Inactivation of E. Coli in Water Using Photocatalytic, Nanostructured Films Synthesized by Aerosol Routes
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation and Characterization of TiO2 Films by ACVD
| Test | Morphology | Temperature of substrate (°C) | Precursor feed rate (μmol/min) | Particleratio (%) | Residence time (ms) | Depositiontime (min) |
|---|---|---|---|---|---|---|
| 1 | Columnar | 500 | 1.53 | 13 | 20 | 50 |
| 2 | Granular | 450 | 4.34 | 30 | 53 | 70 |
| 3 | Branched | 450 | 4.34 | 18 | 30 | 23 |
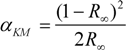
2.2. Estimation of Surface Area of TiO2 Films
2.3. Culture and Sampling Cells
2.4. Light and Material Control Tests
2.5. Photocatalytic and Photoelectrochemical Inactivation of E. coli
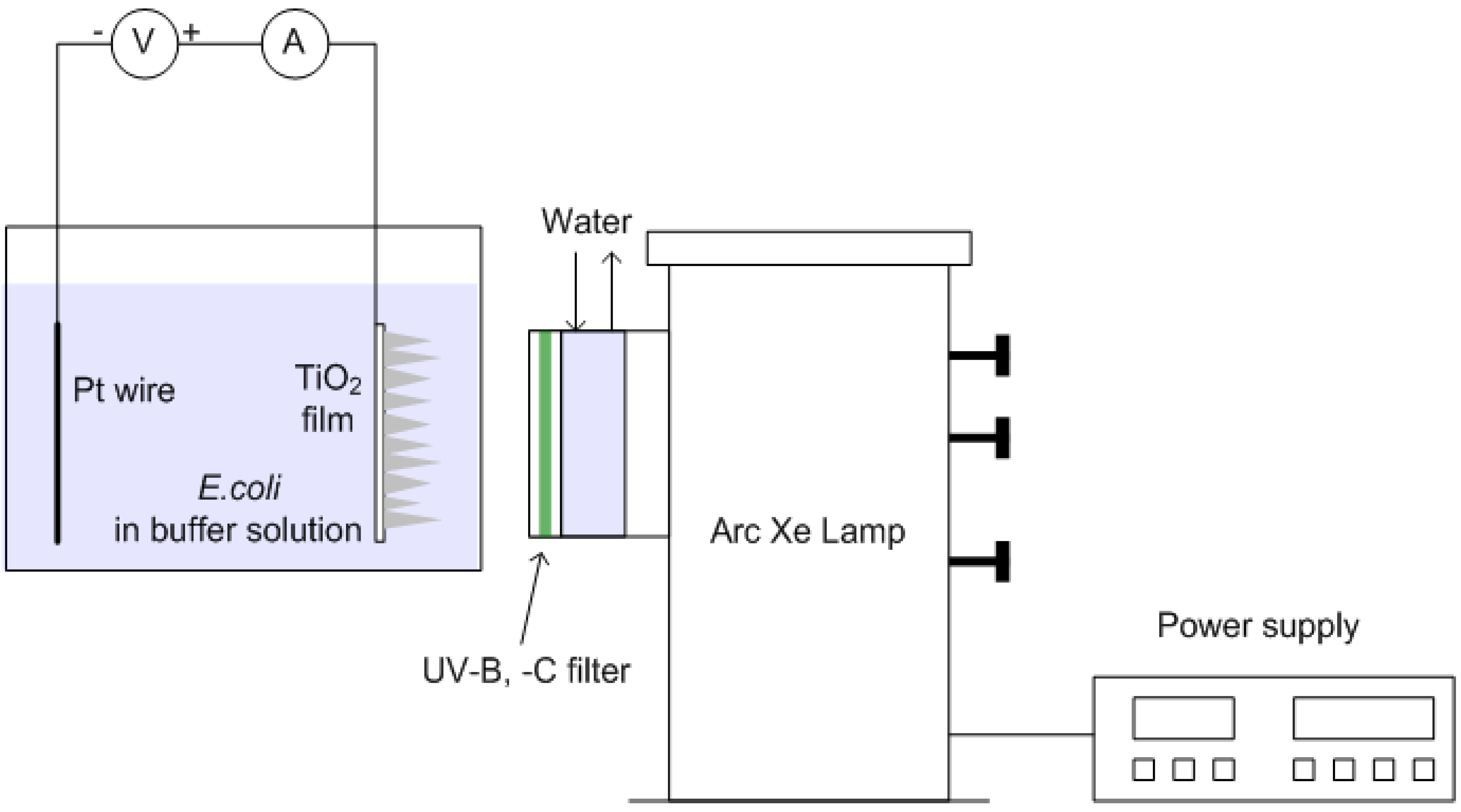
| No. | Test | Objective | Experimental conditions |
|---|---|---|---|
| 1 | Light control test | Examine effect of light on viability of E. coli cells without TiO2 films | Reactor volume: 50 mL |
