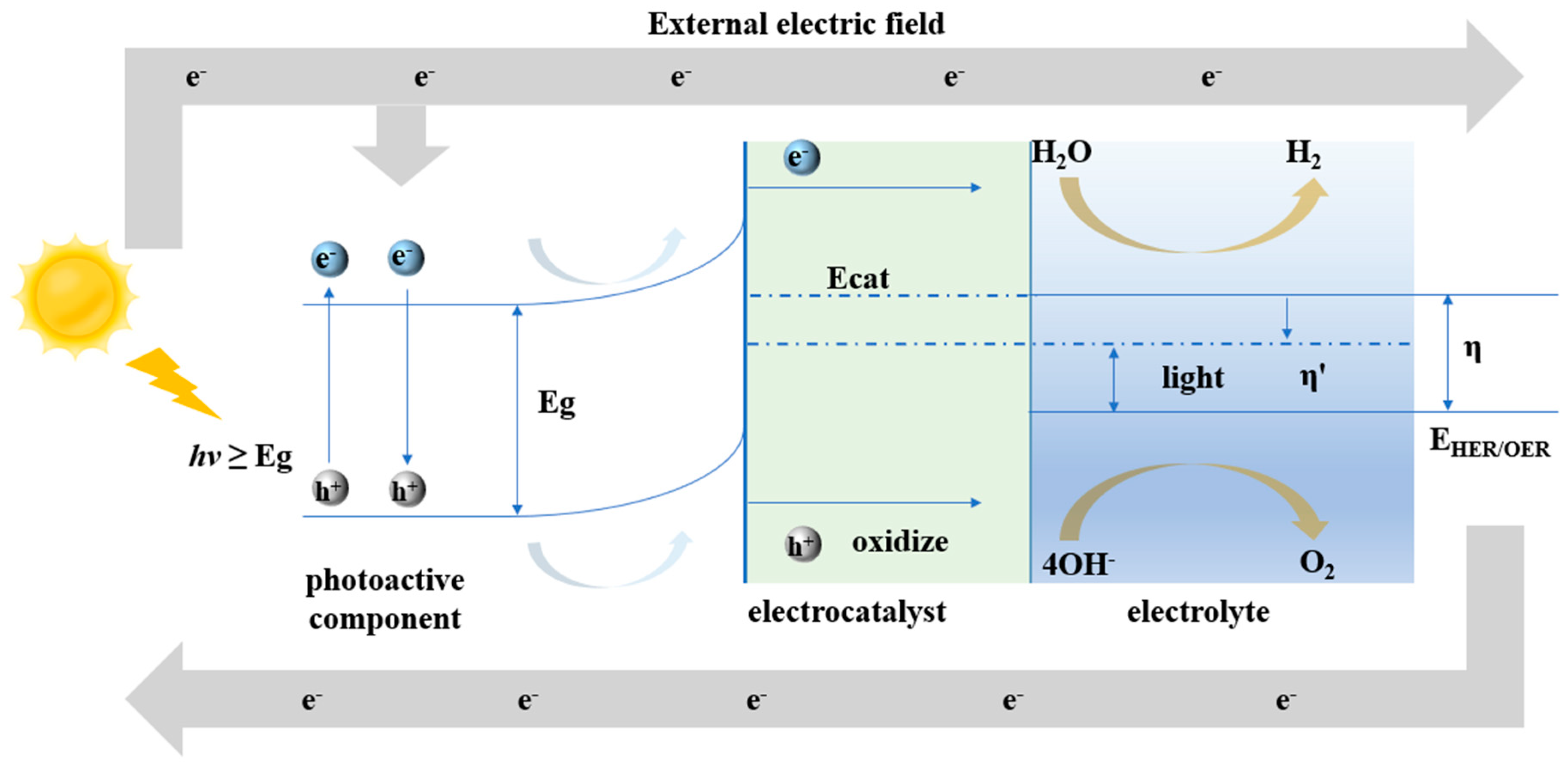
2. Fundamental Principle of Photo-Assisted Electrocatalysis Strategies
3. Photo-Electro Integrated Catalysts
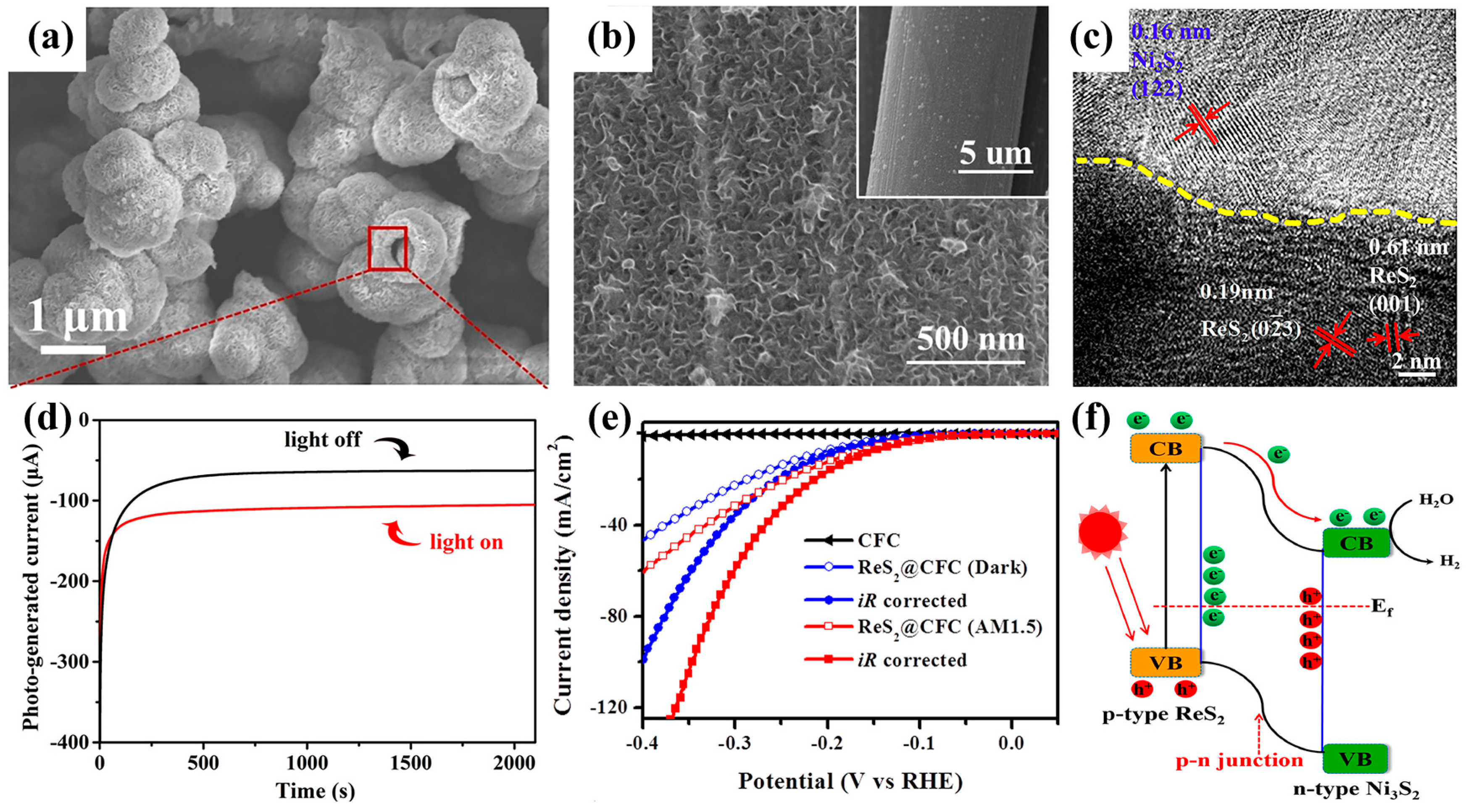
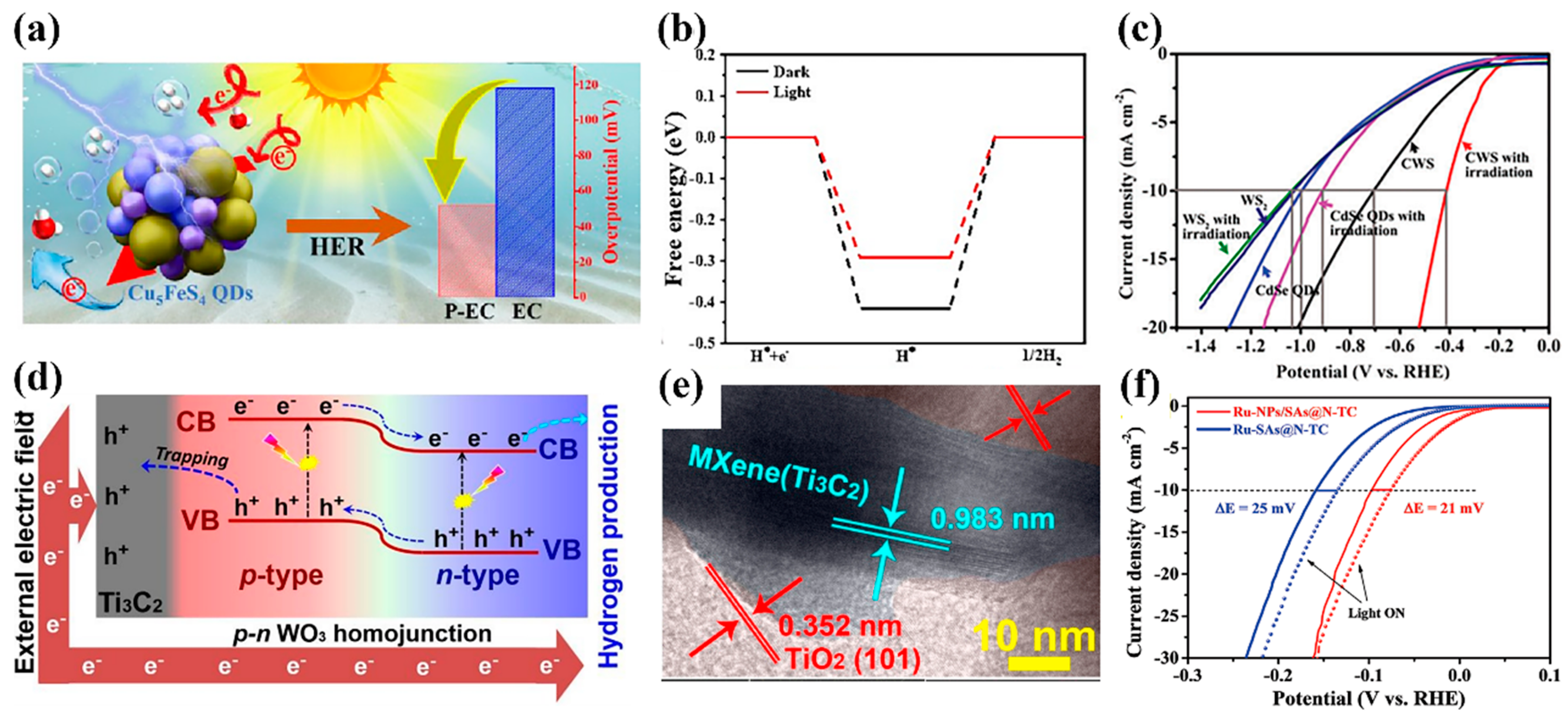
3.1. Photogenerated Carrier-Assisted Electrocatalysts for HER
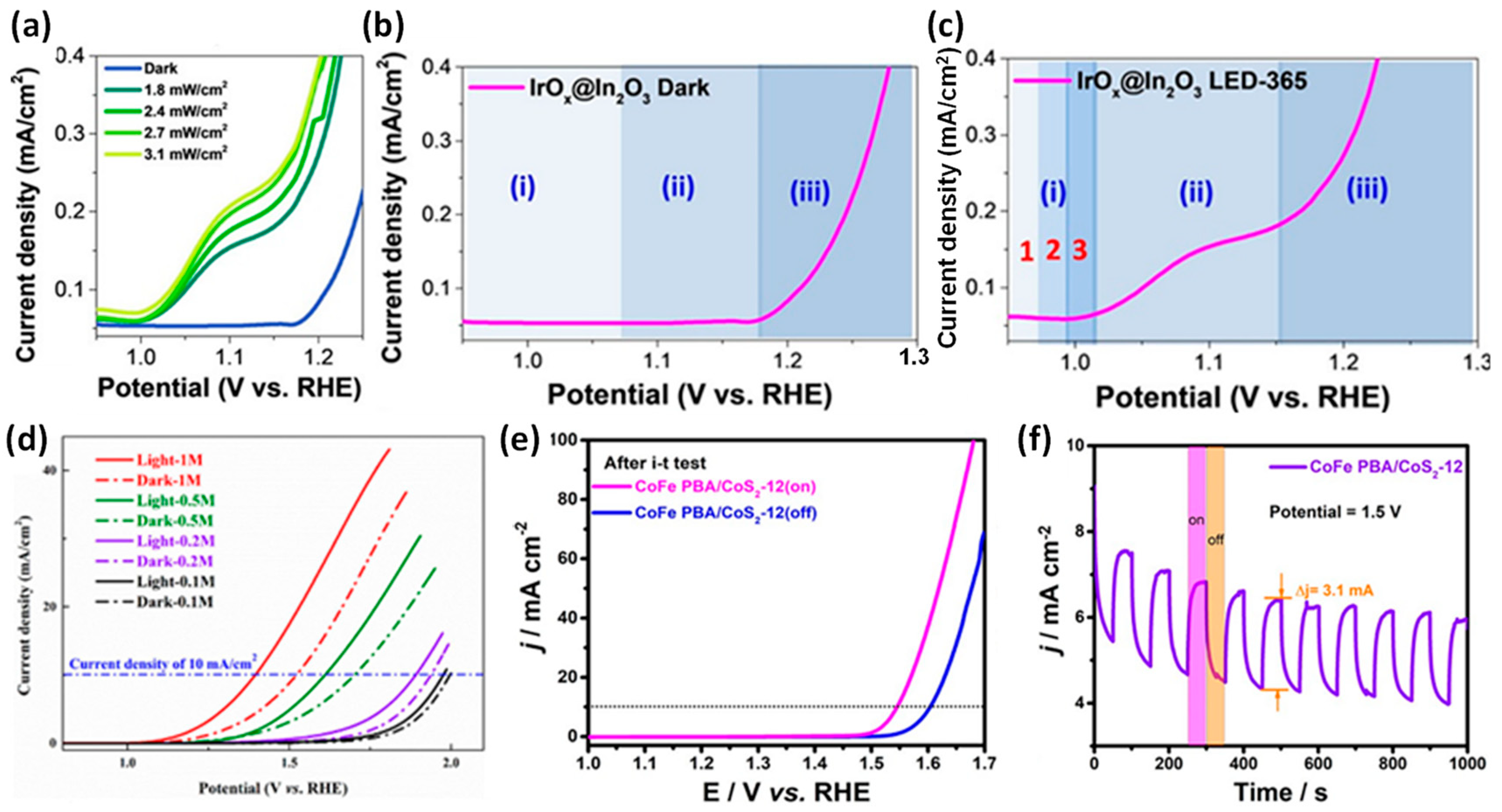
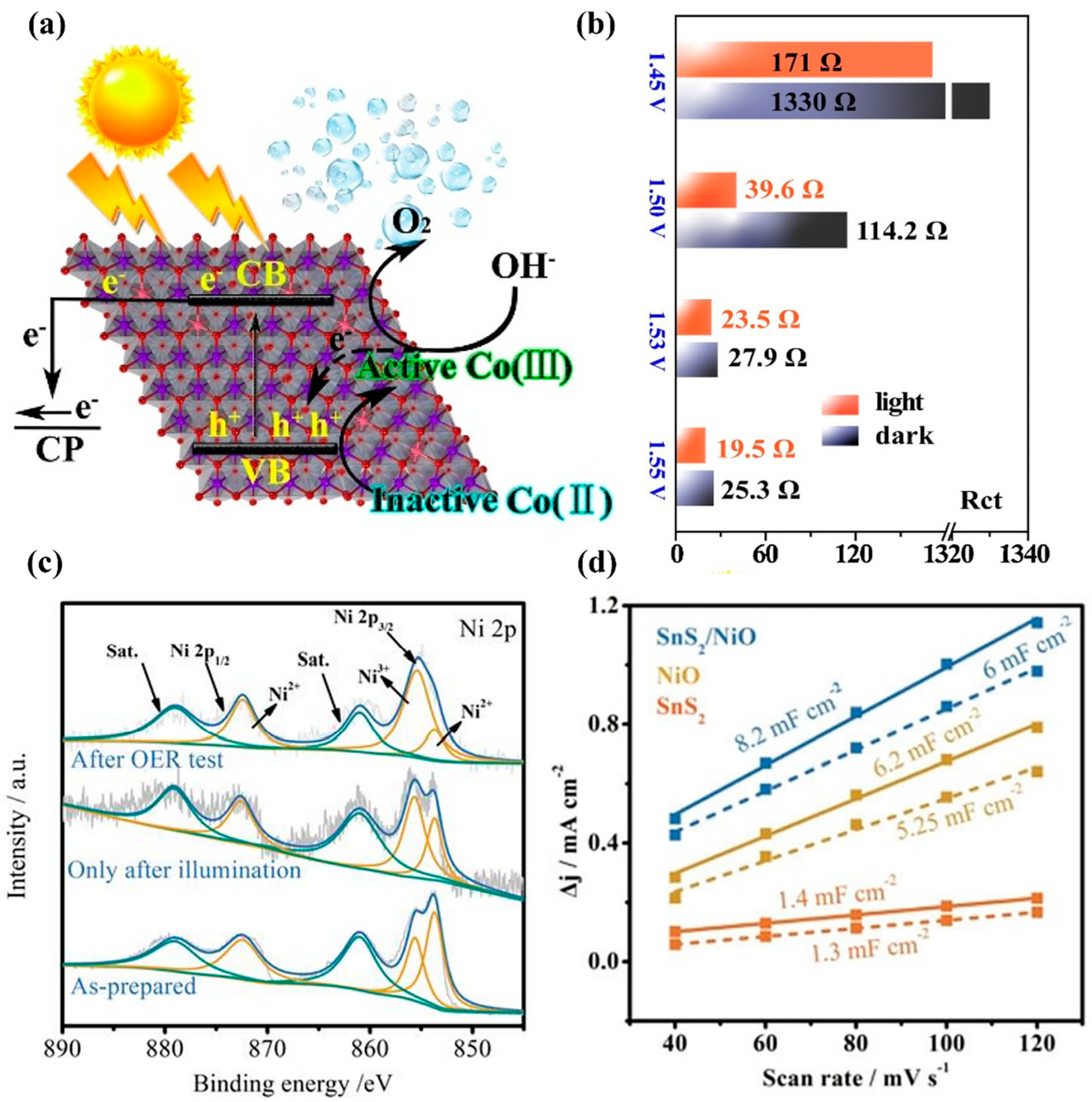
3.2. Photogenerated Carrier-Assisted Electrocatalysts for OER
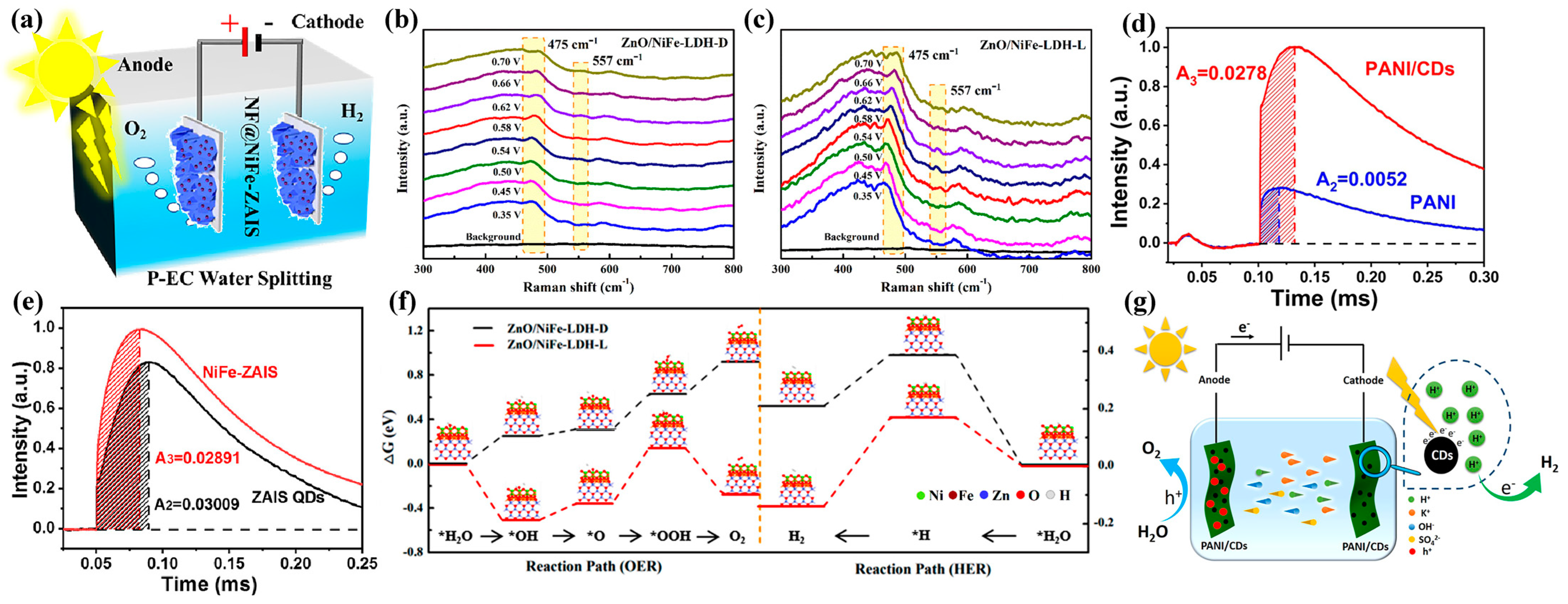
3.3. Photogenerated Carrier-Assisted Electrocatalysts for Overall Water Splitting
4. Conclusions and Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, J.; Hu, J.; Liang, C.; Chang, L.; Du, Y.; Han, X.; Sun, J.; Xu, P. Surface reconstruction of phosphorus-doped cobalt molybdate microarrays in electrochemical water splitting. Chem. Eng. J. 2022, 446, 137094. [Google Scholar] [CrossRef]
- Wang, W.; Xu, X.; Zhou, W.; Shao, Z. Recent Progress in Metal-Organic Frameworks for Applications in Electrocatalytic and Photocatalytic Water Splitting. Adv. Sci. 2017, 4, 1600371. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, L.; Yu, J.; Liu, X.; Zhang, X.; Liu, H.; Zhou, W. Water Splitting: From Electrode to Green Energy System. Nano-Micro Lett. 2020, 12, 131. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Shao, Z.; Jiang, S.P. High-Entropy Materials for Water Electrolysis. Energy Technol. 2022, 10, 2200573. [Google Scholar] [CrossRef]
- You, B.; Sun, Y. Innovative Strategies for Electrocatalytic Water Splitting. Acc. Chem. Res. 2018, 51, 1571–1580. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wei, C.; Zhu, Y.; Huang, H. Emerging chemical driving force in electrocatalytic water splitting. EcoMat 2023, 5, e12294. [Google Scholar] [CrossRef]
- Wang, J.; Gao, Y.; Kong, H.; Kim, J.; Choi, S.; Ciucci, F.; Hao, Y.; Yang, S.; Shao, Z.; Lim, J. Non-precious-metal catalysts for alkaline water electrolysis: Operando characterizations, theoretical calculations, and recent advances. Chem. Soc. Rev. 2020, 49, 9154–9196. [Google Scholar] [CrossRef]
- Wang, Q.; He, R.; Yang, F.; Tian, X.; Sui, H.; Feng, L. An overview of heteroatom doped cobalt phosphide for efficient electrochemical water splitting. Chem. Eng. J. 2023, 456, 141056. [Google Scholar] [CrossRef]
- Yu, Z.-Y.; Duan, Y.; Feng, X.-Y.; Yu, X.; Gao, M.-R.; Yu, S.-H. Clean and Affordable Hydrogen Fuel from Alkaline Water Splitting: Past, Recent Progress, and Future Prospects. Adv. Mater. 2021, 33, 2007100. [Google Scholar] [CrossRef]
- Wang, S.; He, T.; Yun, J.-H.; Hu, Y.; Xiao, M.; Du, A.; Wang, L. New iron-cobalt oxide catalysts promoting BiVO4 films for photoelectrochemical water splitting. Adv. Funct. Mater. 2018, 28, 1802685. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Zhang, M.; Zhou, Y.; Rao, D.; Zhong, C.; Zhang, J.; Han, X.; Hu, W.; Zhang, Y.; et al. Lattice-Strain Engineering of Homogeneous NiS0.5Se0.5 Core-Shell Nanostructure as a Highly Efficient and Robust Electrocatalyst for Overall Water Splitting. Adv. Mater. 2020, 32, e2000231. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xu, G.; Yang, H.; Wang, H.; Xia, B.Y. Rational Design of Transition Metal Phosphide-Based Electrocatalysts for Hydrogen Evolution. Adv. Funct. Mater. 2023, 33, 2208358. [Google Scholar] [CrossRef]
- Abdelghafar, F.; Xu, X.; Jiang, S.P.; Shao, Z. Designing single-atom catalysts toward improved alkaline hydrogen evolution reaction. Mater. Rep. Energy 2022, 2, 100144. [Google Scholar] [CrossRef]
- Duan, Y.; Lee, J.Y.; Xi, S.; Sun, Y.; Ge, J.; Ong, S.J.H.; Chen, Y.; Dou, S.; Meng, F.; Diao, C.; et al. Anodic Oxidation Enabled Cation Leaching for Promoting Surface Reconstruction in Water Oxidation. Angew. Chem. Int. Ed. Engl. 2021, 60, 7418–7425. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Mohamed, A.G.A.; Xie, J.; Wang, Y. Surface evolution of electrocatalysts in energy conversion reactions. Nano Energy 2021, 82, 105745. [Google Scholar] [CrossRef]