| 2 | Material control test | Examine effect of TiO2 films on viability of E. coli cells without irradiation of light | Initial active cell concentration (C0): 108 cells/mL |
| 3 | Photocatalytic inactivation | Study inactivation of E. coli with TiO2 films under irradiation of light without external voltage | Light source: 450 W arc Xe lamp |
| 4 | Photoelectrochemical inactivation | Study inactivation of E. coli with TiO2 films under irradiation of light with external voltage | TiO2 films: columnar, granular, and branched structure |
3. Results and Discussion

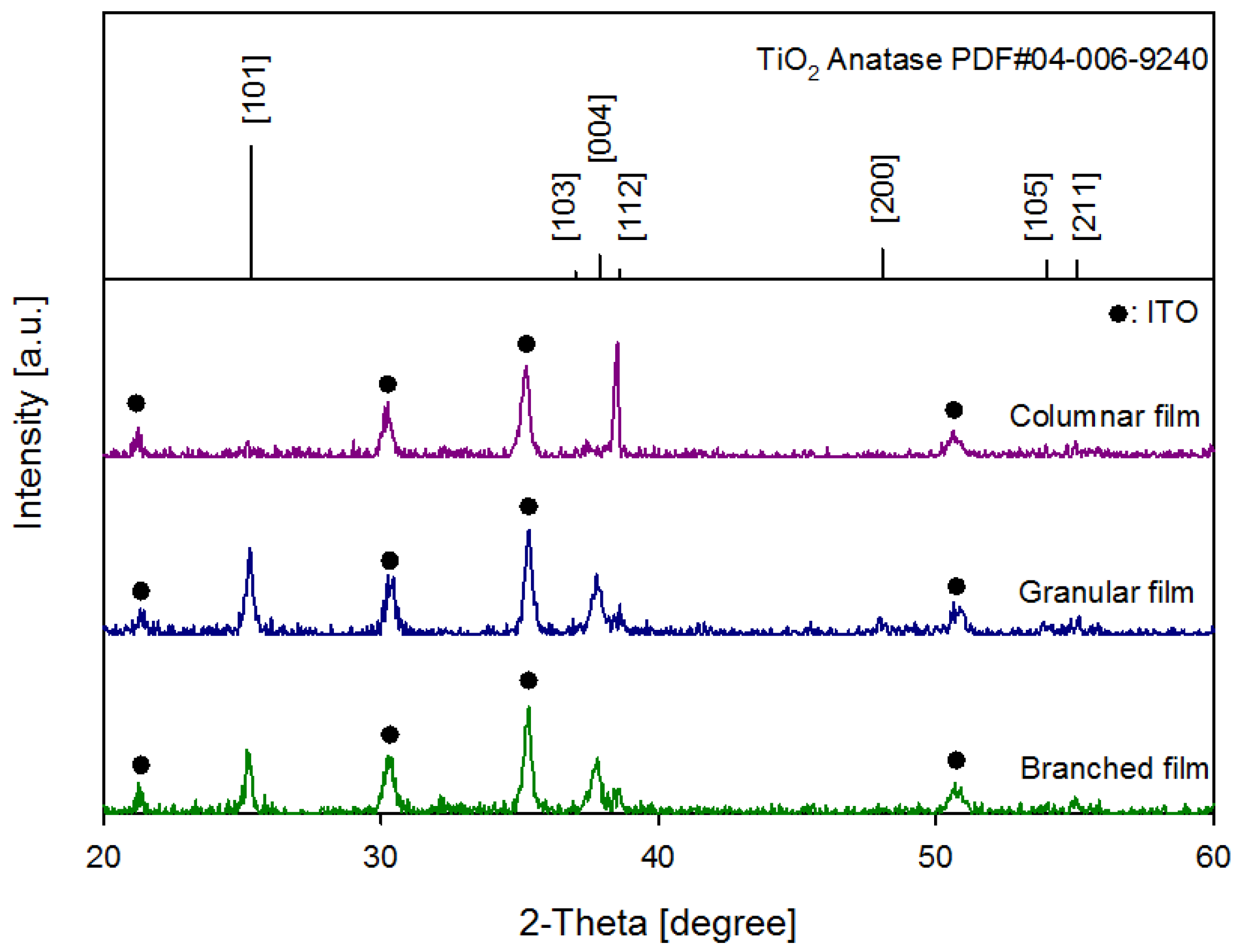
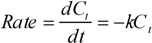
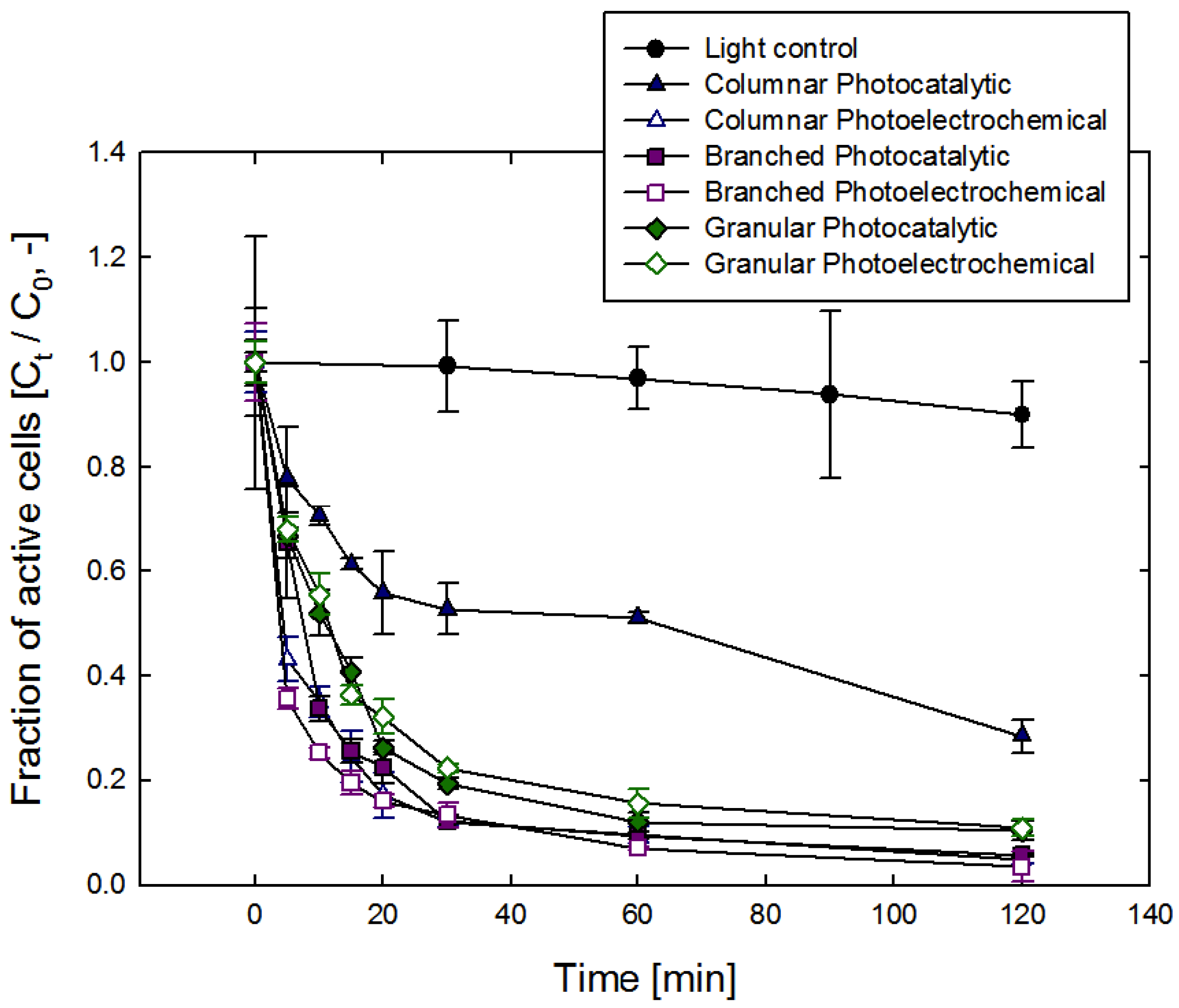
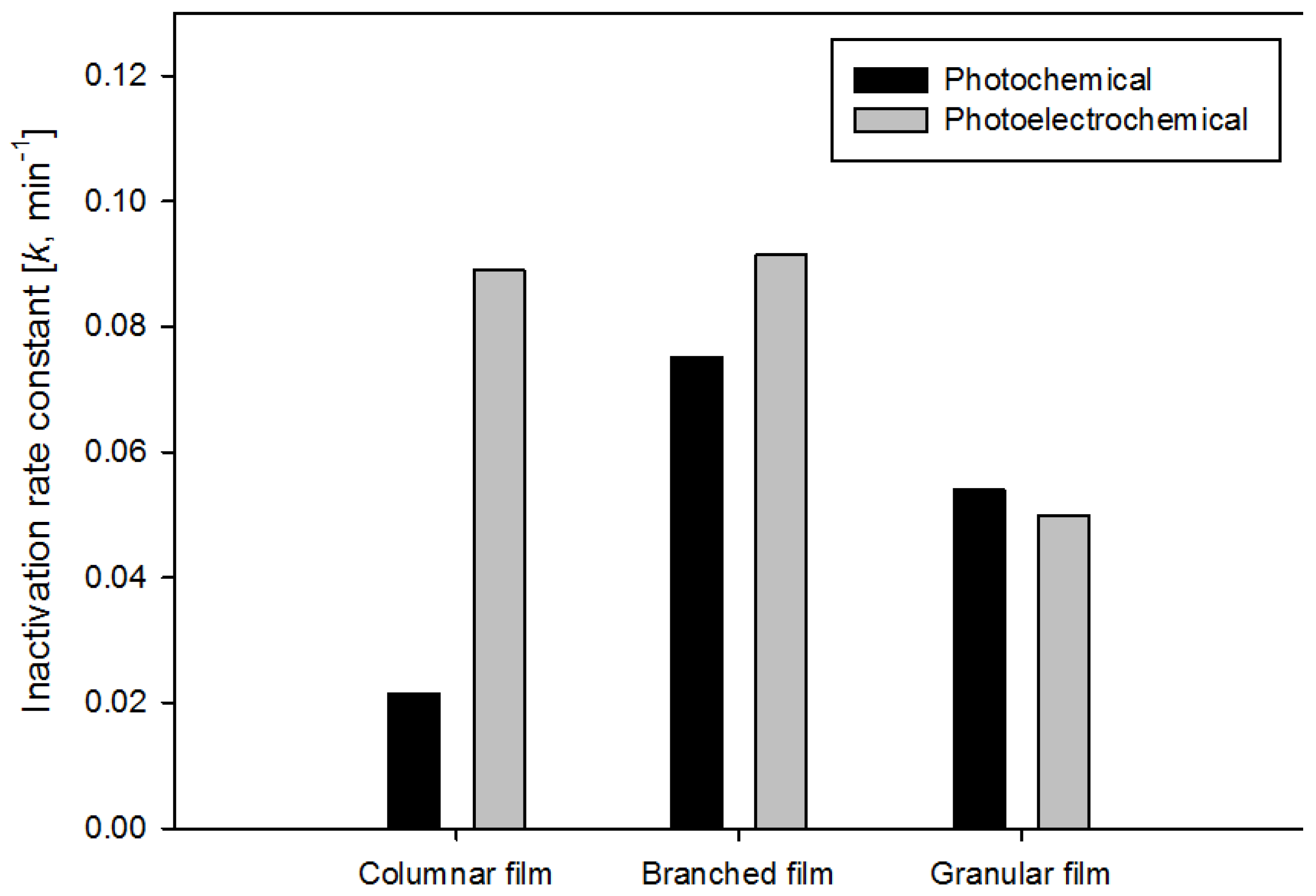
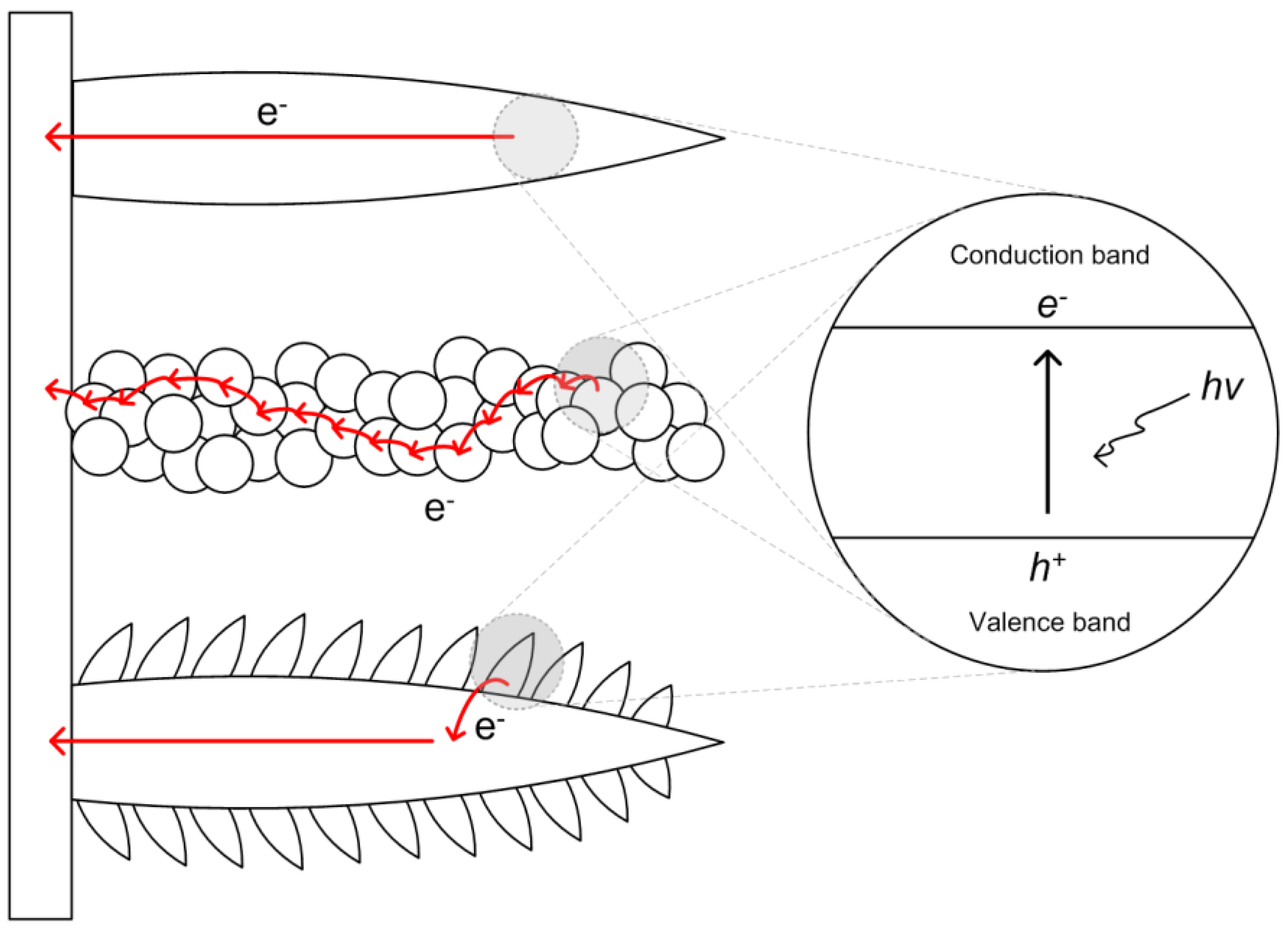
| Columnar film | Granular film-1 | Granular film-2 | Branched film | |
|---|---|---|---|---|
| Total surface area of TiO2 (m2) | 6.88 × 10−4 | 3.18 × 10−3 | 1.69 × 10−2 | 6.34 × 10−3 |
| Normalized rate constant of PC a by surface area (k’, min−1·m−2) | 31.1 | 16.9 | 3.2 | 11.8 |