- Qin, Y.; Wang, Z.; Yu, W.; Sun, Y.; Wang, D.; Lai, J.; Guo, S.; Wang, L. High Valence M-Incorporated PdCu Nanoparticles (M = Ir, Rh, Ru) for Water Electrolysis in Alkaline Solution. Nano Lett. 2021, 21, 5774–5781. [Google Scholar] [CrossRef]
- Xiong, L.; Qiu, Y.; Peng, X.; Liu, Z.; Chu, P.K. Electronic structural engineering of transition metal-based electrocatalysts for the hydrogen evolution reaction. Nano Energy 2022, 104, 107882. [Google Scholar] [CrossRef]
- Yue, L.; Wang, Z.; Wang, D.; Song, W.; Wu, Z.; Zhao, W.; Zhang, L.; Luo, Y.; Sun, S.; Zheng, D.; et al. Aliovalent doping engineering enables multiple modulations of FeS2 anodes to achieve fast and durable sodium storage. J. Mater. Chem. A 2022, 10, 21149–21160. [Google Scholar] [CrossRef]
- Dou, Y.; Yuan, D.; Yu, L.; Zhang, W.; Zhang, L.; Fan, K.; Al-Mamun, M.; Liu, P.; He, C.T.; Zhao, H. Interpolation between W Dopant and Co Vacancy in CoOOH for Enhanced Oxygen Evolution Catalysis. Adv. Mater. 2022, 34, e2104667. [Google Scholar] [CrossRef]
- Li, Z.; Hu, M.; Wang, P.; Liu, J.; Yao, J.; Li, C. Heterojunction catalyst in electrocatalytic water splitting. Coord. Chem. Rev. 2021, 439, 213953. [Google Scholar] [CrossRef]
- Gu, M.; Jiang, L.; Zhao, S.; Wang, H.; Lin, M.; Deng, X.; Huang, X.; Gao, A.; Liu, X.; Sun, P.; et al. Deciphering the Space Charge Effect of the p-n Junction between Copper Sulfides and Molybdenum Selenides for Efficient Water Electrolysis in a Wide pH Range. ACS Nano 2022, 16, 15425–15439. [Google Scholar] [CrossRef]
- Xu, X.; Pan, Y.; Ge, L.; Chen, Y.; Mao, X.; Guan, D.; Li, M.; Zhong, Y.; Hu, Z.; Peterson, V.K.; et al. High-performance perovskite composite electrocatalysts enabled by controllable interface engineering. Small 2021, 17, 2101573. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liang, T.; Kong, D.; Wang, B.; Zhi, L. Strain engineering of two-dimensional materials for advanced electrocatalysts. Mater. Today. Nano 2021, 14, 100111. [Google Scholar] [CrossRef]
- Du, X.; Huang, J.; Zhang, J.; Yan, Y.; Wu, C.; Hu, Y.; Yan, C.; Lei, T.; Chen, W.; Fan, C.; et al. Modulating Electronic Structures of Inorganic Nanomaterials for Efficient Electrocatalytic Water Splitting. Angew. Chem. Int. Ed. 2019, 58, 4484–4502. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Yu, R.; Wang, P.; Pan, X.; Jin, H.; Zheng, D.; Chen, D.; Zhu, J.; Pu, Z.; Wu, J.; et al. Ultra-Fast and In-Depth Reconstruction of Transition Metal Fluorides in Electrocatalytic Hydrogen Evolution Processes. Adv. Sci. 2022, 9, 2103567. [Google Scholar] [CrossRef]
- Zhou, L.; Yang, C.; Zhu, W.; Li, R.; Pang, X.; Zhen, Y.; Wang, C.; Gao, L.; Fu, F.; Gao, Z.; et al. Boosting Alkaline Hydrogen Evolution Reaction via an Unexpected Dynamic Evolution of Molybdenum and Selenium on MoSe2 Electrode. Adv. Energy Mater. 2022, 12, 2202367. [Google Scholar] [CrossRef]
- Lin, R.; Kang, L.; Zhao, T.; Feng, J.; Celorrio, V.; Zhang, G.; Cibin, G.; Kucernak, A.; Brett, D.J.L.; Corà, F.; et al. Identification and manipulation of dynamic active site deficiency-induced competing reactions in electrocatalytic oxidation processes. Energy Environ. Sci. 2022, 15, 2386–2396. [Google Scholar] [CrossRef]
- Liu, D.; Yan, Y.; Li, H.; Liu, D.; Yang, Y.; Li, T.; Du, Y.; Yan, S.; Yu, T.; Zhou, W.; et al. A Template Editing Strategy to Create Interlayer-Confined Active Species for Efficient and Durable Oxygen Evolution Reaction. Adv. Mater. 2023, 35, 2203420. [Google Scholar] [CrossRef]
- Jin, B.; Li, Y.; Wang, J.; Meng, F.; Cao, S.; He, B.; Jia, S.; Wang, Y.; Li, Z.; Liu, X. Promoting Oxygen Evolution Reaction of Co-Based Catalysts (Co3O4, CoS, CoP, and CoN) through Photothermal Effect. Small 2019, 15, 1903847. [Google Scholar] [CrossRef]
- Meng, H.; Xi, W.; Ren, Z.; Du, S.; Wu, J.; Zhao, L.; Liu, B.; Fu, H. Solar-boosted electrocatalytic oxygen evolution via catalytic site remodelling of CoCr layered double hydroxide. Appl. Catal. B 2021, 284, 119707. [Google Scholar] [CrossRef]
- Yao, T.; An, X.; Han, H.; Chen, J.Q.; Li, C. Photoelectrocatalytic Materials for Solar Water Splitting. Adv. Energy. Mater. 2018, 8, 1800210. [Google Scholar] [CrossRef]
- Li, Y.; Xiao, Y.; Wu, C.; Zhang, D.; Huang, J.; Zhang, Z.; He, J.; Li, C. Strategies To Construct n-Type Si-Based Heterojunctions for Photoelectrochemical Water Oxidation. ACS Mater. Lett. 2022, 4, 779–804. [Google Scholar] [CrossRef]
- Yu, J.; González-Cobos, J.; Dappozze, F.; López-Tenllado, F.J.; Hidalgo-Carrillo, J.; Marinas, A.; Vernoux, P.; Caravaca, A.; Guillard, C. WO3-based materials for photoelectrocatalytic glycerol upgrading into glyceraldehyde: Unravelling the synergistic photo- and electro-catalytic effects. Appl. Catal. B 2022, 318, 121843. [Google Scholar] [CrossRef]
- Yang, Q.; Du, J.; Nie, X.; Yang, D.; Bian, L.; Yang, L.; Dong, F.; He, H.; Zhou, Y.; Yang, H. Magnetic Field-Assisted Photoelectrochemical Water Splitting: The Photoelectrodes Have Weaker Nonradiative Recombination of Carrier. ACS Catal. 2021, 11, 1242–1247. [Google Scholar] [CrossRef]
- Ager, J.W.; Shaner, M.R.; Walczak, K.A.; Sharp, I.D.; Ardo, S. Experimental demonstrations of spontaneous, solar-driven photoelectrochemical water splitting. Energy Environ. Sci. 2015, 8, 2811–2824. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; Wang, X.; Zi, G.; Zhou, C.; Liu, B.; Liu, G.; Wang, L.; Huang, W. One-step supramolecular preorganization constructed crinkly graphitic carbon nitride nanosheets with enhanced photocatalytic activity. J. Mater. Sci. Technol. 2022, 104, 155–162. [Google Scholar] [CrossRef]
- Wang, S.; Wang, X.; Liu, B.; Guo, Z.; Ostrikov, K.; Wang, L.; Huang, W. Vacancy defect engineering of BiVO4 photoanodes for photoelectrochemical water splitting. Nanoscale 2021, 13, 17989–18009. [Google Scholar] [CrossRef]
- Wang, S.; Wang, L.; Huang, W. Bismuth-based photocatalysts for solar energy conversion. J. Mater. Chem. A 2020, 8, 24307–24352. [Google Scholar] [CrossRef]
- Zhang, H.; Song, F. Recent advances in photo-assisted electrocatalysts for energy conversion. J. Mater. Chem. A 2021, 9, 27193–27214. [Google Scholar] [CrossRef]
- Yang, D.; Wang, G.; Wang, X. Photo- and thermo-coupled electrocatalysis in carbon dioxide and methane conversion. Sci. China Mater. 2019, 62, 1369–1373. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, M.; Aza, F.; Gao, Q.; Xu, Z.; Li, L.; Wu, L.; Zhang, X.; Chen, Y. Photothermal-effect-promoted interfacial OH− filling and the conversion of carrier type in (Co1−xNix)3C during water oxidation. J. Mater. Chem. A 2022, 10, 8258–8267. [Google Scholar] [CrossRef]
- Han, X.; Yu, Y.; Huang, Y.; Liu, D.; Zhang, B. Photogenerated Carriers Boost Water Splitting Activity over Transition-Metal/Semiconducting Metal Oxide Bifunctional Electrocatalysts. ACS Catal. 2017, 7, 6464–6470. [Google Scholar] [CrossRef]
- Gu, L.; Zhang, C.; Guo, Y.; Gao, J.; Yu, Y.; Zhang, B. Enhancing Electrocatalytic Water Splitting Activities via Photothermal Effect over Bifunctional Nickel/Reduced Graphene Oxide Nanosheets. ACS Sustain. Chem. Eng. 2019, 7, 3710–3714. [Google Scholar] [CrossRef]