| Normalized rate constant of PEC b by surfance area (k’, min−1·m−2) | 129.4 | 15.7 | 2.9 | 14.4 |
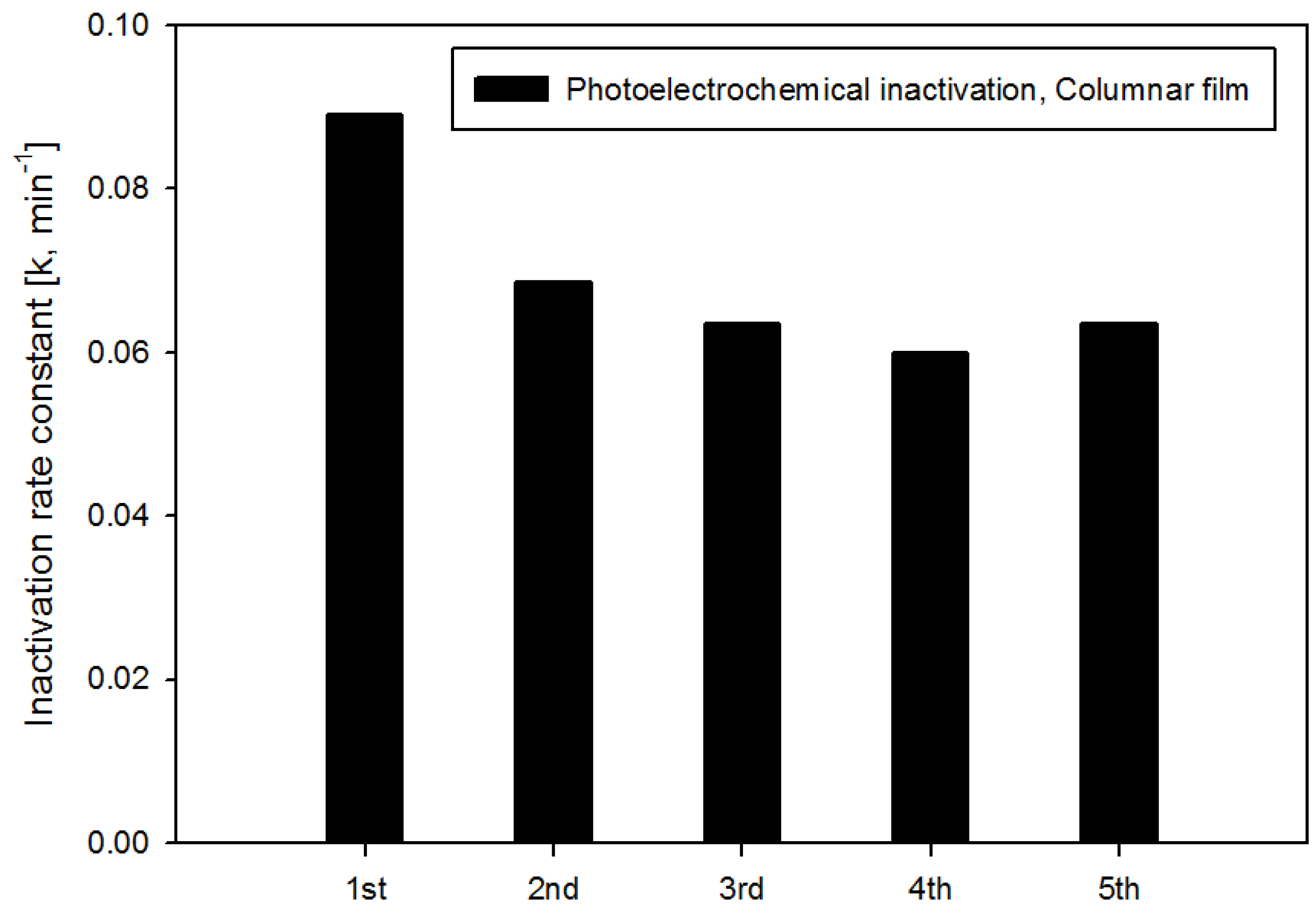
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayers, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar]
- Montgomery, M.A.; Elimelech, M. Water and sanitation in developing countries: Including health in the equation. Environ. Sci. Technol. 2007, 41, 17–24. [Google Scholar] [CrossRef]
- Kemper, K.E. Groundwater-from development to management. Hydrogeol. J. 2003, 12, 3–5. [Google Scholar] [CrossRef]
- Szewzyk, U.; Szewzyk, R.; Manz, W.; Schleifer, K.H. Microbiological safety of drinking water. Annu. Rev. Microbiol. 2000, 54, 81–127. [Google Scholar] [CrossRef]
- Gopal, K.; Tripathy, S.S.; Bersillon, J.L.; Dubey, S.P. Chlorination byproducts, their toxicodynamics and removal from drinking water. J. Hazard. Mater. 2007, 140, 1–6. [Google Scholar] [CrossRef]
- Krasner, S.W.; Weinberg, H.S.; Richardson, S.D.; Pastor, S.J.; Chinn, R.; Sclimenti, M.J.; Onstad, G.D.; Thruston, A.D. Occurrence of a new generation of disinfection byproducts. Environ. Sci. Technol. 2006, 40, 7175–7185. [Google Scholar]
- Zhang, D.; Li, G.; Yu, J.C. Inorganic materials for photocatalytic water disinfection. J. Mater. Chem. 2010, 20, 4529–4536. [Google Scholar] [CrossRef]
- Matsunaga, T.; Tomoda, R.; Nakajima, T.; Wake, H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol. Lett. 1985, 29, 211–214. [Google Scholar] [CrossRef]
- Wei, C.; Lin, W.Y.; Zaina, Z.; Williams, N.E.; Zhu, K.; Kruzic, A.P.; Smith, R.L.; Rajeshwar, K. Bactericidal activity of TiO2 photocatalyst in aqueous media: toward a solar-assisted water disinfection system. Environ. Sci. Technol. 1994, 28, 934–938. [Google Scholar] [CrossRef]
- Hu, C.; Lan, Y.; Qu, J.; Hu, X.; Wang, A. Ag/AgBr/TiO2 visible light photocatalyst for destruction of azodyes and bacteria. J. Phys. Chem. B 2006, 110, 4066–4072. [Google Scholar]
- Hu, C.; Guo, J.; Qu, J.; Hu, X. Photocatalytic degradation of pathogenic bacteria with AgI/TiO2 under visible light irradiation. Langmuir 2007, 23, 4982–4987. [Google Scholar] [CrossRef]