- Mateo, D.; Cerrillo, J.L.; Durini, S.; Gascon, J. Fundamentals and applications of photo-thermal catalysis. Chem. Soc. Rev. 2022, 51, 1547. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Bai, L.; Zhang, X.; Yang, F. Non-Parallel Photo-Assisted Electrocatalysis Mechanism of SnS2/NiO Heterojunction for Efficient Electrocatalytic Oxygen Evolution Reaction. ChemElectroChem 2021, 8, 2087–2093. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, Y.; Li, L.; Li, D.; Jiang, T.; Huang, H.; Jiang, D.; Kang, Z.; Mao, B. Cu5FeS4 quantum dots as a single-component photo-assisted electrocatalyst for efficient hydrogen evolution. J. Mater. Chem. A 2023, 11, 1927–1936. [Google Scholar] [CrossRef]
- Yang, Y.; Ji, Y.; Li, G.; Li, Y.; Jia, B.; Yan, J.; Ma, T.; Liu, S. IrOx@In2O3 Heterojunction from Individually Crystallized Oxides for Weak-Light-Promoted Electrocatalytic Water Oxidation. Angew. Chem. Int. Ed. 2021, 60, 26790–26797. [Google Scholar] [CrossRef]
- Chen, J.; Chen, C.; Qin, M.; Li, B.; Lin, B.; Mao, Q.; Yang, H.; Liu, B.; Wang, Y. Reversible hydrogen spillover in Ru-WO3-x enhances hydrogen evolution activity in neutral pH water splitting. Nat. Commun. 2022, 13, 5382. [Google Scholar] [CrossRef]
- Righi, G.; Plescher, J.; Schmidt, F.-P.; Campen, R.K.; Fabris, S.; Knop-Gericke, A.; Schlögl, R.; Jones, T.E.; Teschner, D.; Piccinin, S. On the origin of multihole oxygen evolution in haematite photoanodes. Nat. Catal. 2022, 5, 888–899. [Google Scholar] [CrossRef]
- Song, J.; Wei, C.; Huang, Z.-F.; Liu, C.; Zeng, L.; Wang, X.; Xu, Z.J. A review on fundamentals for designing oxygen evolution electrocatalysts. Chem. Soc. Rev. 2020, 49, 2196–2214. [Google Scholar] [CrossRef]
- Yang, W.; Prabhakar, R.R.; Tan, J.; Tilley, S.D.; Moon, J. Strategies for enhancing the photocurrent, photovoltage, and stability of photoelectrodes for photoelectrochemical water splitting. Chem. Soc. Rev. 2019, 48, 4979–5015. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Jiang, L.; Wang, B.; Ma, Y. Enhanced electrocatalytic methanol oxidation properties by photo-assisted Fe2O3 nanoplates. Inter. J. Hydrogen Energy 2019, 44, 13214–13220. [Google Scholar] [CrossRef]
- Fang, X.; Cai, S.; Wang, M.; Chen, Z.; Lu, C.; Yang, H. Photogenerated Holes Mediated Nitric Oxide Production for Hypoxic Tumor Treatment. Angew. Chem. Int. Ed. 2021, 60, 7046–7050. [Google Scholar] [CrossRef]
- Malara, F.; Fracchia, M.; Kmentová, H.; Psaro, R.; Vertova, A.; de Souza, D.O.; Aquilanti, G.; Olivi, L.; Ghigna, P.; Minguzzi, A.; et al. Direct Observation of Photoinduced Higher Oxidation States at a Semiconductor/Electrocatalyst Junction. ACS Catal. 2020, 10, 10476–10487. [Google Scholar] [CrossRef]
- Wang, S.; Liu, B.; Wang, X.; Zhang, Y.; Huang, W. Nanoporous MoO3−x/BiVO4 photoanodes promoting charge separation for efficient photoelectrochemical water splitting. Nano Res. 2022, 15, 7026–7033. [Google Scholar] [CrossRef]
- Ding, C.; Shi, J.; Wang, Z.; Li, C. Photoelectrocatalytic Water Splitting: Significance of Cocatalysts, Elec-trolyte, and Interfaces. ACS Catal. 2017, 7, 675–688. [Google Scholar] [CrossRef]
- Kumar, M.; Meena, B.; Subramanyam, P.; Suryakala, D.; Subrahmanyam, C. Recent trends in photoelectrochemical water splitting: The role of cocatalysts. NPG Asia Mater. 2022, 14, 88. [Google Scholar] [CrossRef]
- Li, D.; Shi, J.; Li, C. Transition-Metal-Based Electrocatalysts as Cocatalysts for Photoelectrochemical Water Splitting: A Mini Review. Small 2018, 14, 1704179. [Google Scholar] [CrossRef]
- Pokrant, S.; Dilger, S.; Landsmann, S.; Trottmann, M. Size effects of cocatalysts in photoelectrochemical and photocatalytic water splitting. Mater. Today. Energy. 2017, 5, 158–163. [Google Scholar] [CrossRef]
- Xu, X.; Zhao, H.; Wang, R.; Zhang, Z.; Dong, X.; Pan, J.; Hu, J.; Zeng, H. Identification of few-layer ReS2 as photo-electro integrated catalyst for hydrogen evolution. Nano Energy 2018, 48, 337–344. [Google Scholar] [CrossRef]
- Chang, B.; Zhao, G.; Shao, Y.; Zhang, L.; Huang, B.; Wu, Y.; Hao, X. Photo-enhanced electrocatalysis of sea-urchin shaped Ni3(VO4)2 for the hydrogen evolution reaction. J. Mater. Chem. A 2017, 5, 18038–18043. [Google Scholar] [CrossRef]
- Zhang, Q.; Tan, S.; Mendes, R.G.; Sun, Z.; Chen, Y.; Kong, X.; Xue, Y.; Rümmeli, M.H.; Wu, X.; Chen, S.; et al. Extremely Weak van der Waals Coupling in Vertical ReS2 Nanowalls for High-Current-Density Lithium-Ion Batteries. Adv. Mater. 2016, 28, 2616–2623. [Google Scholar] [CrossRef]
- Zhang, Q.; Fu, L. Novel Insights and Perspectives into Weakly Coupled ReS2 toward Emerging Applications. Chem 2019, 5, 505–525. [Google Scholar] [CrossRef]
- Lu, X.; Liu, R.; Wang, Q.; Xu, C. In Situ Integration of ReS2/Ni3S2 p-n Heterostructure for Enhanced Photoelectrocatalytic Performance. ACS Appl. Mater. Interfaces 2019, 11, 40014–40021. [Google Scholar] [CrossRef]
- Sun, P.; Xing, Z.; Li, Z.; Zhou, W. Recent advances in quantum dots photocatalysts. Chem. Eng. J. 2023, 458, 141399. [Google Scholar] [CrossRef]
- Wu, H.-L.; Li, X.-B.; Tung, C.-H.; Wu, L.-Z. Semiconductor Quantum Dots: An Emerging Candidate for CO2 Photoreduction. Adv. Mater. 2019, 31, 1900709. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Wang, W.; Shi, R.; Tang, H.; Sun, L.; Wang, L.; Liu, Q.; Zhang, T. Recent advances in quantum dot catalysts for hydrogen evolution: Synthesis, characterization, and photocatalytic application. Carbon Energy 2023, e280. [Google Scholar] [CrossRef]
- Yu, Y.; Ma, T.; Huang, H. Semiconducting Quantum Dots for Energy Conversion and Storage. Adv. Funct. Mater. 2023, 2213770. [Google Scholar] [CrossRef]
- Zhong, Y.; Chang, B.; Shao, Y.; Wu, Y.; Hao, X. A photo-responsive electrocatalyst: CdSe quantum dot sensitized WS2 nanosheets for hydrogen evolution in neutral solution. New. J. Chem. 2018, 42, 18021–18027. [Google Scholar] [CrossRef]
- Wang, H.; Lee, J.-M. Recent advances in structural engineering of MXene electrocatalysts. J. Mater. Chem. A 2020, 8, 10604–10624. [Google Scholar] [CrossRef]
- Sun, Y.; Meng, X.; Dall’Agnese, Y.; Dall’Agnese, C.; Duan, S.; Gao, Y.; Chen, G.; Wang, X.-F. 2D MXenes as Co-catalysts in Photocatalysis: Synthetic Methods. Nano-Micro Lett. 2019, 11, 79. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.; Ai, Z.; Shi, D.; Zhong, Y.; Zhang, K.; Shao, Y.; Zhang, L.; Shen, J.; Wu, Y.; Hao, X. p-n tungsten oxide homojunctions for Vis-NIR light-enhanced electrocatalytic hydrogen evolution. J. Mater. Chem. A 2019, 7, 19573–19580. [Google Scholar] [CrossRef]
- Deng, L.; Chang, B.; Shi, D.; Yao, X.; Shao, Y.; Shen, J.; Zhang, B.; Wu, Y.; Hao, X. MXene decorated by phosphorus-doped TiO2 for photo-enhanced electrocatalytic hydrogen evolution reaction. Renew. Energy 2021, 170, 858–865. [Google Scholar] [CrossRef]
- Yan, B.; Liu, D.; Feng, X.; Shao, M.; Zhang, Y. Ru Species Supported on MOF-Derived N-Doped TiO2/C Hybrids as Efficient Electrocatalytic/Photocatalytic Hydrogen Evolution Reaction Catalysts. Adv. Funct. Mater. 2020, 30, 2003007. [Google Scholar] [CrossRef]
- Wan, Z.; He, Q.; Qu, Y.; Dong, J.; Shoko, E.; Yan, P.; Isimjan, T.T.; Yang, X. Designing coral-like Fe2O3-regulated Se-rich CoSe2 heterostructure as a highly active and stable oxygen evolution electrocatalyst for overall water splitting. J. Electroanal. Chem. 2022, 904, 115928. [Google Scholar] [CrossRef]
- Li, L.; Wang, P.; Shao, Q.; Huang, X. Recent Progress in Advanced Electrocatalyst Design for Acidic Oxygen Evolution Reaction. Adv. Mater. 2021, 33, 2004243. [Google Scholar] [CrossRef]
- Li, Z.; Meng, X.; Zhang, Z. Recent development on MoS2-based photocatalysis: A review. J. Photochem. Photobiol. C 2018, 35, 39–55. [Google Scholar] [CrossRef]