- Wu, B.; Zhuang, W.Q.; Sahu, M.; Biswas, P.; Tang, Y.J. Cu-doped TiO(2) nanoparticles enhance survival of Shewanella oneidensis MR-1 under ultraviolet light (UV) exposure. Sci. Total Environ. 2011, 409, 4635–4639. [Google Scholar] [CrossRef]
- Wang, P.; Huang, B.; Qin, X.; Zhang, X.; Dai, Y.; Whangbo, M.H. Ag/AgBr/WO3 3H2O: Visible-light photocatalyst for bacteria destruction. Inorg. Chem. 2009, 48, 10697–10702. [Google Scholar] [CrossRef]
- Mccullagh, C.; Robertson, J.M.C.; Bahnemann, D.W.; Robertson, P.K.J. The application of TiO2 photocatalysis for disinfection of water contaminated with pathogenic micro-organisms: A review. Res. Chem. Intermed. 2007, 33, 359–375. [Google Scholar] [CrossRef]
- Amezaga-Madrid, P.; Nevarez-Moorillon, G.V.; Orrantia-Borunda, E.; Miki-Yoshida, M. Photoinduced bactericidal activity against Pseudomonas aeruginosa by TiO2 based thin films. FEMS Microbiol. Lett. 2002, 211, 183–188. [Google Scholar]