- Ma, Q.; Qiao, H.; Huang, Z.; Liu, F.; Duan, C.; Zhou, Y.; Liao, G.; Qi, X. Photo-assisted electrocatalysis of black phosphorus quantum dots/molybdenum disulfide heterostructure for oxygen evolution reaction. Appl. Surf. Sci. 2021, 562, 150213. [Google Scholar] [CrossRef]
- Wu, X.; Ru, Y.; Bai, Y.; Zhang, G.; Shi, Y.; Pang, H. PBA composites and their derivatives in energy and environmental applications. Coord. Chem. Rev. 2022, 451, 214260. [Google Scholar] [CrossRef]
- Song, X.; Song, S.; Wang, D.; Zhang, H. Prussian Blue Analogs and Their Derived Nanomaterials for Electrochemical Energy Storage and Electrocatalysis. Small Methods. 2021, 5, 2001000. [Google Scholar] [CrossRef]
- Goberna-Ferrón, S.; Hernández, W.Y.; Rodríguez-García, B.; Galán-Mascarós, J.R. Light-Driven Water Oxidation with Metal Hexacyanometallate Heterogeneous Catalysts. ACS Catal. 2014, 41, 637–1641. [Google Scholar] [CrossRef]
- Xu, H.; Shang, H.; Jin, L.; Chen, C.; Wang, C.; Du, Y. Boosting electrocatalytic oxygen evolution over Prussian blue analog/transition metal dichalcogenide nanoboxes by photo-induced electron transfer. J. Mater. Chem. A 2019, 7, 26905–26910. [Google Scholar] [CrossRef]
- Sari, F.N.I.; Chen, H.-S.; Anbalagan, A.K.; Huang, Y.-J.; Haw, S.-C.; Chen, J.-M.; Lee, C.-H.; Su, Y.-H.; Ting, J.-M. V-doped, divacancy-containing β-FeOOH electrocatalyst for high performance oxygen evolution reaction. Chem. Eng. J. 2022, 438, 135515. [Google Scholar] [CrossRef]
- Wang, Y.; Ni, Y.; Wang, X.; Zhang, N.; Li, P.; Dong, J.; Liu, B.; Liu, J.; Cao, M.; Hu, C. Template Electro-Etching-Mediated FeOOH Nanotubes as Highly Efficient Photoactive Electrocatalysts for Oxygen Evolution Reaction. ACS Appl. Energy Mater. 2018, 1, 5718–5725. [Google Scholar] [CrossRef]
- Bai, L.; Jia, S.; Gao, Y.; Li, C.; Chen, X.; Zhou, S.; Han, J.; Yang, F.; Zhang, X.; Lu, S. A p-n WO3/SnSe2 Heterojunction for Efficient Photo-assisted Electrocatalysis of the Oxygen Evolution Reaction. Energy Environ. Mater. 2022, e12456. [Google Scholar] [CrossRef]
- Xiong, B.; Chen, L.; Shi, J. Anion-Containing Noble-Metal-Free Bifunctional Electrocatalysts for Overall Water Splitting. ACS Catal. 2018, 8, 3688–3707. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, K.; Lin, H.; Li, X.; Chan, H.C.; Yang, L.; Gao, Q. MoS2–Ni3S2 Heteronanorods as Efficient and Stable Bifunctional Electrocatalysts for Overall Water Splitting. ACS Catal. 2017, 7, 2357–2366. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, T.; Pohl, D.; Rellinghaus, B.; Dong, R.; Liu, S.; Zhuang, X.; Feng, X. Interface Engineering of MoS2/Ni3S2 Heterostructures for Highly Enhanced Electrochemical Overall-Water-Splitting Activity. Angew. Chem. Int. Ed. 2016, 55, 6702–6707. [Google Scholar] [CrossRef]
- Wang, C.; Shao, X.; Pan, J.; Hu, J.; Xu, X. Redox bifunctional activities with optical gain of Ni3S2 nanosheets edged with MoS2 for overall water splitting. Appl. Catal. B 2020, 268, 118435. [Google Scholar] [CrossRef]
- Yang, S.; Guan, H.; Zhong, Y.; Quan, J.; Luo, N.; Gao, Q.; Xu, Y.; Peng, F.; Zhang, S.; Fang, Y. CdS@Ni3S2 for efficient and stable photo-assisted electrochemical (P-EC) overall water splitting. Chem. Eng. J. 2021, 405, 126231. [Google Scholar] [CrossRef]
- Si, F.; Tang, C.; Gao, Q.; Peng, F.; Zhang, S.; Fang, Y.; Yang, S. Bifunctional CdS@Co9S8/Ni3S2 catalyst for efficient electrocatalytic and photo-assisted electrocatalytic overall water splitting. J. Mater. Chem. A 2020, 8, 3083–3096. [Google Scholar] [CrossRef]
- Dong, Q.; Li, M.; Sun, M.; Si, F.; Gao, Q.; Cai, X.; Xu, Y.; Yuan, T.; Zhang, S.; Peng, F.; et al. Phase-Controllable Growth NixPy Modified CdS@Ni3S2 Electrodes for Efficient Electrocatalytic and Enhanced Photoassisted Electrocatalytic Overall Water Splitting. Small Methods 2021, 8, 2100878. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Han, M.; Chen, R.; Li, S.-L.; Xie, S.-L.; Mao, C.; Bu, X.; Cao, X.-L.; Dong, L.-Z.; Feng, P.; et al. Hexagonal@Cubic CdS Core@Shell Nanorod Photocatalyst for Highly Active Production of H2 with Unprecedented Stability. Adv. Mater. 2016, 28, 8906–8911. [Google Scholar] [CrossRef]
- Guan, H.; Zhang, S.; Cai, X.; Gao, Q.; Yu, X.; Zhou, X.; Peng, F.; Fang, Y.; Yang, S. CdS@Ni3S2 core–shell nanorod arrays on nickel foam: A multifunctional catalyst for efficient electrochemical catalytic, photoelectrochemical and photocatalytic H2 production reaction. J. Mater. Chem. A 2019, 7, 2560–2574. [Google Scholar] [CrossRef]
- Sahoo, D.P.; Das, K.K.; Mansingh, S.; Sultana, S.; Parida, K. Recent progress in first row transition metal Layered double hydroxide (LDH) based electrocatalysts towards water splitting: A review with insights on synthesis. Coord. Chem. Rev. 2022, 469, 214666. [Google Scholar] [CrossRef]
- Wang, Y.; Yan, D.; El Hankari, S.; Zou, Y.; Wang, S. Recent Progress on Layered Double Hydroxides and Their Derivatives for Electrocatalytic Water Splitting. Adv. Sci. 2018, 5, 1800064. [Google Scholar] [CrossRef] [PubMed]
- Zhai, P.; Xia, M.; Wu, Y.; Zhang, G.; Gao, J.; Zhang, B.; Cao, S.; Zhang, Y.; Li, Z.; Fan, Z.; et al. Engineering single-atomic ruthenium catalytic sites on defective nickel-iron layered double hydroxide for overall water splitting. Nat. Commun. 2021, 12, 4587. [Google Scholar] [CrossRef]
- Yang, R.; Zhou, Y.; Xing, Y.; Li, D.; Jiang, D.; Chen, M.; Shi, W.; Yuan, S. Synergistic coupling of CoFe-LDH arrays with NiFe-LDH nanosheet for highly efficient overall water splitting in alkaline media. Appl. Catal. B 2019, 253, 131–139. [Google Scholar] [CrossRef]
- Bodhankar, P.M.; Sarawade, P.B.; Singh, G.; Vinu, A.; Dhawale, D.S. Recent advances in highly active nanostructured NiFe LDH catalyst for electrochemical water splitting. J. Mater. Chem. A 2021, 9, 3180–3208. [Google Scholar] [CrossRef]
- Zhang, D.; Dong, W.; Liu, Y.; Gu, X.; Yang, T.; Hong, Q.; Li, D.; Zhang, D.; Zhou, H.; Huang, H.; et al. Ag-In-Zn-S Quantum Dot-Dominated Interface Kinetics in Ag-In-Zn-S/NiFe LDH Composites toward Efficient Photoassisted Electrocatalytic Water Splitting. ACS Appl. Mater. Interfaces 2021, 13, 42125–42137. [Google Scholar] [CrossRef]
- Yu, J.; Liu, Z.; Yu, F.; Bao, W.; Peng, B.; Wang, G.; Zhang, L.; Xu, Y.; Wang, F. Enhanced photoelectrochemical performance of ZnO/NiFe-layered double hydroxide for water splitting: Experimental and photo-assisted density functional theory calculations. J. Colloid Interface Sci. 2022, 623, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Chen, Z.; Li, Y.; Wu, J.; Wang, X.; Huang, H.; Liu, Y.; Dong, B.; Shao, M.; Kang, Z. Polyaniline/Carbon Dots Composite as a Highly Efficient Metal-Free Dual-Functional Photoassisted Electrocatalyst for Overall Water Splitting. ACS Appl. Mater. Interfaces 2021, 13, 24814–24823. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, S.; Khare, N. Enhanced photosensitization of zinc oxide nanorods using polyaniline for efficient photocatalytic and photoelectrochemical water splitting. Inter. J. Hydrogen Energy 2016, 412, 1088–21098. [Google Scholar] [CrossRef]
- Huang, Z.-F.; Song, J.; Du, Y.; Dou, S.; Sun, L.; Chen, W.; Yuan, K.; Dai, Z.; Wang, X. Optimizing interfacial electronic coupling with metal oxide to activate inert polyaniline for superior electrocatalytic hydrogen generation. Carbon Energy 2019, 1, 77–84. [Google Scholar] [CrossRef]