- Kuhn, K.P.; Chaberny, I.F.; Massholder, K.; Stickler, M.; Benz, V.W.; Sonntag, H.G.; Erdinger, L. Disinfection of surfaces by photocatalytic oxidation with titanium dioxide and UVA light. Chemosphere 2003, 53, 71–77. [Google Scholar] [CrossRef]
- Sunada, K.; Watanabe, T.; Hashimoto, K. Studies on photokilling of bacteria on TiO2 thin film. J. Photochem. Photobiol. A 2003, 156, 227–233. [Google Scholar] [CrossRef]
- Reddy, M.P.; Phil, H.H.; Subrahmanyam, M. Photocatalytic disinfection of Escherichia coli over titanium (IV) oxide supported on Hβ zeolite. Catal. Lett. 2008, 123, 56–64. [Google Scholar]
- Yu, H.; Quan, X.; Zhang, Y.; Ma, N.; Chen, S.; Zhao, H. Electrochemically assisted photocatalytic inactivation of Escherichia coli under visible light using a ZnIn2S4 film electrode. Langmuir 2008, 24, 7599–7604. [Google Scholar] [CrossRef]
- Hayden, S.C.; Allam, N.K.; El-Sayed, M.A. TiO2 Nanotube/CdS hybrid electrodes: Extraordinary enhancement in the inactivation of Escherichia coli. J. Am. Chem. Soc. 2010, 132, 14406–14408. [Google Scholar] [CrossRef]