- Xu, M.; Gao, W.; Zhao, R.; Sun, J.; Han, Y.; Niu, X.; Xu, Z.; Qin, Y.; Wang, Q. Electrodeposition of Metal-Free Polyaniline Electrocatalyst for Efficient Oxygen Evolution in Acid. ChemElectroChem 2022, 9, e202200901. [Google Scholar] [CrossRef]
- Xia, C.; Zhu, S.; Feng, T.; Yang, M.; Yang, B. Evolution and Synthesis of Carbon Dots: From Carbon Dots to Carbonized Polymer Dots. Adv. Sci. 2019, 6, 1901316. [Google Scholar] [CrossRef]
- Kang, Z.; Lee, S.-T. Carbon dots: Advances in nanocarbon applications. Nanoscale 2019, 11, 19214–19224. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; He, Q.; Wang, L.; Zhu, C.; Golani, P.; Handoko, A.D.; Yu, X.; Gao, C.; Ding, M.; Wang, X.; et al. Self-gating in semiconductor electrocatalysis. Nat. Mater. 2019, 18, 1098–1104. [Google Scholar] [CrossRef] [PubMed]


| Catalyst | Electrolyte | Performance Comparison, Light (Dark) | Photocurrent Density | Ref. | |
|---|---|---|---|---|---|
| Overpotential /mV | Tafel Slope /mV dec−1 | ||||
| Ni3(VO4)2 | 0.5 M H2SO4 | 90(122), 10 mA cm−2 | 50(82) | NA | [61] |
| ReS2/carbon fiber clothes | 0.5 M H2SO4 | 167(206), 10 mA cm−2 | 80(83) | NA | [60] |
| ReS2/Ni3S2 | 1.0 M KOH | 106(168), 10 mA cm−2 | 111(118) | 0.058 mA.cm−2 at −0.15 V vs. RHE | [64] |
| Cu5FeS4 QDs | 1.0 M KOH | 52(118), 10 mA cm−2 | 133(143) | NA | [46] |
| CdSe QDs/WS2 | 0.5 M Na2SO4 | 400(1030), 10 mA cm−2 | 56(132) | NA | [69] |
| MXene@P-WO3 | 1.0 M KOH | 44(162), 10 mA cm−2 | 41(102) | 200 μAcm−2 | [72] |
| MXene@P-TiO2 | 1.0 M KOH | 97(138), 10 mA cm−2 | 48.4(73.5) | NA | [73] |
| Ru@N-TiO2/C | 1.0 M KOH | 76(97), 10 mA cm−2 | NA | NA | [74] |
| Catalyst | Electrolyte | Performance Comparison, Light (Dark) | Photocurrent Density | Ref. | |
|---|---|---|---|---|---|
| Overpotential /mV | Tafel Slope /mV dec−1 | ||||
| IrOx@In2O3 | 1 M KOH | 176(190), 10 mA cm−2 210(231), 50 mA cm−2 | 47(55) | NA | [47] |
| BP QDs/MoS2 | 1 M KOH | 180(330), 10 mA cm−2 | 95(108) | NA | [78] |
| CoFe PBA/CoS2 | 1 M KOH | 265(301), 10 mA cm−2 | 59(80) | 6.5 mA cm−2 at 1.6 V | [82] |
| FeOOH/CC | 1 M KOH | 328(352), 10 mA cm−2 | 42(47) | NA | [84] |
| WO3/SnSe2/CoFe-LDH/CNTs | 1 M KOH | 224(291), 10 mA cm−2 | 47(78) | 53 μA cm−2 at 1.23 vs. RHE | [85] |
| CoCr-LDH | 1 M KOH | 338(360), 10 mA cm−2 | 74(85) | 57 μA cm−2 at η = 70 mV | [30] |
| SnS2/NiO | 1 M KOH | 310(388), 10 mA cm−2 | 190(215) | 69.50 μAcm−2 at 1.23 V vs. RHE | [45] |
| Catalyst | Electrolyte | Performance Comparison, Light (Dark) | Photocurrent Density | Ref. | |
|---|---|---|---|---|---|
| Overpotential /mV | Tafel Slope /mV dec−1 | ||||
| MoS2/Ni3S2 | 1 M KOH | 1500(1530), 10 mA cm−2 | NA | NA | [89] |
| CdS/Ni3S2 | 1 M KOH | NA | 75.9(85.8) | NA | [90] |
| CdS/Co9S8/Ni3S2 | 1 M KOH | 285(300), 10 mA cm−2 | 87.2(91.3) | 10 mA cm−2 at 1.56 vs. RHE | [91] |
| CdS/Ni3S2/NixPy | 1 M KOH | NA | 103(108.7) | NA | [92] |
| AgInZnS QDs/NiFe-LDH | 1 M KOH | 1620(1670), 10 mA cm−2 | 105(111.7), 54.6 (71.6) | NA | [100] |
| ZnO/NiFe-LDH | 1 M KOH | 1630(1730), 10 mA cm−2 | 87.03(219.09), 67.28(201.47) | NA | [101] |
| PANI/CDs | 0.5 M K2SO4 | Decrease by 150 mV @30 mA cm−2/65 mV @20 mA cm−2 | 192(283) | 5.12 mA cm−2 at 2.0 V | [102] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Zheng, X.; Zhen, Y.; Liang, Y. Photogenerated Carrier-Assisted Electrocatalysts for Efficient Water Splitting. Catalysts 2023, 13, 712. https://doi.org/10.3390/catal13040712
Li X, Zheng X, Zhen Y, Liang Y. Photogenerated Carrier-Assisted Electrocatalysts for Efficient Water Splitting. Catalysts. 2023; 13(4):712. https://doi.org/10.3390/catal13040712
Chicago/Turabian StyleLi, Xiang, Xueyan Zheng, Yanzhong Zhen, and Yucang Liang. 2023. "Photogenerated Carrier-Assisted Electrocatalysts for Efficient Water Splitting" Catalysts 13, no. 4: 712. https://doi.org/10.3390/catal13040712