- Thimsen, E.; Rastgar, N.; Biswas, P. Nanostructured TiO2 films with controlled morphology synthesized in a single step process: Performance of dye-sensitized solar cells and photo watersplitting. J. Phys. Chem. C 2008, 112, 4134–4140. [Google Scholar] [CrossRef]
- An, W.J.; Thimsen, E.; Biswas, P. Aerosol-chemical vapor deposition method for synthesis of nanostructured metal oxide thin films with controlled morphology. J. Phys. Chem. Lett. 2010, 1, 249–253. [Google Scholar] [CrossRef]
- An, W.J.; Jiang, D.D.; Matthews, J.R.; Borrelli, N.F.; Biswas, P. Thermal conduction effects impacting morphology during synthesis of columnar nanostructured TiO2 thin films. J. Mater. Chem. 2011, 21, 7913–7921. [Google Scholar] [CrossRef]
- Reyes-Coronado, D.; Rodriguez-Gattorno, G.; Espinosa-Pesqueira, M.E.; Cab, C.; Coss, R.; Oskam, G. Phase-pure TiO2 nanoparticles: anatase, brookite and rutile. Nanotechnology 2008, 19. [Google Scholar] [CrossRef]
- Chang, J.C.; Ossoff, S.F.; Lobe, D.C.; Dorfman, M.H.; Dumais, C.M.; Qualls, R.G.; Johnson, J.D. UV inactivation of pathogenic and indicator microorganisms. Appl. Environ. Microbiol. 1985, 49, 1361–1365. [Google Scholar]
- McGuigan, K.G.; Joyce, T.M.; Conroy, R.M. Solar disinfection: use of sunlight to decontaminate drinking water in developing countries. J. Med. Microbiol. 1999, 48, 785–787. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Park, J.; Kettleson, E.; An, W.-J.; Tang, Y.J.; Biswas, P. Inactivation of E. Coli in Water Using Photocatalytic, Nanostructured Films Synthesized by Aerosol Routes. Catalysts 2013, 3, 247-260. https://doi.org/10.3390/catal3010247
Park J, Kettleson E, An W-J, Tang YJ, Biswas P. Inactivation of E. Coli in Water Using Photocatalytic, Nanostructured Films Synthesized by Aerosol Routes. Catalysts. 2013; 3(1):247-260. https://doi.org/10.3390/catal3010247
Chicago/Turabian StylePark, Jinho, Eric Kettleson, Woo-Jin An, Yinjie J. Tang, and Pratim Biswas. 2013. "Inactivation of E. Coli in Water Using Photocatalytic, Nanostructured Films Synthesized by Aerosol Routes" Catalysts 3, no. 1: 247-260. https://doi.org/10.3390/catal3010247




